Experimental In Vivo Models of Candidiasis
Abstract
1. Introduction
2. The Mouse Model
2.1. Induction of Infection
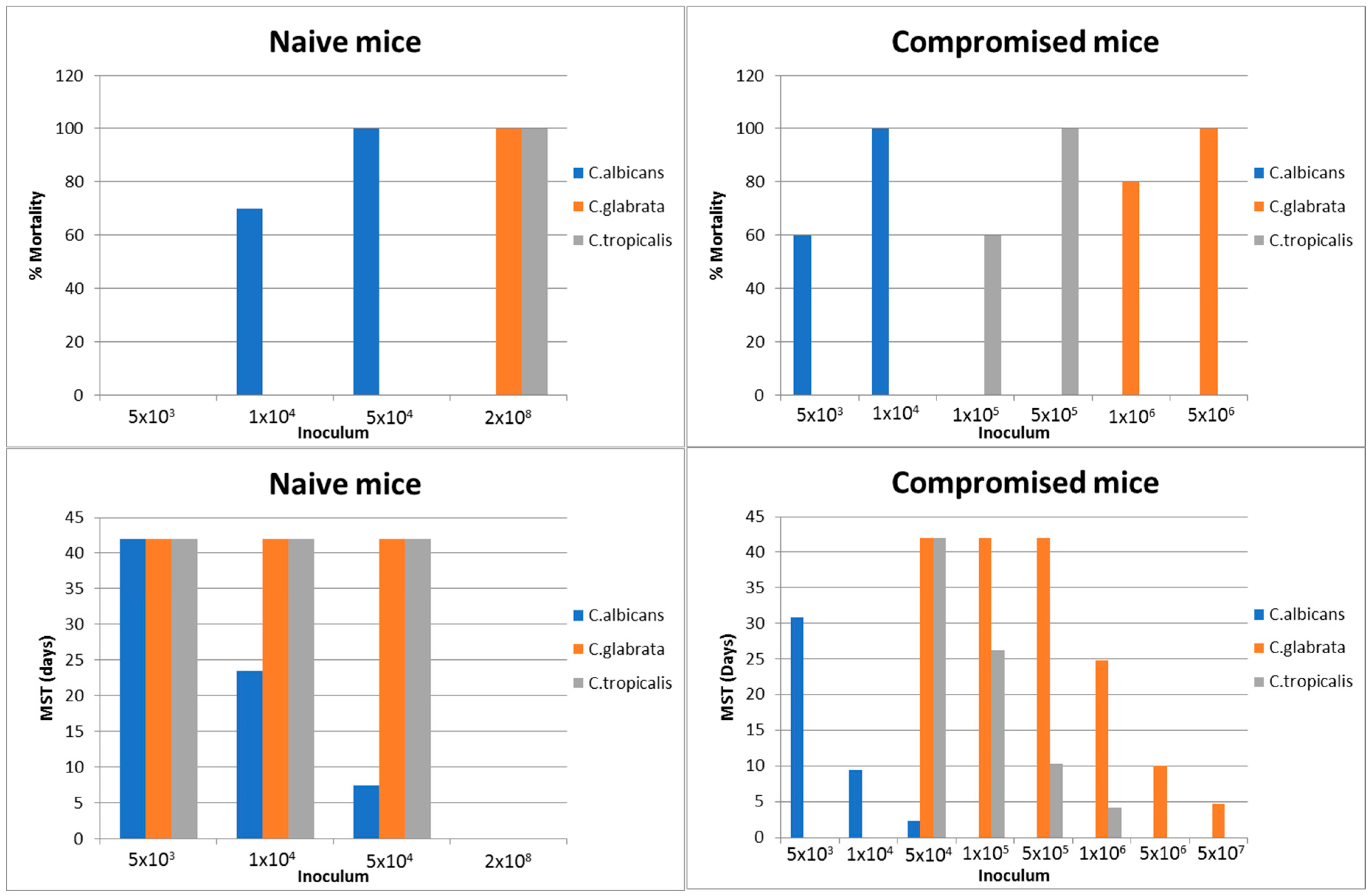
2.1.1. Systemic Infection
2.1.2. Gastrointestinal (GI) Infection
2.1.3. Vaginal Infection
3. The Drosophila melanogaster Model
4. The Caenorhabditis elegans Model
5. The Galleria mellonella Model
6. Summary
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Edwards, J.E. Candida species. In Principles and Practice of Infectious Diseases, 8th ed.; Bennett, J.E., Dolin, R., Blaser, M.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 2879–2894. [Google Scholar]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive candidiasis: A persistent public health problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Global Action Fund for Fungal Infections. Available online: https://www.gaffi.org.
