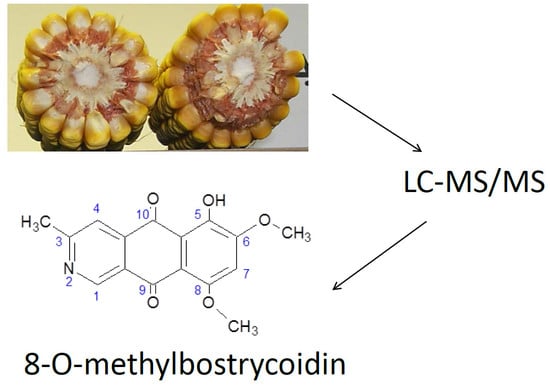
Utilization of High Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry for Characterization of 8-O-methylbostrycoidin Production by Species of the Fungus Fusarium
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fungal Strains and Preparation of Maize Materia
2.3. Extraction of 8-O-methylbostrycoidin from Maize
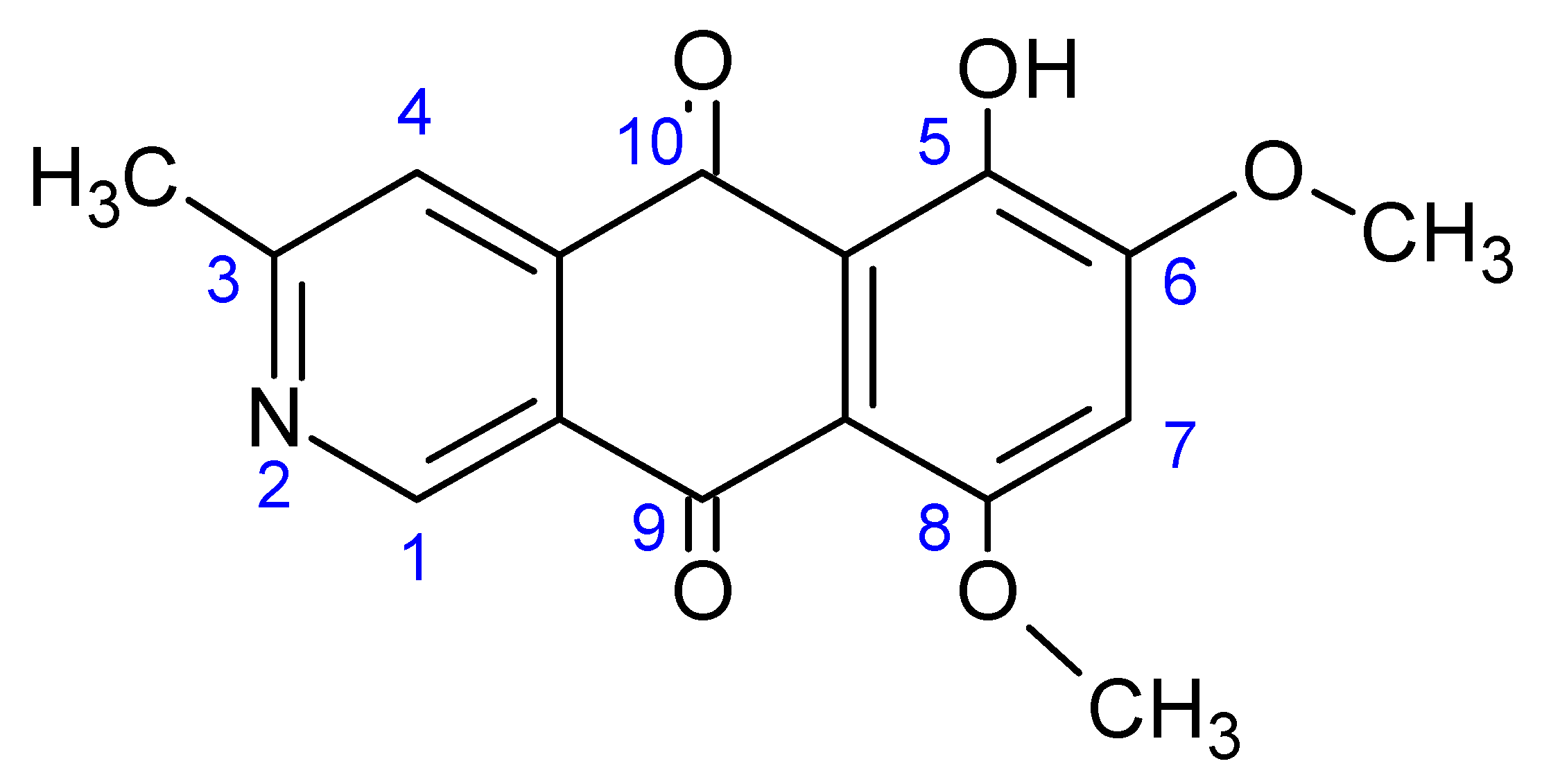
2.4. ESI-MS and MS/MS
2.5. HPLC-MS/MS
2.6. Spike/Recovery Experiments and Method Validation
2.7. Field Test
3. Results
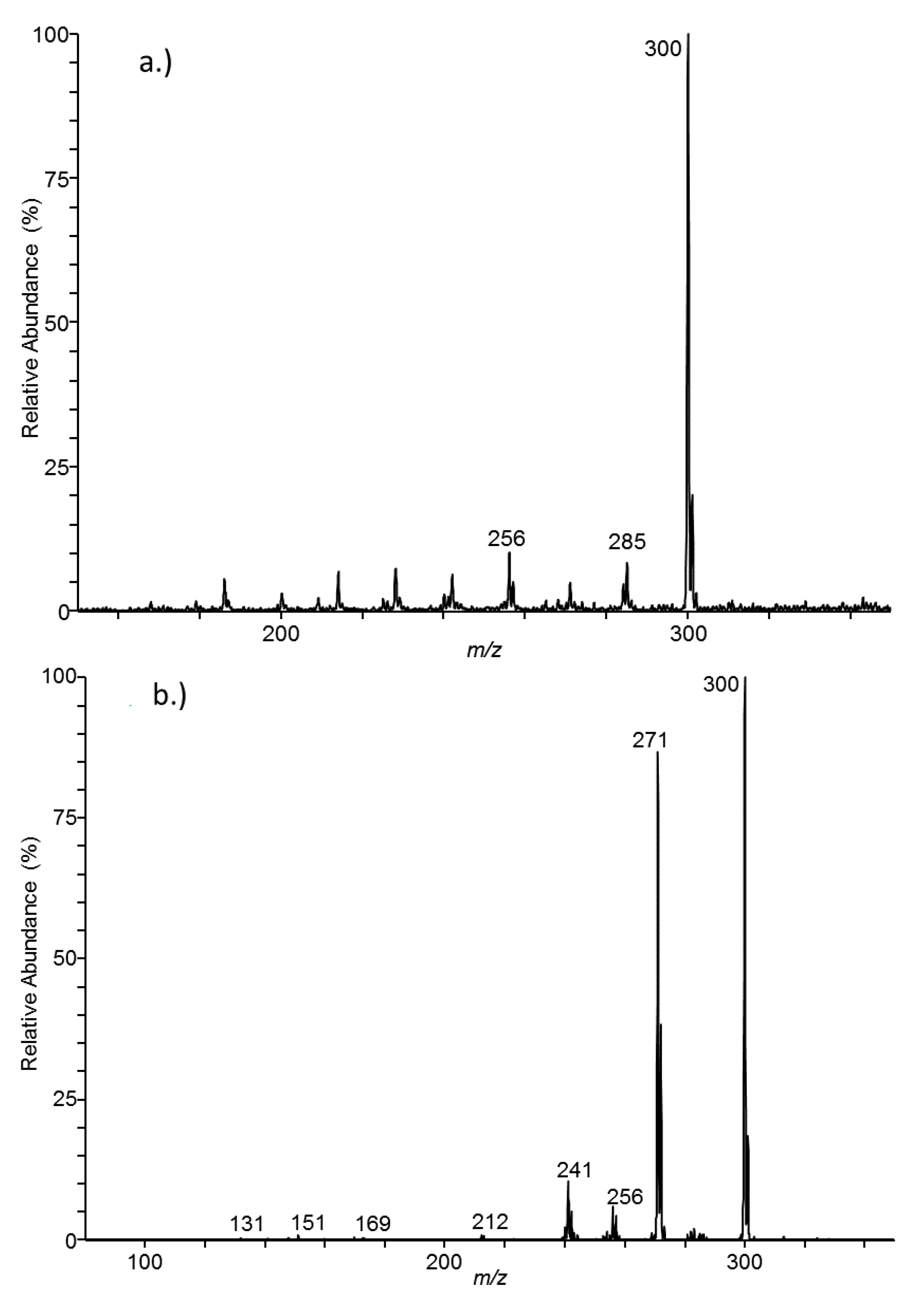
3.1. MS and MS/MS by Flow Injection
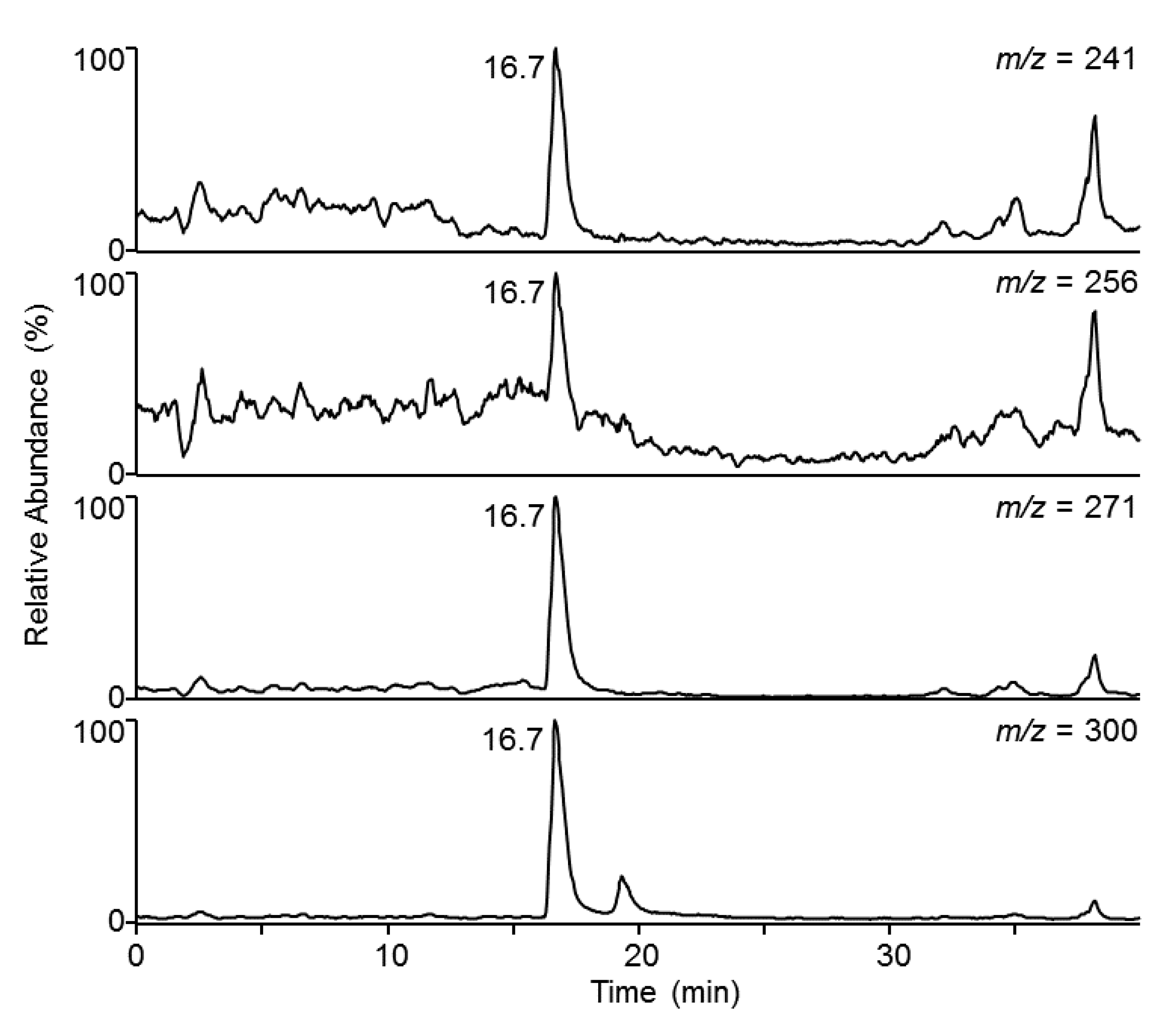
3.2. HPLC Coupled to MS
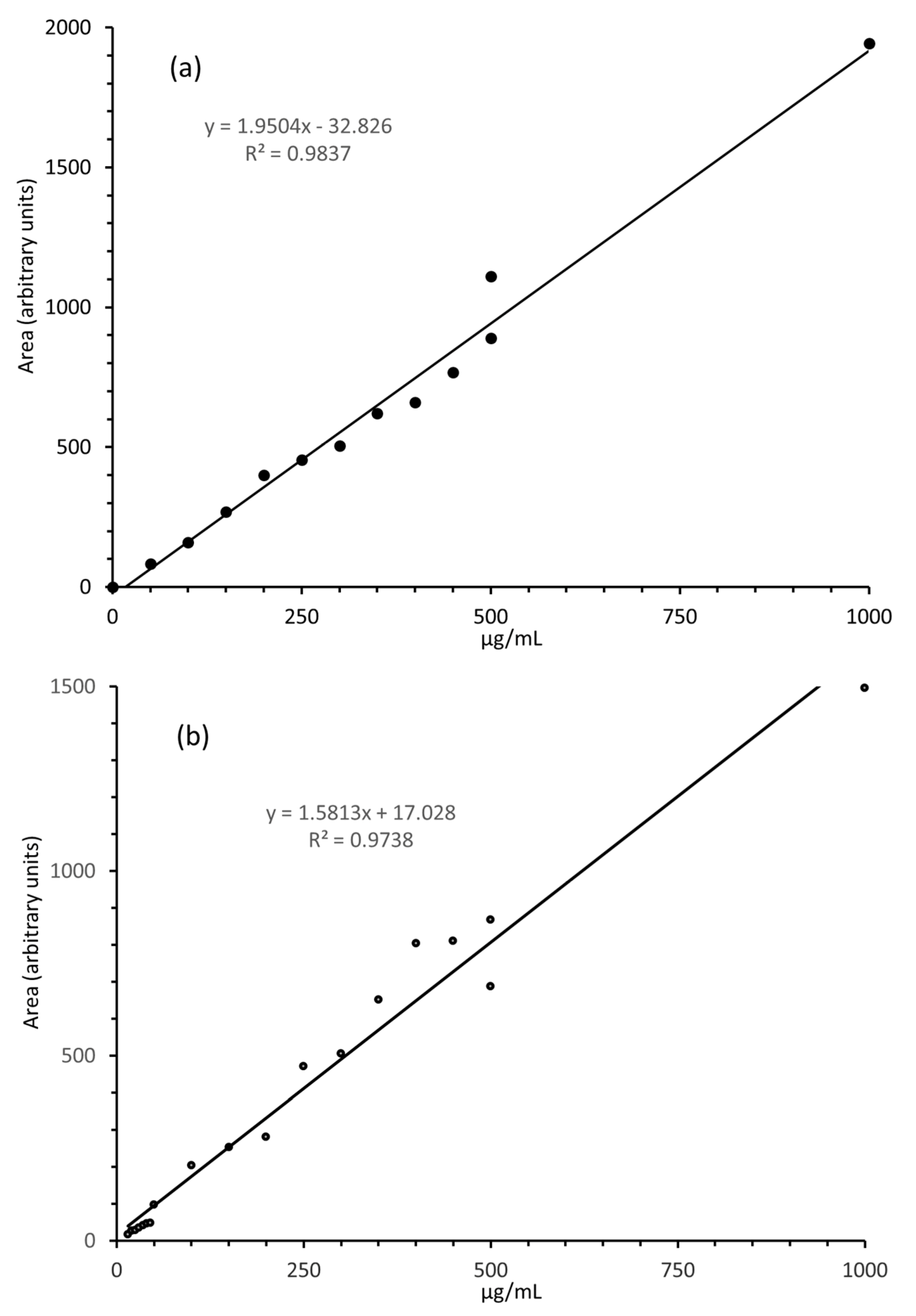
3.3. LC-MS/MS Quantitation
3.4. Spike Recovery Studies
3.5. Matrix Interference
3.6. Determination of 8-O-methylbostrycoidin from Field Grown Maize
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Steyn, P.S.; Wessels, P.L.; Marasas, W.F.O. Pigments from Fusarium moniliforme sheldon. Structure and 13C nuclear magnetic resonance assignments of an azaanthraquinone and three naphthoquinones. Tetrahedron 1979, 35, 1551–1555. [Google Scholar] [CrossRef]
