COVID-19-Associated Pulmonary Aspergillosis in Russia
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
- CAPA develops mainly on the background of diabetes (33%), hematological or oncological diseases (31%), and COPD (13%).
- The probability of CAPA significantly increases with lymphocytopenia for >10 days (OR = 8.156 (3.056–21.771), p = 0.001), decompensated diabetes (29% vs. 7%, (OR = 5.688 (1.991–16.246), p = 0.001), use of steroids at a prednisone-equivalent dose > 60 mg/day (OR = 4.493 (1.896–10.647), p = 0.001) and monoclonal antibodies to IL-1ß and IL-6 (OR = 2.880 (1.272–6.518), p = 0.01)
- CAPA is characterized by pulmonary involvement (100%), and rarely characterized by trachea and bronchi (7%).
- Severe disease course with prolonged (median—15.5 (5–60) days) stays in the ICU (71%), mechanical ventilation (52%) and ARDS (31%) are typical in CAPA patients.
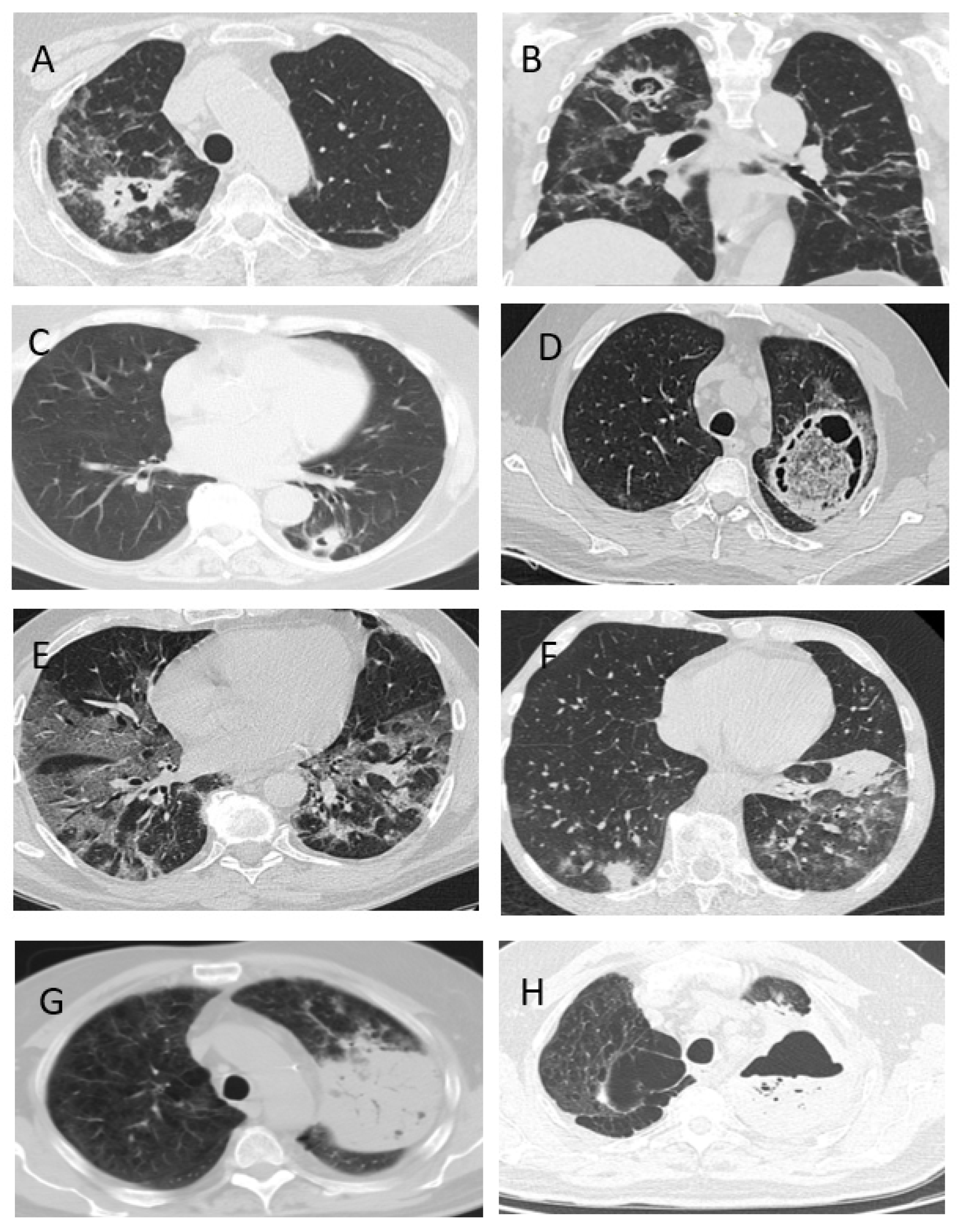
- The clinical signs of CAPA are nonspecific, but typically include: fever (98%), cough (89%) and hemoptysis (36%). The radiological signs of CAPA are foci of consolidation (89%) and destruction (47%), and hydrothorax (26%).