- Sobel, J.D. Vulvovaginal candidosis. Lancet 2007, 369, 1961–1971. [Google Scholar] [CrossRef]
- Romani, L. Animal Models for Candidiasis. In Current Protocols in Immunology; Unit 19.6; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1999. [Google Scholar]
- Chamilos, G.; Lionakis, M.S.; Lewis, R.E.; Lopez-Ribot, J.L.; Saville, S.P.; Albert, N.D.; Halder, G.; Kontoyiannis, D.P. Drosophila melanogaster as a facile model for large-scale studies of virulence mechanisms and antifungal drug efficacy in Candida species. J. Infect. Dis. 2006, 193, 1014–1022. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, B.B.; O’Brien, E.; El Khoury, J.B.; Mylonakis, E. Methods for using Galleria mellonella as a model host to study fungal pathogenesis. Virulence 2010, 1, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Brenner, S. In the Beginning Was the Worm. Genetics 2009, 182, 413–415. [Google Scholar] [CrossRef] [PubMed]
- Joly, V.; Yeni, P. Rodent models of Candida sepsis. In Handbook of Animal Models of Infection; Zak, O., Sande, M., Eds.; Academic Press: Cambridge, MA, USA, 1999; pp. 650–657. [Google Scholar]
- Inbred Strains of Mice: BALB. Available online: http://www.informatics.jax.org/inbred_strains/mouse/docs/BALB.shtml (accessed on 1 December 2017).
- Aurora’s Guide to Mouse Colony Management at MIT. Available online: https://ki.mit.edu/files/ki/cfile/sbc/escell/mouseManagement.pdf (accessed on 1 December 2017).
- Semis, R.; Mendlovic, S.; Polacheck, I.; Segal, E. Activity of an Intralipid formulation of nystatin in murine systemic candidiasis. Int. J. Antimicrob. Agents 2011, 38, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Segal, E.; Josef-Lev, A. Induction of Candidal vaginitis in diabetic mice and attempts to prevent the infection. J. Med. Vet. Mycol. 1995, 33, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Segal, E.; Baranetz, T.; Sandovsky-Losica, H.; Gov, Y.; Teicher, S.; Dayan, D. Experimental oral murine candidiasis and attempts of prevention. J. Med. Mycol. 1999, 9, 55–59. [Google Scholar]
- Frenkel, M.; Mandelltat, M.; Alastruey-Izquierdo, A.; Mendlovic, S.; Semis, R.; Segal, E. Pathogenicity of Candida albicans isolates from bloodstream and mucosal candidiasis assessed in mice and Galleria mellonella. J. Mycol. Med. 2016, 26, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sandovsky losica, H.; Barrnea, L.; Segal, E. Fatal Systemic Candidiasis of Gastrointestinal Origin-an Experimental-Model in Mice Compromised by Anticancer Treatment. J. Med. Vet. Mycol. 1992, 30, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Segal, E.; Gottfried, L.; Lehrer, N. Candidal Vaginitis in Hormone-Treated Mice—Prevention by a Chitin Extract. Mycopathologia 1988, 102, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Lionakis, M.S.; Lim, J.K.; Lee, C.C.; Murphy, P.M. Organ-specific innate immune responses in a mouse model of invasive candidiasis. J. Innate Immunity 2011, 3, 180–199. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.A.; Radwan, R.R.; Raafat, A.I.; Ali, A.E. Antifungal activity of oral (Tragacanth/acrylic acid) Amphotericin B carrier for systemic candidiasis: In vitro and in vivo study. Drug Deliv. Transl. Res. 2018, 8, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.; Segal, E.; Eylan, E. Protective Immunity against Murine Candidiasis Elicited by Candida-albicans Ribosomal Fractions. Infect. Immunity 1981, 31, 874–878. [Google Scholar]
