Detecting Azole-Antifungal Resistance in Aspergillus fumigatus by Pyrosequencing
Abstract
1. Diagnosis of Aspergillosis
2. Azole Resistance
3. Molecular Techniques for Discerning cyp51A Resistance Polymorphisms
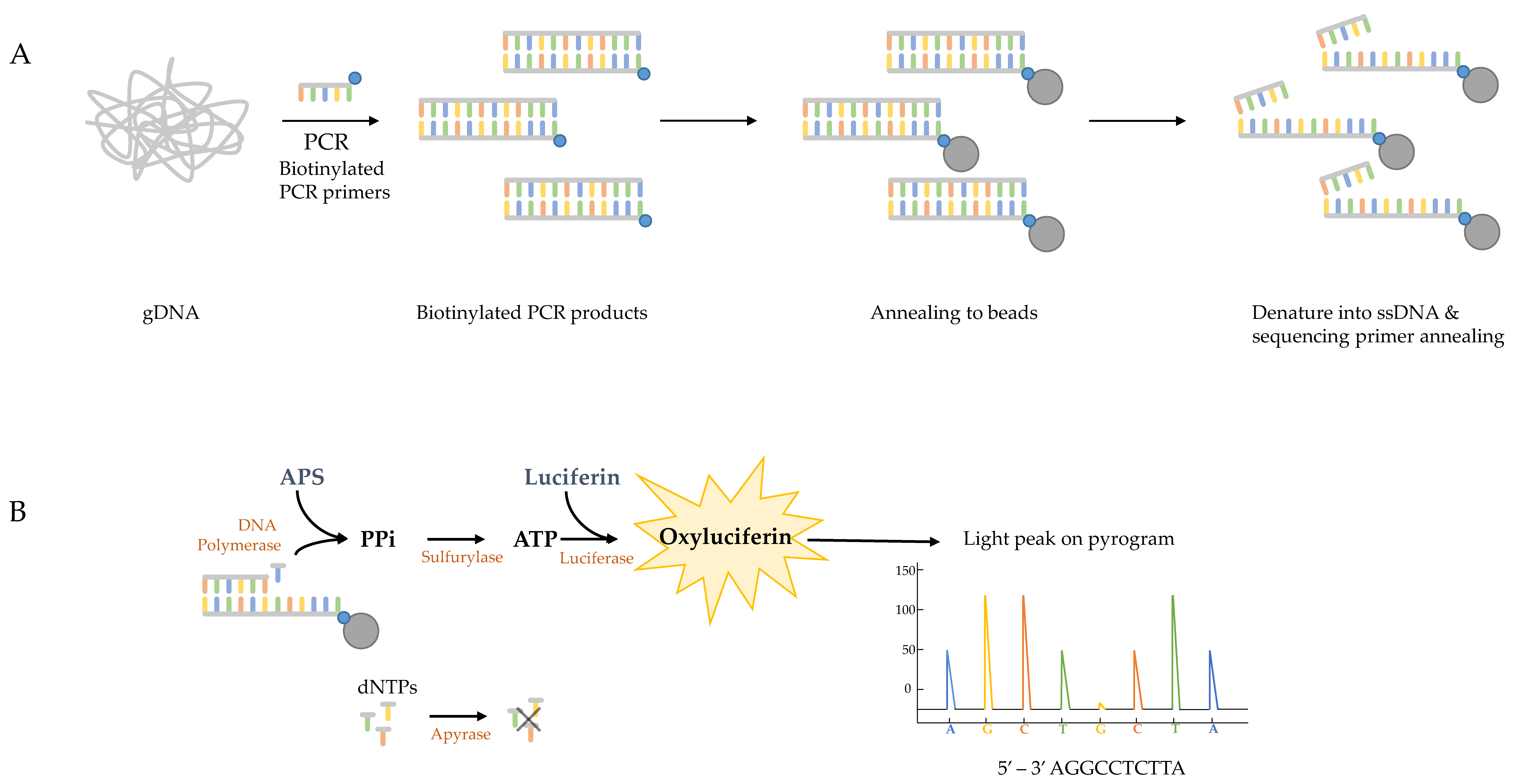
4. The Pyrosequencing Method
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brown, G.D.; Denning, D.W.; Gow, N.A.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden killers: Human fungal infections. Sci. Transl. Med. 2012, 4. [Google Scholar] [CrossRef] [PubMed]
- Taccone, F.S.; Van den Abeele, A.M.; Bulpa, P.; Misset, B.; Meersseman, W.; Cardoso, T.; Paiva, J.A.; Blasco-Navalpotro, M.; De Laere, E.; Dimopoulos, G.; et al. Epidemiology of invasive aspergillosis in critically ill patients: Clinical presentation, underlying conditions, and outcomes. Crit. Care 2015, 19, 7. [Google Scholar] [CrossRef] [PubMed]
- Kosmidis, C.; Denning, D.W. The clinical spectrum of pulmonary aspergillosis. Thorax 2015, 70, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Meersseman, W.; Vandecasteele, S.J.; Wilmer, A.; Verbeken, E.; Peetermans, W.E.; Van Wijngaerden, E. Invasive aspergillosis in critically ill patients without malignancy. Am. J. Respir. Crit. Care Med. 2004, 170, 621–625. [Google Scholar] [CrossRef]
- Mayr, A.; Lass-Florl, C. Epidemiology and antifungal resistance in invasive Aspergillosis according to primary disease: Review of the literature. Eur. J. Med. Res. 2011, 16, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Patterson, T.F.; Thompson, G.R.; Denning, D.W.; Fishman, J.A.; Hadley, S.; Herbrecht, R.; Kontoyiannis, D.P.; Marr, K.A.; Morrison, V.A.; Nguyen, M.H.; et al. Practice Guidelines for the Diagnosis and Management of Aspergillosis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2016, 63, e1–e60. [Google Scholar] [CrossRef]
- Heinz, W.J.; Buchheidt, D.; Christopeit, M.; Von Lilienfeld-Toal, M.; Cornely, O.A.; Einsele, H.; Karthaus, M.; Link, H.; Mahlberg, R.; Neumann, S.; et al. Diagnosis and empirical treatment of fever of unknown origin (FUO) in adult neutropenic patients: Guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Medical Oncology (DGHO). Ann. Hematol. 2017, 96, 1775–1792. [Google Scholar] [CrossRef]
- Ullmann, A.J.; Aguado, J.M.; Arikan-Akdagli, S.; Denning, D.W.; Groll, A.H.; Lagrou, K.; Lass-Florl, C.; Lewis, R.E.; Munoz, P.; Verweij, P.E.; et al. Diagnosis and management of Aspergillus diseases: Executive summary of the 2017 ESCMID-ECMM-ERS guideline. Clin. Microbiol. Infect. 2018, 24, e1–e38. [Google Scholar] [CrossRef]
- Ruhnke, M.; Behre, G.; Buchheidt, D.; Christopeit, M.; Hamprecht, A.; Heinz, W.; Heussel, C.P.; Horger, M.; Kurzai, O.; Karthaus, M.; et al. Diagnosis of invasive fungal diseases in haematology and oncology: 2018 update of the recommendations of the infectious diseases working party of the German society for hematology and medical oncology (AGIHO). Mycoses 2018, 61, 796–813. [Google Scholar] [CrossRef]
- Rath, P.M.; Steinmann, J. Overview of Commercially Available PCR Assays for the Detection of Aspergillus spp. DNA in Patient Samples. Front. Microbiol. 2018, 9, 740. [Google Scholar] [CrossRef]
- Cruciani, M.; Mengoli, C.; Barnes, R.; Donnelly, J.P.; Loeffler, J.; Jones, B.L.; Klingspor, L.; Maertens, J.; Morton, C.O.; White, L.P. Polymerase chain reaction blood tests for the diagnosis of invasive aspergillosis in immunocompromised people. Cochrane Database Syst. Rev. 2019, 9, CD009551. [Google Scholar] [CrossRef]
- Avni, T.; Levy, I.; Sprecher, H.; Yahav, D.; Leibovici, L.; Paul, M. Diagnostic accuracy of PCR alone compared to galactomannan in bronchoalveolar lavage fluid for diagnosis of invasive pulmonary aspergillosis: A systematic review. J. Clin. Microbiol. 2012, 50, 3652–3658. [Google Scholar] [CrossRef] [PubMed]
- Fraczek, M.G.; Kirwan, M.B.; Moore, C.B.; Morris, J.; Denning, D.W.; Richardson, M.D. Volume dependency for culture of fungi from respiratory secretions and increased sensitivity of Aspergillus quantitative PCR. Mycoses 2014, 57, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Vergidis, P.; Moore, C.B.; Novak-Frazer, L.; Richardson, R.; Walker, A.; Denning, D.W.; Richardson, M.D. High-volume culture and quantitative real-time PCR for the detection of Aspergillus in sputum. Clin. Microbiol. Infect. 2019. [Google Scholar] [CrossRef] [PubMed]
- Cornely, O.A.; Koehler, P.; Arenz, D.S.C.M. EQUAL Aspergillosis Score 2018: An ECMM score derived from current guidelines to measure QUALity of the clinical management of invasive pulmonary aspergillosis. Mycoses 2018, 61, 833–836. [Google Scholar] [CrossRef]
- Van der Linden, J.W.; Arendrup, M.C.; Warris, A.; Lagrou, K.; Pelloux, H.; Hauser, P.M.; Chryssanthou, E.; Mellado, E.; Kidd, S.E.; Tortorano, A.M.; et al. Prospective multicenter international surveillance of azole resistance in Aspergillus fumigatus. Emerg. Infect. Dis. 2015, 21, 1041–1044. [Google Scholar] [CrossRef]
- Verweij, P.E.; Chowdhary, A.; Melchers, W.J.; Meis, J.F. Azole Resistance in Aspergillus fumigatus: Can We Retain the Clinical Use of Mold-Active Antifungal Azoles? Clin. Infect. Dis. 2016, 62, 362–368. [Google Scholar] [CrossRef]
- Garcia-Rubio, R.; Cuenca-Estrella, M.; Mellado, E. Triazole Resistance in Aspergillus Species: An Emerging Problem. Drugs 2017, 77, 599–613. [Google Scholar] [CrossRef]
- Fisher, M.C.; Hawkins, N.J.; Sanglard, D.; Gurr, S.J. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 2018, 360, 739–742. [Google Scholar] [CrossRef]
- Perlin, D.S.; Rautemaa-Richardson, R.; Alastruey-Izquierdo, A. The global problem of antifungal resistance: Prevalence, mechanisms, and management. Lancet Infect. Dis. 2017, 17, e383–e392. [Google Scholar] [CrossRef]
- Buil, J.B.; Snelders, E.; Denardi, L.B.; Melchers, W.J.G.; Verweij, P.E. Trends in Azole Resistance in Aspergillus fumigatus, the Netherlands, 1994–2016. Emerg. Infect. Dis. 2019, 25, 176–178. [Google Scholar] [CrossRef]
