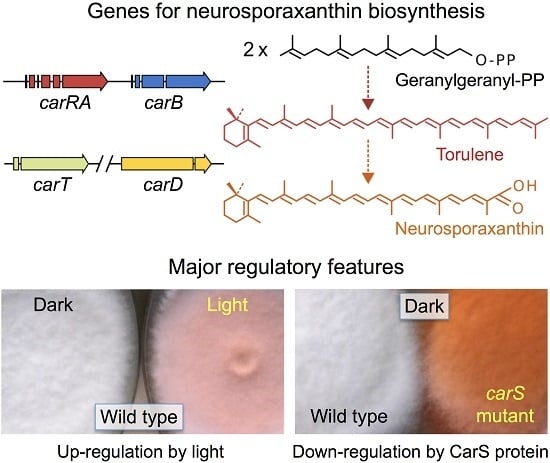
Carotenoid Biosynthesis in Fusarium
Abstract
:1. Introduction
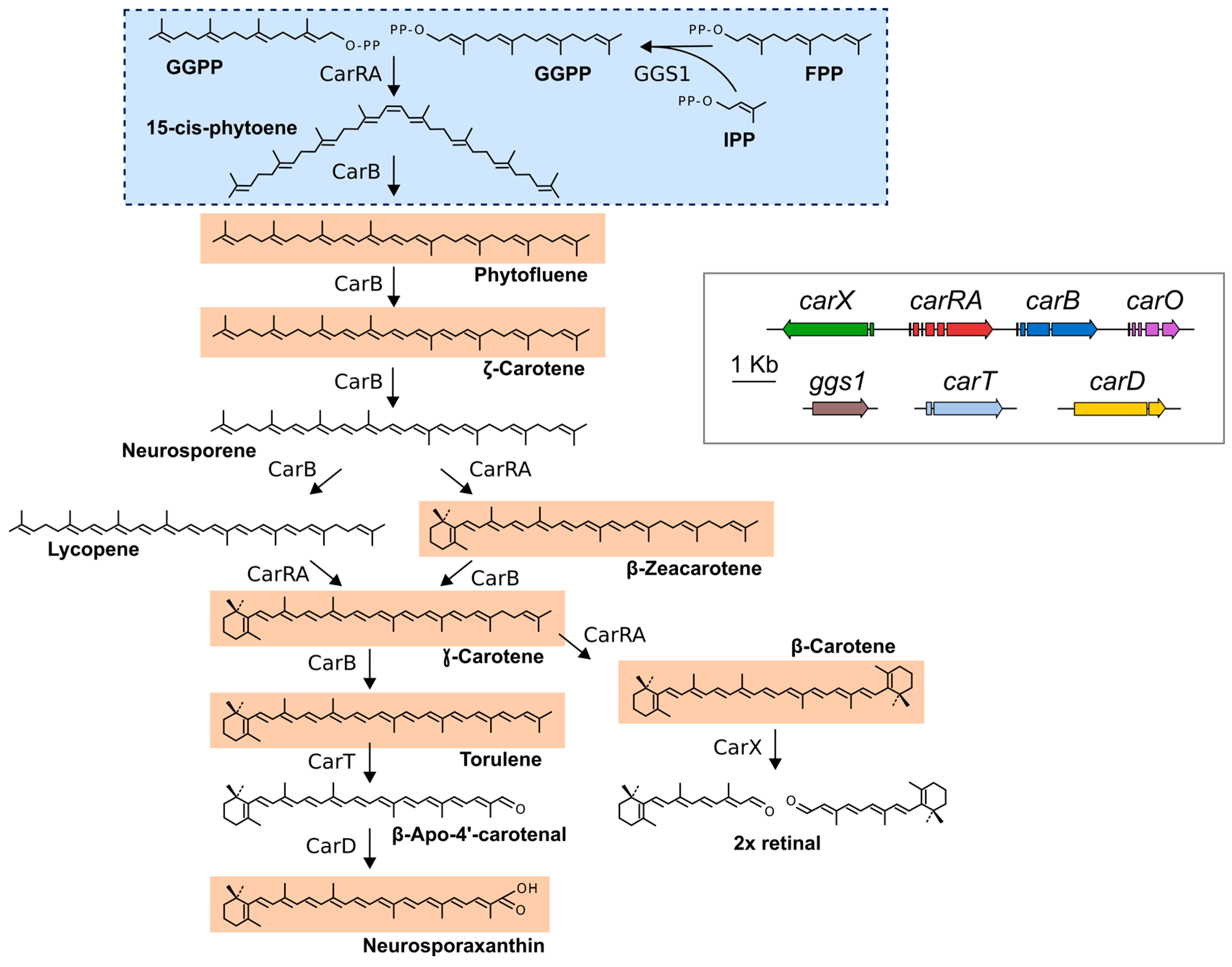
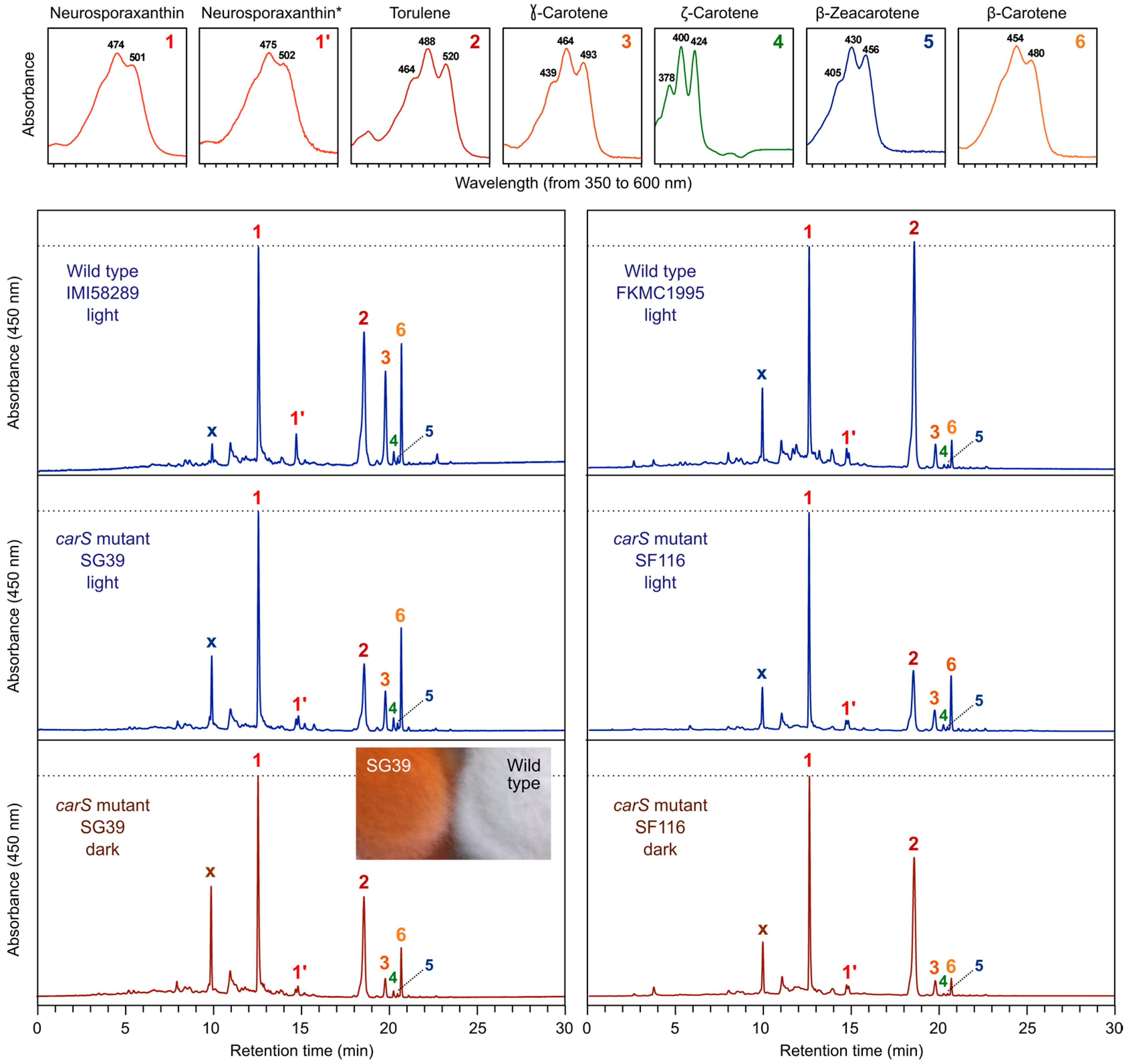
2. Fusarium Carotenoids
3. Genes and Enzymes of Carotenoid Metabolism in Fusarium
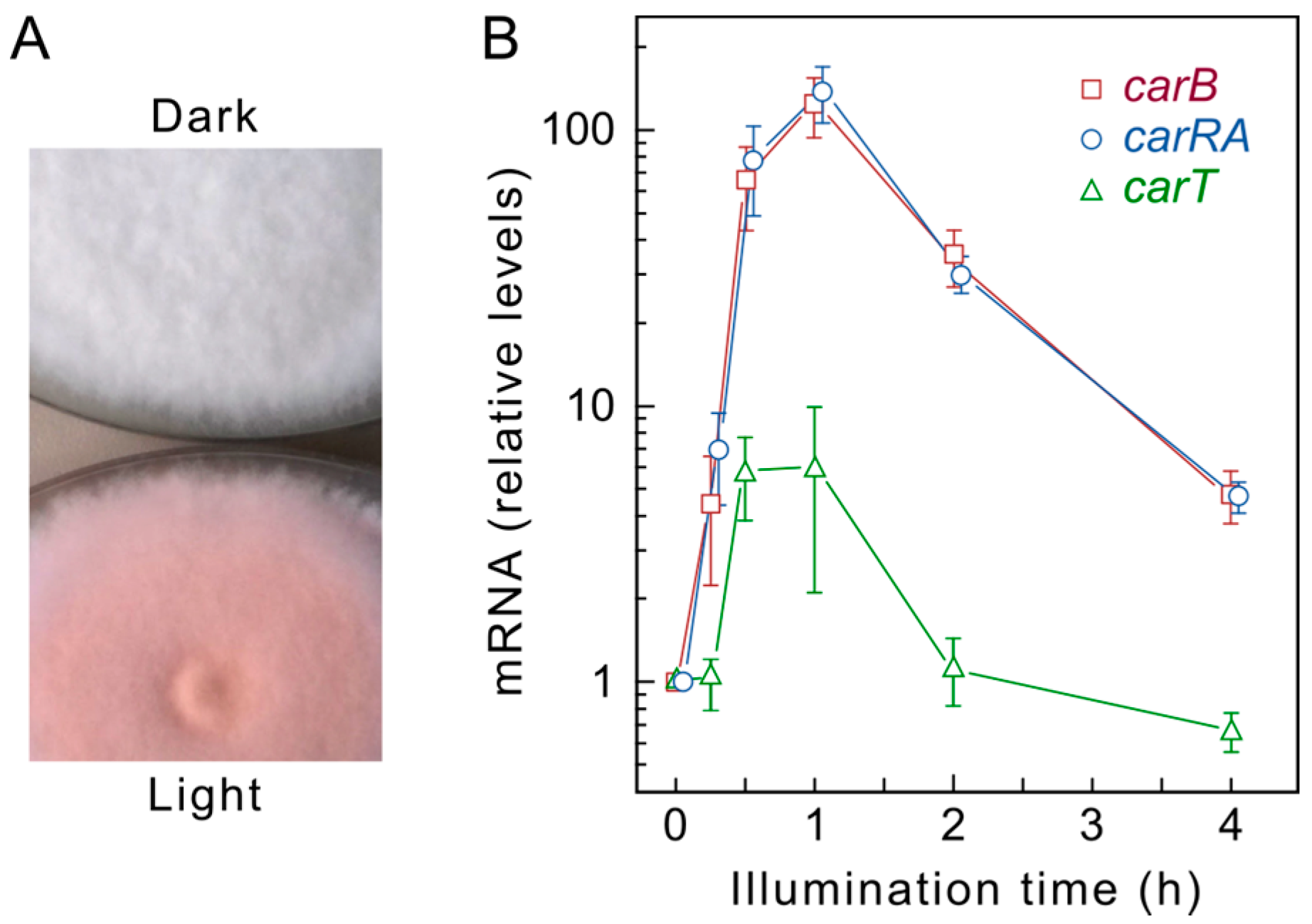
4. Regulation by Light
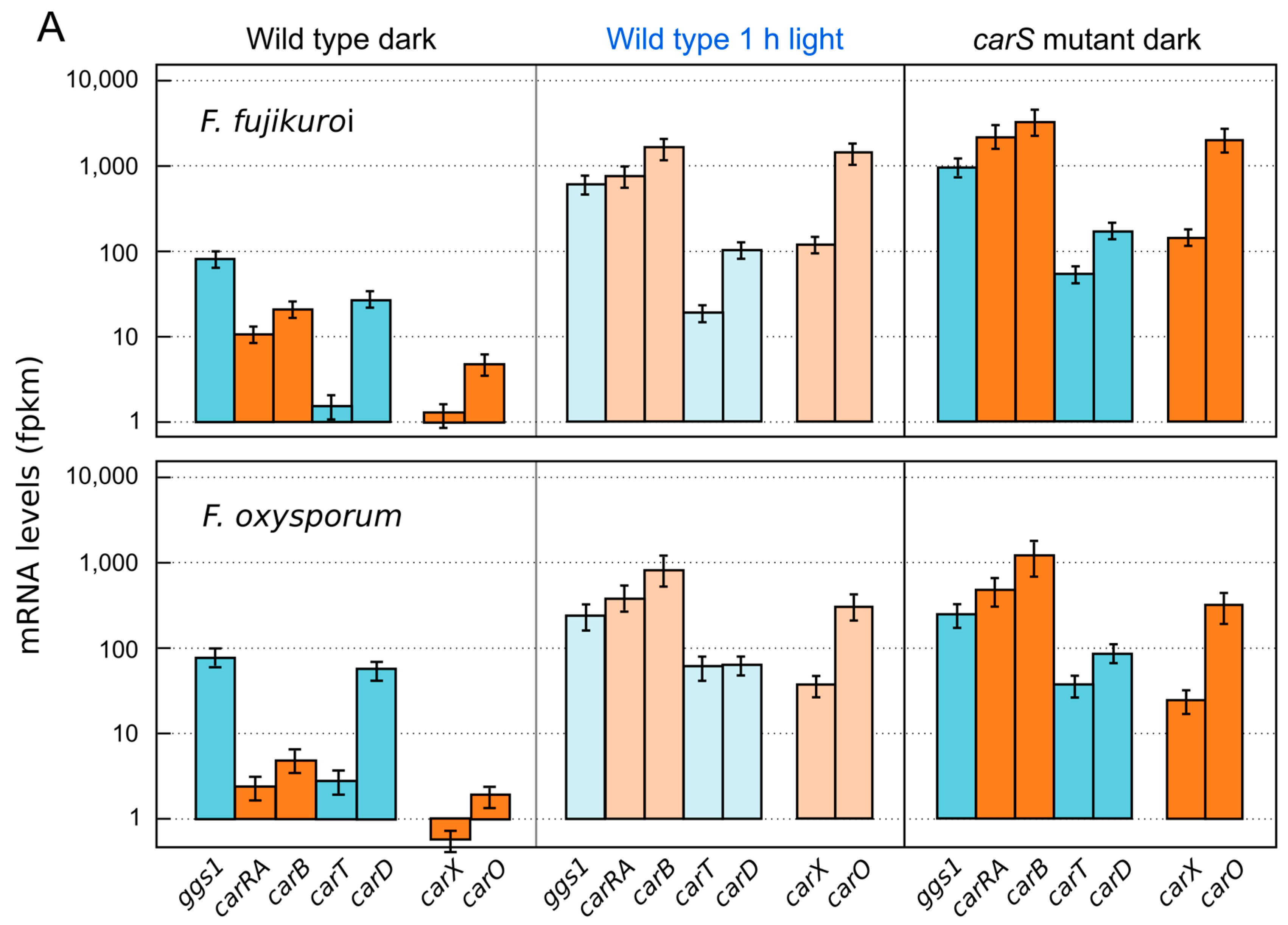
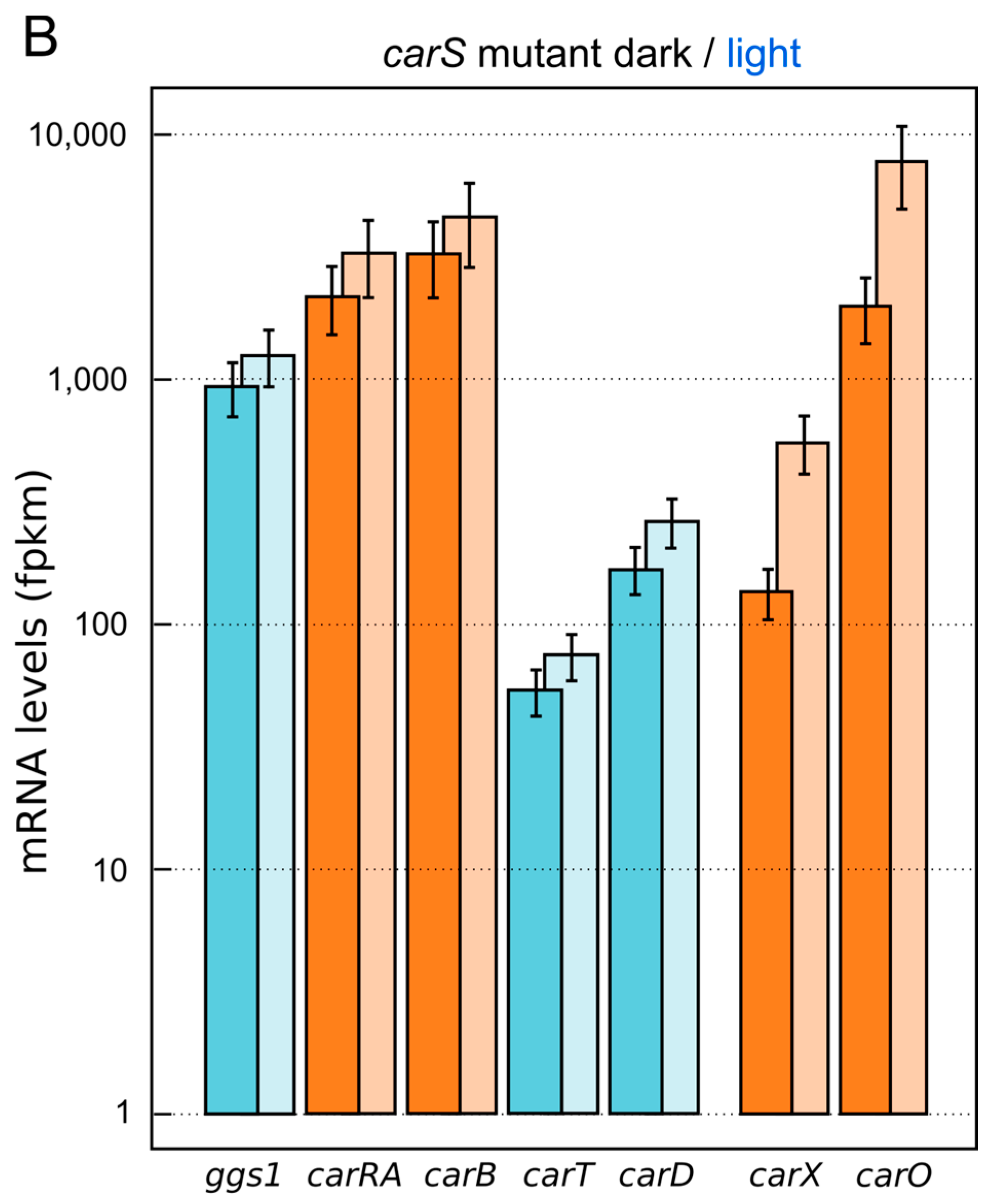
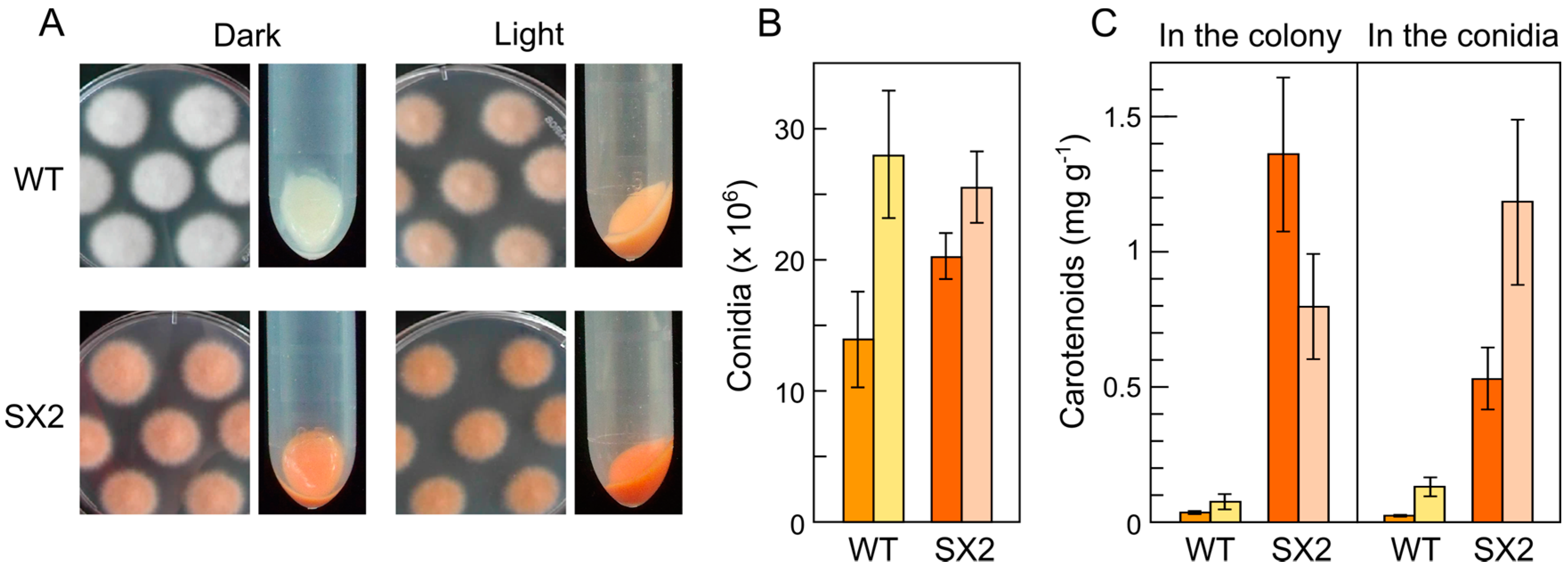
5. Down-Regulation of Carotenoid Production: The CarS Gene
6. Regulation by Light-Independent Factors
7. Conclusions and Future Prospects
Acknowledgments
Conflicts of Interest
References
- Booth, C. The Genus Fusarium; Commonwealth Agricultural Bureaux for the Commonwealth Mycological Institute: Farnham Royal, UK, 1971. [Google Scholar]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Professional: Ames, IA, USA, 2006. [Google Scholar]
- Carlile, M.J. A study of the factors influencing non-genetic variation in a strain of Fusarium oxysporum. J. Gen. Microbiol. 1956, 14, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Britton, G.; Liaaen-Jensen, S.; Pfander, H. Carotenoids; Birkhäuser Verlag: Basel, Switzerland, 1998; Volumes 1 and 2. [Google Scholar]
- Britton, G.; Liaaen-Jensen, S.; Pfander, H. Carotenoids: Handbook; Birkhauser Verlag: Basel, Switzerland, 2004. [Google Scholar]