- The most effective method of diagnosing CAPA is the GM test in BAL.
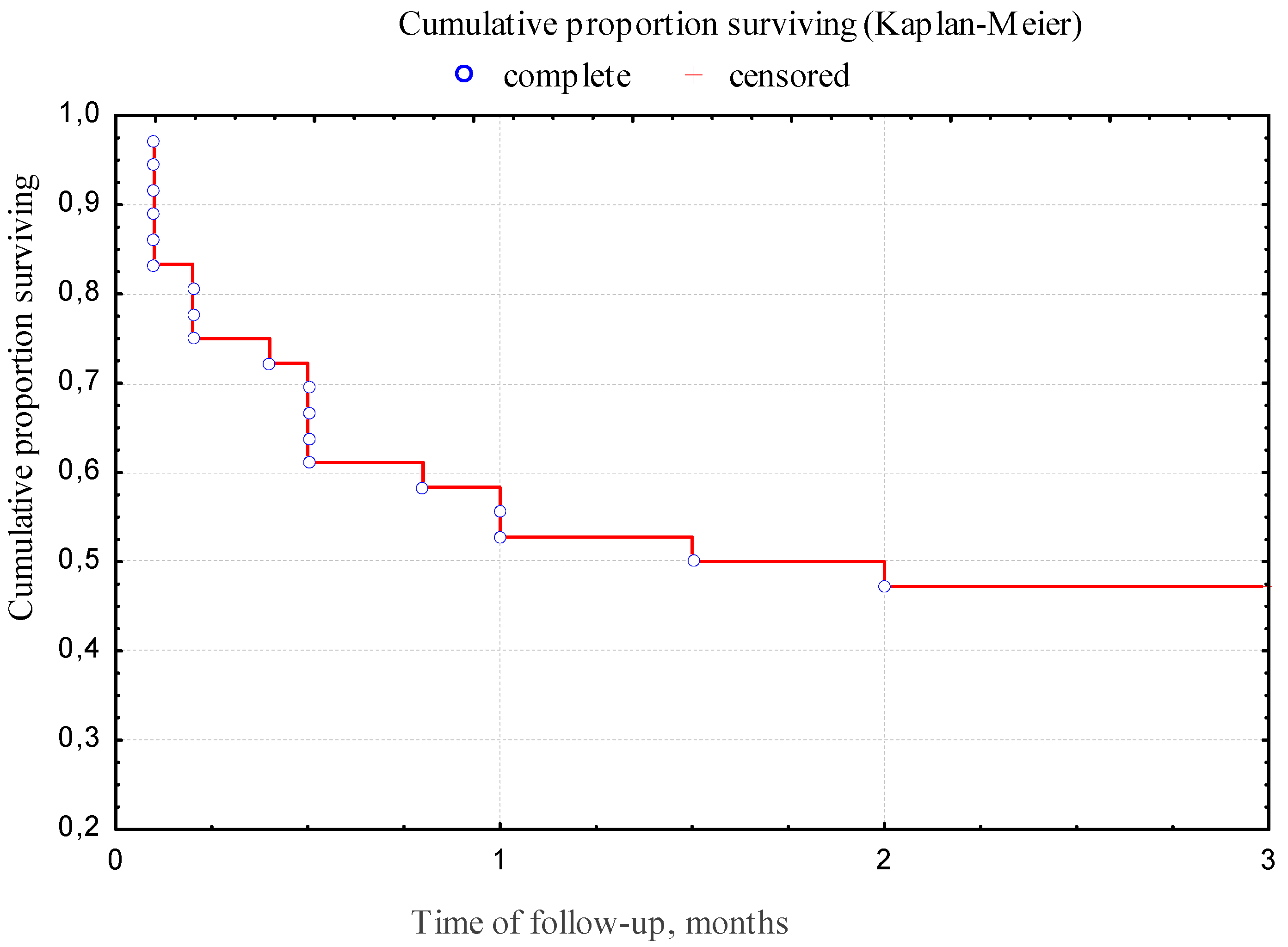
- The overall 12-week survival rate of patients with CAPA was 47.2%.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schauwvlieghe, A.F.A.D.; Rijnders, B.J.A.; Philips, N.; Verwijs, R.; Vanderbeke, L.; Van Tienen, C.; Lagrou, K.; Verweij, P.E.; Van De Veerdonk, F.L.; Gommers, D.; et al. Invasive aspergillosis in patients admitted to the intensive care unit with severe influenza: A retrospective cohort study. Lancet Respir. Med. 2018, 6, 782–792. [Google Scholar] [CrossRef]
- Marr, K.A.; Platt, A.; Tornheim, J.A.; Zhang, S.X.; Datta, K.; Cardozo, C.; Garcia-Vidal, C. Aspergillosis Complicating Severe Coronavirus Disease. Emerg. Infect. Dis. 2021, 27, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Koehler, P.; Cornely, O.A.; Böttiger, B.W.; Dusse, F.; Eichenauer, D.A.; Fuchs, F.; Hallek, M.; Jung, N.; Klein, F.; Persigehl, T.; et al. COVID-19 associated pulmonary aspergillosis. Mycoses 2020, 63, 528–534. [Google Scholar] [CrossRef] [PubMed]
- van Arkel, A.L.; Rijpstra, T.A.; Belderbos, H.N.; Van Wijngaarden, P.; Verweij, P.E.; Bentvelsen, R.G. COVID-19 associated pulmonary aspergillosis. Am. J. Respir. Crit. Care Med. 2020, 202, 132–135. [Google Scholar] [CrossRef]
- Alanio, A.; Dellière, S.; Fodil, S.; Bretagne, S.; Mégarbane, B. Prevalence of putative invasive pulmonary aspergillosis in critically ill patients with COVID-19. Lancet Respir. Med. 2020, 8, e48–e49. [Google Scholar] [CrossRef]
- Nasir, N.; Farooqi, J.; Mahmood, S.F.; Jabeen, K. COVID-19-associated pulmonary aspergillosis (CAPA) in patients admitted with severe COVID-19 pneumonia: An observational study from Pakistan. Mycoses 2020, 63, 766–770. [Google Scholar] [CrossRef]