- Meis, J.F.; Chowdhary, A.; Rhodes, J.L.; Fisher, M.C.; Verweij, P.E. Clinical implications of globally emerging azole resistance in Aspergillus fumigatus. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016, 371, 20150460. [Google Scholar] [CrossRef] [PubMed]
- De Greeff, S.C.; Mouton, J.W. Nethmap 2015: Consumption of Antimicrobial Agents and Antimicrobial Resistance Among Medically Important Bacteria in the Netherlands/MARAN 2015: Monitoring of Antimicrobial Resistance and Antibiotic Usage in Animals in the Netherlands in 2014; Rijksinstituut Voor Volksgezondheid en Milieu: Catharijnesingel, The Netherlands, 2015. [Google Scholar]
- De Greeff, S.C.; Mouton, J.W.; Schoffelen, A.F.; Verduin, C.M. NethMap 2019: Consumption of Antimicrobial Agents and Antimicrobial Resistance Among Medically Important Bacteria in the Netherlands/Maran 2019: Monitoring of Antimicrobial Resistance and Antibiotic Usage in Animals in the Netherlands in 2018; Rijksinstituut Voor Volksgezondheid en Milieu: Catharijnesingel, The Netherlands, 2019. [Google Scholar]
- Abdolrasouli, A.; Petrou, M.A.; Park, H.; Rhodes, J.L.; Rawson, T.M.; Moore, L.S.P.; Donaldson, H.; Holmes, A.H.; Fisher, M.C.; Armstrong-James, D. Surveillance for Azole-Resistant Aspergillus fumigatus in a Centralized Diagnostic Mycology Service, London, United Kingdom, 1998–2017. Front. Microbiol. 2018, 9, 2234. [Google Scholar] [CrossRef] [PubMed]
- Bueid, A.; Howard, S.J.; Moore, C.B.; Richardson, M.D.; Harrison, E.; Bowyer, P.; Denning, D.W. Azole antifungal resistance in Aspergillus fumigatus: 2008 and 2009. J. Antimicrob. Chemother. 2010, 65, 2116–2118. [Google Scholar] [CrossRef] [PubMed]
- Howard, S.J.; Cerar, D.; Anderson, M.J.; Albarrag, A.; Fisher, M.C.; Pasqualotto, A.C.; Laverdiere, M.; Arendrup, M.C.; Perlin, D.S.; Denning, D.W. Frequency and evolution of Azole resistance in Aspergillus fumigatus associated with treatment failure. Emerg. Infect. Dis. 2009, 15, 1068–1076. [Google Scholar] [CrossRef]
- Denning, D.W.; Park, S.; Lass-Florl, C.; Fraczek, M.G.; Kirwan, M.; Gore, R.; Smith, J.; Bueid, A.; Moore, C.B.; Bowyer, P.; et al. High-frequency triazole resistance found in nonculturable Aspergillus fumigatus from lungs of patients with chronic fungal disease. Clin. Infect. Dis. 2011, 52, 1123–1129. [Google Scholar] [CrossRef]
- Astvad, K.M.T.; Jensen, R.H.; Hassan, T.M.; Mathiasen, E.G.; Thomsen, G.M.; Pedersen, U.G.; Christensen, M.; Hilberg, O.; Arendrup, M.C. First detection of TR46/Y121F/T289A and TR34/L98H alterations in Aspergillus fumigatus isolates from azole-naive patients in Denmark despite negative findings in the environment. Antimicrob. Agents Chemother. 2014, 58, 5096–5101. [Google Scholar] [CrossRef]
- Choukri, F.; Botterel, F.; Sitterlé, E.; Bassinet, L.; Foulet, F.; Guillot, J.; Costa, J.M.; Fauchet, N.; Dannaoui, E. Prospective evaluation of azole resistance in Aspergillus fumigatus clinical isolates in France. Med. Mycol. 2015, 53, 593–596. [Google Scholar] [CrossRef][Green Version]
- Rivero-Menendez, O.; Soto-Debran, J.C.; Medina, N.; Lucio, J.; Mellado, E.; Alastruey-Izquierdo, A. Molecular Identification, Antifungal Susceptibility Testing, and Mechanisms of Azole Resistance in Aspergillus Species Received within a Surveillance Program on Antifungal Resistance in Spain. Antimicrob. Agents Chemother. 2019, 63, e00865-19. [Google Scholar] [CrossRef]
- Sewell, T.R.; Zhu, J.; Rhodes, J.; Hagen, F.; Meis, J.F.; Fisher, M.C.; Jombart, T. Nonrandom Distribution of Azole Resistance across the Global Population of Aspergillus fumigatus. MBio 2019, 10, e00392-19. [Google Scholar] [CrossRef]
- Vermeulen, E.; Lagrou, K.; Verweij, P.E. Azole resistance in Aspergillus fumigatus: A growing public health concern. Curr. Opin. Infect. Dis. 2013, 26, 493–500. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Gil, V.G.; Gutierrez, F.; Lindner, J.R.; Albataineh, M.T.; McCarthy, D.I.; Sanders, C.; Fan, H.; Fothergill, A.W.; Sutton, D.A. First Detection of TR34 L98H and TR46 Y121F T289A Cyp51 Mutations in Aspergillus fumigatus Isolates in the United States. J. Clin. Microbiol. 2016, 54, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Feng, C.-L.; Chen, F.; He, Q.; Su, X.; Shi, Y. Triazole Resistance in Aspergillus fumigatus Clinical Isolates Obtained in Nanjing, China. Chin. Med. J. (Engl.) 2017, 130, 665–668. [Google Scholar] [CrossRef] [PubMed]
- Resendiz Sharpe, A.; Lagrou, K.; Meis, J.F.; Chowdhary, A.; Lockhart, S.R.; Verweij, P.E. Triazole resistance surveillance in Aspergillus fumigatus. Med. Mycol. 2018, 56, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Verweij, P.E.; Lestrade, P.P.A.; Melchers, W.J.G.; Meis, J.F. Azole resistance surveillance in Aspergillus fumigatus: Beneficial or biased? J. Antimicrob. Chemother. 2016, 71, 2079–2082. [Google Scholar] [CrossRef] [PubMed]
- Arikan-Akdagli, S.; Ghannoum, M.; Meis, J.F. Antifungal Resistance: Specific Focus on Multidrug Resistance in Candida auris and Secondary Azole Resistance in Aspergillus fumigatus. J. Fungi 2018, 4, 129. [Google Scholar] [CrossRef]
- Guinea, J.; Verweij, P.E.; Meletiadis, J.; Mouton, J.W.; Barchiesi, F.; Arendrup, M.C. Subcommittee on Antifungal Susceptibility Testing of the E.E.C.f.A.S.T. How to: EUCAST recommendations on the screening procedure E.Def 10.1 for the detection of azole resistance in Aspergillus fumigatus isolates using four-well azole-containing agar plates. Clin. Microbiol. Infect. 2019, 25, 681–687. [Google Scholar] [CrossRef]
- Lestrade, P.P.; Van der Velden, W.; Bouwman, F.; Stoop, F.J.; Blijlevens, N.M.A.; Melchers, W.J.G.; Verweij, P.E.; Donnelly, J.P. Epidemiology of invasive aspergillosis and triazole-resistant Aspergillus fumigatus in patients with haematological malignancies: A single-centre retrospective cohort study. J. Antimicrob. Chemother. 2018, 73, 1389–1394. [Google Scholar] [CrossRef]