- Schulz, B.; Weber, K.; Schmidt, A.; Borg-von Zepelin, M.; Ruhnke, M. Difference in virulence between fluconazole-susceptible and fluconazole-resistant Candida albicans in a mouse model. Mycoses 2011, 54, E522–E530. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, M.; Capilla, J.; Castanheira, M.; Martin-Vicente, A.; Sutton, D.A.; Fothergill, A.W.; Wiederhold, N.P.; Guarro, J. Voriconazole minimum inhibitory concentrations are predictive of treatment outcome in experimental murine infections by Candida glabrata. Int. J. Antimicrob. Agents 2016, 47, 286–288. [Google Scholar] [CrossRef] [PubMed]
- Semis, R.; Nili, S.S.; Munitz, A.; Zaslavsky, Z.; Polacheck, I.; Segal, E. Pharmacokinetics, tissue distribution and immunomodulatory effect of intralipid formulation of nystatin in mice. J. Antimicrob. Chemother. 2012, 67, 1716–1721. [Google Scholar] [CrossRef] [PubMed]
- Segal, E.; Elad, D. Fungal vaccines and immunotherapy. J. Mycol. Med. 2006, 16, 134–151. [Google Scholar] [CrossRef]
- Segal, E. Testing Antifungal Vaccines in an Animal Model of Invasive Candidiasis and in Human Mucosal Candidiasis. Methods Mol. Biol. 2017, 1625, 343–353. [Google Scholar] [PubMed]
- Ahmad, E.; Zia, Q.; Fatima, M.T.; Owais, M.; Saleemuddin, M. Vaccine potential of plasma bead-based dual antigen delivery system against experimental murine candidiasis. Int. J. Biol. Macromol. 2015, 81, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Saville, S.P.; Lazzell, A.L.; Chaturvedi, A.K.; Monteagudo, C.; Lopez-Ribot, J.L. Efficacy of a Genetically Engineered Candida albicans tet-NRG1 Strain as an Experimental Live Attenuated Vaccine against Hematogenously Disseminated Candidiasis. Clin. Vaccine Immunol. 2009, 16, 430–432. [Google Scholar] [CrossRef] [PubMed]
- Segal, E. Inhibition of Candida adhesion to prevent candidiasis. In Toward Anti Adhesion Therapy for Microbial Diseases of Advances in Experimental Medicine and Biology; Kahane, I., Ofek, I., Eds.; Plenum Press: New York, NY, USA; London, UK, 1996; Volume 408, pp. 197–206. [Google Scholar]
- Shadkchan, Y.; Segal, E. Antifungal activity of amphotericin B-lipid admixtures in experimental systemic candidosis in naive mice. J. Antimicrob. Chemother. 1999, 44, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Shadkchan, Y.; Segal, E. Treatment of experimental candidosis with amphotericin B-Intralipid admixtures in immunocompromised mice. J. Antimicrob. Chemother. 2001, 48, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Taschdjian, C.L.; Reiss, F.; Kozin, P.J. Experimental vaginal candidiasis in mice; its implications for superficial candidiasis in humans. J. Investig. Dermatol. 1960, 34, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Peer, A.K.; Hoosen, A.A.; Seedat, M.A.; van den Ende, J.; Omar, M.A. Vaginal Yeast Infections in Diabetic Women. S. Afr. Med. J. 1993, 83, 727–729. [Google Scholar] [PubMed]