- Ferreira, J.C.; Ho, Y.-L.; Besen, B.A.M.P.; Malbouisson, L.M.S.; Taniguchi, L.U.; Mendes, P.V.; Costa, E.L.V.; Park, M.; Daltro-Oliveira, R.; Roepke, R.M.L.; et al. Additional file 1 of Protective ventilation and outcomes of critically ill patients with COVID-19: A cohort study. Ann. Intensive Care 2021, 11, 11. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Northwell COVID-19 Research Consortium. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized with COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Karagiannidis, C.; Mostert, C.; Hentschker, C.; Voshaar, T.; Malzahn, J.; Schillinger, G.; Klauber, J.; Janssens, U.; Marx, G.; Weber-Carstens, S.; et al. Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: An observational study. Lancet Respir. Med. 2020, 8, 853–862. [Google Scholar] [CrossRef]
- Bartoletti, M.; Pascale, R.; Cricca, M.; Rinaldi, M.; Maccaro, A.; Bussini, L.; Fornaro, G.; Tonetti, T.; Pizzilli, G.; Francalanci, E.; et al. Epidemiology of Invasive Pulmonary Aspergillosis Among Intubated Patients With COVID-19: A Prospective Study. Clin. Infect. Dis. 2020, ciaa1065. [Google Scholar] [CrossRef]
- Gusarov, V.G.; Zamyatin, M.N.; Kamyshova, D.A.; Fomina, V.S.; Abovich, Y.A.; Lovtsevich, N.V.; Bronov, O.Y.; Petrova, L.V.; Sysoeva, T.S.; Vasilashko, V.I.; et al. Invasive pulmonary aspergillosis in COVID-19 patients. J. Infektologii 2021, 13, 38–49. [Google Scholar] [CrossRef]
- Donnelly, J.P.; Chen, S.C.; Kauffman, C.A.; Steinbach, W.J.; Baddley, J.W.; Verweij, P.E.; Clancy, C.J.; Wingard, J.R.; Lockhart, S.R.; Groll, A.H.; et al. Revision and Update of the Consensus Definitions of Invasive Fungal Dis-ease From the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin. Infect. Dis. 2020, 71, 1367–1376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koehler, P.; Bassetti, M.; Chakrabarti, A.; Chen, S.C.A.; Colombo, A.L.; Hoenigl, M.; Klimko, N.; Lass-Flörl, C.; Oladele, R.O.; Vinh, D.C.; et al. Defining and managing COVID-19-associated pulmonary aspergillosis: The 2020 ECMM/ISHAM consensus criteria for research and clinical guidance. Lancet Infect. Dis. 2021, 21, e149–e162. [Google Scholar] [CrossRef]