- Sandmann, G.; Misawa, N. Fungal carotenoids. In The Mycota X. Industrial Applications; Osiewacz, H.D., Ed.; Springer: Berlin, Germany, 2002; pp. 247–262. [Google Scholar]
- Avalos, J.; Díaz-Sánchez, V.; García-Martínez, J.; Castrillo, M.; Ruger-Herreros, M.; Limón, M.C. Carotenoids. In Biosynthesis and Molecular Genetics of Fungal Secondary Metabolites; Martín, J.F., García-Estrada, C., Zeilinger, S., Eds.; Springer: New York, NY, USA, 2014; pp. 149–185. [Google Scholar]
- Phadwal, K. Carotenoid biosynthetic pathway: Molecular phylogenies and evolutionary behavior of crt genes in eubacteria. Gene 2005, 345, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Boronat, A.; Rodríguez-Concepción, M. Terpenoid biosynthesis in prokaryotes. Adv. Biochem. Eng. Biotechnol. 2015, 148, 3–18. [Google Scholar] [PubMed]
- Mandrioli, M.; Rivi, V.; Nardelli, A.; Manicardi, G.C. Genomic and cytogenetic localization of the carotenoid genes in the aphid genome. Cytogenet. Genome Res. 2016, 149, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Domonkos, I.; Kis, M.; Gombos, Z.; Ughy, B. Carotenoids, versatile components of oxygenic photosynthesis. Prog. Lipid Res. 2013, 52, 539–561. [Google Scholar] [CrossRef] [PubMed]