- Glittenberg, M.T.; Silas, S.; MacCallum, D.M.; Gow, N.A.; Ligoxygakis, P. Wild-type Drosophila melanogaster as an alternative model system for investigating the pathogenicity of Candida albicans. Dis. Model. Mech. 2011, 4, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Alarco, A.M.; Marcil, A.; Chen, J.; Suter, B.; Thomas, D.; Whiteway, M. Immune-deficient Drosophila melanogaster: A model for the innate immune response to human fungal pathogens. J. Immunol. 2004, 172, 5622–5628. [Google Scholar] [CrossRef] [PubMed]
- San-Blas, G.; Travassos, L.R.; Fries, B.C.; Goldman, D.L.; Casadevall, A.; Carmona, A.K.; Barros, T.F.; Puccia, R.; Hostetter, M.K.; Shanks, S.G.; et al. Fungal morphogenesis and virulence. Med. Mycol. 2000, 38, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Camejo, L.A.; Maldonado-Morales, G.; Bayman, P. Differential Microbial Diversity in Drosophila melanogaster: Are Fruit Flies Potential Vectors of Opportunistic Pathogens? Int. J. Microbiol. 2017, 2017, 8526385. [Google Scholar] [CrossRef] [PubMed]
- Sulston, J.E.; Brenner, S. The DNA of Caenorhabditis elegans. Genetics 1974, 77, 95–104. [Google Scholar] [PubMed]
- Ewbank, J.J.; Pujol, N. Local and long-range activation of innate immunity by infection and damage in C. elegans. Curr. Opin. Immunol. 2016, 38, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ewbank, J.J.; Zugasti, O. C. elegans: Model host and tool for antimicrobial drug discovery. Dis. Model. Mech. 2011, 4, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Pukkila-Worley, R.; Feinbaum, R.L.; McEwan, D.L.; Conery, A.L.; Ausubel, F.M. The evolutionarily conserved mediator subunit MDT-15/MED15 links protective innate immune responses and xenobiotic detoxification. PLoS Pathog. 2014, 10, e1004143. [Google Scholar] [CrossRef] [PubMed]
- Scorzoni, L.; de Lucas, M.P.; Mesa-Arango, A.C.; Fusco-Almeida, A.M.; Lozano, E.; Cuenca-Estrella, M.; Mendes-Giannini, M.J.; Zaragoza, O. Antifungal efficacy during Candida krusei infection in non-conventional models correlates with the yeast in vitro susceptibility profile. PLoS ONE 2013, 8, e60047. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, M.R.B.; Tonk, M.; Schreiber, C.; Salzig, D.; Czermak, P.; Vilcinskas, A.; Rahnamaeian, M. The potential of the Galleria mellonella innate immune system is maximized by the co-presentation of diverse antimicrobial peptides. Biol. Chem. 2016, 397, 939–945. [Google Scholar]
- Ramarao, N.; Nielsen-Leroux, C.; Lereclus, D. The Insect Galleria mellonella as a Powerful Infection Model to Investigate Bacterial Pathogenesis. J. Vis. Exp. 2012, e4392. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.J.; Muhammed, M.; Kasperkovitz, P.V.; Vyas, J.M.; Mylonakis, E. Fusarium pathogenesis investigated using Galleria mellonella as a heterologous host. Fungal Biol. 2011, 115, 1279–1289. [Google Scholar] [CrossRef] [PubMed]
- Slater, J.L.; Gregson, L.; Denning, D.W.; Warn, P.A. Pathogenicity of Aspergillus fumigatus mutants assessed in Galleria mellonella matches that in mice. Med. Mycol. 2011, 49, S107–S113. [Google Scholar] [CrossRef] [PubMed]
- Amorim-Vaz, S.; Delarze, E.; Ischer, F.; Sanglard, D.; Coste, A.T. Examining the virulence of Candida albicans transcription factor mutants using Galleria mellonella and mouse infection models. Front. Microbiol. 2015, 6, 367. [Google Scholar] [CrossRef] [PubMed]
