The Consequences of Our Changing Environment on Life Threatening and Debilitating Fungal Diseases in Humans
Abstract
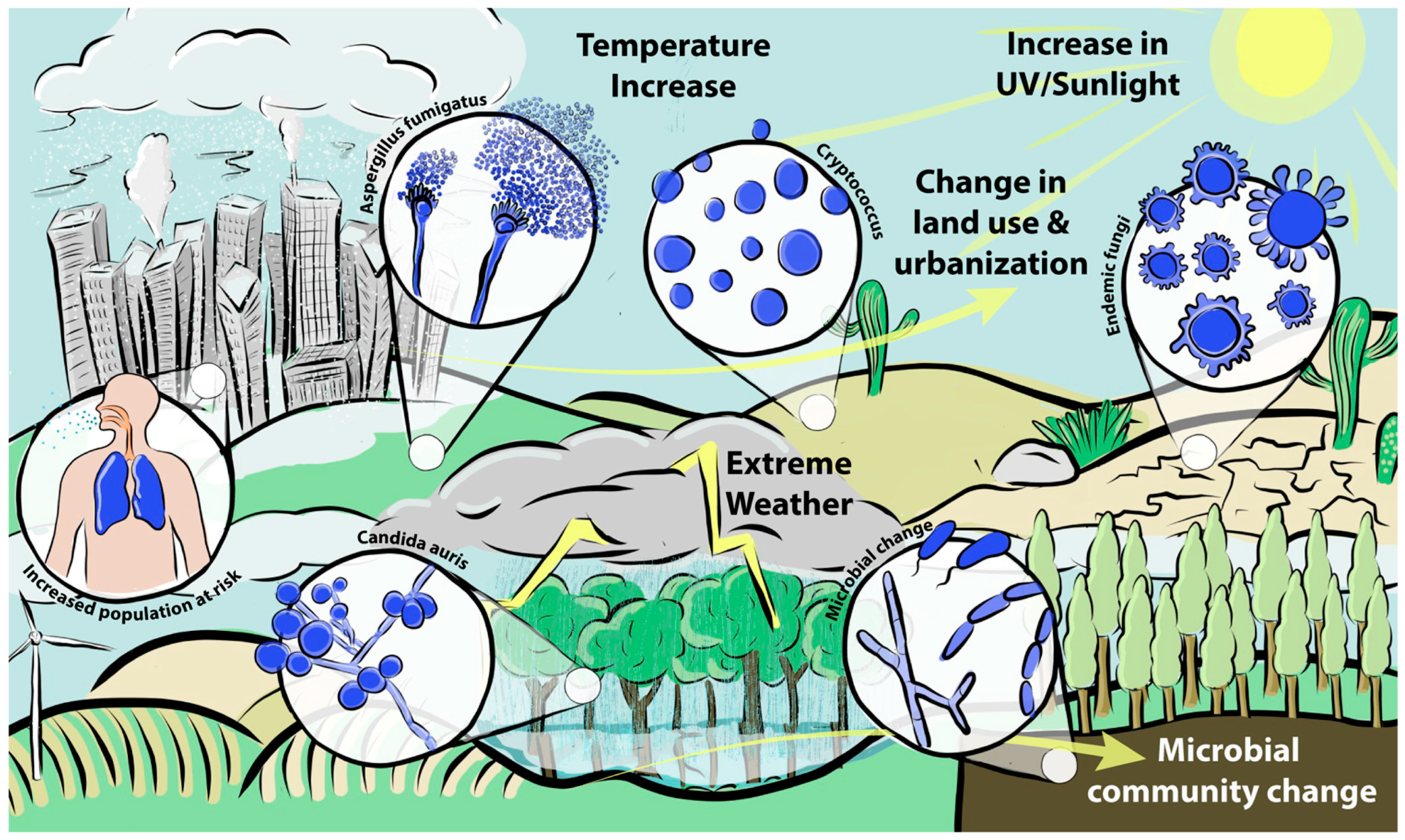
1. Orientation
2. The Ability to Infect Requires Pathogens to Adapt to a Hostile Host Environment
3. How Climate Change Could Affect Species Commonly Associated with Infections of Humans
3.1. Cryptococcus
3.2. Aspergillus
3.3. Mucorales
3.4. Candida
4. The Spectre of Newly Arising Fungal Pathogens
5. Geographic Spread of Endemic Mycoses
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Hansen, J.; Sato, M.; Ruedy, R.; Lo, K.; Lea, D.W.; Medina-Elizade, M. Global temperature change. Proc. Natl. Acad. Sci. USA 2006, 103, 14288–14293. [Google Scholar] [CrossRef]
- Pachauri, R.K.; Allen, M.R.; Barros, V.R.; Broome, J.; Cramer, W.; Christ, R.; Church, J.A.; Clarke, L.; Dahe, Q.; Dasgupta, P.; et al. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Pachauri, R., Meyer, L., Eds.; IPCC: Geneva, Switzerland, 2014; 151p. [Google Scholar]
- Hansen, J.; Sato, M.; Kharecha, P.; Russell, G.; Lea, D.W.; Siddall, M. Climate change and trace gases. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2007, 365, 1925–1954. [Google Scholar] [CrossRef] [PubMed]
- Seneviratne, S.I.; Donat, M.G.; Mueller, B.; Alexander, L.V. No pause in the increase of hot temperature extremes. Nat. Clim. Chang. 2014, 4, 161–163. [Google Scholar] [CrossRef]
- Hansen, J.; Sato, M. Regional climate change and national responsibilities. Environ. Res. Lett. 2016, 11, 034009. [Google Scholar] [CrossRef]
- Karl, T.R.; Trenberth, K.E. Modern global climate change. Science 2003, 302, 1719–1723. [Google Scholar] [CrossRef]
- Tabari, H. Climate change impact on flood and extreme precipitation increases with water availability. Sci. Rep. 2020, 10, 13768. [Google Scholar] [CrossRef]
- Bonfils, C.J.W.; Santer, B.D.; Fyfe, J.C.; Marvel, K.; Phillips, T.J.; Zimmerman, S.R.H. Human influence on joint changes in temperature, rainfall and continental aridity. Nat. Clim. Chang. 2020, 10, 726–731. [Google Scholar] [CrossRef]
- Zeng, Z.; Ziegler, A.D.; Searchinger, T.; Yang, L.; Chen, A.; Ju, K.; Piao, S.; Li, L.Z.X.; Ciais, P.; Chen, D.; et al. A reversal in global terrestrial stilling and its implications for wind energy production. Nat. Clim. Chang. 2019, 9, 979–985. [Google Scholar] [CrossRef]
- McKenzie, R.L.; Aucamp, P.J.; Bais, A.F.; Björn, L.O.; Ilyas, M.; Madronich, S. Ozone depletion and climate change: Impacts on UV radiation. Photochem. Photobiol. Sci. 2011, 10, 182–198. [Google Scholar] [CrossRef]
- Weiskerger, C.J.; Brandão, J.; Ahmed, W.; Aslan, A.; Avolio, L.; Badgley, B.D.; Boehm, A.B.; Edge, T.A.; Fleisher, J.M.; Heaney, C.D.; et al. Impacts of a changing earth on microbial dynamics and human health risks in the continuum between beach water and sand. Water Res. 2019, 162, 456–470. [Google Scholar] [CrossRef]
- Wang, D.Y.C.; Kumar, S.; Hedges, S.B. Divergence time estimates for the early history of animal phyla and the origin of plants, animals and fungi. Proc. R. Soc. B Boil. Sci. 1999, 266, 163–171. [Google Scholar] [CrossRef]
- Tedersoo, L.; Bahram, M.; Põlme, S.; Kõljalg, U.; Yorou, N.S.; Wijesundera, R.L.C.; Ruiz, L.V.; Vasco-Palacios, A.M.; Thu, P.Q.; Suija, A.; et al. Global diversity and geography of soil fungi. Science 2014, 346, 1256688. [Google Scholar] [CrossRef]
- Kubicek, C.P.; Druzhinina, I.S. (Eds.) Fungi in Extreme Environments. In Environmental and Microbial Relationships; Springer: Berlin/Heidelberg, Germany, 2007; pp. 85–103. [Google Scholar]
- Hawksworth, D.L.; Lücking, R. Fungal diversity revisited: 2.2 to 3.8 million species. In The Fungal Kingdom; Heitman, J., Howlett, B.J., Crous, P.W., Stukenbrock, E.H., James, T.Y., Gow, N.A.R., Eds.; Wiley Online Library: Hoboken, NJ, USA, 2017; pp. 79–95. [Google Scholar] [CrossRef]
- Henkel, T.W.; Aime, M.C.; Chin, M.M.L.; Miller, S.L.; Vilgalys, R.; Smith, M.E. Ectomycorrhizal fungal sporocarp diversity and discovery of new taxa in Dicymbe monodominant forests of the Guiana Shield. Biodivers. Conserv. 2011, 21, 2195–2220. [Google Scholar] [CrossRef]
- López-Quintero, C.A.; Straatsma, G.; Franco-Molano, A.E.; Boekhout, T. Macrofungal diversity in Colombian Amazon forests varies with regions and regimes of disturbance. Biodivers. Conserv. 2012, 21, 2221–2243. [Google Scholar] [CrossRef]
- Truong, C.; Mujic, A.B.; Healy, R.; Kuhar, F.; Furci, G.; Torres, D.; Niskanen, T.; Sandoval-Leiva, P.A.; Fernández, N.; Escobar, J.M.; et al. How to know the fungi: Combining field inventories and DNA-barcoding to document fungal diversity. New Phytol. 2017, 214, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Kwon-Chung, K.; Bennett, J. Medical mycology. Revista do Instituto de Medicina Tropical de São Paulo 1992, 34, 504. [Google Scholar] [CrossRef]
- Naranjo-Ortiz, M.A.; Gabaldón, T. Fungal evolution: Major ecological adaptations and evolutionary transitions. Biol. Rev. 2019, 94, 1443–1476. [Google Scholar] [CrossRef] [PubMed]
- Robert, V.A.; Casadevall, A. Vertebrate Endothermy Restricts Most Fungi as Potential Pathogens. J. Infect. Dis. 2009, 200, 1623–1626. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A. Thermal restriction as an antimicrobial function of fever. PLoS Pathog. 2016, 12, e1005577. [Google Scholar] [CrossRef] [PubMed]
- Romani, L. Immunity to fungal infections. Nat. Rev. Immunol. 2011, 11, 275–288. [Google Scholar] [CrossRef]
- Malavia, D.; Crawford, A.; Wilson, D. Nutritional immunity and fungal pathogenesis: The struggle for micronutrients at the host–pathogen interface. Adv. Microb. Physiol. 2017, 70, 85–103. [Google Scholar] [PubMed]
- Beattie, S.R.; Mark, K.M.; Thammahong, A.; Ries, L.N.A.; Dhingra, S.; Caffrey-Carr, A.K.; Cheng, C.; Black, C.C.; Bowyer, P.; Bromley, M.J.; et al. Filamentous fungal carbon catabolite repression supports metabolic plasticity and stress responses es-sential for disease progression. PLoS Pathog. 2017, 13, e1006340. [Google Scholar] [CrossRef] [PubMed]
- Schrettl, M.; Bignell, E.; Kragl, C.; Joechl, C.; Rogers, T.; Arst, H.N., Jr.; Haynes, K.; Haas, H. Siderophore biosynthesis but not reductive iron assimilation is essential for Aspergillus fumigatus virulence. J. Exp. Med. 2004, 200, 1213–1219. [Google Scholar] [CrossRef]
- König, A.; Müller, R.; Mogavero, S.; Hube, B. Fungal factors involved in host immune evasion, modulation and exploitation during infection. Cell. Microbiol. 2021, 23, 13272. [Google Scholar] [CrossRef] [PubMed]
- Moyes, D.L.; Wilson, D.; Richardson, J.P.; Mogavero, S.; Tang, S.X.; Wernecke, J.; Höfs, S.; Gratacap, R.L.; Robbins, J.; Runglall, M.; et al. Candidalysin is a fungal peptide toxin critical for mucosal infection. Nat. Cell Biol. 2016, 532, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Gaston, K.J.; Fuller, R.A. Commonness, population depletion and conservation biology. Trends Ecol. Evol. 2008, 23, 14–19. [Google Scholar] [CrossRef]
