The Cell Wall Integrity Signaling Pathway and Its Involvement in Secondary Metabolite Production
Abstract
:1. Fungal Secondary Metabolites
2. Translating Stress to Stress Response
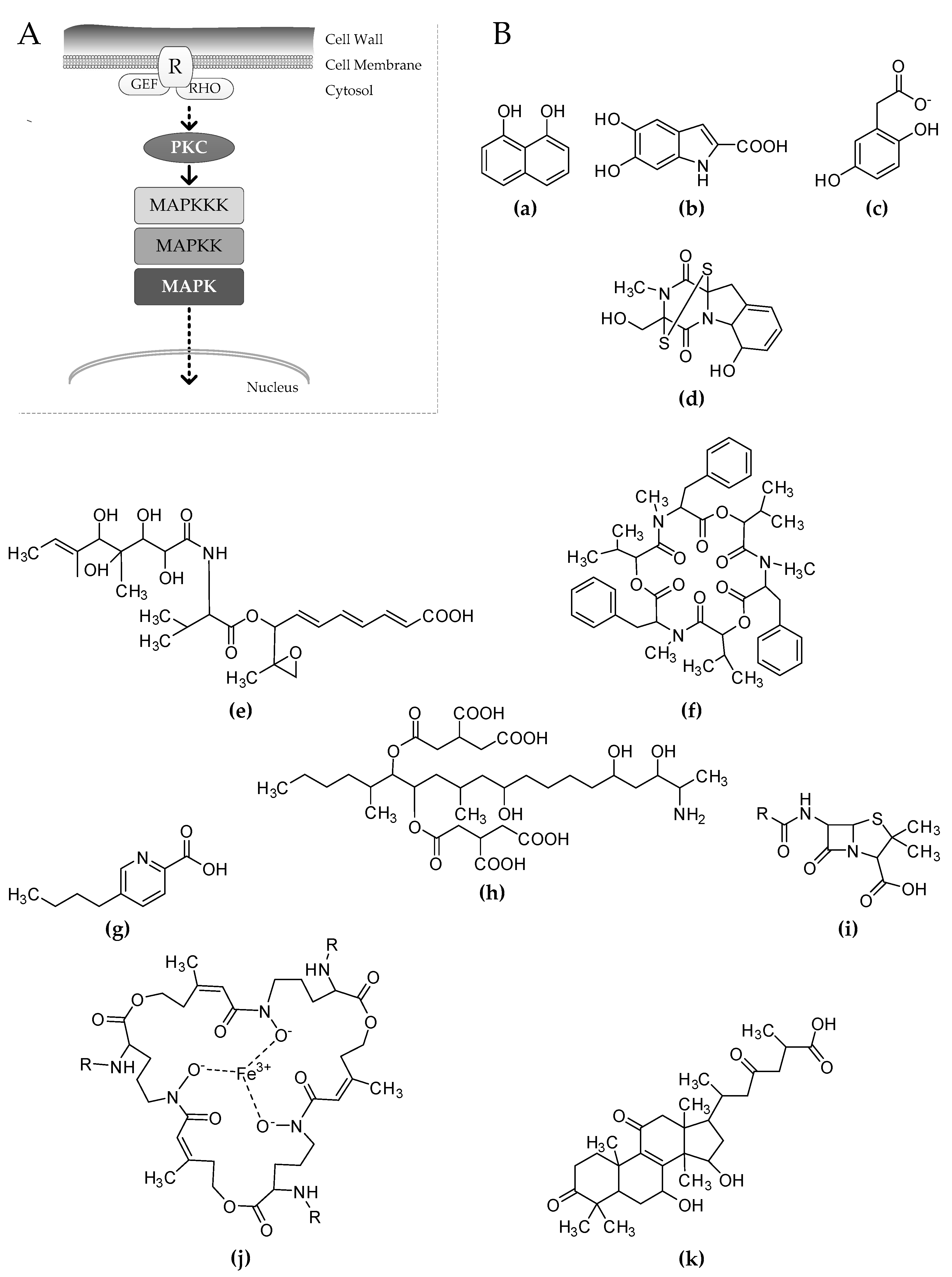
3. The Cell Wall Integrity Pathway Affects the Production of Melanins
4. Mycotoxins, Antibiotics, and Virulence Determinants Affected by the CWI Signaling
5. Perspectives
Acknowledgments
Conflicts of Interest
References
- Brakhage, A.A. Regulation of Fungal Secondary Metabolism. Nat. Rev. Microbiol. 2013, 11, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Haas, H. Fungal Siderophore Metabolism with a Focus on Aspergillus fumigatus. Nat. Prod. Rep. 2014, 31, 1266–1276. [Google Scholar] [CrossRef] [PubMed]
- Hertweck, C. The Biosynthetic Logic of Polyketide Diversity. Angew. Chem. Int. Ed. Engl. 2009, 48, 4688–4716. [Google Scholar] [CrossRef] [PubMed]
- Macheleidt, J.; Mattern, D.J.; Fischer, J.; Netzker, T.; Weber, J.; Schroeckh, V.; Valiante, V.; Brakhage, A.A. Regulation and Role of Fungal Secondary Metabolites. Annu. Rev. Genet. 2016, 50, 371–392. [Google Scholar] [CrossRef] [PubMed]
- Nosanchuk, J.D.; Stark, R.E.; Casadevall, A. Fungal Melanin: What Do We Know About Structure? Front. Microbiol. 2015, 6, 1463. [Google Scholar] [CrossRef] [PubMed]
- Heinekamp, T.; Thywissen, A.; Macheleidt, J.; Keller, S.; Valiante, V.; Brakhage, A.A. Aspergillus fumigatus Melanins: Interference with the Host Endocytosis Pathway and Impact on Virulence. Front. Microbiol. 2012, 3, 440. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.; Hall, M.N. Nutrient Sensing and TOR Signaling in Yeast and Mammals. EMBO J. 2017, 36, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Rispail, N.; Soanes, D.M.; Ant, C.; Czajkowski, R.; Grunler, A.; Huguet, R.; Perez-Nadales, E.; Poli, A.; Sartorel, E.; Valiante, V.; et al. Comparative Genomics of Map Kinase and Calcium-Calcineurin Signalling Components in Plant and Human Pathogenic Fungi. Fungal Genet. Biol. 2009, 46, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Turra, D.; Segorbe, D.; di Pietro, A. Protein Kinases in Plant-Pathogenic Fungi: Conserved Regulators of Infection. Annu. Rev. Phytopathol. 2014, 52, 267–288. [Google Scholar] [CrossRef] [PubMed]
- Bruder Nascimento, A.C.; Reis, T.F.D.; de Castro, P.A.; Hori, J.I.; Bom, V.L.; de Assis, L.J.; Ramalho, L.N.; Rocha, M.C.; Malavazi, I.; Brown, N.A.; et al. Mitogen Activated Protein Kinases Saka (Hog1) and Mpkc Collaborate for Aspergillus fumigatus Virulence. Mol. Microbiol. 2016, 100, 841–859. [Google Scholar] [CrossRef] [PubMed]
- Altwasser, R.; Baldin, C.; Weber, J.; Guthke, R.; Kniemeyer, O.; Brakhage, A.A.; Linde, J.; Valiante, V. Network Modeling Reveals Cross Talk of Map Kinases during Adaptation to Caspofungin Stress in Aspergillus fumigatus. PLoS ONE 2015, 10, e0136932. [Google Scholar] [CrossRef] [PubMed]