- Hirakawa, M.P.; Martinez, D.A.; Sakthikumar, S.; Anderson, M.Z.; Berlin, A.; Gujja, S.; Zeng, Q.D.; Zisson, E.; Wang, J.M.; Greenberg, J.M.; et al. Genetic and phenotypic intra-species variation in Candida albicans. Genome Res. 2015, 25, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.; Thomas, D.Y.; Whiteway, M.; Kavanagh, K. Correlation between virulence of Candida albicans mutants in mice and Galleria mellonella larvae. FEMS Immunol. Med. Microbiol. 2002, 34, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Ames, L.; Duxbury, S.; Pawlowska, B.; Ho, H.L.; Haynes, K.; Bates, S. Galleria mellonella as a host model to study Candida glabrata virulence and antifungal efficacy. Virulence 2017, 8, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Astvad, K.M.T.; Meletiadis, J.; Whalley, S.; Arendrup, M.C. Fluconazole Pharmacokinetics in Galleria mellonella Larvae and Performance Evaluation of a Bioassay Compared to Liquid Chromatography-Tandem Mass Spectrometry for Hemolymph Specimens. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Mesa-Arango, A.C.; Forastiero, A.; Bernal-Martinez, L.; Cuenca-Estrella, M.; Mellado, E.; Zaragoza, O. The non-mammalian host Galleria mellonella can be used to study the virulence of the fungal pathogen Candida tropicalis and the efficacy of antifungal drugs during infection by this pathogenic yeast. Med. Mycol. 2013, 51, 461–472. [Google Scholar] [CrossRef] [PubMed]
- De Verno, P.J.; Aston, W.P.; Chadwick, J.S. Transfer of Immunity against Pseudomonas-aeruginosa P11-1 in Galleria-mellonella Larvae. Dev. Comp. Immunol. 1983, 7, 423–434. [Google Scholar] [CrossRef]
| Days After Inoculation | Diabetic Mice with Estrus (% Infected) | Diabetic Mice Without Estrus (% Infected) | Naïve Mice |
|---|---|---|---|
| 1 | 100 | 92 | 70 |
| 3 | 100 | 92 | 20 |
| 5 | 83 | 58 | 10 |
| 7 | 83 | 55 | 0 |
| 14 | 67 | 55 | 0 |
| 21 | 50 | 55 | 0 |
| 28 | 50 | 55 | 0 |
| 35 | 33 | 55 | 0 |
| Mouse Model | Galleria Model | |||
|---|---|---|---|---|
| Strain | MST (Days) | SD | MST (Days) | SD |
| S2 | 28.27 | 4.9 | 2.75 | 1.95 |
| S11 | 27.87 | 4.7 | 2.00 | 0.97 |
| S19 | 27.67 | 4.0 | 1.90 | 0.80 |
| S5 | 27.5 | 5.9 | 2.18 | 1.20 |
| S14 | 12.6 | 9.3 | 1.70 | 0.75 |
| CBS | 4.47 | 2.2 | 1.31 | 0.51 |
| M33 | 16.47 | 6.7 | 3.33 | 2.08 |
| M32 | 17.93 | 9.5 | 2.65 | 1.37 |
| M42 | 21.67 | 9.4 | 1.40 | 1.35 |
| M29 | 24.64 | 8.5 | 2.95 | 1.84 |
| M39 | 27.53 | 5.5 | 1.40 | 0.78 |
| M26 | 29.53 | 1.8 | 2.45 | 1.25 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segal, E.; Frenkel, M. Experimental In Vivo Models of Candidiasis. J. Fungi 2018, 4, 21. https://doi.org/10.3390/jof4010021
Segal E, Frenkel M. Experimental In Vivo Models of Candidiasis. Journal of Fungi. 2018; 4(1):21. https://doi.org/10.3390/jof4010021
Chicago/Turabian StyleSegal, Esther, and Michael Frenkel. 2018. "Experimental In Vivo Models of Candidiasis" Journal of Fungi 4, no. 1: 21. https://doi.org/10.3390/jof4010021
APA StyleSegal, E., & Frenkel, M. (2018). Experimental In Vivo Models of Candidiasis. Journal of Fungi, 4(1), 21. https://doi.org/10.3390/jof4010021