- Cameron, D.W.; Deutscher, K.R.; Feutrill, G.I. Synthesis of bostrycoidin and 8-O-methlylbostrycoidin. Tetrahedron Lett. 1980, 21, 5089–5090. [Google Scholar] [CrossRef]
- Cameron, D.W.; Deutscher, K.R.; Feutrill, G.I. Nucleophilic alkenes. Ix. Addition of 1,1-dimethoxyethene to azanaphthoquinones: Synthesis of bostrycoidin and 8-O-methylbostrycoidin. Aust. J. Chem. 1982, 35, 1439. [Google Scholar] [CrossRef]
- Tatum, J.H.; Baker, R.A.; Berry, R.E. Naphthoquinones produced by Fusarium oxysporum isolated from citrus. Phytochemistry 1985, 24, 457–459. [Google Scholar] [CrossRef]
- Deng, C.M.; Liu, S.X.; Huang, C.H.; Pang, J.Y.; Lin, Y.C. Secondary metabolites of a mangrove endophytic fungus Aspergillus terreus (no. Gx7-3b) from the South China Sea. Mar. Drugs 2013, 11, 2616–2624. [Google Scholar] [CrossRef] [PubMed]
- Vesonder, R.F.; Gasdorf, H.; Peterson, R.E. Comparison of the cytotoxicities of Fusarium metabolites and Alternaria metabolite AAL-toxin to cultured mammalian cell lines. Arch. Environ. Contam. Toxicol. 1993, 24, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, J.; Motohashi, K.; Sakamoto, K.; Hashimoto, S.; Yamanouchi, M.; Tanaka, H.; Takahashi, T.; Takagi, M.; Shin-ya, K. Screening and evaluation of new inhibitors of hepatic glucose production. J. Antibiot. (Tokyo) 2009, 62, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Awakawa, T.; Kaji, T.; Wakimoto, T.; Abe, I. A heptaketide naphthaldehyde produced by a polyketide synthase from Nectria haematococca. Bioorg. Med. Chem. Lett. 2012, 22, 4338–4340. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.W.; Butchko, R.A.E.; Busman, M.; Proctor, R.H. Identification of gene clusters associated with fusaric acid, fusarin, and perithecial pigment production in Fusarium verticillioides. Fungal Genet. Biol. 2012, 49, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Studt, L.; Wiemann, P.; Kleigrewe, K.; Humpf, H.U.; Tudzynski, B. Biosynthesis of fusarubins accounts for pigmentation of Fusarium fujikuroi perithecia. Appl. Environ. Microbiol. 