- Sala, O.E. Global Biodiversity Scenarios for the Year 2100. Science 2000, 287, 1770–1774. [Google Scholar] [CrossRef] [PubMed]
- Crowley, T.J.; Berner, R.A. CO2 and climate change. Science 2001, 292, 870–872. [Google Scholar] [CrossRef] [PubMed]
- Berdugo, M.; Delgado-Baquerizo, M.; Soliveres, S.; Hernández-Clemente, R.; Zhao, Y.; Gaitán, J.J.; Gross, N.; Saiz, H.; Maire, V.; Lehmann, A.; et al. Global ecosystem thresholds driven by aridity. Science 2020, 367, 787–790. [Google Scholar] [CrossRef]
- Lynch, J.P.; Clair, S.B.S. Mineral stress: The missing link in understanding how global climate change will affect plants in real world soils. Field Crop. Res. 2004, 90, 101–115. [Google Scholar] [CrossRef]
- Corwin, D.L. Climate change impacts on soil salinity in agricultural areas. Eur. J. Soil Sci. 2021, 72, 842–862. [Google Scholar] [CrossRef]
- Cavicchioli, R.; Ripple, W.J.; Timmis, K.N.; Azam, F.; Bakken, L.R.; Baylis, M.; Behrenfeld, M.J.; Boetius, A.; Boyd, P.W.; Classen, A.T.; et al. Scientists’ warning to humanity: Microorganisms and climate change. Nat. Rev. Genet. 2019, 17, 569–586. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, E.A.; Hasselquist, D.; Nilsson, J.-Å.; Westerdahl, H.; Cornwallis, C.K. Wetter climates select for higher immune gene diversity in resident, but not migratory, songbirds. Proc. R. Soc. B Boil. Sci. 2020, 287, 20192675. [Google Scholar] [CrossRef] [PubMed]
- Khawcharoenporn, T.; Apisarnthanarak, A.; Mundy, L.M. Non-neoformans Cryptococcal Infections: A Systematic Review. Infection 2007, 35, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Rajasingham, R.; Smith, R.M.; Park, B.J.; Jarvis, J.N.; Govender, N.P.; Chiller, T.M.; Denning, D.W.; Loyse, A.; Boulware, D.R. Global burden of disease of HIV-associated cryptococcal meningitis: An updated analysis. Lancet Infect. Dis. 2017, 17, 873–881. [Google Scholar] [CrossRef]
- Kneale, M.; Bartholomew, J.S.; Davies, E.; Denning, D.W. Global access to antifungal therapy and its variable cost. J. Antimicrob. Chemother. 2016, 71, 3599–3606. [Google Scholar] [CrossRef] [PubMed]
- Lieber, M.; Chin-Hong, P.; Whittle, H.J.; Hogg, R.; Weiser, S.D. The Synergistic Relationship Between Climate Change and the HIV/AIDS Epidemic: A Conceptual Framework. AIDS Behav. 2021, 1–12. [Google Scholar] [CrossRef]
- Baker, R.E. Climate change drives increase in modeled HIV prevalence. Clim. Chang. 2020, 163, 237–252. [Google Scholar] [CrossRef]
- Kidd, S.E.; Chow, Y.; Mak, S.; Bach, P.J.; Chen, H.; Hingston, A.O.; Kronstad, J.W.; Bartlett, K.H. Characterization of environmental sources of the human and animal pathogen Cryptococcus gattii in British Columbia, Canada, and the Pacific Northwest of the United States. Appl. Environ. Microbiol. 2007, 73, 1433–1443. [Google Scholar] [CrossRef]
- Ellis, D.H.; Pfeiffer, T.J. Natural habitat of Cryptococcus neoformans var. gattii. J. Clin. Microbiol. 1990, 28, 1642–1644. [Google Scholar] [CrossRef]
- Acheson, E.S.; Galanis, E.; Bartlett, K.; Klinkenberg, B. Climate Classification System-Based Determination of Temperate Climate Detection of Cryptococcus gattii sensu lato. Emerg. Infect. Dis. 2019, 25, 1723–1726. [Google Scholar] [CrossRef] [PubMed]
- Engelthaler, D.M.; Casadevall, A. On the emergence of Cryptococcus gattii in the Pacific Northwest: Ballast tanks, tsunamis, and black swans. mBio 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Granados, D.P.; Castañeda, E. Influence of climatic conditions on the isolation of members of the Cryptococcus neoformans species complex from trees in Colombia from 1992–2004. FEMS. Yeast. Res. 2006, 6, 636–644. [Google Scholar] [CrossRef]
- Kidd, S.E.; Bach, P.J.; Hingston, A.O.; Mak, S.; Chow, Y.; MacDougall, L.; Kronstad, J.W.; Bartlett, K.H. Cryptococcus gattii Dispersal Mechanisms, British Columbia, Canada. Emerg. Infect. Dis. 2007, 13, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, D.H.; Sorrell, T.C.; Allworth, A.M.; Heath, C.H.; McGregor, A.R.; Papanaoum, K.; Richards, M.J.; Gottlieb, T. Cryptococcal disease of the CNS in immunocompetent hosts: Influence of cryptococcal variety on clinical manifestations and outcome. Clin. Infect. Dis. 1995, 20, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Mak, S.; Klinkenberg, B.; Bartlett, K.; Fyfe, M. Ecological niche modeling of Cryptococcus gattii in British Columbia, Canada. Environ. Health Perspect. 2010, 118, 653–658. [Google Scholar] [CrossRef]
- D’Souza, C.A.; Kronstad, J.W.; Taylor, G.; Warren, R.; Yuen, M.; Hu, G.; Jung, W.H.; Sham, A.; Kidd, S.E.; Tangen, K.; et al. Genome Variation in Cryptococcus gattii, an Emerging Pathogen of Immunocompetent Hosts. mBio 2011, 2, e00342-10. [Google Scholar] [CrossRef]
- Byrnes, E.J.; Li, W.; Lewit, Y.; Ma, H.; Voelz, K.; Ren, P.; Carter, D.A.; Chaturvedi, V.; Bildfell, R.J.; May, R.C.; et al. Emergence and Pathogenicity of Highly Virulent Cryptococcus gattii Genotypes in the Northwest United States. PLoS Pathog. 2010, 6, e1000850. [Google Scholar] [CrossRef]
- Fraser, J.A.; Giles, S.S.; Wenink, E.C.; Geunes-Boyer, S.G.; Wright, J.R.; Diezmann, S.; Allen, A.; Stajich, J.E.; Dietrich, F.S.; Perfect, J.R.; et al. Same-sex mating and the origin of the Vancouver Island Cryptococcus gattii outbreak. Nat. Cell Biol. 2005, 437, 1360–1364. [Google Scholar] [CrossRef]
- DaCal, M.; Bradford, M.A.; Plaza, C.; Maestre, F.T.; García-Palacios, P. Soil microbial respiration adapts to ambient temperature in global drylands. Nat. Ecol. Evol. 2019, 3, 232–238. [Google Scholar] [CrossRef]
- Bradford, M.A.; McCulley, R.L.; Crowther, T.W.; Oldfield, E.E.; Wood, S.A.; Fierer, N. Cross-biome patterns in soil microbial respiration predictable from evolutionary theory on thermal adaptation. Nat. Ecol. Evol. 2019, 3, 223–231. [Google Scholar] [CrossRef]
- Fernandes, K.E.; Dwyer, C.; Campbell, L.T.; Carter, D.A. Species in the Cryptococcus gattii complex differ in capsule and cell size following growth under capsule-inducing conditions. Msphere 2016, 1, e00350-16. [Google Scholar] [CrossRef]
- Kozel, T.R.; Pfrommer, G.S.T.; Guerlain, A.S.; Highison, B.A.; Highison, G.J. Role of the Capsule in Phagocytosis of Cryptococcus neoformans. Clin. Infect. Dis. 1988, 10, S436–S439. [Google Scholar] [CrossRef]
- Bojarczuk, A.; Miller, K.A.; Hotham, R.; Lewis, A.; Ogryzko, N.V.; Kamuyango, A.A.; Frost, H.; Gibson, R.H.; Stillman, E.; May, R.C.; et al. Cryptococcus neoformans Intracellular Proliferation and Capsule Size Determines Early Macrophage Control of Infection. Sci. Rep. 2016, 6, 21489. [Google Scholar] [CrossRef] [PubMed]
- Robertson, E.J.; Najjuka, G.; Rolfes, M.A.; Akampurira, A.; Jain, N.; Anantharanjit, J.; Hohenberg, M.v.; Tassieri, M.; Carlsson, A.; Meya, D.B.; et al. Cryptococcus neoformans ex vivo capsule size is associated with intracranial pressure and host immune response in HIV-associated cryptococcal meningitis. J. Infect. Dis. 2014, 209, 74–82. [Google Scholar] [CrossRef]