- Kohno, S.; Izumikawa, K.; Ogawa, K.; Kurashima, A.; Okimoto, N.; Amitani, R.; Kakeya, H.; Niki, Y.; Miyazaki, Y. Intravenous micafungin versus voriconazole for chronic pulmonary aspergillosis: A multicenter trial in Japan. J. Infect. 2010, 61, 410–418. [Google Scholar] [CrossRef]
- Chong, G.M.; Van der Beek, M.T.; Von dem Borne, P.A.; Boelens, J.; Steel, E.; Kampinga, G.A.; Span, L.F.; Lagrou, K.; Maertens, J.A.; Dingemans, G.J.; et al. PCR-based detection of Aspergillus fumigatus Cyp51A mutations on bronchoalveolar lavage: A multicentre validation of the AsperGenius assay(R) in 201 patients with haematological disease suspected for invasive aspergillosis. J. Antimicrob. Chemother. 2016, 71, 3528–3535. [Google Scholar] [CrossRef]
- Meersseman, W.; Lagrou, K.; Maertens, J.; Wilmer, A.; Hermans, G.; Vanderschueren, S.; Spriet, I.; Verbeken, E.; Van Wijngaerden, E. Galactomannan in bronchoalveolar lavage fluid: A tool for diagnosing aspergillosis in intensive care unit patients. Am. J. Respir. Crit. Care Med. 2008, 177, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Tudela, J.L.; Alcazar-Fuoli, L.; Alastruey-Izquierdo, A.; Monzon, A.; Mellado, E.; Cuenca-Estrella, M. Time of incubation for antifungal susceptibility testing of Aspergillus fumigatus: Can MIC values be obtained at 24 h? Antimicrob. Agents Chemother. 2007, 51, 4502–4504. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lestrade, P.P.A.; Meis, J.F.; Melchers, W.J.G.; Verweij, P.E. Triazole resistance in Aspergillus fumigatus: Recent insights and challenges for patient management. Clin. Microbiol. Infect. 2019, 25, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, D.; Arai, T.; Takahashi, H.; Kusuya, Y.; Watanabe, A.; Kamei, K. Non-cyp51A Azole-Resistant Aspergillus fumigatus Isolates with Mutation in HMG-CoA Reductase. Emerg. Infect. Dis. 2018, 24, 1889–1897. [Google Scholar] [CrossRef] [PubMed]
- Buied, A.; Moore, C.B.; Denning, D.W.; Bowyer, P. High-level expression of cyp51B in azole-resistant clinical Aspergillus fumigatus isolates. J. Antimicrob. Chemother. 2013, 68, 512–514. [Google Scholar] [CrossRef] [PubMed]
- Sharma, C.; Nelson-Sathi, S.; Singh, A.; Radhakrishna Pillai, M.; Chowdhary, A. Genomic perspective of triazole resistance in clinical and environmental Aspergillus fumigatus isolates without cyp51A mutations. Fungal Genet. Biol. 2019, 132, 103265. [Google Scholar] [CrossRef]
- Dhingra, S.; Cramer, R.A. Regulation of Sterol Biosynthesis in the Human Fungal Pathogen Aspergillus fumigatus: Opportunities for Therapeutic Development. Front. Microbiol. 2017, 8, 92. [Google Scholar] [CrossRef]
- Rybak, J.M.; Ge, W.; Wiederhold, N.P.; Parker, J.E.; Kelly, S.L.; Rogers, P.D.; Fortwendel, J.R. Mutations in hmg1, Challenging the Paradigm of Clinical Triazole Resistance in Aspergillus fumigatus. MBio 2019, 10, e00437-19. [Google Scholar] [CrossRef]
- Jahanshiri, Z.; Shams-Ghahfarokhi, M.; Asghari-Paskiabi, F.; Saghiri, R.; Razzaghi-Abyaneh, M. α-Bisabolol inhibits Aspergillus fumigatus Af239 growth via affecting microsomal ∆(24)-sterol methyltransferase as a crucial enzyme in ergosterol biosynthesis pathway. World J. Microbiol. Biotechnol. 2017, 33, 55. [Google Scholar] [CrossRef]
- Leonardelli, F.; Macedo, D.; Dudiuk, C.; Cabeza, M.S.; Gamarra, S.; Garcia-Effron, G. Aspergillus fumigatus Intrinsic Fluconazole Resistance Is Due to the Naturally Occurring T301I Substitution in Cyp51Ap. Antimicrob. Agents Chemother. 2016, 60, 5420–5426. [Google Scholar] [CrossRef]
- Van der Linden, J.W.M.; Snelders, E.; Kampinga, G.A.; Rijnders, B.J.A.; Mattsson, E.; Debets-Ossenkopp, Y.J.; Kuijper, E.J.; Van Tiel, F.H.; Melchers, W.J.G.; Verweij, P.E. Clinical implications of azole resistance in Aspergillus fumigatus, The Netherlands, 2007-2009. Emerg. Infect. Dis. 2011, 17, 1846–1854. [Google Scholar] [CrossRef] [PubMed]
- Camps, S.M.; Van der Linden, J.W.; Li, Y.; Kuijper, E.J.; Van Dissel, J.T.; Verweij, P.E.; Melchers, W.J. Rapid induction of multiple resistance mechanisms in Aspergillus fumigatus during azole therapy: A case study and review of the literature. Antimicrob. Agents Chemother. 2012, 56, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Snelders, E.; Camps, S.M.; Karawajczyk, A.; Schaftenaar, G.; Kema, G.H.; Van der Lee, H.A.; Klaassen, C.H.; Melchers, W.J.; Verweij, P.E. Triazole fungicides can induce cross-resistance to medical triazoles in Aspergillus fumigatus. PLoS ONE 2012, 7, e31801. [Google Scholar] [CrossRef] [PubMed]
- Snelders, E.; Camps, S.M.; Karawajczyk, A.; Rijs, A.J.; Zoll, J.; Verweij, P.E.; Melchers, W.J. Genotype-phenotype complexity of the TR46/Y121F/T289A cyp51A azole resistance mechanism in Aspergillus fumigatus. Fungal Genet. Biol. 2015, 82, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Snelders, E.; Van der Lee, H.A.; Kuijpers, J.; Rijs, A.J.; Varga, J.; Samson, R.A.; Mellado, E.; Donders, A.R.; Melchers, W.J.; Verweij, P.E. Emergence of azole resistance in Aspergillus fumigatus and spread of a single resistance mechanism. PLoS Med. 2008, 5, e219. [Google Scholar] [CrossRef] [PubMed]
- Mellado, E.; Garcia-Effron, G.; Alcazar-Fuoli, L.; Melchers, W.J.; Verweij, P.E.; Cuenca-Estrella, M.; Rodriguez-Tudela, J.L. A new Aspergillus fumigatus resistance mechanism conferring in vitro cross-resistance to azole antifungals involves a combination of cyp51A alterations. Antimicrob. Agents Chemother. 2007, 51, 1897–1904. [Google Scholar] [CrossRef]
- Buil, J.B.; Hagen, F.; Chowdhary, A.; Verweij, P.E.; Meis, J.F. Itraconazole, Voriconazole, and Posaconazole CLSI MIC Distributions for Wild-Type and Azole-Resistant Aspergillus fumigatus Isolates. J. Fungi 2018, 4, 103. [Google Scholar] [CrossRef]
- Van der Linden, J.W.; Camps, S.M.; Kampinga, G.A.; Arends, J.P.; Debets-Ossenkopp, Y.J.; Haas, P.J.; Rijnders, B.J.; Kuijper, E.J.; Van Tiel, F.H.; Varga, J.; et al. Aspergillosis due to voriconazole highly resistant Aspergillus fumigatus and recovery of genetically related resistant isolates from domiciles. Clin. Infect. Dis. 2013, 57, 513–520. [Google Scholar] [CrossRef]