- Arastehfar, A.; Carvalho, A.; Van De Veerdonk, F.L.; Jenks, J.D.; Koehler, P.; Krause, R.; Cornely, O.A.; Perlin, D.S.; Lass-Flörl, C.; Hoenigl, M. COVID-19 Associated Pulmonary Aspergillosis (CAPA)—From Immunology to Treatment. J. Fungi 2020, 6, 91. [Google Scholar] [CrossRef] [PubMed]
- Clancy, C.J.; Nguyen, M.H. Coronavirus Disease 2019, Superinfections, and Antimicrobial Development: What Can We Expect? Clin. Infect. Dis. 2020, 71, 2736–2743. [Google Scholar] [CrossRef]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Wang, W.; et al. Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis. 2020, 71, 762–768. [Google Scholar] [CrossRef]
- Salmanton-Garcia, J.; Sprute, R.; Stemler, J.; Bartoletti, M.; Dupont, D.; Valerio, M.; Garcia-Vidal, C.; Falces-Romero, I.; Machado, M.; de la Villa, S.; et al. COVID-19–Associated Pulmonary Aspergillosis, March–August 2020. Emerg. Infect. Dis. 2021, 27, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Mitaka, H.; Kuno, T.; Takagi, H.; Patrawalla, P. Incidence and mortality of COVID-19-associated pulmonary aspergillosis: A systematic review and meta-analysis. Mycoses 2021, 64, 993–1001. [Google Scholar] [CrossRef]
- Dellière, S.; Dudoignon, E.; Fodil, S.; Voicu, S.; Collet, M.; Oillic, P.-A.; Salmona, M.; Dépret, F.; Ghelfenstein-Ferreira, T.; Plaud, B.; et al. Risk factors associated with COVID-19-associated pulmonary aspergillosis in ICU patients: A French multicentric retrospective cohort. Clin. Microbiol. Infect. 2020, 27, 790.e1–790.e5. [Google Scholar] [CrossRef]
- Gangneux, J.P.; Dannaoui, E.; Fekkar, A.; Luyt, C.E.; Botterel, F.; De Prost, N.; Tadié, J.-M.; Reizine, F.; Pharm, S.H.; Timsit, J.-F.; et al. Characterization of Fungal Infections in COVID-19 Infected Mechanically Ventilated Patients in I.C.U. the MY-CO-VID Clinical Trial. Available online: https://clinicaltrials.gov/ct2/show/study/NCT04368221 (accessed on 8 October 2021).
