Breakthrough Mucormycosis Developing on Mucorales-Active Antifungals Portrays a Poor Prognosis in Patients with Hematologic Cancer
Abstract
1. Introduction
2. Patients and Methods
2.1. Study Design
2.2. Characteristics Assessed and Definitions
2.3. Outcomes and Statistical Analysis
3. Results
3.1. Patients and Clinical Characteristics
3.2. Outcomes
4. Discussion
5. Conclusions and Outlook
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Farmakiotis, D.; Kontoyiannis, D.P. Mucormycoses. Infect. Dis. Clin. N. Am. 2016, 30, 143–163. [Google Scholar] [CrossRef]
- Kontoyiannis, D.P.; Lionakis, M.S.; Lewis, R.E.; Chamilos, G.; Healy, M.; Perego, C.; Safdar, A.; Kantarjian, H.; Champlin, R.; Walsh, T.J.; et al. Zygomycosis in a Tertiary-Care Cancer Center in the Era ofAspergillus-Active Antifungal Therapy: A Case-Control Observational Study of 27 Recent Cases. J. Infect. Dis. 2005, 191, 1350–1360. [Google Scholar] [CrossRef]
- Halpern, A.B.; Lyman, G.H.; Walsh, T.J.; Kontoyiannis, D.P.; Walter, R.B. Primary antifungal prophylaxis during curative-intent therapy for acute myeloid leukemia. Blood 2015, 126, 2790–2797. [Google Scholar] [CrossRef] [PubMed]
- Sipsas, N.V.; Kontoyiannis, D.P. Clinical Issues Regarding Relapsing Aspergillosis and the Efficacy of Secondary Antifungal Prophylaxis in Patients with Hematological Malignancies. Clin. Infect. Dis. 2006, 42, 1584–1591. [Google Scholar] [CrossRef] [PubMed]
- Chou, L.S.; Lewis, R.E.; Ippoliti, C.; Champlin, R.E.; Kontoyiannis, D.P. Caspofungin as Primary Antifungal Prophylaxis in Stem Cell Transplant Recipients. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2007, 27, 1644–1650. [Google Scholar] [CrossRef] [PubMed]
- Auberger, J.; Lass-Flörl, C.; Aigner, M.; Clausen, J.; Gastl, G.; Nachbaur, D. Invasive fungal breakthrough infections, fungal colonization and emergence of resistant strains in high-risk patients receiving antifungal prophylaxis with posaconazole: Real-life data from a single-centre institutional retrospective observational study. J. Antimicrob. Chemother. 2012, 67, 2268–2273. [Google Scholar] [CrossRef]