- Chowdhary, A.; Kathuria, S.; Xu, J.; Sharma, C.; Sundar, G.; Singh, P.K.; Gaur, S.N.; Hagen, F.; Klaassen, C.H.; Meis, J.F. Clonal expansion and emergence of environmental multiple-triazole-resistant Aspergillus fumigatus strains carrying the TR(3)(4)/L98H mutations in the cyp51A gene in India. PLoS ONE 2012, 7, e52871. [Google Scholar] [CrossRef]
- Tsitsopoulou, A.; Posso, R.; Vale, L.; Bebb, S.; Johnson, E.; White, P.L. Determination of the Prevalence of Triazole Resistance in Environmental Aspergillus fumigatus Strains Isolated in South Wales, UK. Front. Microbiol. 2018, 9, 1395. [Google Scholar] [CrossRef]
- Chowdhary, A.; Sharma, C.; Van den Boom, M.; Yntema, J.B.; Hagen, F.; Verweij, P.E.; Meis, J.F. Multi-azole-resistant Aspergillus fumigatus in the environment in Tanzania. J. Antimicrob. Chemother. 2014, 69, 2979–2983. [Google Scholar] [CrossRef] [PubMed]
- Rocchi, S.; Poncot, M.; Morin-Crini, N.; Laboissiere, A.; Valot, B.; Godeau, C.; Lechenault-Bergerot, C.; Reboux, G.; Crini, G.; Millon, L. Determination of azole fungal residues in soils and detection of Aspergillus fumigatus-resistant strains in market gardens of Eastern France. Env. Sci. Pollut. Res. Int. 2018, 25, 32015–32023. [Google Scholar] [CrossRef]
- Riat, A.; Plojoux, J.; Gindro, K.; Schrenzel, J.; Sanglard, D. Azole Resistance of Environmental and Clinical Aspergillus fumigatus Isolates from Switzerland. Antimicrob. Agents Chemother. 2018, 62, e02088-17. [Google Scholar] [CrossRef] [PubMed]
- Bader, O.; Tunnermann, J.; Dudakova, A.; Tangwattanachuleeporn, M.; Weig, M.; Gross, U. Environmental isolates of azole-resistant Aspergillus fumigatus in Germany. Antimicrob. Agents Chemother. 2015, 59, 4356–4359. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, E.; Maertens, J.; Schoemans, H.; Lagrou, K. Azole-resistant Aspergillus fumigatus due to TR46/Y121F/T289A mutation emerging in Belgium, July 2012. Euro Surveill. 2012, 17, 20326. [Google Scholar] [PubMed]
- Chowdhary, A.; Kathuria, S.; Randhawa, H.S.; Gaur, S.N.; Klaassen, C.H.; Meis, J.F. Isolation of multiple-triazole-resistant Aspergillus fumigatus strains carrying the TR/L98H mutations in the cyp51A gene in India. J. Antimicrob. Chemother. 2012, 67, 362–366. [Google Scholar] [CrossRef]
- Chowdhary, A.; Sharma, C.; Kathuria, S.; Hagen, F.; Meis, J.F. Azole-resistant Aspergillus fumigatus with the environmental TR46/Y121F/T289A mutation in India. J. Antimicrob. Chemother. 2014, 69, 555–557. [Google Scholar] [CrossRef]
- Lockhart, S.R.; Frade, J.P.; Etienne, K.A.; Pfaller, M.A.; Diekema, D.J.; Balajee, S.A. Azole resistance in Aspergillus fumigatus isolates from the ARTEMIS global surveillance study is primarily due to the TR/L98H mutation in the cyp51A gene. Antimicrob. Agents Chemother. 2011, 55, 4465–4468. [Google Scholar] [CrossRef]
- Mortensen, K.L.; Mellado, E.; Lass-Flörl, C.; Rodriguez-Tudela, J.L.; Johansen, H.K.; Arendrup, M.C. Environmental study of azole-resistant Aspergillus fumigatus and other aspergilli in Austria, Denmark, and Spain. Antimicrob. Agents Chemother. 2010, 54, 4545–4549. [Google Scholar] [CrossRef]
- Bowyer, P.; Denning, D.W. Environmental fungicides and triazole resistance in Aspergillus. Pest. Manag Sci. 2014, 70, 173–178. [Google Scholar] [CrossRef]
- Buil, J.B.; Hare, R.K.; Zwaan, B.J.; Arendrup, M.C.; Melchers, W.J.G.; Verweij, P.E. The fading boundaries between patient and environmental routes of triazole resistance selection in Aspergillus fumigatus. PLoS Pathog 2019, 15, e1007858. [Google Scholar] [CrossRef] [PubMed]
- Bromley, M.J.; Van Muijlwijk, G.; Fraczek, M.G.; Robson, G.; Verweij, P.E.; Denning, D.W.; Bowyer, P. Occurrence of azole-resistant species of Aspergillus in the UK environment. J. Glob. Antimicrob. Resist. 2014, 2, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Verweij, P.E.; Snelders, E.; Kema, G.H.J.; Mellado, E.; Melchers, W.J.G. Azole resistance in Aspergillus fumigatus: A side-effect of environmental fungicide use? Lancet Infect. Dis. 2009, 9, 789–795. [Google Scholar] [CrossRef]
- Ren, J.; Jin, X.; Zhang, Q.; Zheng, Y.; Lin, D.; Yu, Y. Fungicides induced triazole-resistance in Aspergillus fumigatus associated with mutations of TR46/Y121F/T289A and its appearance in agricultural fields. J. Hazard. Mater. 2017, 326, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Jørgensen, L.N.; Arendrup, M.C. Azole-Resistant Invasive Aspergillosis: Relationship to Agriculture. Curr. Fungal Infect. Rep. 2012, 6, 178. [Google Scholar] [CrossRef]
- Schoustra, S.E.; Debets, A.J.M.; Rijs, A.J.M.M.; Zhang, J.; Snelders, E.; Leendertse, P.C.; Melchers, W.J.G.; Rietveld, A.G.; Zwaan, B.J.; Verweij, P.E. Environmental Hotspots for Azole Resistance Selection of Aspergillus fumigatus, the Netherlands. Emerg. Infect. Dis. 2019, 25, 1347–1353. [Google Scholar] [CrossRef]
- Moore, C.B.; Novak-Frazer, L.; Muldoon, E.; Dunn, K.W.; Masania, R.; Richardson, M.D.; Rautemaa-Richardson, R. First isolation of the pan-azole-resistant Aspergillus fumigatus cyp51A TR46/Y121F/T289A mutant in a UK patient. Int. J. Antimicrob. Agents 2017, 49, 512–514. [Google Scholar] [CrossRef]
- Da Silva Ferreira, M.E.; Capellaro, J.L.; Dos Reis Marques, E.; Malavazi, I.; Perlin, D.; Park, S.; Anderson, J.B.; Colombo, A.L.; Arthington-Skaggs, B.A.; Goldman, M.H.; et al. In vitro evolution of itraconazole resistance in Aspergillus fumigatus involves multiple mechanisms of resistance. Antimicrob. Agents Chemother. 2004, 48, 4405–4413. [Google Scholar] [CrossRef]
- Ferreira, M.E.d.S.; Colombo, A.L.; Paulsen, I.; Ren, Q.; Wortman, J.; Huang, J.; Goldman, M.H.S.; Goldman, G.H. The ergosterol biosynthesis pathway, transporter genes, and azole resistance in Aspergillus fumigatus. Med. Mycol. 2005, 43, S313–S319. [Google Scholar] [CrossRef]