- Blomhoff, R.; Blomhoff, H.K. Overview of retinoid metabolism and function. J. Neurobiol. 2006, 66, 606–630. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.; Limón, M.C. Biological roles of fungal carotenoids. Curr. Genet. 2015, 61, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Rohdich, F.; Kis, K.; Bacher, A.; Eisenreich, W. The non-mevalonate pathway of isoprenoids: Genes, enzymes and intermediates. Curr. Opin. Chem. Biol. 2001, 5, 535–540. [Google Scholar] [CrossRef]
- Rau, W.; Zehender, C. Die Carotinoide von Fusarium aquaeductuum Lagh. Arch. Mikrobiol. 1959, 32, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Zalokar, M. Isolation of an acidic pigment in Neurospora. Arch. Biochem. Biophys. 1957, 70, 568–571. [Google Scholar] [CrossRef]
- Aasen, A.J.; Jensen, S.L. Fungal carotenoids II. The structure of the carotenoid acid neurosporaxanthin. Acta Chem. Scand. 1965, 19, 1843–1853. [Google Scholar] [CrossRef] [PubMed]
- Valadon, L.R.G.; Mummery, R.S. Biosynthesis of neurosporaxanthin. Microbios 1969, 1A, 3–8. [Google Scholar]
- Valadon, L.R.G.; Osman, M.; Mummery, R.S.; Jerebzoff-Quintin, S.; Jerebzoff, S. The effect of monochromatic radiation in the range 350 to 750 nm on the carotenogenesis in Verticillium agaricinum. Physiol. Plant. 1982, 56, 199–203. [Google Scholar] [CrossRef]