2012, 78, 4468–4480. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.S.; Fotso, J.; Leslie, J.F.; Wu, Y.; VanderVelde, D.; Thakur, R.A. Characterization of bostrycoidin: An analytical analog of zearalenone. J. Food Sci. 2004, 69, C227–C232. [Google Scholar] [CrossRef]
- Nelson, P.E.; Desjardins, A.E.; Plattner, R.D. Fumonisins, mycotoxins produced by Fusarium species: Biology, chemistry, and significance. Annu. Rev. Phytopathol. 1993, 31, 233–252. [Google Scholar] [CrossRef] [PubMed]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Publishing: Ames, IA, USA, 2006. [Google Scholar]
- Geiser, D.M.; Jimenez-Gasco, M.D.M.; Kang, S.; Makalowska, I.; Veeraraghavan, N.; Ward, T.J.; Zhang, N.; Kuldau, G.A.; O’Donnell, K. Fusarium-ID v. 1.0: A DNA sequence database for identifying Fusarium. Eur. J. Plant Pathol. 2004, 110, 473–480. [Google Scholar] [CrossRef]
- Seo, J.A.; Proctor, R.H.; Plattner, R.D. Characterization of four clustered and coregulated genes associated with fumonisin biosynthesis in Fusarium verticillioides. Fungal Genet. Biol. 2001, 34, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Reid, L.; Hamilton, R.; Mather, D. Screening maize for resistance to gibberella ear rot. In Technical Bulletin 1996-5e; Agriculture & Agri-food Canada: Ottawa, ON, Canada, 1996; pp. 1–40. [Google Scholar]
- Desjardins, A.E.; Plattner, R.D. Fumonisin B1-nonproducing strains of Fusarium verticillioides cause maize (Zea mays) ear infection and ear rot. J. Agric. Food Chem. 2000, 48, 5773–5780. [Google Scholar] [CrossRef] [PubMed]
- Matuszewski, B.K.; Constanzer, M.L.; Chavez-Eng, C.M. Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS. Anal. Chem. 2003, 75, 3019–3030. [Google Scholar] [CrossRef] [PubMed]
| Strain | Disease Score 1 | Level of 8-O-methylbostrycoidin (µg/g) 1 |
|---|---|---|
| AMR 5 | 4.9 (1.9) | 0.5 (0.9) |
| AMR 10 | 4.4 (1.6) | 2.6 (4.8) |
| Control inoculation | 1.3 (0.9) | 0 (–) |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Busman, M. Utilization of High Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry for Characterization of 8-O-methylbostrycoidin Production by Species of the Fungus Fusarium. J. Fungi 2017, 3, 43. https://doi.org/10.3390/jof3030043
Busman M. Utilization of High Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry for Characterization of 8-O-methylbostrycoidin Production by Species of the Fungus Fusarium. Journal of Fungi. 2017; 3(3):43. https://doi.org/10.3390/jof3030043
Chicago/Turabian StyleBusman, Mark. 2017. "Utilization of High Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry for Characterization of 8-O-methylbostrycoidin Production by Species of the Fungus Fusarium" Journal of Fungi 3, no. 3: 43. https://doi.org/10.3390/jof3030043
APA StyleBusman, M. (2017). Utilization of High Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry for Characterization of 8-O-methylbostrycoidin Production by Species of the Fungus Fusarium. Journal of Fungi, 3(3), 43. https://doi.org/10.3390/jof3030043