- Mann, P.A.; Parmegiani, R.M.; Wei, S.Q.; Mendrick, C.A.; Li, X.; Loebenberg, D.; DiDomenico, B.; Hare, R.S.; Walker, S.S.; McNicholas, P.M. Mutations in Aspergillus fumigatus resulting in reduced susceptibility to posaconazole appear to be restricted to a single amino acid in the cytochrome P450 14alpha-demethylase. Antimicrob. Agents Chemother. 2003, 47, 577–581. [Google Scholar] [CrossRef]
- Kidd, S.E.; Goeman, E.; Meis, J.F.; Slavin, M.A.; Verweij, P.E. Multi-triazole-resistant Aspergillus fumigatus infections in Australia. Mycoses 2015, 58, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Mellado, E.; Garcia-Effron, G.; Alcazar-Fuoli, L.; Cuenca-Estrella, M.; Rodriguez-Tudela, J.L. Substitutions at methionine 220 in the 14alpha-sterol demethylase (Cyp51A) of Aspergillus fumigatus are responsible for resistance in vitro to azole antifungal drugs. Antimicrob. Agents Chemother. 2004, 48, 2747–2750. [Google Scholar] [CrossRef] [PubMed]
- Bader, O.; Weig, M.; Reichard, U.; Lugert, R.; Kuhns, M.; Christner, M.; Held, J.; Peter, S.; Schumacher, U.; Buchheidt, D.; et al. cyp51A-Based mechanisms of Aspergillus fumigatus azole drug resistance present in clinical samples from Germany. Antimicrob. Agents Chemother. 2013, 57, 3513–3517. [Google Scholar] [CrossRef] [PubMed]
- Gregson, L.; Goodwin, J.; Johnson, A.; McEntee, L.; Moore, C.B.; Richardson, M.; Hope, W.W.; Howard, S.J. In vitro susceptibility of Aspergillus fumigatus to isavuconazole: Correlation with itraconazole, voriconazole, and posaconazole. Antimicrob. Agents Chemother. 2013, 57, 5778–5780. [Google Scholar] [CrossRef]
- Krishnan Natesan, S.; Wu, W.; Cutright, J.L.; Chandrasekar, P.H. In vitro-in vivo correlation of voriconazole resistance due to G448S mutation (cyp51A gene) in Aspergillus fumigatus. Diagn. Microbiol. Infect. Dis. 2012, 74, 272–277. [Google Scholar] [CrossRef]
- Bellete, B.; Raberin, H.; Morel, J.; Flori, P.; Hafid, J.; Manhsung, R.T. Acquired resistance to voriconazole and itraconazole in a patient with pulmonary aspergilloma. Med. Mycol. 2010, 48, 197–200. [Google Scholar] [CrossRef]
- Albarrag, A.M.; Anderson, M.J.; Howard, S.J.; Robson, G.D.; Warn, P.A.; Sanglard, D.; Denning, D.W. Interrogation of related clinical pan-azole-resistant Aspergillus fumigatus strains: G138C, Y431C, and G434C single nucleotide polymorphisms in cyp51A, upregulation of cyp51A, and integration and activation of transposon Atf1 in the cyp51A promoter. Antimicrob. Agents Chemother. 2011, 55, 5113–5121. [Google Scholar] [CrossRef]
- Alanio, A.; Sitterle, E.; Liance, M.; Farrugia, C.; Foulet, F.; Botterel, F.; Hicheri, Y.; Cordonnier, C.; Costa, J.M.; Bretagne, S. Low prevalence of resistance to azoles in Aspergillus fumigatus in a French cohort of patients treated for haematological malignancies. J. Antimicrob. Chemother. 2011, 66, 371–374. [Google Scholar] [CrossRef]
- Diaz-Guerra, T.M.; Mellado, E.; Cuenca-Estrella, M.; Rodriguez-Tudela, J.L. A point mutation in the 14alpha-sterol demethylase gene cyp51A contributes to itraconazole resistance in Aspergillus fumigatus. Antimicrob. Agents Chemother. 2003, 47, 1120–1124. [Google Scholar] [CrossRef]
- Ballard, E.; Melchers, W.J.G.; Zoll, J.; Brown, A.J.P.; Verweij, P.E.; Warris, A. In-host microevolution of Aspergillus fumigatus: A phenotypic and genotypic analysis. Fungal Genet. Biol. FG B 2018, 113, 1–13. [Google Scholar] [CrossRef]
- Escribano, P.; Peláez, T.; Muñoz, P.; Bouza, E.; Guinea, J. Is azole resistance in Aspergillus fumigatus a problem in Spain? Antimicrob. Agents Chemother. 2013, 57, 2815–2820. [Google Scholar] [CrossRef] [PubMed]
- Rivero-Menendez, O.; Alastruey-Izquierdo, A.; Mellado, E.; Cuenca-Estrella, M. Triazole Resistance in Aspergillus spp.: A Worldwide Problem? J. Fungi 2016, 2, 21. [Google Scholar] [CrossRef] [PubMed]
- Hodiamont, C.J.; Dolman, K.M.; Ten Berge, I.J.; Melchers, W.J.; Verweij, P.E.; Pajkrt, D. Multiple-azole-resistant Aspergillus fumigatus osteomyelitis in a patient with chronic granulomatous disease successfully treated with long-term oral posaconazole and surgery. Med. Mycol. 2009, 47, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Perez-Cantero, A.; Lopez-Fernandez, L.; Guarro-Artigas, J.; Capilla, J. Update and recent advances on azole resistance mechanisms in Aspergillus. Int. J. Antimicrob. Agents 2019. [Google Scholar] [CrossRef] [PubMed]
- Lescar, J.; Meyer, I.; Akshita, K.; Srinivasaraghavan, K.; Verma, C.; Palous, M.; Mazier, D.; Datry, A.; Fekkar, A. Aspergillus fumigatus harbouring the sole Y121F mutation shows decreased susceptibility to voriconazole but maintained susceptibility to itraconazole and posaconazole. J. Antimicrob. Chemother. 2014, 69, 3244–3247. [Google Scholar] [CrossRef] [PubMed]
- Snelders, E.; Karawajczyk, A.; Schaftenaar, G.; Verweij, P.E.; Melchers, W.J.G. Azole resistance profile of amino acid changes in Aspergillus fumigatus CYP51A based on protein homology modeling. Antimicrob. Agents Chemother. 2010, 54, 2425–2430. [Google Scholar] [CrossRef]
- Howard, S.J.; Webster, I.; Moore, C.B.; Gardiner, R.E.; Park, S.; Perlin, D.S.; Denning, D.W. Multi-azole resistance in Aspergillus fumigatus. Int. J. Antimicrob. Agents 2006, 28, 450–453. [Google Scholar] [CrossRef]
- Liu, M.; Zeng, R.; Zhang, L.; Li, D.; Lv, G.; Shen, Y.; Zheng, H.; Zhang, Q.; Zhao, J.; Zheng, N.; et al. Multiple cyp51A-based mechanisms identified in azole-resistant isolates of Aspergillus fumigatus from China. Antimicrob. Agents Chemother. 2015, 59, 4321–4325. [Google Scholar] [CrossRef]
- Snelders, E.; Huis In t Veld, R.A.G.; Rijs, A.J.M.M.; Kema, G.H.J.; Melchers, W.J.G.; Verweij, P.E. Possible environmental origin of resistance of Aspergillus fumigatus to medical triazoles. Appl. Env. Microbiol. 2009, 75, 4053–4057. [Google Scholar] [CrossRef]
- Hare, R.K.; Gertsen, J.B.; Astvad, K.M.T.; Degn, K.B.; Løkke, A.; Stegger, M.; Andersen, P.S.; Kristensen, L.; Arendrup, M.C. In Vivo Selection of a Unique Tandem Repeat Mediated Azole Resistance Mechanism (TR(120)) in Aspergillus fumigatus cyp51A, Denmark. Emerg. Infect. Dis. 2019, 25, 577–580. [Google Scholar] [CrossRef]