- Saito, H. Regulation of Cross-Talk in Yeast Mapk Signaling Pathways. Curr. Opin. Microbiol. 2010, 13, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Torres, L.; Martin, H.; Garcia-Saez, M.I.; Arroyo, J.; Molina, M.; Sanchez, M.; Nombela, C. A Protein Kinase Gene Complements the Lytic Phenotype of Saccharomyces cerevisiae Lyt2 Mutants. Mol. Microbiol. 1991, 5, 2845–2854. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.E. Regulation of Cell Wall Biogenesis in Saccharomyces cerevisiae: The Cell Wall Integrity Signaling Pathway. Genetics 2011, 189, 1145–1175. [Google Scholar] [CrossRef] [PubMed]
- Hamel, L.P.; Nicole, M.C.; Duplessis, S.; Ellis, B.E. Mitogen-Activated Protein Kinase Signaling in Plant-Interacting Fungi: Distinct Messages from Conserved Messengers. Plant Cell 2012, 24, 1327–1351. [Google Scholar] [CrossRef] [PubMed]
- Eisenman, H.C.; Casadevall, A. Synthesis and Assembly of Fungal Melanin. Appl. Microbiol. Biotechnol. 2012, 93, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Langfelder, K.; Jahn, B.; Gehringer, H.; Schmidt, A.; Wanner, G.; Brakhage, A.A. Identification of a Polyketide Synthase Gene (Pksp) of Aspergillus fumigatus Involved in Conidial Pigment Biosynthesis and Virulence. Med. Microbiol. Immunol. 1998, 187, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Akamatsu, H.O.; Chilvers, M.I.; Stewart, J.E.; Peever, T.L. Identification and Function of a Polyketide Synthase Gene Responsible for 1,8-Dihydroxynaphthalene-Melanin Pigment Biosynthesis in Ascochyta Rabiei. Curr. Genet. 2010, 56, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, J. Dhn Melanin Biosynthesis in the Plant Pathogenic Fungus Botrytis cinerea Is Based on Two Developmentally Regulated Key Enzyme (Pks)-Encoding Genes. Mol. Microbiol. 2016, 99, 729–748. [Google Scholar] [CrossRef] [PubMed]
- Brakhage, A.A.; Liebmann, B. Aspergillus fumigatus Conidial Pigment and Camp Signal Transduction: Significance for Virulence. Med. Mycol. 2005, 43 (Suppl. S1), S75–S82. [Google Scholar] [CrossRef] [PubMed]
- Alspaugh, J.A.; Perfect, J.R.; Heitman, J. Cryptococcus neoformans Mating and Virulence Are Regulated by the G-Protein Alpha Subunit Gpa1 and Camp. Genes Dev. 1997, 11, 3206–3217. [Google Scholar] [CrossRef] [PubMed]
- Calvo, A.M.; Wilson, R.A.; Bok, J.W.; Keller, N.P. Relationship between Secondary Metabolism and Fungal Development. Microbiol. Mol. Biol. Rev. 2002, 66, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Garcia, R.; Bravo, E.; Diez-Muniz, S.; Nombela, C.; Rodriguez-Pena, J.M.; Arroyo, J. A Novel Connection between the Cell Wall Integrity and the Pka Pathways Regulates Cell Wall Stress Response in Yeast. Sci. Rep. 2017, 7, 5703. [Google Scholar] [CrossRef] [PubMed]
- Muller, S.; Baldin, C.; Groth, M.; Guthke, R.; Kniemeyer, O.; Brakhage, A.A.; Valiante, V. Comparison of Transcriptome Technologies in the Pathogenic Fungus Aspergillus fumigatus Reveals Novel Insights into the Genome and Mpka Dependent Gene Expression. BMC Genom. 2012, 13, 519. [Google Scholar] [CrossRef] [PubMed]