- Rausch, C.R.; DiPippo, A.J.; Bose, P.; Kontoyiannis, D.P. Breakthrough Fungal Infections in Patients with Leukemia Receiving Isavuconazole. Clin. Infect. Dis. 2018, 67, 1610–1613. [Google Scholar] [CrossRef]
- Cahuayme-Zuniga, L.; Lewis, R.E.; Mulanovich, V.E.; Kontoyiannis, D.P. Weekly liposomal amphotericin B as secondary prophylaxis for invasive fungal infections in patients with hematological malignancies. Med. Mycol. 2012, 50, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, J.P.; Chen, S.C.; Kauffman, C.A.; Steinbach, W.J.; Baddley, J.W.; Verweij, P.E.; Clancy, C.J.; Wingard, J.R.; Lockhart, S.R.; Groll, A.H.; et al. Revision and Update of the Consensus Definitions of Invasive Fungal Disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin. Infect. Dis. 2020, 71, 1367–1376. [Google Scholar] [CrossRef]
- Cornely, O.A.; Hoenigl, M.; Lass-Flörl, C.; Chen, S.C.-A.; Kontoyiannis, D.P.; Morrissey, C.O.; Thompson, G.R., III. Defining breakthrough invasive fungal infection—Position paper of the mycoses study group education and research consortium and the European Confederation of Medical Mycology. Mycoses 2019, 62, 716–729. [Google Scholar] [CrossRef]
- Chamilos, G.; Lewis, R.E.; Kontoyiannis, D.P. Delaying Amphotericin B–Based Frontline Therapy Significantly Increases Mortality among Patients with Hematologic Malignancy Who Have Zygomycosis. Clin. Infect. Dis. 2008, 47, 503–509. [Google Scholar] [CrossRef]
- Kyvernitakis, A.; Torres, H.; Jiang, Y.; Chamilos, G.; Lewis, R.; Kontoyiannis, D. Initial use of combination treatment does not impact survival of 106 patients with haematologic malignancies and mucormycosis: A propensity score analysis. Clin. Microbiol. Infect. 2016, 22, 811.e1–811.e8. [Google Scholar] [CrossRef] [PubMed]
- Salmanton-García, J.; Koehler, P.; Kindo, A.; Falces-Romero, I.; García-Rodríguez, J.; Ráčil, Z.; Chen, S.C.-A.; Klimko, N.; Desoubeaux, G.; Thompson, I.G.R.; et al. Needles in a haystack: Extremely rare invasive fungal infections reported in FungiScope®—Global Registry for Emerging Fungal Infections. J. Infect. 2020, 81, 802–815. [Google Scholar] [CrossRef] [PubMed]