- Strobel, I.; Breitenbach, J.; Scheckhuber, C.Q.; Osiewacz, H.D.; Sandmann, G. Carotenoids and carotenogenic genes in Podospora anserina: Engineering of the carotenoid composition extends the life span of the mycelium. Curr. Genet. 2009, 55, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Bindl, E.; Lang, W.; Rau, W. Untersuchungen über die lichtabhängige Carotinoidsynthese. VI. Zeitlicher Verlauf der Synthese der einzelnen Carotinoide bei Fusarium aquaeductuum unter verschiedenen Induktionsbedingungen. Planta 1970, 94, 156–174. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.; Cerdá-Olmedo, E. Carotenoid mutants of Gibberella fujikuroi. Curr. Genet. 1987, 25, 1837–1841. [Google Scholar] [CrossRef]
- Sakaki, H.; Kaneno, H.; Sumiya, Y.; Tsushima, M.; Miki, W.; Kishimoto, N.; Fujita, T.; Matsumoto, S.; Komemushi, S.; Sawabe, A. A new carotenoid glycosyl ester isolated from a marine microorganism, Fusarium strain T-1. J. Nat. Prod. 2002, 65, 1683–1684. [Google Scholar] [CrossRef] [PubMed]
- Woitek, S.; Unkles, S.E.; Kinghorn, J.R.; Tudzynski, B. 3-Hydroxy-3-methylglutaryl-CoA reductase gene of Gibberella fujikuroi: Isolation and characterization. Curr. Genet. 1997, 31, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Homann, V.; Mende, K.; Arntz, C.; Ilardi, V.; Macino, G.; Morelli, G.; Bose, G.; Tudzynski, B. The isoprenoid pathway: Cloning and characterization of fungal FPPS genes. Curr. Genet. 1996, 30, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Mende, K.; Homann, V.; Tudzynski, B. The geranylgeranyl diphosphate synthase gene of Gibberella fujikuroi: Isolation and expression. Mol. Gen. Genet. 