- Pool, A.; Lowder, L.; Wu, Y.; Forrester, K.; Rumbaugh, J. Neurovirulence of Cryptococcus neoformans determined by time course of capsule accumulation and total volume of capsule in the brain. J. NeuroVirol. 2013, 19, 228–238. [Google Scholar] [CrossRef]
- Rosas, Á.L.; Casadevall, A. Melanization affects susceptibility of Cryptococcus neoformans to heat and cold. FEMS Microbiol. Lett. 1997, 153, 265–272. [Google Scholar] [CrossRef]
- Grishkan, I. Ecological Stress: Melanization as a Response in Fungi to Radiation. In Extremophiles Handbook; Horiko-shi, K., Ed.; Springer: Tokyo, Japan, 2011; pp. 1135–1145. [Google Scholar]
- Dadachova, E.; Bryan, R.A.; Huang, X.; Moadel, T.; Schweitzer, A.D.; Aisen, P.; Nosanchuk, J.D.; Casadevall, A. Ionizing Radiation Changes the Electronic Properties of Melanin and Enhances the Growth of Melanized Fungi. PLoS ONE 2007, 2, e457. [Google Scholar] [CrossRef]
- Firacative, C.; Duan, S.; Meyer, W. Galleria mellonella model identifies highly virulent strains among all major molec-ular types of Cryptococcus gattii. PLoS ONE 2014, 9, e105076. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, K.E.; Brockway, A.; Haverkamp, M.; Cuomo, C.A.; van Ogtrop, F.; Perfect, J.R.; Carter, D.A. Phenotypic variability correlates with clinical outcome in Cryptococcus isolates obtained from Botswanan HIV/AIDS patients. mBio 2018, 9, e02016-18. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, H.R.; de Oliveira, G.P.; de Oliveira Frazão, S.; de Melo Gorgonha, K.C.; Rosa, C.P.; Garcez, E.M.; Junior, J.L.; Correia, A.F.; de Freitas, W.F.; Paes, H.C.; et al. Faster Cryptococcus melanization increases virulence in experimental and human cryptococcosis. bioRxiv 2020. [Google Scholar] [CrossRef]
- Singaravelan, N.; Grishkan, I.; Beharav, A.; Wakamatsu, K.; Ito, S.; Nevo, E. Adaptive melanin response of the soil fungus Aspergillus niger to UV radiation stress at “Evolution Canyon”, Mount Carmel, Israel. PLoS ONE 2008, 3, e2993. [Google Scholar] [CrossRef]
- Latge, J.P.; Chamilos, G. Aspergillus fumigatus and Aspergillosis in 2019. Clin. Microbiol. Rev. 2019, 33. [Google Scholar] [CrossRef]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and Multi-National Prevalence of Fungal Diseases—Estimate Precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Reinmuth-Selzle, K.; Kampf, C.J.; Lucas, K.; Lang-Yona, N.; Fröhlich-Nowoisky, J.; Shiraiwa, M.; Lakey, P.S.J.; Lai, S.; Liu, F.; Kunert, A.T.; et al. Air Pollution and Climate Change Effects on Allergies in the Anthropocene: Abundance, Interaction, and Modification of Allergens and Adjuvants. Environ. Sci. Technol. 2017, 51, 4119–4141. [Google Scholar] [CrossRef]
- Lang-Yona, N.; Shuster-Meiseles, T.; Mazar, Y.; Yarden, O.; Rudich, Y. Impact of urban air pollution on the allergenicity of Aspergillus fumigatus conidia: Outdoor exposure study supported by laboratory experiments. Sci. Total. Environ. 2016, 541, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Bertuzzi, M.; Hayes, G.E.; Icheoku, U.J.; Van Rhijn, N.; Denning, D.W.; Osherov, N.; Bignell, E.M. Anti-Aspergillus Activities of the Respiratory Epithelium in Health and Disease. J. Fungi 2018, 4, 8. [Google Scholar] [CrossRef]
- Denning, D.W.; Pleuvry, A.; Cole, D.C. Global burden of chronic pulmonary aspergillosis as a sequel to pulmonary tu-berculosis. Bull. World Health Organ. 2011, 89, 864–872. [Google Scholar] [CrossRef]
- Gelaw, Y.A.; Yu, W.; Magalhães, R.J.S.; Assefa, Y.; Williams, G. Effect of Temperature and Altitude Difference on Tuberculosis Notification: A Systematic Review. J. Glob. Infect. Dis. 2019, 11, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, F.M.D.C.; Martins, E.D.S.; Pedrosa, D.M.A.S.; Evangelista, M.D.S.N. Relationship between climatic factors and air quality with tuberculosis in the Federal District, Brazil, 2003–2012. Braz. Infec. Dis. 2017, 21, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Sleeman, K.E.; de Brito, M.; Etkind, S.; Nkhoma, K.; Guo, P.; Higginson, I.J.; Gomes, B.; Harding, R. The escalating global burden of serious health-related suffering: Projections to 2060 by world regions, age groups, and health conditions. Lancet Glob. Health 2019, 7, e883–e892. [Google Scholar] [CrossRef]
- Ayres, J.G.; Forsberg, B.; Annesi-Maesano, I.; Dey, R.; Ebi, K.L.; Helms, P.J.; Medina-Ramon, M.; Windt, M.; Forastiere, F.; on behalf of the Environment and Health Committee of the European Respiratory Society. Climate change and respiratory disease: European Respiratory Society position statement. Eur. Respir. J. 2009, 34, 295–302. [Google Scholar] [CrossRef]
- Lin, S.-J.; Schranz, J.; Teutsch, S.M. Aspergillosis Case-Fatality Rate: Systematic Review of the Literature. Clin. Infect. Dis. 2001, 32, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Lestrade, P.P.; Buil, J.B.; van der Beek, M.T.; Kuijper, J.; van Dijk, K.; Kampinga, G.A.; Rijnders, B.J.A.; Vonk, A.G.; Greeff, S.C.d.; Schoffelen, A.F.; et al. Paradoxal trends in azole-resistant Aspergillus fumigatus in a national multicenter surveillance program, the Netherlands, 2013–2018. Emerg. Infect. Dis. 2020, 26, 1447. [Google Scholar] [CrossRef]
- Brockhurst, M.A.; Harrison, F.; Veening, J.-W.; Harrison, E.; Blackwell, G.; Iqbal, Z.; MacLean, C. Assessing evolutionary risks of resistance for new antimicrobial therapies. Nat. Ecol. Evol. 2019, 3, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, T.; Van Rhijn, N.; Fraczek, M.; Gsaller, F.; Davies, E.; Carr, P.; Gago, S.; Fortune-Grant, R.; Rahman, S.; Gilsenan, J.M.; et al. The negative cofactor 2 complex is a key regulator of drug resistance in Aspergillus fumigatus. Nat. Commun. 2020, 11, 1–16. [Google Scholar] [CrossRef]
- Gsaller, F.; Hortschansky, P.; Furukawa, T.; Carr, P.D.; Rash, B.; Capilla, J.; Müller, C.; Bracher, F.; Bowyer, P.; Haas, H.; et al. Sterol Biosynthesis and Azole Tolerance Is Governed by the Opposing Actions of SrbA and the CCAAT Binding Complex. PLoS Pathog. 2016, 12, e1005775. [Google Scholar]
- Camps, S.M.T.; Rijs, A.J.M.M.; Klaassen, C.H.W.; Meis, J.F.; O’Gorman, C.M.; Dyer, P.S.; Melchers, W.J.G.; Verweij, P.E. Molecular Epidemiology of Aspergillus fumigatus Isolates Harboring the TR34/L98H Azole Resistance Mechanism. J. Clin. Microbiol. 2012, 50, 2674–2680. [Google Scholar] [CrossRef]
- Caspeta, L.; Chen, Y.; Ghiaci, P.; Feizi, A.; Buskov, S.; Hallström, B.M.; Petranovic, D.; Nielsen, J. Altered sterol composition renders yeast thermotolerant. Science 2014, 346, 75–78. [Google Scholar] [CrossRef]
- Swan, T.M.; Watson, K. Stress tolerance in a yeast sterol auxotroph: Role of ergosterol, heat shock proteins and trehalose. FEMS Microbiol. Lett. 1998, 169, 191–197. [Google Scholar] [CrossRef]