- Wurster, S.; Lewis, R.E.; Albert, N.D.; Kontoyiannis, D.P. Preexposure to Isavuconazole Increases the Virulence of Mucorales but Not As-pergillus fumigatus in a Drosophila melanogaster Infection Model. Antimicrob. Agents Chemother. 2019, 63, e01896-18. [Google Scholar] [PubMed]
- Singh, N.; Aguado, J.M.; Bonatti, H.; Forrest, G.; Gupta, K.L.; Safdar, N.; John, G.T.; Pursell, K.J.; Muñoz, P.; Patel, R.; et al. Zygomycosis in Solid Organ Transplant Recipients: A Prospective, Matched Case-Control Study to Assess Risks for Disease and Outcome. J. Infect. Dis. 2009, 200, 1002–1011. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.E.; Cahyame-Zuniga, L.; Leventakos, K.; Chamilos, G.; Ben-Ami, R.; Tamboli, P.; Tarrand, J.; Bodey, G.P.; Luna, M.; Kontoyiannis, D.P. Epidemiology and sites of involvement of invasive fungal infections in patients with haematological malignancies: A 20-year autopsy study. Mycoses 2013, 56, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, F.; Kontoyiannis, D.P. Therapeutic Challenges of Non-Aspergillus Invasive Mold Infections in Immunosuppressed Patients. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef]
- Kontoyiannis, D.P.; Selleslag, D.; Mullane, K.; A Cornely, O.; Hope, W.; Lortholary, O.; Croos-Dabrera, R.; Lademacher, C.; Engelhardt, M.; Patterson, T.F. Impact of unresolved neutropenia in patients with neutropenia and invasive aspergillosis: A post hoc analysis of the SECURE trial. J. Antimicrob. Chemother. 2018, 73, 757–763. [Google Scholar] [CrossRef]
- Campo, M.; Lewis, R.E.; Kontoyiannis, D.P. Invasive fusariosis in patients with hematologic malignancies at a cancer center: 1998–2009. J. Infect. 2010, 60, 331–337. [Google Scholar] [CrossRef]
- McCarthy, M.W.; Kontoyiannis, D.P.; A Cornely, O.; Perfect, J.R.; Walsh, T.J. Novel Agents and Drug Targets to Meet the Challenges of Resistant Fungi. J. Infect. Dis. 2017, 216, S474–S483. [Google Scholar] [CrossRef]
- Dadwal, S.S.; Kontoyiannis, D.P. Recent advances in the molecular diagnosis of mucormycosis. Expert Rev. Mol. Diagn. 2018, 18, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Kontoyiannis, D.P.; Lewis, R.E.; Lortholary, O.; Spellberg, B.; Petrikkos, G.; Roillides, E.; Ibrahim, A.; Walsh, T.J. Future Directions in Mucormycosis Research. Clin. Infect. Dis. 2012, 54, S79–S85. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | All Patients (n = 103) | Mucorales-Active Antifungals (n = 16) | Other Mold-Active Antifungals (n = 87) | p-Value | |