- Valiante, V.; Baldin, C.; Hortschansky, P.; Jain, R.; Thywissen, A.; Strassburger, M.; Shelest, E.; Heinekamp, T.; Brakhage, A.A. The Aspergillus fumigatus Conidial Melanin Production Is Regulated by the Bifunctional Bhlh Devr and Mads-Box Rlma Transcription Factors. Mol. Microbiol. 2016, 102, 321–335. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.C.; Fabri, J.H.; de Godoy, K.F.; de Castro, P.A.; Hori, J.I.; da Cunha, A.F.; Arentshorst, M.; Ram, A.F.; van den Hondel, C.A.; Goldman, G.H.; et al. Aspergillus fumigatus Mads-Box Transcription Factor Rlma Is Required for Regulation of the Cell Wall Integrity and Virulence. G3 Genes Genomes Genet. 2016, 6, 2983–3002. [Google Scholar] [CrossRef] [PubMed]
- Dodou, E.; Treisman, R. The Saccharomyces cerevisiae Mads-Box Transcription Factor Rlm1 Is a Target for the Mpk1 Mitogen-Activated Protein Kinase Pathway. Mol. Cell. Biol. 1997, 17, 1848–1859. [Google Scholar] [CrossRef] [PubMed]
- Eliahu, N.; Igbaria, A.; Rose, M.S.; Horwitz, B.A.; Lev, S. Melanin Biosynthesis in the Maize Pathogen Cochliobolus heterostrophus Depends on Two Mitogen-Activated Protein Kinases, Chk1 and Mps1, and the Transcription Factor Cmr1. Eukaryot. Cell 2007, 6, 421–429. [Google Scholar] [CrossRef] [PubMed]
- So, K.K.; Ko, Y.H.; Chun, J.; Kim, J.M.; Kim, D.H. Mutation of the Slt2 Ortholog from Cryphonectria parasitica Results in Abnormal Cell Wall Integrity and Sectorization with Impaired Pathogenicity. Sci. Rep. 2017, 7, 9038. [Google Scholar] [CrossRef] [PubMed]
- Yago, J.I.; Lin, C.H.; Chung, K.R. The Slt2 Mitogen-Activated Protein Kinase-Mediated Signalling Pathway Governs Conidiation, Morphogenesis, Fungal Virulence and Production of Toxin and Melanin in the Tangerine Pathotype of Alternaria alternata. Mol. Plant Pathol. 2011, 12, 653–665. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Scher, K.; Mukherjee, M.; Pardovitz-Kedmi, E.; Sible, G.V.; Singh, U.S.; Kale, S.P.; Mukherjee, P.K.; Horwitz, B.A. Overlapping and Distinct Functions of Two Trichoderma virens Map Kinases in Cell-Wall Integrity, Antagonistic Properties and Repression of Conidiation. Biochem. Biophys. Res. Commun. 2010, 398, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Soulie, M.C.; Perrino, C.; Fillinger, S. The Osmosensing Signal Transduction Pathway from Botrytis cinerea Regulates Cell Wall Integrity and Map Kinase Pathways Control Melanin Biosynthesis with Influence of Light. Fungal Genet. Biol. 2011, 48, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Gerik, K.J.; Donlin, M.J.; Soto, C.E.; Banks, A.M.; Banks, I.R.; Maligie, M.A.; Selitrennikoff, C.P.; Lodge, J.K. Cell Wall Integrity Is Dependent on the Pkc1 Signal Transduction Pathway in Cryptococcus neoformans. Mol. Microbiol. 2005, 58, 393–408. [Google Scholar] [CrossRef] [PubMed]