- Jenks, J.D.; Spiess, B.; Buchheidt, D.; Hoenigl, M. (New) Methods for Detection of Aspergillus fumigatus Resistance in Clinical Samples. Curr. Fungal Infect. Rep. 2019, 13, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Buil, J.B.; Zoll, J.; Verweij, P.E.; Melchers, W.J.G. Molecular Detection of Azole-Resistant Aspergillus fumigatus in Clinical Samples. Front. Microbiol. 2018, 9, 515. [Google Scholar] [CrossRef] [PubMed]
- Dudakova, A.; Spiess, B.; Tangwattanachuleeporn, M.; Sasse, C.; Buchheidt, D.; Weig, M.; Groß, U.; Bader, O. Molecular Tools for the Detection and Deduction of Azole Antifungal Drug Resistance Phenotypes in Aspergillus Species. Clin. Microbiol. Rev. 2017, 30, 1065–1091. [Google Scholar] [CrossRef] [PubMed]
- Spiess, B.; Seifarth, W.; Merker, N.; Howard, S.J.; Reinwald, M.; Dietz, A.; Hofmann, W.K.; Buchheidt, D. Development of novel PCR assays to detect azole resistance-mediating mutations of the Aspergillus fumigatus cyp51A gene in primary clinical samples from neutropenic patients. Antimicrob. Agents Chemother. 2012, 56, 3905–3910. [Google Scholar] [CrossRef] [PubMed]
- Bernal-Martínez, L.; Gago, S.; Buitrago, M.J.; Gomez-Lopez, A.; Rodríguez-Tudela, J.L.; Cuenca-Estrella, M. Analysis of performance of a PCR-based assay to detect DNA of Aspergillus fumigatus in whole blood and serum: A comparative study with clinical samples. J. Clin. Microbiol. 2011, 49, 3596–3599. [Google Scholar] [CrossRef]
- Cuenca-Estrella, M.; Meije, Y.; Diaz-Pedroche, C.; Gomez-Lopez, A.; Buitrago, M.J.; Bernal-Martinez, L.; Grande, C.; Juan, R.S.; Lizasoain, M.; Rodriguez-Tudela, J.L.; et al. Value of serial quantification of fungal DNA by a real-time PCR-based technique for early diagnosis of invasive Aspergillosis in patients with febrile neutropenia. J. Clin. Microbiol. 2009, 47, 379–384. [Google Scholar] [CrossRef][Green Version]
- Postina, P.; Skladny, J.; Boch, T.; Cornely, O.A.; Hamprecht, A.; Rath, P.M.; Steinmann, J.; Bader, O.; Miethke, T.; Dietz, A.; et al. Comparison of Two Molecular Assays for Detection and Characterization of Aspergillus fumigatus Triazole Resistance and Cyp51A Mutations in Clinical Isolates and Primary Clinical Samples of Immunocompromised Patients. Front. Microbiol. 2018, 9, 555. [Google Scholar] [CrossRef]
- Zhao, Y.; Garnaud, C.; Brenier-Pinchart, M.-P.; Thiébaut-Bertrand, A.; Saint-Raymond, C.; Camara, B.; Hamidfar, R.; Cognet, O.; Maubon, D.; Cornet, M.; et al. Direct Molecular Diagnosis of Aspergillosis and CYP51A Profiling from Respiratory Samples of French Patients. Front. Microbiol. 2016, 7, 1164. [Google Scholar] [CrossRef]
- Bernal-Martinez, L.; Gil, H.; Rivero-Menendez, O.; Gago, S.; Cuenca-Estrella, M.; Mellado, E.; Alastruey-Izquierdo, A. Development and Validation of a High-Resolution Melting Assay To Detect Azole Resistance in Aspergillus fumigatus. Antimicrob. Agents Chemother. 2017, 61, e01083-17. [Google Scholar] [CrossRef]
- Tuohy, M.J.; Reja, V.; Park, S.; Perlin, D.S.; Wnek, M.; Procop, G.W.; Yen-Lieberman, B. Use of a high-resolution melt assay to characterize codon 54 of the cyp51A gene of Aspergillus fumigatus on a Rotor-Gene 6000 instrument. Antimicrob. Agents Chemother. 2010, 54, 2248–2251. [Google Scholar] [CrossRef]
- Alanio, A.; Bretagne, S. Performance evaluation of multiplex PCR including Aspergillus-not so simple! Med. Mycol. 2017, 55, 56–62. [Google Scholar] [CrossRef]
- Schauwvlieghe, A.F.A.D.; Vonk, A.G.; Buddingh, E.P.; Hoek, R.A.S.; Dalm, V.A.; Klaassen, C.H.W.; Rijnders, B.J.A. Detection of azole-susceptible and azole-resistant Aspergillus coinfection by cyp51A PCR amplicon melting curve analysis. J. Antimicrob. Chemother. 2017, 72, 3047–3050. [Google Scholar] [CrossRef][Green Version]
- White, P.L.; Posso, R.B.; Barnes, R.A. Analytical and Clinical Evaluation of the PathoNostics AsperGenius Assay for Detection of Invasive Aspergillosis and Resistance to Azole Antifungal Drugs Directly from Plasma Samples. J. Clin. Microbiol. 2017, 55, 2356–2366. [Google Scholar] [CrossRef]
- Chong, G.L.; Van de Sande, W.W.; Dingemans, G.J.; Gaajetaan, G.R.; Vonk, A.G.; Hayette, M.P.; Van Tegelen, D.W.; Simons, G.F.; Rijnders, B.J. Validation of a new Aspergillus real-time PCR assay for direct detection of Aspergillus and azole resistance of Aspergillus fumigatus on bronchoalveolar lavage fluid. J. Clin. Microbiol. 2015, 53, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Montesinos, I.; Argudin, M.A.; Hites, M.; Ahajjam, F.; Dodemont, M.; Dagyaran, C.; Bakkali, M.; Etienne, I.; Jacobs, F.; Knoop, C.; et al. Culture-Based Methods and Molecular Tools for Azole-Resistant Aspergillus fumigatus Detection in a Belgian University Hospital. J. Clin. Microbiol. 2017, 55, 2391–2399. [Google Scholar] [CrossRef] [PubMed]
- Dannaoui, E.; Gabriel, F.; Gaboyard, M.; Lagardere, G.; Audebert, L.; Quesne, G.; Godichaud, S.; Verweij, P.E.; Accoceberry, I.; Bougnoux, M.E. Molecular Diagnosis of Invasive Aspergillosis and Detection of Azole Resistance by a Newly Commercialized PCR Kit. J. Clin. Microbiol. 2017, 55, 3210–3218. [Google Scholar] [CrossRef] [PubMed]
- Mikulska, M.; Furfaro, E.; De Carolis, E.; Drago, E.; Pulzato, I.; Borghesi, M.L.; Zappulo, E.; Raiola, A.M.; Grazia, C.D.; Del Bono, V.; et al. Use of Aspergillus fumigatus real-time PCR in bronchoalveolar lavage samples (BAL) for diagnosis of invasive aspergillosis, including azole-resistant cases, in high risk haematology patients: The need for a combined use with galactomannan. Med. Mycol. 2019, 57, 987–996. [Google Scholar] [CrossRef]
- Balashov, S.V.; Gardiner, R.; Park, S.; Perlin, D.S. Rapid, high-throughput, multiplex, real-time PCR for identification of mutations in the cyp51A gene of Aspergillus fumigatus that confer resistance to itraconazole. J. Clin. Microbiol. 2005, 43, 214–222. [Google Scholar] [CrossRef]