1997, 255, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Tudzynski, B. Gibberellin biosynthesis in fungi: Genes, enzymes, evolution, and impact on biotechnology. Appl. Microbiol. Biotechnol. 2005, 66, 597–611. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Martín, R.; Cerdá-Olmedo, E.; Avalos, J. Homologous recombination and allele replacement in transformants of Fusarium fujikuroi. Mol. Gen. Genet. 2000, 263, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Schmidhauser, T.J.; Lauter, F.R.; Russo, V.E.; Yanofsky, C. Cloning, sequence, and photoregulation of al-1, a carotenoid biosynthetic gene of Neurospora crassa. Mol. Cell. Biol. 1990, 10, 5064–5070. [Google Scholar] [CrossRef] [PubMed]
- Prado-Cabrero, A.; Schaub, P.; Díaz-Sánchez, V.; Estrada, A.F.; Al-Babili, S.; Avalos, J. Deviation of the neurosporaxanthin pathway towards β-carotene biosynthesis in Fusarium fujikuroi by a point mutation in the phytoene desaturase gene. FEBS J. 2009, 276, 4582–4597. [Google Scholar] [CrossRef] [PubMed]
- Hausmann, A.; Sandmann, G. A single five-step desaturase is involved in the carotenoid biosynthesis pathway to β-carotene and torulene in Neurospora crassa. Fungal Genet. Biol. 2000, 30, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Schmidhauser, T.J.; Lauter, F.R.; Schumacher, M.; Zhou, W.; Russo, V.E.; Yanofsky, C. Characterization of al-2, the phytoene synthase gene of Neurospora crassa. Cloning, sequence analysis, and photoregulation. J. Biol. Chem. 1994, 269, 12060–12066. [Google Scholar] [PubMed]
- Linnemannstöns, P.; Prado, M.M.; Fernández-Martín, R.; Tudzynski, B.; Avalos, J. A carotenoid biosynthesis gene cluster in Fusarium fujikuroi: the genes carB and carRA. Mol. Genet. Genomics 2002, 267, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Verdoes, J.C.; Krubasik, P.; Sandmann, G.; van Ooyen, A.J.J. Isolation and functional characterisation of a novel type of carotenoid biosynthetic gene from Xanthophyllomyces dendrorhous. Mol. Gen. Genet. 1999, 262, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Arrach, N.; Schmidhauser, T.J.; Avalos, J. Mutants of the carotene cyclase domain of al-2 from Neurospora crassa. Mol. Genet. Genom. 2002, 266, 914–921. [Google Scholar]
- Díaz-Sánchez, V.; Estrada, A.F.; Trautmann, D.; Limón, M.C.; Al-Babili, S.; Avalos, J. Analysis of al-2 mutations in Neurospora. PLoS ONE 2011, 6, e21948. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.; Kiser, P.D.; von Lintig, J.; Palczewski, K. Structural basis of carotenoid cleavage: From bacteria to mammals. Arch. Biochem. Biophys. 