- Ngamskulrungroj, P.; Price, J.; Sorrell, T.; Perfect, J.R.; Meyer, W. Cryptococcus gattii Virulence Composite: Candidate Genes Revealed by Microarray Analysis of High and Less Virulent Vancouver Island Outbreak Strains. PLoS ONE 2011, 6, e16076. [Google Scholar] [CrossRef] [PubMed]
- Park, G.; Pan, S.; Borkovich, K.A. Mitogen-Activated Protein Kinase Cascade Required for Regulation of Development and Secondary Metabolism in Neurospora crassa. Eukaryot. Cell 2008, 7, 2113–2122. [Google Scholar] [CrossRef] [PubMed]
- Valiante, V.; Jain, R.; Heinekamp, T.; Brakhage, A.A. The Mpka Map Kinase Module Regulates Cell Wall Integrity Signaling and Pyomelanin Formation in Aspergillus fumigatus. Fungal Genet. Biol. 2009, 46, 909–918. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, G.; Kenmochi, Y.; Takano, Y.; Sweigard, J.; Farrall, L.; Furusawa, I.; Horino, O.; Kubo, Y. Novel Fungal Transcriptional Activators, Cmr1p of Colletotrichum lagenarium and Pig1p of Magnaporthe Grisea, Contain Cys2his2 Zinc Finger and Zn(Ii)2cys6 Binuclear Cluster DNA-Binding Motifs and Regulate Transcription of Melanin Biosynthesis Genes in a Developmentally Specific Manner. Mol. Microbiol. 2000, 38, 940–954. [Google Scholar] [PubMed]
- Breunig, J.S.; Hackett, S.R.; Rabinowitz, J.D.; Kruglyak, L. Genetic Basis of Metabolome Variation in Yeast. PLoS Genet. 2014, 10, e1004142. [Google Scholar] [CrossRef] [PubMed]
- Bannister, A.J.; Kouzarides, T. Regulation of Chromatin by Histone Modifications. Cell Res. 2011, 21, 381–395. [Google Scholar] [CrossRef] [PubMed]
- Nutzmann, H.W.; Fischer, J.; Scherlach, K.; Hertweck, C.; Brakhage, A.A. Distinct Amino Acids of Histone H3 Control Secondary Metabolism in Aspergillus nidulans. Appl. Environ. Microbiol. 2013, 79, 6102–6109. [Google Scholar] [CrossRef] [PubMed]
- Scharf, D.H.; Brakhage, A.A.; Mukherjee, P.K. Gliotoxin—Bane or Boon? Environ. Microbiol. 2016, 18, 1096–1109. [Google Scholar] [CrossRef] [PubMed]
- Hillmann, F.; Novohradska, S.; Mattern, D.J.; Forberger, T.; Heinekamp, T.; Westermann, M.; Winckler, T.; Brakhage, A.A. Virulence Determinants of the Human Pathogenic Fungus Aspergillus fumigatus Protect against Soil Amoeba Predation. Environ. Microbiol. 2015, 17, 2858–2869. [Google Scholar] [CrossRef] [PubMed]
- Cramer, R.A., Jr.; Gamcsik, M.P.; Brooking, R.M.; Najvar, L.K.; Kirkpatrick, W.R.; Patterson, T.F.; Balibar, C.J.; Graybill, J.R.; Perfect, J.R.; Abraham, S.N.; et al. Disruption of a Nonribosomal Peptide Synthetase in Aspergillus fumigatus Eliminates Gliotoxin Production. Eukaryot. Cell 2006, 5, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, L.B.; de Francisco, T.M.; Gasparetto, J.C.; Campos, F.R.; Pontarolo, R. Development and Validation of an Hplc-Ms/Ms Method for the Early Diagnosis of Aspergillosis. PLoS ONE 2014, 9, e92851. [Google Scholar] [CrossRef] [PubMed]