- Garcia-Effron, G.; Dilger, A.; Alcazar-Fuoli, L.; Park, S.; Mellado, E.; Perlin, D.S. Rapid detection of triazole antifungal resistance in Aspergillus fumigatus. J. Clin. Microbiol. 2008, 46, 1200–1206. [Google Scholar] [CrossRef]
- Zhao, Y.; Stensvold, C.R.; Perlin, D.S.; Arendrup, M.C. Azole resistance in Aspergillus fumigatus from bronchoalveolar lavage fluid samples of patients with chronic diseases. J. Antimicrob. Chemother. 2013, 68, 1497–1504. [Google Scholar] [CrossRef]
- Trama, J.P.; Mordechai, E.; Adelson, M.E. Detection of Aspergillus fumigatus and a mutation that confers reduced susceptibility to itraconazole and posaconazole by real-time PCR and pyrosequencing. J. Clin. Microbiol. 2005, 43, 906–908. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Novak-Frazer, L.H.D.; Hill, S.; Masania, R.; Denning, D.W.; Moore, C.; Rautemaa-Richardson, R.; Richardson, M. Profiling Aspergillus Fumigatus Cyp51a Polymorphisms by Pyrosequencing Reveals Triazole Resistance When Susceptibility Testing is Not Possible; ECCMID: Madrid, Spain, 2018. [Google Scholar]
- Brown, L.; Rautemaa-Richardson, R.; Novak-Frazer, L.; Hill, S.; Hassan, D.; Richardson, M.D.; Gangneux, J.-P.; Lortholary, O.; Cornely, O.A.; Pagano, L. Identification of Cyp51A-mediated triazole resistance in Aspergillus fumigatus using pyrosequencing: An audit of UK National Aspergillosis Centre cases. In Proceedings of the 9th Trends in Medical Mycology, Nice, France, 11–14 October 2019. [Google Scholar]
- Florea, A. Pyrosequencing and Its Application in Epigenetic Clinical Diagnostics. Epigenetic Biomarkers and Diagnostics; Academic Press: Cambridge, UK, 2016. [Google Scholar]
- Wiederhold, N.P.; Grabinski, J.L.; Garcia-Effron, G.; Perlin, D.S.; Lee, S.A. Pyrosequencing to detect mutations in FKS1 that confer reduced echinocandin susceptibility in Candida albicans. Antimicrob. Agents Chemother. 2008, 52, 4145–4148. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Royo, J.L.; Galan, J.J. Pyrosequencing for SNP genotyping. Methods Mol. Biol. 2009, 578, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Ronaghi, M. Pyrosequencing sheds light on DNA sequencing. Genome Res. 2001, 11, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Qiagen. PyroMark® Q24 Advanced and PyroMark Q24 Advanced CpG Reagents Handbook. Available online: https://www.qiagen.com/us/products/instruments-and-automation/pyrosequencing/consumables/pyromark-q24-advanced-accessories-and-reagents/#orderinginformation (accessed on 9 December 2019).
- Camps, S.M.T.; Dutilh, B.E.; Arendrup, M.C.; Rijs, A.J.M.M.; Snelders, E.; Huynen, M.A.; Verweij, P.E.; Melchers, W.J.G. Discovery of a HapE mutation that causes azole resistance in Aspergillus fumigatus through whole genome sequencing and sexual crossing. PLoS ONE 2012, 7, e50034. [Google Scholar] [CrossRef] [PubMed]
- Ballard, E.; Weber, J.; Melchers, W.J.G.; Tammireddy, S.; Whitfield, P.D.; Brakhage, A.A.; Brown, A.J.P.; Verweij, P.E.; Warris, A. Recreation of in-host acquired single nucleotide polymorphisms by CRISPR-Cas9 reveals an uncharacterised gene playing a role in Aspergillus fumigatus azole resistance via a non-cyp51A mediated resistance mechanism. Fungal Genet. Biol. FG B 2019, 130, 98–106. [Google Scholar] [CrossRef]
- Hortschansky, P.; Haas, H.; Huber, E.M.; Groll, M.; Brakhage, A.A. The CCAAT-binding complex (CBC) in Aspergillus species. Biochim. Biophys. Acta Gene Regul. Mech. 2017, 1860, 560–570. [Google Scholar] [CrossRef]
- Wei, X.; Chen, P.; Gao, R.; Li, Y.; Zhang, A.; Liu, F.; Lu, L. Screening and Characterization of a Non-cyp51A Mutation in an Aspergillus fumigatus cox10 Strain Conferring Azole Resistance. Antimicrob. Agents Chemother. 2016, 61, e02101–e02116. [Google Scholar] [CrossRef]
- Fraczek, M.G.; Bromley, M.; Buied, A.; Moore, C.B.; Rajendran, R.; Rautemaa, R.; Ramage, G.; Denning, D.W.; Bowyer, P. The cdr1B efflux transporter is associated with non-cyp51a-mediated itraconazole resistance in Aspergillus fumigatus. J. Antimicrob. Chemother. 2013, 68, 1486–1496. [Google Scholar] [CrossRef]
- Meneau, I.; Coste, A.T.; Sanglard, D. Identification of Aspergillus fumigatus multidrug transporter genes and their potential involvement in antifungal resistance. Med. Mycol. 2016, 54, 616–627. [Google Scholar] [CrossRef]
- Chen, P.; Liu, J.; Zeng, M.; Sang, H. Exploring the molecular mechanism of azole resistance in Aspergillus fumigatus. J. Mycol. Med. 2019. [Google Scholar] [CrossRef]
- Yang, B.; Damaschke, N.; Yao, T.; McCormick, J.; Wagner, J.; Jarrard, D. Pyrosequencing for accurate imprinted allele expression analysis. J. Cell. Biochem. 2015, 116, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Castillo, F.Y.; Loera-Muro, A.; Jacques, M.; Garneau, P.; Avelar-González, F.J.; Harel, J.; Guerrero-Barrera, A.L. Waterborne pathogens: Detection methods and challenges. Pathogens 2015, 4, 307. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, J.F., Jr.; Fouad, A.F.; Rôças, I.N. Pyrosequencing as a tool for better understanding of human microbiomes. J. Oral Microbiol. 2012, 4. [Google Scholar] [CrossRef]
- Chikamatsu, K.; Aono, A.; Hata, H.; Igarashi, Y.; Takaki, A.; Yamada, H.; Sakashita, K.; Mitarai, S. Evaluation of PyroMark Q24 pyrosequencing as a method for the identification of mycobacteria. Diagn. Microbiol. Infect. Dis. 2018, 90, 35–39. [Google Scholar] [CrossRef]
- Lau, H.; Deng, Y.-M.; Xu, X.; Sessions, W.; Barr, I.G. Rapid detection of new B/Victoria-lineage haemagglutinin variants of influenza B viruses by pyrosequencing. Diagn. Microbiol. Infect. Dis. 2019, 93, 311–317. [Google Scholar] [CrossRef]
- Kumar, D.; Mehta, A.; Panigrahi, M.K.; Nath, S.; Saikia, K.K. NPM1 Mutation Analysis in Acute Myeloid Leukemia: Comparison of Three Techniques-Sanger Sequencing, Pyrosequencing, and Real-Time Polymerase Chain Reaction. Turk. J. Haematol. 2018, 35, 49–53. [Google Scholar] [CrossRef]
- Chronic Pulmonary Aspergillus Annual Report 2018/2019. Available online: https://aspergillosis.org/nac-reports/ (accessed on 13 December 2019).