2013, 539, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Thewes, S.; Prado-Cabrero, A.; Prado, M.M.; Tudzynski, B.; Avalos, J. Characterization of a gene in the car cluster of Fusarium fujikuroi that codes for a protein of the carotenoid oxygenase family. Mol. Genet. Genom. 2005, 274, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Prado-Cabrero, A.; Scherzinger, D.; Avalos, J.; Al-Babili, S. Retinal biosynthesis in fungi: Characterization of the carotenoid oxygenase CarX from Fusarium fujikuroi. Eukaryot. Cell 2007, 6, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Estrada, A.F.; Avalos, J. Regulation and targeted mutation of opsA, coding for the NOP-1 opsin orthologue in Fusarium fujikuroi. J. Mol. Biol. 2009, 387, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Sánchez, V.; Limón, M.C.; Schaub, P.; Al-Babili, S.; Avalos, J. A RALDH-like enzyme involved in Fusarium verticillioides development. Fungal Genet. Biol. 2016, 86, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Prado-Cabrero, A.; Estrada, A.F.; Al-Babili, S.; Avalos, J. Identification and biochemical characterization of a novel carotenoid oxygenase: Elucidation of the cleavage step in the Fusarium carotenoid pathway. Mol. Microbiol. 2007, 64, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Saelices, L.; Youssar, L.; Holdermann, I.; Al-Babili, S.; Avalos, J. Identification of the gene responsible for torulene cleavage in the Neurospora carotenoid pathway. Mol. Genet. Genom. 