- Rutsaert, L.; Steinfort, N.; Van Hunsel, T.; Bomans, P.; Naesens, R.; Mertes, H.; Dits, H.; Van Regenmortel, N. COVID-19-associated invasive pulmonary aspergillosis. Ann. Intensiv. Care 2020, 10, 528–534. [Google Scholar] [CrossRef]
- Janssen, N.A.F.; Nyga, R.; Vanderbeke, L.; Jacobs, C.; Ergün, M.; Buil, J.B.; van Dijk, K.; Altenburg, J.; Bouman, C.S.C.; van der Spoel, H.I.; et al. Multinational Observational Cohort Study of COVID-19—Associated Pulmonary Aspergillosis. Emerg. Infect. Dis. 2021, 27, 2892. [Google Scholar] [CrossRef] [PubMed]
- Prattes, J.; Wauters, J.; Giacobbe, D.R.; Salmanton-García, J.; Maertens, J.; Bourgeois, M.; Reynders, M.; Rutsaert, L.; Van Regenmortel, N.; Lormans, P.; et al. Risk factors and outcome of pulmonary aspergillosis in critically ill coronavirus disease 2019 patients—a multinational observational study by the European Confederation of Medical Mycology. Clin. Microbiol. Infect. 2021, 27, 2892. [Google Scholar] [CrossRef]
- White, P.L.; Dhillon, R.; Cordey, A.; Hughes, H.; Faggian, F.; Soni, S.; Pandey, M.; Whitaker, H.; May, A.; Morgan, M.; et al. A National Strategy to Diagnose Coronavirus Disease 2019—Associated Invasive Fungal Disease in the Intensive Care Unit. Clin. Infect. Dis. 2021, 73, e1634–e1644. [Google Scholar] [CrossRef]
- Wang, J.; Yang, Q.; Zhang, P.; Sheng, J.; Zhou, J.; Qu, T. Clinical characteristics of invasive pulmonary aspergillosis in patients with COVID-19 in Zhejiang, China: A retrospective case series. Crit. Care 2020, 24, 4. [Google Scholar] [CrossRef] [PubMed]
- Verweij, P.E.; Brüggemann, R.J.M.; Azoulay, E.; Bassetti, M.; Blot, S.; Buil, J.B.; Calandra, T.; Chiller, T.; Clancy, C.J.; Cornely, O.A.; et al. Taskforce report on the diagnosis and clinical management of COVID-19 associated pulmonary aspergillosis. Intensiv. Care Med. 2021, 47, 819–834. [Google Scholar] [CrossRef] [PubMed]
- Lahmer, T.; Kriescher, S.; Herner, A.; Rothe, K.; Spinner, C.D.; Schneider, J.; Mayer, U.; Neuenhahn, M.; Hoffmann, D.; Geisler, F.; et al. Invasive pulmonary aspergillosis in critically ill patients with severe COVID-19 pneumonia: Results from the prospective AspCOVID-19 study. PLoS ONE 2021, 16, e0238825. [Google Scholar] [CrossRef]
- Hatzl, S.; Reisinger, A.C.; Posch, F.; Prattes, J.; Stradner, M.; Pilz, S.; Eller, P.; Schoerghuber, M.; Toller, W.; Gorkiewicz, G.; et al. Antifungal prophylaxis for prevention of COVID-19-associated pulmonary asper-gillosis in critically ill patients: An observational study. Crit. Care. 2021, 25, 335. [Google Scholar] [CrossRef] [PubMed]
| CAPA | COVID-19 without IA | p-Value | |||
|---|---|---|---|---|---|
| n = 45 | % | n = 90 | % | ||
| Demographics | |||||
| males | 31 | 69% | 60 | 67% | 0.66 |
| females | 14 | 31% | 30 | 33% | |
| age median (years) | 34–82 62 | 25–82 63 | 0.67 | ||
| background diseases | |||||
| hematological diseases | 9 | 20% | 11 | 12% | 0.2 |
| lymphomas acute leukemia MM CLL others | 6 1 2 - - | 13% 2% 4% | 6 1 1 2 1 | 7% 1% 1% 1% | |
| oncology | 5 | 11% | 5 | 5.5% | 0.1 |
| active hematological/oncological disease | 11 | 24% | 2 | 2% | 0.03 |
| DM decompensated DM | 15 13 | 33% 29% | 17 6 | 19% 7% | 0.06 0.0003 |
| COPD | 6 | 13% | 5 | 5.5% | 0.09 |
| ARF/CRF | 5 | 11% | 8 | 9% | 0.6 |
| COVID-IA | COVID-19 without IA | p-Value | |||
|---|---|---|---|---|---|
| n/N | % | n/N | % | ||