- Winkelstroter, L.K.; Dolan, S.K.; Reis, T.F.D.; Bom, V.L.; de Castro, P.A.; Hagiwara, D.; Alowni, R.; Jones, G.W.; Doyle, S.; Brown, N.A.; et al. Systematic Global Analysis of Genes Encoding Protein Phosphatases in Aspergillus fumigatus. G3 Genes Genomes Genet. 2015, 5, 1525–1539. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.G.; Kim, S.S.; Yu, J.H.; Shin, K.S. Characterization of Gprk Encoding a Putative Hybrid G-Protein-Coupled Receptor in Aspergillus fumigatus. PLoS ONE 2016, 11, e0161312. [Google Scholar] [CrossRef] [PubMed]
- Johnk, B.; Bayram, O.; Abelmann, A.; Heinekamp, T.; Mattern, D.J.; Brakhage, A.A.; Jacobsen, I.D.; Valerius, O.; Braus, G.H. Scf Ubiquitin Ligase F-Box Protein Fbx15 Controls Nuclear Co-Repressor Localization, Stress Response and Virulence of the Human Pathogen Aspergillus fumigatus. PLoS Pathog. 2016, 12, e1005899. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Valiante, V.; Remme, N.; Docimo, T.; Heinekamp, T.; Hertweck, C.; Gershenzon, J.; Haas, H.; Brakhage, A.A. The Map Kinase Mpka Controls Cell Wall Integrity, Oxidative Stress Response, Gliotoxin Production and Iron Adaptation in Aspergillus fumigatus. Mol. Microbiol. 2011, 82, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Tanaka, T.; Hatta, R.; Yamamoto, M.; Akimitsu, K.; Tsuge, T. Dissection of the Host Range of the Fungal Plant Pathogen Alternaria alternata by Modification of Secondary Metabolism. Mol. Microbiol. 2004, 52, 399–411. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Li, M.; Sun, F.; Xi, P.; Sun, L.; Zhang, L.; Jiang, Z. Mitogen-Activated Protein Kinases Are Associated with the Regulation of Physiological Traits and Virulence in Fusarium oxysporum f. sp. Cubense. PLoS ONE 2015, 10, e0122634. [Google Scholar] [CrossRef] [PubMed]
- Niehaus, E.M.; von Bargen, K.W.; Espino, J.J.; Pfannmuller, A.; Humpf, H.U.; Tudzynski, B. Characterization of the Fusaric Acid Gene Cluster in Fusarium fujikuroi. Appl. Microbiol. Biotechnol. 2014, 98, 1749–1762. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.; Moretti, A.; Castella, G.; Kostecki, M.; Golinski, P.; Ritieni, A.; Chelkowski, J. Beauvericin Production by Fusarium Species. Appl. Environ. Microbiol. 1998, 64, 3084–3088. [Google Scholar] [PubMed]
- Wang, E.; Norred, W.P.; Bacon, C.W.; Riley, R.T.; Merrill, A.H., Jr. Inhibition of Sphingolipid Biosynthesis by Fumonisins. Implications for Diseases Associated with Fusarium moniliforme. J. Biol. Chem. 1991, 266, 14486–14490. [Google Scholar] [PubMed]
- Lee, H.J.; Ryu, D. Worldwide Occurrence of Mycotoxins in Cereals and Cereal-Derived Food Products: Public Health Perspectives of Their Co-Occurrence. J. Agric. Food Chem. 2017, 65, 7034–7051. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S.; van der Westhuizen, L.; Sewram, V. Biomarkers of Exposure to Fumonisin Mycotoxins: A Review. Food Addit. Contam. 2007, 24, 1196–1201. [Google Scholar] [CrossRef] [PubMed]