- Rybak, J.M.; Ge, W.; Wiederhold, N.P.; Parker, J.E.; Kelly, S.L.; Rogers, P.D.; Fortwendel, J.R. Mutations in hmg1, Challenging the Paradigm of Clinical Triazole Resistance in Aspergillus fumigatus. mBio 2019, 10, e00437-19. [Google Scholar] [CrossRef] [PubMed]
- Van Rhijn, N. A Genome Scale Census of Virulence Factors in the Major Mould Pathogen of Human Lungs, Aspergillus fumigatus. Ph.D. Thesis, University of Manchester, Manchester, UK, 2019. Available online: https://www.escholar.manchester.ac.uk/jrul/item/?pid=uk-ac-man-scw:322852 (accessed on 1 May 2021).
- Lanz, B.; Dietz, S.; Swanson, T. The expansion of modern agriculture and global biodiversity decline: An integrated as-sessment. Ecolog. Econ. 2018, 144, 260–277. [Google Scholar] [CrossRef]
- Garrett, K.; Nita, M.; De Wolf, E.; Esker, P.; Gomez-Montano, L.; Sparks, A. Plant Pathogens as Indicators of Climate Change; Elsevier BV: Amsterdam, The Netherlands, 2021; pp. 499–513. [Google Scholar]
- Chen, C.-C.; McCarl, B.A. An Investigation of the Relationship between Pesticide Usage and Climate Change. Clim. Chang. 2001, 50, 475–487. [Google Scholar] [CrossRef]
- Rhodes, L.A.; McCarl, B.A. An Analysis of Climate Impacts on Herbicide, Insecticide, and Fungicide Expenditures. Agronomy 2020, 10, 745. [Google Scholar] [CrossRef]
- Snelders, E.; Veld, R.A.G.H.I.; Rijs, A.J.M.M.; Kema, G.H.J.; Melchers, W.J.G.; Verweij, P.E. Possible Environmental Origin of Resistance of Aspergillus fumigatus to Medical Triazoles. Appl. Environ. Microbiol. 2009, 75, 4053–4057. [Google Scholar] [CrossRef] [PubMed]
- Bueid, A.; Howard, S.J.; Moore, C.B.; Richardson, M.D.; Harrison, E.; Bowyer, P.; Denning, D.W. Azole antifungal resistance in Aspergillus fumigatus: 2008 and 2009. J. Antimicrob. Chemother. 2010, 65, 2116–2118. [Google Scholar] [CrossRef]
- Melo, A.M.; Stevens, D.A.; Tell, L.A.; Veríssimo, C.; Sabino, R.; Xavier, M.O. Aspergillosis, Avian Species and the One Health Perspective: The Possible Importance of Birds in Azole Resistance. Microorganisms 2020, 8, 2037. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.C.; Tsai, H.-F.; Karos, M.; Kwon-Chung, K. THTA, a thermotolerance gene of Aspergillus fumigatus. Fungal Genet. Biol. 2004, 41, 888–896. [Google Scholar] [CrossRef]
- Kim, J.H.; Mahoney, N.; Chan, K.L.; Campbell, B.C.; Haff, R.P.; Stanker, L.H. Use of benzo analogs to enhance antimycotic activity of strobilurin for control of aflatoxigenic fungal pathogens. Front. Microbiol. 2014, 5, 87. [Google Scholar] [CrossRef] [PubMed]
- Kwon-Chung, K.J.; Sugui, J.A. Aspergillus fumigatus—What Makes the Species a Ubiquitous Human Fungal Pathogen? PLoS Pathog. 2013, 9, e1003743. [Google Scholar] [CrossRef]
- Paterson, R.R.M.; Lima, N. Thermophilic Fungi to Dominate Aflatoxigenic/Mycotoxigenic Fungi on Food under Global Warming. Int. J. Environ. Res. Public Health 2017, 14, 199. [Google Scholar] [CrossRef]
- Chilaka, C.A.; De Kock, S.; Phoku, J.Z.; Mwanza, M.; Egbuta, M.A.; Dutton, M.F. Fungal and mycotoxin contamination of South African commercial maize. J. Food Agric. Environ. 2012, 10, 296–303. [Google Scholar]
- El-Maghraby, O.; Abdel-Sater, M. Mycoflora and natural occurrence of mycotoxins in tobacco from cigarettes in Egypt. Zentralblatt für Mikrobiol. 1993, 148, 253–264. [Google Scholar] [CrossRef]
- Abdel-Hafez, A.; Saber, S.M. Mycoflora and mycotoxin of hazelnut (Corylus avellana L.) and walnut (Juglans regia L.) seeds in Egypt. Zentralblatt für Mikrobiol. 1993, 148, 137–147. [Google Scholar] [CrossRef]
- Krishnan, S.; Manavathu, E.K.; Chandrasekar, P.H. Aspergillus flavus: An emerging non-fumigatus Aspergillus species of significance. Mycoses 2009, 52, 206–222. [Google Scholar] [CrossRef] [PubMed]
- Skiada, A.; Lass-Floerl, C.; Klimko, N.; Ibrahim, A.; Roilides, E.; Petrikkos, G. Challenges in the diagnosis and treatment of mu-cormycosis. Med. Mycol. 2018, 56 (Suppl. 1), S93–S101. [Google Scholar] [CrossRef]
- Skiada, A.; Pagano, L.; Groll, A.; Zimmerli, S.; Dupont, B.; Lagrou, K.; Lass-Florl, C.; Bouza, E.; Klimko, N.; Gaustad, P.; et al. Zygomycosis in Europe: Analysis of 230 cases accrued by the registry of the European Confederation of Medical Mycology (ECMM) Working Group on Zygomycosis between 2005 and 2007. Clin. Microbiol. Infect. 2011, 17, 1859–1867. [Google Scholar] [CrossRef]
- Prakash, H.; Chakrabarti, A. Global Epidemiology of Mucormycosis. J. Fungi 2019, 5, 26. [Google Scholar] [CrossRef]
- Borgharkar, S.S.; Das, S.S. Real-world evidence of glycemic control among patients with type 2 diabetes mellitus in India: The TIGHT study. BMJ Open Diabetes Res. Care 2019, 7, e000654. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Dall, T.M.; Tan, E.; Byrne, E.; Iacobucci, W.; Chakrabarti, R.; Loh, F.E. Diabetes diagnosis and management among insured adults across metropolitan areas in the U.S. Prev. Med. Rep. 2018, 10, 227–233. [Google Scholar] [CrossRef]
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef]
- Aguiree, F.; Brown, A.; Cho, N.H.; Dahlquist, G.; Dodd, S.; Dunning, T.; Hirst, M.; Hwang, C.; Magliano, D.; Patterson, C.; et al. IDF Diabetes Atlas, 6th ed.; International Diabetes Federation: Basel, Switzerland, 2013. [Google Scholar]
- Alastruey-Izquierdo, A.; Hoffmann, K.; De Hoog, G.S.; Rodriguez-Tudela, J.L.; Voigt, K.; Bibashi, E.; Walther, G. Species Recognition and Clinical Relevance of the Zygomycetous Genus Lichtheimia (syn. Absidia Pro Parte, Mycocladus). J. Clin. Microbiol. 2010, 48, 2154–2170. [Google Scholar] [CrossRef]
- Richardson, M. The ecology of the Zygomycetes and its impact on environmental exposure. Clin. Microbiol. Infect. 2009, 15, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Sivagnanam, S.; Sengupta, D.J.; Hoogestraat, D.; Jain, R.; Stednick, Z.; Fredricks, D.N.; Hendrie, P.; Whimbey, E.; Podczervinski, S.T.; Krantz, E.M.; et al. Seasonal clustering of sinopulmonary mucormycosis in patients with hematologic malignancies at a large comprehensive cancer center. Antimicrob. Resist. Infect. Control. 2017, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Roden, M.M.; Zaoutis, T.E.; Buchanan, W.L.; Knudsen, T.A.; Sarkisova, T.A.; Schaufele, R.L.; Sein, M.; Sein, T.; Chiou, C.C.; Chu, J.H.; et al. Epidemiology and Outcome of Zygomycosis: A Review of 929 Reported Cases. Clin. Infect. Dis. 2005, 41, 634–653. [Google Scholar] [CrossRef]
- Zahoor, B.; Kent, S.; Wall, D. Cutaneous mucormycosis secondary to penetrative trauma. Injury 2016, 47, 1383–1387. [Google Scholar] [CrossRef][Green Version]
- Neblett Fanfair, R.; Benedict, K.; Bos, J.; Bennett, S.D.; Lo, Y.C.; Adebanjo, T.; Etienne, K.; Deak, E.; Derado, G.; Shieh, W.J.; et al. Necrotizing cutaneous mucormycosis after a tornado in Joplin, Missouri, in 2011. N. Engl. Med. 2012, 367, 2214–2225. [Google Scholar] [CrossRef] [PubMed]