|---|---|---|---|---|---|
| Age (years), median (range) | 52 (18–76) | 57 (25–75) | 52 (18–76) | 0.26 | |
| Gender, male | 67 (65) | 11 (69) | 56 (64) | 0.74 | |
| Race | 0.45 | ||||
| White | 86 (84) | 13 (81) | 73 (84) | ||
| Black | 11 (11) | 3 (19) | 8 (9) | ||
| Asian | 6 (6) | 0 (0) | 6 (7) | ||
| Ethnicity | 0.12 | ||||
| Hispanic | 14 (14) | 0 (0) | 14 (16) | ||
| Non-Hispanic | 89 (86) | 16 (100) | 73 (84) | ||
| Underlying malignancy | >0.99 | ||||
| Leukemia/MDS | 92 (89) | 15 (94) | 77 (89) | ||
| Lymphoma/myeloma | 11 (11) | 1 (6) | 10 (11) | ||
| Status of primary disease | 0.51 | ||||
| Active | 80 (78) | 14 (88) | 66 (76) | ||
| In remission | 23 (22) | 2 (12) | 21 (24) | ||
| Allogeneic HSCT recipients | 50 (49) | 5 (31) | 45 (52) | 0.13 | |
| GvHD | 40/50 (80) | 5/5 (100) | 35/45 (78) | 0.57 | |
| Active | 23/40 (58) | 3/5 (60) | 20/35 (57) | ||
| Chronic | 17/40 (43) | 2/5 (40) | 15/35 (43) | ||
| Neutropenia at diagnosis | 65 (63) | 12 (75) | 53 (61) | 0.28 | |
| Neutrophil recovery after neutropenia | 41/65 (63) | 6/12 (50) | 35/53 (66) | 0.33 | |
| Duration of neutropenia at diagnosis (days), median (IQR) | 21 (12–52) | 29 (20–49) | 18 (11–54) | 0.26 | |
| Corticosteroid use | 36 (35) | 5 (31) | 31 (36) | 0.74 | |
| History of diabetes mellitus | 42 (41) | 2 (13) | 40 (46) | 0.012 | |
| Type of infection | 0.58 | ||||
| Localized infection | 19 (18) | 2 (13) | 17 (20) | ||
| Sinopulmonary infection | 65 (63) | 12 (75) | 53 (61) | ||
| Disseminated infection | 19 (18) | 2 (13) | 17 (20) | ||
| Genus isolated | 0.77 | ||||
| Rhizopus spp. | 61 (59) | 10 (63) | 51 (59) | ||
| Mucor spp. | 18 (17) | 3 (19) | 15 (17) | ||
| Rhizomucor spp. | 1 (12) | 2 (13) | 10 (11) | ||
| Cunninghamella spp. | 8 (8) | 0 | 8 (9) | ||
| Absidia/Lichtheimia spp. | 4 (4) | 1 (6) | 3 (3) | ||
| APACHE II score at diagnosis, median (IQR) | 14 (12–17) | 16 (13–18) | 14 (12–17) | 0.31 | |
| ICU admission at diagnosis | 13 (13) | 2 (13) | 11 (13) | >0.99 | |