- Chowdhary, A.; Kathuria, S.; Xu, J.; Meis, J.F. Emergence of azole-resistant aspergillus fumigatus strains due to agricultural azole use creates an increasing threat to human health. PLoS Pathog. 2013, 9, e1003633. [Google Scholar] [CrossRef]
- Hagiwara, D.; Watanabe, A.; Kamei, K.; Goldman, G.H. Epidemiological and Genomic Landscape of Azole Resistance Mechanisms in Aspergillus Fungi. Front. Microbiol. 2016, 7, 1382. [Google Scholar] [CrossRef]
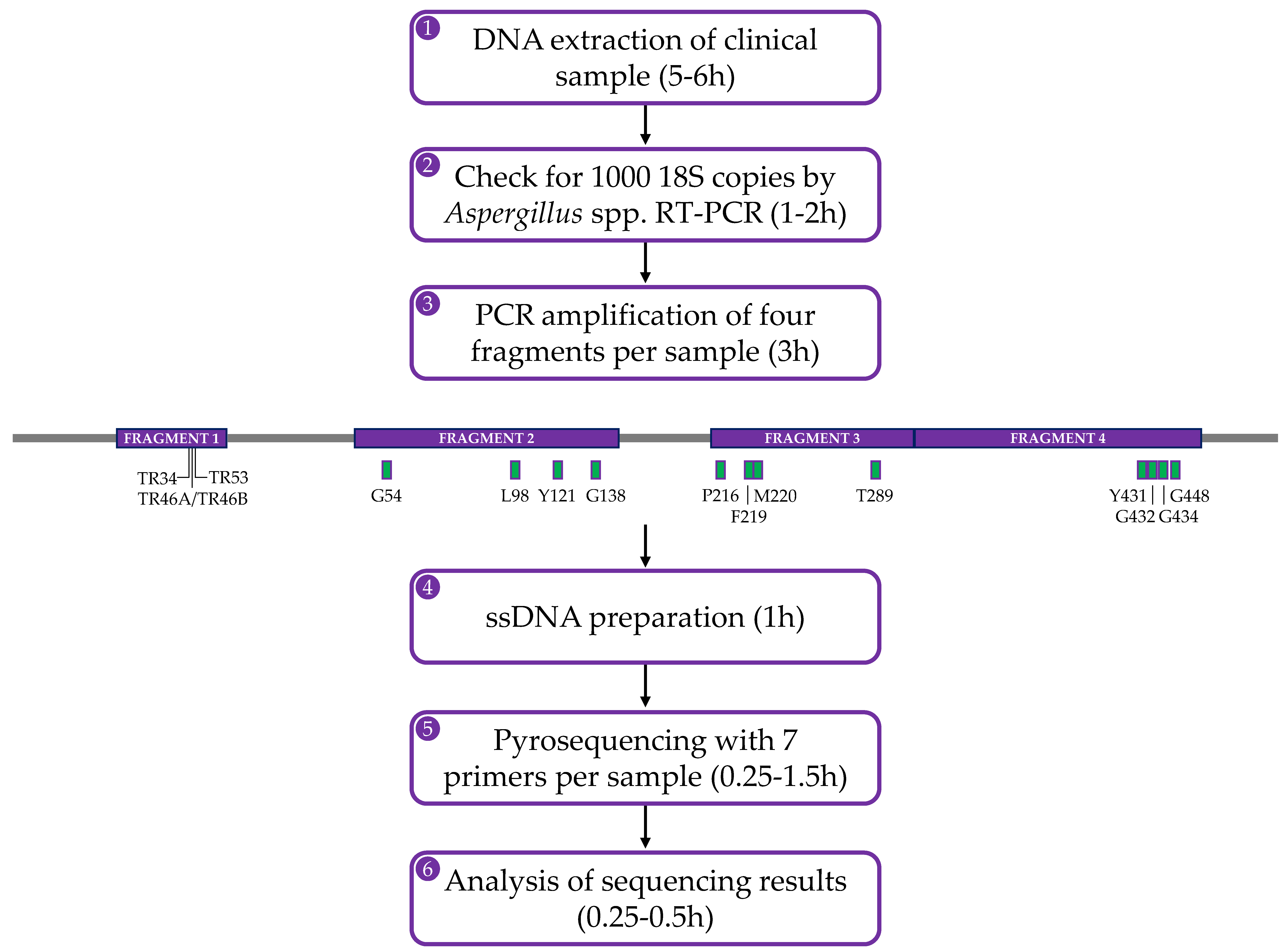
| cyp51A Polymorphism | Reported Azole Resistance | Comment | Reference(s) |
|---|---|---|---|
| TR34/L98H | pan-azole | Most common resistance mechanism. Only displays azole-resistant phenotype in combination with L98H substitution. | [27,28,57,58,59,61,63] |
| TR46/Y121F/T289A | voriconazole, posaconazole | Variable susceptibility to itraconazole. The single point mutation T289A does not result in azole resistance, whereas the single mutation TR46 has a slight reduced pan-azole susceptibility. | [56,59,60,67,69,79] |
| TR53 | itraconazole, voriconazole | Not yet reported in combination with single point mutations | [95] |
| TR120/F46Y/M172V/E427K | pan-azole | A clinical case of infection with azole-resistance acquired during long-term azole treatment | [102] |
| N22 | itraconazole | Voriconazole and posaconazole susceptibilities not reported | [80,81] |
| G54 | itraconazole, posaconazole | - | [27,54,80,81,82,83,91,92] |
| Y121 | voriconazole | - | [56,97] |
| G138 | pan-azole | - | [27,86,89,99] |
| Q141 | pan-azole | Reported in combination with TR34/L98H | [98,101] |
| H147 | pan-azole | Reported in combination with G448 | [27] |
| P216 | itraconazole, posaconazole | - | [27,54,92] |
| F219 | itraconazole, posaconazole | - | [54,85] |
| M220 | itraconazole, posaconazole | Variable susceptibility to voriconazole | [26,27,80,81,84,92] |
| M236 | itraconazole | - | [81] |
| A284 | pan-azole | The single point mutation results in reduced pan-azole susceptibility. Pan-azole resistance is reported in combination with TR34/L98H. | [26,98] |
| S297 | pan-azole | Reported only in combination with TR34/L98H | [57,58,100] |
| P394 | itraconazole | Voriconazole and posaconazole susceptibilities not reported | [80] |
| Y431 | pan-azole | - | [27,83,86,89] |
| G432 | itraconazole, voriconazole | - | [90,100] |
| G434 | pan-azole | - | [27,86,89] |
| T440 | itraconazole | Voriconazole and posaconazole susceptibilities not reported | [80,81] |
| G448 | itraconazole | Variable susceptibility to voriconazole and posiconazole | [27,83,86,87,88,93] |
| Y491 | itraconazole | Voriconazole and posaconazole susceptibilities not reported | [80,81] |
| F495 | pan-azole | Reported only in combination with TR34/L98H | [58,100] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van der Torre, M.H.; Novak-Frazer, L.; Rautemaa-Richardson, R. Detecting Azole-Antifungal Resistance in Aspergillus fumigatus by Pyrosequencing. J. Fungi 2020, 6, 12. https://doi.org/10.3390/jof6010012
van der Torre MH, Novak-Frazer L, Rautemaa-Richardson R. Detecting Azole-Antifungal Resistance in Aspergillus fumigatus by Pyrosequencing. Journal of Fungi. 2020; 6(1):12. https://doi.org/10.3390/jof6010012
Chicago/Turabian Stylevan der Torre, Mireille H., Lilyann Novak-Frazer, and Riina Rautemaa-Richardson. 2020. "Detecting Azole-Antifungal Resistance in Aspergillus fumigatus by Pyrosequencing" Journal of Fungi 6, no. 1: 12. https://doi.org/10.3390/jof6010012
APA Stylevan der Torre, M. H., Novak-Frazer, L., & Rautemaa-Richardson, R. (2020). Detecting Azole-Antifungal Resistance in Aspergillus fumigatus by Pyrosequencing. Journal of Fungi, 6(1), 12. https://doi.org/10.3390/jof6010012