- Rocha, L.O.; Barroso, V.M.; Andrade, L.J.; Pereira, G.H.; Ferreira-Castro, F.L.; Duarte, A.P.; Michelotto, M.D.; Correa, B. Fum Gene Expression Profile and Fumonisin Production by Fusarium verticillioides Inoculated in Bt and Non-Bt Maize. Front. Microbiol. 2015, 6, 1503. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, J.; Tao, H.; Dang, X.; Wang, Y.; Chen, M.; Zhai, Z.; Yu, W.; Xu, L.; Shim, W.B.; et al. Fvbck1, a Component of Cell Wall Integrity Map Kinase Pathway, Is Required for Virulence and Oxidative Stress Response in Sugarcane Pokkah Boeng Pathogen. Front. Microbiol. 2015, 6, 1096. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Choi, Y.E.; Zou, X.; Xu, J.R. The Fvmk1 Mitogen-Activated Protein Kinase Gene Regulates Conidiation, Pathogenesis, and Fumonisin Production in Fusarium verticillioides. Fungal Genet. Biol. 2011, 48, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Caballero Ortiz, S.; Trienens, M.; Rohlfs, M. Induced Fungal Resistance to Insect Grazing: Reciprocal Fitness Consequences and Fungal Gene Expression in the Drosophila-Aspergillus Model System. PLoS ONE 2013, 8, e74951. [Google Scholar] [CrossRef] [PubMed]
- Doll, K.; Chatterjee, S.; Scheu, S.; Karlovsky, P.; Rohlfs, M. Fungal Metabolic Plasticity and Sexual Development Mediate Induced Resistance to Arthropod Fungivory. Proc. Biol. Sci. 2013, 280. [Google Scholar] [CrossRef] [PubMed]
- Chiang, Y.M.; Szewczyk, E.; Nayak, T.; Davidson, A.D.; Sanchez, J.F.; Lo, H.C.; Ho, W.Y.; Simityan, H.; Kuo, E.; Praseuth, A.; et al. Molecular Genetic Mining of the Aspergillus Secondary Metabolome: Discovery of the Emericellamide Biosynthetic Pathway. Chem. Biol. 2008, 15, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Valiante, V.; Mattern, D.J.; Schuffler, A.; Horn, F.; Walther, G.; Scherlach, K.; Petzke, L.; Dickhaut, J.; Guthke, R.; Hertweck, C.; et al. Discovery of an Extended Austinoid Biosynthetic Pathway in Aspergillus calidoustus. ACS Chem. Biol. 2017, 12, 1227–1234. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, M.; Sprote, P.; Brakhage, A.A. Protein Kinase C (Pkca) of Aspergillus nidulans Is Involved in Penicillin Production. Appl. Environ. Microbiol. 2006, 72, 2957–2970. [Google Scholar] [CrossRef] [PubMed]
- Haas, H.; Eisendle, M.; Turgeon, B.G. Siderophores in Fungal Physiology and Virulence. Annu. Rev. Phytopathol. 2008, 46, 149–187. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Bok, J.W.; Guzman-de-Pena, D.; Keller, N.P. Requirement of Spermidine for Developmental Transitions in Aspergillus nidulans. Mol. Microbiol. 2002, 46, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Gsaller, F.; Hortschansky, P.; Beattie, S.R.; Klammer, V.; Tuppatsch, K.; Lechner, B.E.; Rietzschel, N.; Werner, E.R.; Vogan, A.A.; Chung, D.; et al. The Janus Transcription Factor Hapx Controls Fungal Adaptation to Both Iron Starvation and Iron Excess. EMBO J. 2014, 33, 2261–2276. [Google Scholar] [CrossRef] [PubMed]