- Andresen, D.; Donaldson, A.; Choo, L.; Knox, A.; Klaassen, M.; Ursic, C.; Vonthethoff, L.; Krilis, S.; Konecny, P. Multifocal cutaneous mucormycosis complicating polymicrobial wound infections in a tsunami survivor from Sri Lanka. Lancet 2005, 365, 876–878. [Google Scholar] [CrossRef]
- Brooks, H.E.; Carbin, G.W.; Marsh, P.T. Increased variability of tornado occurrence in the United States. Science. 2014, 346, 349–352. [Google Scholar] [CrossRef]
- Gensini, V.A.; Brooks, H.E. Spatial trends in United States tornado frequency. npj Clim. Atmospheric Sci. 2018, 1, 38. [Google Scholar] [CrossRef]
- McGuire, B. Waking the Giant: How a Changing Climate Triggers Earthquakes, Tsunamis, and Volcanoes; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Pugh, D.; Woodworth, P. Sea-Level Science: Understanding Tides, Surges, Tsunamis and Mean Sea-Level Changes; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of Invasive Candidiasis: A Persistent Public Health Problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Zilberberg, M.D.; Shorr, A.F.; Kollef, M.H. Secular Trends in Candidemia-Related Hospitalization in the United States, 2000–2005. Infect. Control. Hosp. Epidemiol. 2008, 29, 978–980. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, F.; Lockhart, S.R.; Berkow, E.L.; Calandra, T. Changes in the epidemiological landscape of invasive candidiasis. J. Antimicrob. Chemother. 2018, 73, i4–i13. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, S.R.; Iqbal, N.; Cleveland, A.A.; Farley, M.M.; Harrison, L.H.; Bolden, C.B.; Baughman, W.; Stein, B.; Hollick, R.; Park, B.J.; et al. Species identification and antifungal susceptibility testing of Candida bloodstream isolates from population-based surveillance studies in two US cities from 2008 to 2011. J. Clin. Microbiol. 2012, 50, 3435–3442. [Google Scholar] [CrossRef]
- Lyon, G.M.; Karatela, S.; Sunay, S.; Adiri, Y.; for the Candida Surveillance Study Investigators. Antifungal Susceptibility Testing of Candida Isolates from the Candida Surveillance Study. J. Clin. Microbiol. 2010, 48, 1270–1275. [Google Scholar] [CrossRef]
- Price, M.F.; LaRocco, M.T.; Gentry, L.O. Fluconazole susceptibilities of Candida species and distribution of species recovered from blood cultures over a 5-year period. Antimicrob. Agents Chemother. 1994, 38, 1422–1424. [Google Scholar] [CrossRef]
- Satoh, K.; Makimura, K.; Hasumi, Y.; Nishiyama, Y.; Uchida, K.; Yamaguchi, H. Candida auris sp. nov., a novel ascomycetous yeast isolated from the external ear canal of an inpatient in a Japanese hospital. Microbiol. Immunol. 2009, 53, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Desnos-Ollivier, M.; Fekkar, A.; Bretagne, S. Earliest case of Candida auris infection imported in 2007 in Europe from India prior to the 2009 description in Japan. J. Med. Mycol. 2021, 8, 101139. [Google Scholar] [CrossRef]
- Abdolrasouli, A.; Farrer, R.A.; Cuomo, C.A.; Aanensen, D.M.; Armstrong-James, D.; Fisher, M.C.; Schelenz, S. Genomic epidemiology of the UK outbreak of the emerging human fungal pathogen Candida auris. Emerg. Microb. Infect. 2018, 7, 1–12. [Google Scholar]
- Schelenz, S.; Hagen, F.; Rhodes, J.L.; Abdolrasouli, A.; Chowdhary, A.; Hall, A.; Ryan, L.; Shackleton, J.; Trimlett, R.; Meis, J.F.; et al. First hospital outbreak of the globally emerging Candida auris in a European hospital. Antimicrob. Resist. Infect. Control. 2016, 5, 1–7. [Google Scholar] [CrossRef]
- World Health Organization. Global Antimicrobial Resistance Surveillance System (GLASS): Early Implementation Protocol for Inclusion of Candida spp.; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Arensman, K.; Miller, J.L.; Chiang, A.; Mai, N.; Levato, J.; LaChance, E.; Anderson, M.; Beganovic, M.; Pena, J.D. Clinical Outcomes of Patients Treated for Candida auris Infections in a Multisite Health System, Illinois, USA. Emerg. Infect. Dis. 2020, 26, 876. [Google Scholar] [CrossRef]
- Rhodes, J.; Fisher, M.C. Global epidemiology of emerging Candida auris. Curr. Opin. Microbiol. 2019, 52, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Arora, P.; Singh, P.; Wang, Y.; Yadav, A.; Pawar, K.; Singh, A.; Padmavati, G.; Xu, J.; Chowdhary, A. Environmental Isolation of Candida auris from the Coastal Wetlands of Andaman Islands, India. mBio 2021, 12, e03181-20. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, S.R.; Etienne, K.A.; Vallabhaneni, S.; Farooqi, J.; Chowdhary, A.; Govender, N.P.; Colombo, A.L.; Calvo, B.; Cuomo, C.A.; Desjardins, C.A.; et al. Simultaneous emergence of multidrug-resistant Candida auris on 3 continents confirmed by whole-genome sequencing and epidemiological analyses. Clin. Infect. Dis. 2017, 64, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Welsh, R.M.; Sexton, D.J.; Forsberg, K.; Vallabhaneni, S.; Litvintseva, A. Insights into the Unique Nature of the East Asian Clade of the Emerging Pathogenic Yeast Candida auris. J. Clin. Microbiol. 2019, 57. [Google Scholar] [CrossRef]
- Casadevall, A.; Fu, M.S.; Guimaraes, A.J.; Albuquerque, P. The ‘Amoeboid Predator-Fungal Animal Virulence’ Hypothesis. J. Fungi. 2019, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Kontoyiannis, D.P.; Robert, V. On the Emergence of Candida auris: Climate Change, Azoles, Swamps, and Birds. mBio 2019, 10, 01397-19. [Google Scholar] [CrossRef]
- Jackson, B.R.; Chow, N.; Forsberg, K.; Litvintseva, A.P.; Lockhart, S.R.; Welsh, R.; Vallabhaneni, S.; Chiller, T. On the Origins of a Species: What Might Explain the Rise of Candida auris? J. Fungi 2019, 5, 58. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.Z.; Schwartz, I.S. Emerging Fungal Infections: New Patients, New Patterns, and New Pathogens. J. Fungi 2019, 5, 67. [Google Scholar] [CrossRef]
- Schwartz, I.S.; Kenyon, C.; Lehloenya, R.; Claasens, S.; Spengane, Z.; Prozesky, H.; Burton, R.; Parker, A.; Wasserman, S.; Meintjes, G.; et al. AIDS-Related Endemic Mycoses in Western Cape, South Africa, and Clinical Mimics: A Cross-Sectional Study of Adults With Advanced HIV and Recent-Onset, Widespread Skin Lesions. Open Forum Infect. Dis. 2017, 4. [Google Scholar] [CrossRef]
- Barros MB, D.L.; Schubach TM, P.; Gutierrez Galhardo, M.C.; Schubach AD, O.; Monteiro PC, F.; Reis, R.S.; Zancopé-Oliveira, R.M.; Lazéra, M.D.S.; Cuzzi-Maya, T.; Blanco, T.C.M.; et al. Sporotrichosis: An emergent zoonosis in Rio de Janeiro. Memórias do Instituto Oswaldo Cruz 2001, 96, 777–779. [Google Scholar] [CrossRef]
- Sanchotene, K.O.; Madrid, I.M.; Klafke, G.B.; Bergamashi, M.; Terra PP, D.; Rodrigues, A.M.; Camargo, Z.P.D.; Xavier, M.O. Sporothrix brasiliensis outbreaks and the rapid emergence of feline sporotrichosis. Mycoses 2015, 58, 652–658. [Google Scholar] [CrossRef]