| ICU/Hospice at any time during treatment | 79 (77) | 15 (94) | 64 (74) | 0.11 | |
| Days from symptom onset to treatment initiation, median (IQR) | 6 (3–11) | 4 (2–10) | 7 (3–12) | 0.16 | |
| Initial treatment strategy | 0.88 | ||||
| AMB + POSA + CAS | 31 (30) | 5 (31) | 26 (30) | ||
| AMB + POSA | 26 (25) | 6 (38) | 20 (23) | ||
| AMB + CAS | 21 (20) | 2 (13) | 19 (22) | ||
| AMB | 1 (15) | 2 (13) | 13 (15) | ||
| AMB + ISA | 8 (8) | 1 (6) | 7 (8) | ||
| POSA | 2 (2) | 0 (0) | 2 (2) | ||
| Surgical treatment of mucormycosis | 47 (46) | 3 (19) | 44 (51) | 0.019 |
| Anti-Fungal | Year | Daily Dose | Form | Serum Level | Age/Sex | Cancer | Days ANC <500 a | ANC <500 at Dx? | ANC Reco-Very b | Antifungal Indication | Days AntiFungal c | BT-MCR Location | Pathogen | Treatment Strategy | Day 42 Outcome d |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AMB | 2004 | 5 mg/kg | IV | N/D | 29/M | Burkitt’s | 34 | yes | no | Treatment e | 7 | Localized | Rhizopus spp. | AMB | dead |
| ISA | 2015 | 372 mg | tab | N/D | 61/F | R/R AML | 11 | yes | yes | 1º PPx (NP) | 34 | Pulmonary | Mucor spp. | AMB + PSOA | alive |
| ISA | 2016 | 372 mg | tab | N/D | 75/M | act AML | 24 | yes | no | 1º PPx (NP) | 14 | Disseminated | Rhizopus spp. | AMB + POSA | dead |
| ISA | 2016 | 372 mg | tab | N/D | 47/M | act ALL | 110 | yes | yes | 1º PPx | 37 | Sinusitis | Rhizopus spp. | AMB + POSA | dead |
| ISA | 2016 | 372 mg | tab | N/D | 61/M | R/R CLL | 191 | yes | no | Treatment f | 243 | Pulmonary | Rhizomucor spp. | AMB + POSA | dead |
| ISA | 2017 | 372 mg | tab | N/D | 60/F | R/R ALL | 47 | yes | no | Treatment g | 151 | Pulmonary | Rhizopus spp. | AMB + CAS + POSA | dead |
| ISA | 2018 | 372 mg | tab | N/D | 45/M | R/R CLL | 0 | no | - | 1º PPx (GC) | 60 | Sinusitis | Rhizopus spp. | AMB + POSA | alive |
| ISA | 2019 | 372 mg | tab | N/D | 54/M | act AML | 21 | yes | no | 1º PPx | 106 | Pulmonary | Mucor spp. | AMB + POSA | dead |
| ISA | 2019 | 372 mg | tab | N/D | 49/F | R/R AML | 51 | yes | yes | Treatment h | 16 | Sinusitis | Rhizopus spp. | AMB + CAS + POSA | alive |
| ISA | 2019 | 372 mg | tab | N/D | 65/F | act AML | 44 | yes | no | Treatment i | 27 | Sinusitis | Rhizopus spp. | AMB + CAS + POSA | dead |