- Schrettl, M.; Beckmann, N.; Varga, J.; Heinekamp, T.; Jacobsen, I.D.; Jochl, C.; Moussa, T.A.; Wang, S.; Gsaller, F.; Blatzer, M.; et al. Hapx-Mediated Adaption to Iron Starvation Is Crucial for Virulence of Aspergillus fumigatus. PLoS Pathog. 2010, 6, e1001124. [Google Scholar] [CrossRef] [PubMed]
- Hajjaj, H.; Mace, C.; Roberts, M.; Niederberger, P.; Fay, L.B. Effect of 26-Oxygenosterols from Ganoderma lucidum and Their Activity as Cholesterol Synthesis Inhibitors. Appl. Environ. Microbiol. 2005, 71, 3653–3658. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Sun, Z.; Ren, A.; Shi, L.; Shi, D.; Li, X.; Zhao, M. The Mitogen-Activated Protein Kinase Glslt2 Regulates Fungal Growth, Fruiting Body Development, Cell Wall Integrity, Oxidative Stress and Ganoderic Acid Biosynthesis in Ganoderma lucidum. Fungal Genet. Biol. 2017, 104, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Netzker, T.; Fischer, J.; Weber, J.; Mattern, D.J.; Konig, C.C.; Valiante, V.; Schroeckh, V.; Brakhage, A.A. Microbial Communication Leading to the Activation of Silent Fungal Secondary Metabolite Gene Clusters. Front. Microbiol. 2015, 6, 299. [Google Scholar] [CrossRef] [PubMed]
- Canals, D.; Perry, D.M.; Jenkins, R.W.; Hannun, Y.A. Drug Targeting of Sphingolipid Metabolism: Sphingomyelinases and Ceramidases. Br. J. Pharmacol. 2011, 163, 694–712. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, K.; Hohmann, S. Synthetic Biology: Lessons from Engineering Yeast Mapk Signalling Pathways. Mol. Microbiol. 2013, 88, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Chen, C.Y.; Levin, D.E. Saccharomyces cerevisiae Pkc1 Encodes a Protein Kinase C (Pkc) Homolog with a Substrate Specificity Similar to That of Mammalian Pkc. J. Biol. Chem. 1994, 269, 16829–16836. [Google Scholar] [PubMed]
- Katayama, T.; Ohta, A.; Horiuchi, H. Protein Kinase C Regulates the Expression of Cell Wall-Related Genes in Rlma-Dependent and Independent Manners in Aspergillus nidulans. Biosci. Biotechnol. Biochem. 2015, 79, 321–330. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valiante, V. The Cell Wall Integrity Signaling Pathway and Its Involvement in Secondary Metabolite Production. J. Fungi 2017, 3, 68. https://doi.org/10.3390/jof3040068
Valiante V. The Cell Wall Integrity Signaling Pathway and Its Involvement in Secondary Metabolite Production. Journal of Fungi. 2017; 3(4):68. https://doi.org/10.3390/jof3040068
Chicago/Turabian StyleValiante, Vito. 2017. "The Cell Wall Integrity Signaling Pathway and Its Involvement in Secondary Metabolite Production" Journal of Fungi 3, no. 4: 68. https://doi.org/10.3390/jof3040068
APA StyleValiante, V. (2017). The Cell Wall Integrity Signaling Pathway and Its Involvement in Secondary Metabolite Production. Journal of Fungi, 3(4), 68. https://doi.org/10.3390/jof3040068