- Cruz, I.L.R.; Freitas, D.F.S.; de Macedo, P.M.; Gutierrez-Galhardo, M.C.; Valle, A.C.F.D.; Almeida, M.D.A.; Coelho, R.A.; Brito-Santos, F.; Figueiredo-Carvalho, M.H.G.; Zancopé-Oliveira, R.M.; et al. Evolution of virulence-related phenotypes of Sporothrix brasiliensis isolates from patients with chronic sporotrichosis and acquired immunodeficiency syndrome. Braz. J. Microbiol. 2021, 52, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Hussein, A.S.; El Mofty, H.; Hassanien, M. Climate change and predicted trend of fungal keratitis in Egypt. East. Mediterr. Health J. 2011, 17, 468–473. [Google Scholar] [CrossRef]
- Gower, E.W.; Keay, L.J.; Oechsler, R.A.; Iovieno, A.; Alfonso, E.C.; Jones, D.B.; Colby, K.; Tuli, S.S.; Patel, S.R.; Lee, S.M.; et al. Trends in Fungal Keratitis in the United States, 2001 to 2007. Ophthalmology 2010, 117, 2263–2267. [Google Scholar] [CrossRef]
- Lalitha, P.; Prajna, N.V.; Manoharan, G.; Srinivasan, M.; Mascarenhas, J.; Das, M.; D’Silva, S.S.; Porco, T.C.; Keenan, J.D. Trends in bacterial and fungal keratitis in South India, 2002–2012. Br. J. Ophthalmol. 2015, 99, 192–194. [Google Scholar] [CrossRef] [PubMed]
- Burge, H.A. An update on pollen and fungal spore aerobiology. J. Allergy Clin. Immunol. 2002, 110, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.W.; Fernandez, M.M.; Rebong, R.A.; Carlson, A.N.; Kim, T.; Afshari, N.A. Microbiological profiles of fungal keratitis: A 10-year study at a tertiary referral center. J. Ophthalmic Inflamm. Infect. 2016, 6, 5. [Google Scholar] [CrossRef]
- Tuft, S.J.; Tullo, A.B. Fungal keratitis in the United Kingdom 2003–2005. Eye 2008, 23, 1308–1313. [Google Scholar] [CrossRef]
- Abdolrasouli, A.; Bercusson, A.C.; Rhodes, J.L.; Hagen, F.; Buil, J.B.; Tang, A.Y.Y.; de Boer, L.L.; Shah, A.; Milburn, A.J.; Elborn, J.S.; et al. Airway persistence by the emerging multi-azole-resistant Rasamsonia argillacea complex in cystic fibrosis. Mycoses 2018, 61, 665–673. [Google Scholar] [CrossRef]
- Abdolrasouli, A.; Gibani, M.M.; de Groot, T.; Borman, A.M.; Hoffman, P.; Azadian, B.S.; Mughal, N.; Moore, L.S.P.; Johnson, E.M.; Meis, J.F. A pseudo-outbreak of Rhinocladiella similis in a bronchoscopy unit of a tertiary care teaching hospital in London, United Kingdom. Mycoses 2020, 64, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, N.; Kubat, R.C.; Poplin, V.; Adenis, A.A.; Denning, D.W.; Wright, L.; McCotter, O.; Schwartz, I.S.; Jackson, B.R.; Chiller, T.; et al. Re-drawing the Maps for Endemic Mycoses. Mycopathologia 2020, 185, 843–865. [Google Scholar] [PubMed]
- Brown, E.M.; McTaggart, L.R.; Dunn, D.; Pszczolko, E.; Tsui, K.G.; Morris, S.K.; Stephens, D.; Kus, J.V.; Richardson, S.E. Epidemiology and Geographic Distribution of Blastomycosis, Histoplasmosis, and Coccidioidomycosis, Ontario, Canada, 1990–2015. Emerg. Infect. Dis. 2018, 24, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Benedict, K.; Thompson, G.R.; Deresinski, S.; Chiller, T. Mycotic Infections Acquired outside Areas of Known Endemicity, United States. Emerg. Infect. Dis. 2015, 21, 1935–1941. [Google Scholar] [CrossRef]
- Silva, F.B.; Santos, J.R.N.; Da Silva, L.C.; Gomes, W.C.; Villis, P.C.M.; Gomes, E.D.S.; Pinheiro, E.D.A.D.; Azevedo, C.D.M.P.E.S.D.; Dias, R.D.S.; de Andrade Monteiro, C.; et al. Climate drivers of hospitalizations for mycoses in Brazil. Sci. Rep. 2019, 9, 6902. [Google Scholar] [CrossRef]
- Comrie, A.C. Climate Factors Influencing Coccidioidomycosis Seasonality and Outbreaks. Environ. Health Perspect. 2005, 113, 688–692. [Google Scholar] [CrossRef]
- Comrie, A.C.; Glueck, M.F. Assessment of climate-coccidioidomycosis model: Model sensitivity for assessing climatologic effects on the risk of acquiring coccidioidomycosis. Ann. N. Y. Acad. Sci. 2007, 1111, 83–95. [Google Scholar] [CrossRef]
- Baddley, J.W. Geographic Distribution of Endemic Fungal Infections among Older Persons, United States. Emerg. Infect. Dis. 2011, 17, 1664–1669. [Google Scholar] [CrossRef]
- Wilson, L.; Ting, J.; Lin, H.; Shah, R.; MacLean, M.; Peterson, M.W.; Stockamp, N.; Libke, R.; Brown, P. The Rise of Valley Fever: Prevalence and Cost Burden of Coccidioidomycosis Infection in California. Int. J. Environ. Res. Public Health 2019, 16, 1113. [Google Scholar] [CrossRef]
- Bezold, C.P.; Khan, M.A.; Adame, G.; Brady, S.; Sunenshine, R.; Komatsu, K. Notes from the Field: Increase in Coccidioidomycosis—Arizona, October 2017–March 2018. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 1246–1247. [Google Scholar] [CrossRef]
- Cooksey, G.S.; Nguyen, A.; Knutson, K.; Tabnak, F.; Benedict, K.; McCotter, O.; Jain, S.; Vugia, D. Notes from the Field: Increase in Coccidioidomycosis—California, 2016. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 833–834. [Google Scholar] [CrossRef] [PubMed]
- Sondermeyer, G.L.; Lee, L.A.; Gilliss, D.; Vugia, D.J. Coccidioidomycosis-Associated Deaths in California, 2000–2013. Public Health Rep. 2016, 131, 531–535. [Google Scholar] [CrossRef]
- Huang, J.Y.; Bristow, B.; Shafir, S.; Sorvillo, F. Coccidioidomycosis-associated Deaths, United States, 1990–2008. Emerg. Infect. Dis. 2012, 18, 1723–1728. [Google Scholar] [CrossRef]
- Park, B.J.; Sigel, K.; Vaz, V.; Komatsu, K.; McRill, C.; Phelan, M.; Colman, T.; Comrie, A.C.; Warnock, D.W.; Galgiani, J.N.; et al. An Epidemic of Coccidioidomycosis in Arizona Associated with Climatic Changes, 1998–2001. J. Infect. Dis. 2005, 191, 1981–1987. [Google Scholar] [CrossRef] [PubMed]
- Tamerius, J.D.; Comrie, A.C. Coccidioidomycosis Incidence in Arizona Predicted by Seasonal Precipitation. PLoS ONE 2011, 6, e21009. [Google Scholar] [CrossRef]
- Weaver, E.A.; Kolivras, K.N. Investigating the Relationship Between Climate and Valley Fever (Coccidioidomycosis). EcoHealth 2018, 15, 840–852. [Google Scholar] [CrossRef]
- McCotter, O.Z.; Benedict, K.; Engelthaler, D.M.; Komatsu, K.; Lucas, K.D.; Mohle-Boetani, J.C.; Oltean, H.; Vugia, D.; Chiller, T.M.; Cooksey, G.L.S.; et al. Update on the Epidemiology of coccidioidomycosis in the United States. Med. Mycol. 2019, 57, S30–S40. [Google Scholar] [CrossRef] [PubMed]
- Engelthaler, D.M.; Roe, C.C.; Hepp, C.M.; Teixeira, M.; Driebe, E.M.; Schupp, J.M.; Gade, L.; Waddell, V.; Komatsu, K.; Arathoon, E.; et al. Local population structure and patterns of Western Hemisphere dispersal for Coccidioides spp., the fungal cause of Valley Fever. mBio 2016, 7, e00550-16. [Google Scholar] [CrossRef]
- Turabelidze, G.; Aggu-Sher, R.K.; Jahanpour, E.; Hinkle, C.J. Coccidioidomycosis in a State Where It Is Not Known to Be Endemic—Missouri, 2004–2013. MMWR. Morb. Mortal. Wkly. Rep. 2015, 64, 636–639. [Google Scholar] [PubMed]
- Litvintseva, A.P.; Marsden-Haug, N.; Hurst, S.; Hill, H.; Gade, L.; Driebe, E.M.; Ralston, C.; Roe, C.; Barker, B.M.; Goldoft, M.; et al. Valley Fever: Finding New Places for an Old Disease: Coccidioides immitis Found in Washington State Soil Associated with Recent Human Infection. Clin. Infect. Dis. 2014, 60, e1–e3. [Google Scholar] [CrossRef]