| POSA | 2000 | 800 mg | liquid | N/D | 29/F | AML (CR) | 0 | no | - | Treatment j | 211 | Disseminated | Rhizopus spp. | AMB | dead |
| POSA | 2009 | 600 mg | liquid | N/D | 52/M | CLL (CR) | 0 | no | - | 1º PPx (GC) | 61 | Sinusitis | Rhizopus spp. | AMB + CAS | dead |
| POSA | 2012 | 800 mg | liquid | 685 ng/dL | 70/M | R/R AML | 20 | yes | yes | 1º PPx (NP) k | 54 | Pulmonary | Rhizomucor spp. | AMB + CAS | dead |
| POSA | 2016 | 300 mg | tab | 1750 ng/dL | 61/M | R/R AML | 14 | yes | yes | 1º PPx (NP) | 17 | Cutaneous | Rhizopus spp. | AMB + CAS + POSA | alive |
| POSA | 2017 | 300 mg | tab | 2520 ng/dL | 25/M | R/R ALL | 20 | yes | yes | 1º PPx (NP) | 437 | Sinusitis | Absidia spp. | AMB + CAS + POSA | alive |
| POSA | 2019 | 300 mg | tab | 2180 ng/dL | 68/M | act AML | 0 | no | - | 1º PPx (NP) | 63 | Pulmonary | Mucor spp. | AMB + ISA | dead |
| Characteristic | All Patients (n = 103) | Mucorales-Active Antifungals (n = 16) | Other Mold-Active Antifungals (n = 87) | p-Value |
|---|---|---|---|---|
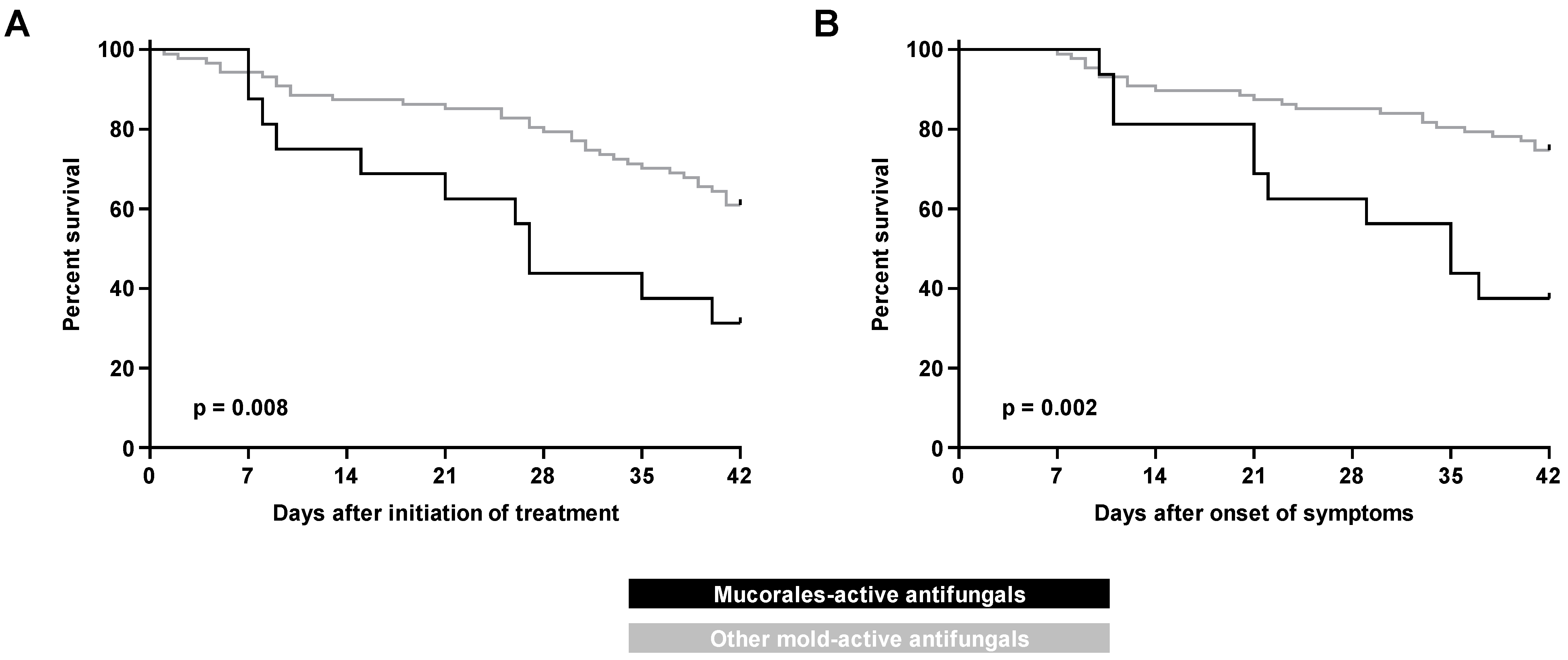
| 42-Day mortality from treatment initiation | 45 (44) | 11 (69) | 34 (39) | 0.028 |
| 84-Day mortality from treatment initiation | 64 (62) | 16 (100) | 48 (55) | 0.0007 |
| 42-Day mortality from symptom onset | 32 (31) | 10 (63) | 22 (25) | 0.006 |
| 84-Day mortality from symptom onset | 61 (59) | 16 (100) | 45 (52) | 0.0003 |
| Days from treatment initiation to death, median (IQR) | 44 (27–110) | 27 (12–50) | 49 (30–146) | 0.007 |
| Days from symptom onset to death, median (IQR) | 55 (34–114) | 35 (22–59) | 61 (40–151) | 0.007 |
| (A) Mortality within 42 Days of Treatment Initiation | ||||
| Predictors | Hazard Ratio | 95% Confidence Interval | p-Value | |
| ICU at diagnosis | 2.46 | 1.07 to 5.68 | 0.034 | |
| APACHE II score at diagnosis | Every 1-unit increase | 1.21 | 1.12 to 1.30 | <0.0001 |
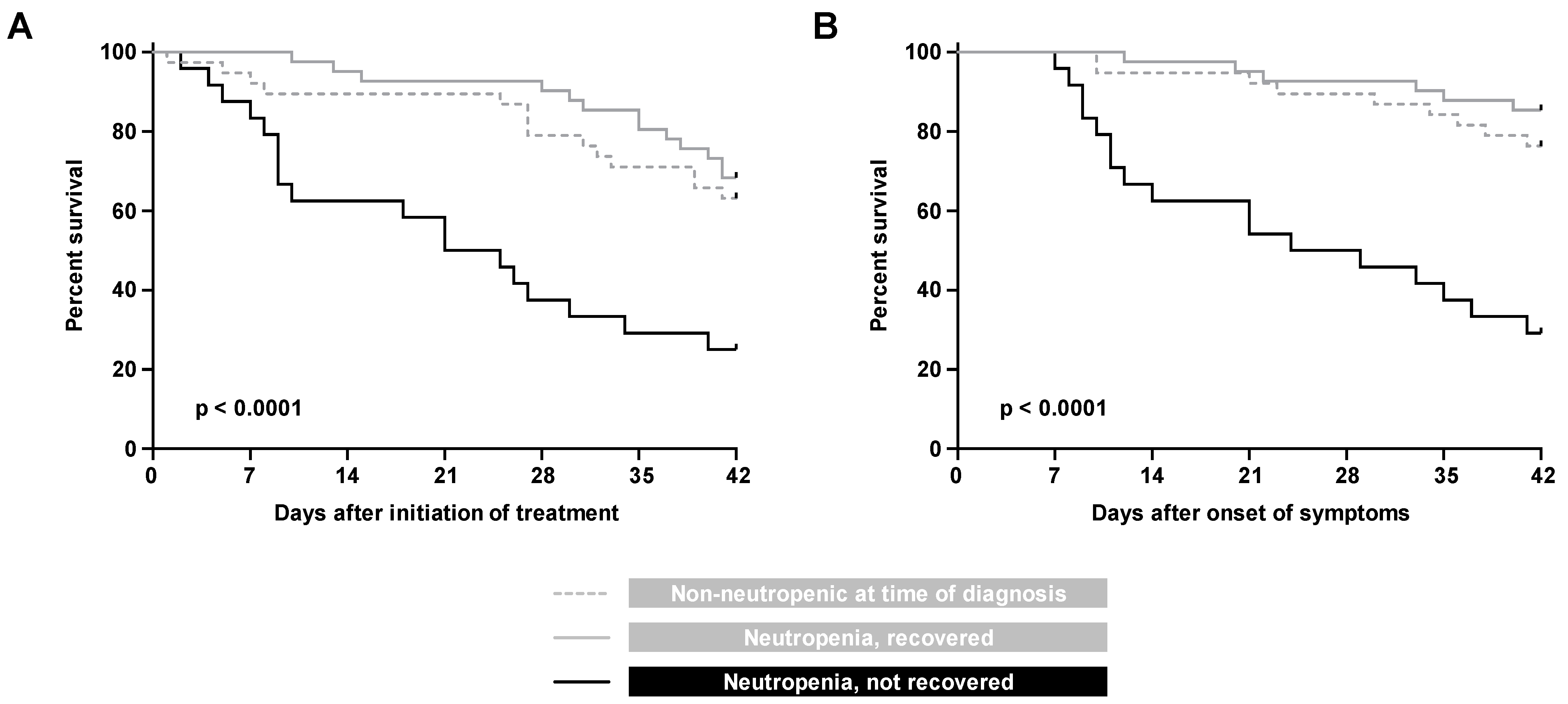
| Neutropenia status | <0.001 | |||
| No neutropenia at diagnosis | Reference | |||
| Neutropenia, recovered | 0.83 | 0.37 to 1.84 | ||
| Neutropenia, not recovered | 3.25 | 1.53 to 6.90 | ||
| Antifungal prophylaxis | 0.015 | |||