| neutropenia <0.5 × 109/L | 4/45 | 9% | 0/90 | 0.006 | |
| duration (min-max)/ Me (days) | 5–25 10 | - | |||
| lymphocytopenia <1.0 × 109/L | 38/43 | 88% | 68/88 | 77% | 0.1 |
| duration (min-max)/ Me (days) | 5–100 15 | 2–42 9 | 0.00002 | ||
| lymphocytopenia >10 days | 29/35 | 83% | 32/86 | 37% | 0.006 |
| glucocorticoids (GCS): | 38/43 | 88% | 77/88 | 88% | 0.7 |
| GCS >60 mg/d in prednisone-equivalent dose | 17/37 | 46% | 14/88 | 16% | 0.01 |
| inhibitors of receptors IL-1β and IL-6 | 18/43 | 42% | 16/80 | 20% | 0.01 |
| Risk Factors | CAPA | COVID-19 without IA | OR (95% CI) | p-Value |
|---|---|---|---|---|
| n/N(%) | n/N(%) | |||
| decompensated DM | 13/45 (29%) | 6/90 (7%) | 5.688 (1.991–16.246) | 0.001 |
| lymphocytopenia >10 days | 29/35 (83%) | 32/86 (37%) | 8.156 (3.056–21.771) | 0.0001 |
| GCS >60 mg/d in prednisone-equivalent dose | 17/37 (46%) | 14/88 (16%) | 4.493 (1.896–10.647) | 0.001 |
| inhibitors of receptors IL-1β and IL-6 | 18/43 (42%) | 16/80 (20%) | 2.880 (1.272–6.518) | 0.01 |
| Features | CAPA n = 45 | COVID-19 without IA n = 90 | p-Value | ||
|---|---|---|---|---|---|
| N | % | n | % | ||
| fever | 44 | 98% | 62/73 | 85% | 0.007 |
| cough | 40 | 89% | 42/53 | 72% | 0.002 |
| chest pain | 10/42 | 24% | 4/45 | 9% | 0.05 |
| respiratory failure 2-3-4 (requiring O2 or ventilation) | 28 | 62% | 54 | 60% | 0.7 |
| ARDS | 14 | 31% | 16 | 18% | 0.02 |
| hemoptysis | 16 | 36% | 3/87 | 3% | 0.0001 |
| ICU | 32 | 71% | 57 | 63% | 0.4 |
| total days in ICU Me | 5–60 15.5 | 1–55 6 | 0.0004 | ||
| mechanical ventilation | 14 | 52% | 8/54 | 15% | 0.004 |
| CT-signs | |||||
| bilateral lesion | 42 | 93% | 75/80 | 94% | 0.8 |
| infiltrations | 40 | 89% | 37/63 | 59% | 0.004 |
| the “frosted glass” symptom | 33 | 73% | 64/80 | 80% | 0.3 |
| destruction cavity | 21 | 47% | 1 | 1% | 0.00001 |
| the “halo” symptom | - | - | - | - | |
| hydrothorax | 10/38 | 26% | 10/88 | 11% | 0.03 |
| Method | Result | |
|---|---|---|
| n | % | |
| microscopy (+) | 11 | 24% |
| culture (+) | 14 | 31% |
| GM in blood (+) | 3 | 7% |
| GM in BAL (+) | 25 | 56% |
| histology (+) | 3 | 7% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shadrivova, O.; Gusev, D.; Vashukova, M.; Lobzin, D.; Gusarov, V.; Zamyatin, M.; Zavrazhnov, A.; Mitichkin, M.; Borzova, Y.; Kozlova, O.; et al. COVID-19-Associated Pulmonary Aspergillosis in Russia. J. Fungi 2021, 7, 1059. https://doi.org/10.3390/jof7121059
Shadrivova O, Gusev D, Vashukova M, Lobzin D, Gusarov V, Zamyatin M, Zavrazhnov A, Mitichkin M, Borzova Y, Kozlova O, et al. COVID-19-Associated Pulmonary Aspergillosis in Russia. Journal of Fungi. 2021; 7(12):1059. https://doi.org/10.3390/jof7121059
Chicago/Turabian StyleShadrivova, Olga, Denis Gusev, Maria Vashukova, Dmitriy Lobzin, Vitaliy Gusarov, Mikhail Zamyatin, Anatoliy Zavrazhnov, Mikhail Mitichkin, Yulia Borzova, Olga Kozlova, and et al. 2021. "COVID-19-Associated Pulmonary Aspergillosis in Russia" Journal of Fungi 7, no. 12: 1059. https://doi.org/10.3390/jof7121059
APA StyleShadrivova, O., Gusev, D., Vashukova, M., Lobzin, D., Gusarov, V., Zamyatin, M., Zavrazhnov, A., Mitichkin, M., Borzova, Y., Kozlova, O., Desyatik, E., Burygina, E., Ignatyeva, S., Oganesyan, E., Vasilyeva, N., Klimko, N., & Working Group. (2021). COVID-19-Associated Pulmonary Aspergillosis in Russia. Journal of Fungi, 7(12), 1059. https://doi.org/10.3390/jof7121059