- Gorris, M.E.; Cat, L.A.; Zender, C.S.; Treseder, K.K.; Randerson, J.T. Coccidioidomycosis Dynamics in Relation to Climate in the Southwestern United States. GeoHealth 2018, 2, 6–24. [Google Scholar] [CrossRef]
- Gorris, M.E.; Treseder, K.K.; Zender, C.S.; Randerson, J.T. Expansion of Coccidioidomycosis Endemic Regions in the United States in Response to Climate Change. GeoHealth 2019, 3, 308–327. [Google Scholar] [CrossRef]
- Tong, D.Q.; Wang, J.X.L.; Gill, T.E.; Lei, H.; Wang, B. Intensified dust storm activity and Valley fever infection in the southwestern United States. Geophys. Res. Lett. 2017, 44, 4304–4312. [Google Scholar] [CrossRef]
- Griffin, D.W. Atmospheric Movement of Microorganisms in Clouds of Desert Dust and Implications for Human Health. Clin. Microbiol. Rev. 2007, 20, 459–477. [Google Scholar] [CrossRef] [PubMed]
- Cayan, D.; Dettinger, M.; Pierce, D.; Das, T.; Knowles, N.; Ralph, F.; Sumargo, E. Natural Variability, Anthropogenic Climate Change, and Impacts on Water Availability and Flood Extremes in the Western United States. Pestic. Agric. Environ. 2016, 17–42. [Google Scholar] [CrossRef]
- World Health Organization. Ending the Neglect to Attain the Sustainable Development Goals: A Road Map for Neglected Trop-Ical Diseases; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Prado, M.; Silva MB, D.; Laurenti, R.; Travassos, L.R.; Taborda, C.P. Mortality due to systemic mycoses as a primary cause of death or in association with AIDS in Brazil: A re-view from 1996 to 2006. Memórias do Instituto Oswaldo Cruz 2009, 104, 513–521. [Google Scholar] [CrossRef]
- Teixeira MD, M.; Theodoro, R.C.; Oliveira FF, M.D.; Machado, G.C.; Hahn, R.C.; Bagagli, E.; San-Blas, G.; Soares Felipe, M.S. Paracoccidioides lutzii sp. nov.: Biological and clinical implications. Med. Mycol. 2014, 52, 19–28. [Google Scholar]
- Restrepo, A.; McEwen, J.G.; Castaneda, E. The habitat of Paracoccidioides brasiliensis: How far from solving the riddle? Med. Mycol. 2001, 39, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R. New Trends in Paracoccidioidomycosis Epidemiology. J. Fungi 2017, 3, 1. [Google Scholar] [CrossRef] [PubMed]
- Vieira, G.D.D.; Alves, T.D.C.; Lima, S.M.D.D.; Camargo, L.M.A.; Sousa, C.M.D. Paracoccidioidomycosis in a western Brazilian Amazon State: Clinical-epidemiologic profile and spatial distribution of the disease. Rev. Soc. Bras. Med. Trop. 2014, 47, 63–68. [Google Scholar] [CrossRef]
- Coutinho, Z.F.; Wanke, B.; Travassos, C.; Oliveira, R.M.; Xavier, D.R.; Coimbra, C.E.A., Jr. Hospital morbidity due to paracoccidioidomycosis in Brazil (1998–2006). Trop. Med. Int. Health 2015, 20, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Cermeño, J.; Cermeño, J.; Godoy, G.; Hernández, I.; Orellán, Y.; Blanco, Y.; Penna, S.; García, L.; Mender, T.; Gonsálvez, M.; et al. Epidemiological study of paracoccidioidomycosis and histoplasmosis in a suburb of San Felix city, Bolivar state, Venezuela. Investig. Clin. 2009, 50, 213–220. [Google Scholar] [PubMed]
- Barrozo, L.V.; Mendes, R.P.; Marques, S.A.; Benard, G.; Silva, M.E.S.; Bagagli, E. Climate and acute/subacute paracoccidioidomycosis in a hyper-endemic area in Brazil. Int. J. Epidemiol. 2009, 38, 1642–1649. [Google Scholar] [CrossRef]
- Conti-Diaz, I.; MacKinnon, J.; Furcolow, M. Effect of drying on Paracoccidioides brasiliensis. Med. Mycol. 1971, 9, 69–78. [Google Scholar] [CrossRef]
- Simões, L.B.; Marques, S.A.; Bagagli, E. Distribution of paracoccidioidomycosis: Determination of ecologic correlates through spatial analyses. Med. Mycol. 2004, 42, 517–523. [Google Scholar] [CrossRef]
- Barrozo, L.V.; Benard, G.; Silva, M.E.S.; Bagagli, E.; Marques, S.A.; Mendes, R.P. First Description of a Cluster of Acute/Subacute Paracoccidioidomycosis Cases and Its Association with a Climatic Anomaly. PLoS Negl. Trop. Dis. 2010, 4, e643. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Luo, X.; Yang, Y.-M.; Sun, W.; Cane, M.A.; Cai, W.; Yeh, S.-W.; Liu, J. Historical change of El Niño properties sheds light on future changes of extreme El Niño. Proc. Natl. Acad. Sci. USA 2019, 116, 22512–22517. [Google Scholar] [CrossRef]
- DiNezio, P.N.; Puy, M.; Thirumalai, K.; Jin, F.-F.; Tierney, J.E. Emergence of an equatorial mode of climate variability in the Indian Ocean. Sci. Adv. 2020, 6, eaay7684. [Google Scholar] [CrossRef]
- Restrepo, A.; Baumgardner, D.J.; Bagagli, E.; Cooper, C.R.; McGinnis, M.R.; Lazera, M.S.; Barbosa, F.H.; Bosco, S.M.G.; Camargo, Z.P.; Coelho, K.I.R.; et al. Clues to the presence of pathogenic fungi in certain environments. Med. Mycol. 2000, 38, 67–77. [Google Scholar] [CrossRef][Green Version]
- Baumgardner, D.J.; Paretsky, D.P.; Baeseman, Z.J.; Schreiber, A. Effects of season and weather on blastomycosis in dogs: Northern Wisconsin, USA. Med. Mycol. 2011, 49, 49–55. [Google Scholar] [CrossRef]
- Klein, B.S.; Vergeront, J.M.; Disalvo, A.F.; Kaufman, L.; Davis, J.P. Two outbreaks of blastomycosis along rivers in Wisconsin. Isolation of Blastomyces dermatitidis from riverbank soil and evidence of its transmission along waterways. Am. Rev. Respir. Dis. 1987, 136, 1333–1338. [Google Scholar] [CrossRef] [PubMed]
- Proctor, M.E.; Klein, B.S.; Jones, J.M.; Davis, J.P. Cluster of pulmonary blastomycosis in a rural community: Evidence for multiple high-risk environ-mental foci following a sustained period of diminished precipitation. Mycopathologia 2002, 153, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Pfister, J.R.; Archer, J.R.; Hersil, S.; Boers, T.; Reed, K.D.; Meece, J.K.; Anderson, J.L.; Burgess, J.W.; Sullivan, T.D.; Klein, B.S.; et al. Non-Rural Point Source Blastomycosis Outbreak Near a Yard Waste Collection Site. Clin. Med. Res. 2010, 9, 57–65. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baumgardner, D.J.; Egan, G.; Giles, S.; Laundre, B. An outbreak of blastomycosis on a United States Indian reservation. Wilderness Environ. Med. 2002, 13, 250–252. [Google Scholar] [CrossRef]
- Diffenbaugh, N.S.; Swain, D.L.; Touma, D. Anthropogenic warming has increased drought risk in California. Proc. Natl. Acad. Sci. USA 2015, 112, 3931–3936. [Google Scholar] [CrossRef]
- Williams, A.P.; Cook, E.R.; Smerdon, J.E.; Cook, B.I.; Abatzoglou, J.T.; Bolles, K.; Baek, S.H.; Badger, A.M.; Livneh, B. Large contribution from anthropogenic warming to an emerging North American megadrought. Science 2020, 368, 314–318. [Google Scholar] [CrossRef]
- Marvel, K.; Cook, B.I.; Bonfils, C.J.W.; Durack, P.J.; Smerdon, J.E.; Williams, A.P. Twentieth-century hydroclimate changes consistent with human influence. Nat. Cell Biol. 2019, 569, 59–65. [Google Scholar] [CrossRef]
- USGCRP. Climate Science Special Report: Fourth National Climate Assessment; Wuebbles, D.J., Fahey, D.W., Hibbard, K.A., Arnold, J.R., DeAngelo, B., Doherty, S., Easterling, D.R., Edmonds, J., Edmonds, T., Hall, T., et al., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2017; Volume I, p. 470.