| Mucorales-active | 2.40 | 1.19 to 4.86 | ||
| Other mold-active | Reference | |||
| (B) Mortality within 42 Days of Symptom Onset | ||||
| Predictors | Hazard Ratio | 95% Confidence Interval | p-Value | |
| ICU at diagnosis | 4.71 | 2.05 to 10.85 | <0.001 | |
| APACHE II score at diagnosis | Every 1-unit increase | 1.13 | 1.04 to 1.23 | 0.005 |
| Neutropenia status | <0.0001 | |||
| No neutropenia at diagnosis | Reference | |||
| Neutropenia, recovered | 0.67 | 0.23 to 2.00 | ||
| Neutropenia, not recovered | 9.63 | 3.57 to 25.99 | ||
| Antifungal prophylaxis | <0.001 | |||
| Mucorales-active | 4.63 | 1.91 to 11.23 | ||
| Other mold-active | Reference | |||
| Treatment * | <0.0001 | |||
| Amphotericin B + caspofungin | 8.15 | 3.09 to 21.48 | ||
| Others | Reference | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Axell-House, D.B.; Wurster, S.; Jiang, Y.; Kyvernitakis, A.; Lewis, R.E.; Tarrand, J.J.; Raad, I.I.; Kontoyiannis, D.P. Breakthrough Mucormycosis Developing on Mucorales-Active Antifungals Portrays a Poor Prognosis in Patients with Hematologic Cancer. J. Fungi 2021, 7, 217. https://doi.org/10.3390/jof7030217
Axell-House DB, Wurster S, Jiang Y, Kyvernitakis A, Lewis RE, Tarrand JJ, Raad II, Kontoyiannis DP. Breakthrough Mucormycosis Developing on Mucorales-Active Antifungals Portrays a Poor Prognosis in Patients with Hematologic Cancer. Journal of Fungi. 2021; 7(3):217. https://doi.org/10.3390/jof7030217
Chicago/Turabian StyleAxell-House, Dierdre B., Sebastian Wurster, Ying Jiang, Andreas Kyvernitakis, Russell E. Lewis, Jeffrey J. Tarrand, Issam I. Raad, and Dimitrios P. Kontoyiannis. 2021. "Breakthrough Mucormycosis Developing on Mucorales-Active Antifungals Portrays a Poor Prognosis in Patients with Hematologic Cancer" Journal of Fungi 7, no. 3: 217. https://doi.org/10.3390/jof7030217
APA StyleAxell-House, D. B., Wurster, S., Jiang, Y., Kyvernitakis, A., Lewis, R. E., Tarrand, J. J., Raad, I. I., & Kontoyiannis, D. P. (2021). Breakthrough Mucormycosis Developing on Mucorales-Active Antifungals Portrays a Poor Prognosis in Patients with Hematologic Cancer. Journal of Fungi, 7(3), 217. https://doi.org/10.3390/jof7030217