- Wheat, L.J.; Azar, M.M.; Bahr, N.C.; Spec, A.; Relich, R.F.; Hage, H. Histoplasmosis. Infect. Dis. Clin. N. Am. 2016, 30, 207–227. [Google Scholar] [CrossRef]
- Manos, N.E.; Ferebee, S.H.; Kerschbaum, W.F. Geographic Variation in the Prevalence of Histoplasmin Sensitivity. Dis. Chest 1956, 29, 649–668. [Google Scholar] [CrossRef]
- Benedict, K.; Mody, R.K. Epidemiology of Histoplasmosis Outbreaks, United States, 1938–2013. Emerg. Infect. Dis. 2016, 22, 370–378. [Google Scholar] [CrossRef]
- Benedict, K.; Derado, G.; Mody, R.K. Histoplasmosis-Associated Hospitalizations in the United States, 2001–2012. Open Forum Infect. Dis. 2016, 3, ofv219. [Google Scholar] [CrossRef]
- Nett, R.J.; Skillman, D.; Riek, L.; Davis, B.; Blue, S.R.; Sundberg, E.E.; Merriman, J.R.; Hahn, C.G.; Park, B.J. Histoplasmosis in Idaho and Montana, USA, 2012–2013. Emerg. Infect. Dis. 2015, 21, 1071–1072. [Google Scholar] [CrossRef] [PubMed]
- Fungus That Causes Histoplasmosis May Be Endemic in Montana. JAMA 2013, 310, 2608. [CrossRef]
- Armstrong, P.A.; Jackson, B.R.; Haselow, D.; Fields, V.; Ireland, M.; Austin, C.; Signs, K.; Al, P.A.A.E.; Patel, R.; Ellis, P.; et al. Multistate Epidemiology of Histoplasmosis, United States, 2011–20141. Emerg. Infect. Dis. 2017, 24, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Maiga, A.W.; Deppen, S.; Scaffidi, B.K.; Baddley, J.; Aldrich, M.C.; Dittus, R.S.; Grogan, E.L. Mapping Histoplasma capsulatum Exposure, United States. Emerg. Infect. Dis. 2018, 24, 1835–1839. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.; Beale, M.; Hagen, F.; Fisher, M.; Terra, P.; de Hoog, S.; Brilhante, R.; Cordeiro, R.D.A.; Rocha, M.F.G.; Sidrim, J.J.C.; et al. The global epidemiology of emerging Histoplasma species in recent years. Stud. Mycol. 2020, 97, 100095. [Google Scholar] [CrossRef]
- Jones, G.; Jacobs, D.S.; Kunz, T.H.; Willig, M.R.; Racey, P.A. Carpe noctem: The importance of bats as bioindicators. Endanger. Species Res. 2009, 8, 93–115. [Google Scholar] [CrossRef]
- Crick, H.Q.P. The impact of climate change on birds. Ibis 2004, 146, 48–56. [Google Scholar] [CrossRef]
- Williamson, C.E.; Zepp, R.G.; Lucas, R.M.; Madronich, S.; Austin, A.T.; Ballaré, C.L.; Norval, M.; Sulzberger, B.; Bais, A.F.; McKenzie, R.L.; et al. Solar ultraviolet radiation in a changing climate. Nat. Clim. Chang. 2014, 4, 434–441. [Google Scholar] [CrossRef]
- Campbell, C.C.; Berliner, M.D. Virulence Differences in Mice of Type A and B Histoplasma capsulatum Yeasts Grown in Continuous Light and Total Darkness. Infect. Immun. 1973, 8, 677–678. [Google Scholar] [CrossRef]
- Medoff, G.; Maresca, B.; Lambowitz, A.M.; Kobayashi, G.; Painter, A.; Sacco, M.; Carratu, L. Correlation between pathogenicity and temperature sensitivity in different strains of Histoplasma capsulatum. J. Clin. Investig. 1986, 78, 1638–1647. [Google Scholar] [CrossRef] [PubMed]
- Schauwvlieghe, A.F.A.D.; Rijnders, B.J.A.; Philips, N.; Verwijs, R.; Vanderbeke, L.; Van Tienen, C.; Lagrou, K.; Verweij, P.E.; Van De Veerdonk, F.L.; Gommers, D.; et al. Invasive aspergillosis in patients admitted to the intensive care unit with severe influenza: A retrospective cohort study. Lancet Respir. Med. 2018, 6, 782–792. [Google Scholar] [CrossRef]
- Van Arkel, A.L.; Rijpstra, T.A.; Belderbos, H.N.A.; van Wijngaarden, P.; Verweij, P.E.; Bentvelsen, R.G. COVID-19–associated pulmonary aspergillosis. Am. J. Respir. Crit. Care Med. 2020, 202, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Armstrong-James, D.; Youngs, J.; Bicanic, T.; Abdolrasouli, A.; Denning, D.W.; Johnson, E.; Mehra, V.; Pagliuca, T.; Patel, B.; Rhodes, J.; et al. Confronting and mitigating the risk of COVID-19 associated pulmonary aspergillosis. Eur. Respir. J. 2020, 56, 2002554. [Google Scholar] [CrossRef] [PubMed]
- United Nations. World Population Prospects: The 2004 Revision. CD-ROM Edition Extended Dataset; United National Publication: New York, NY, USA, 2005. [Google Scholar]
- Alirol, E.; Getaz, L.; Stoll, B.; Chappuis, F.; Loutan, L. Urbanisation and infectious diseases in a globalised world. Lancet Infect. Dis. 2011, 11, 131–141. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Rhijn, N.; Bromley, M. The Consequences of Our Changing Environment on Life Threatening and Debilitating Fungal Diseases in Humans. J. Fungi 2021, 7, 367. https://doi.org/10.3390/jof7050367
van Rhijn N, Bromley M. The Consequences of Our Changing Environment on Life Threatening and Debilitating Fungal Diseases in Humans. Journal of Fungi. 2021; 7(5):367. https://doi.org/10.3390/jof7050367
Chicago/Turabian Stylevan Rhijn, Norman, and Michael Bromley. 2021. "The Consequences of Our Changing Environment on Life Threatening and Debilitating Fungal Diseases in Humans" Journal of Fungi 7, no. 5: 367. https://doi.org/10.3390/jof7050367
APA Stylevan Rhijn, N., & Bromley, M. (2021). The Consequences of Our Changing Environment on Life Threatening and Debilitating Fungal Diseases in Humans. Journal of Fungi, 7(5), 367. https://doi.org/10.3390/jof7050367