2007, 278, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.M.; Lee, J.; Lee, Y.W. Characterization of carotenoid biosynthetic genes in the ascomycete Gibberella zeae. FEMS Microbiol. Lett. 2010, 302, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Goldie, A.H.; Subden, R.E. The neutral carotenoids of wild-type and mutant strains of Neurospora crassa. Biochem. Genet. 1973, 10, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Sandmann, G. Photoregulation of carotenoid biosynthesis in mutants of Neurospora crassa: Activities of enzymes involved in the synthesis and conversion of phytoene. Z. Naturforsch. 1993, 48c, 570–574. [Google Scholar]
- Estrada, A.F.; Youssar, L.; Scherzinger, D.; Al-Babili, S.; Avalos, J. The ylo-1 gene encodes an aldehyde dehydrogenase responsible for the last reaction in the Neurospora carotenoid pathway. Mol. Microbiol. 2008, 69, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Sánchez, V.; Estrada, A.F.; Trautmann, D.; Al-Babili, S.; Avalos, J. The gene carD encodes the aldehyde dehydrogenase responsible for neurosporaxanthin biosynthesis in Fusarium fujikuroi. FEBS J. 2011, 278, 3164–3176. [Google Scholar] [CrossRef] [PubMed]
- Rau, W. Über den Einfluss der Temperatur auf die lichtabhängige Carotinoidbildung von Fusarium aquaeductuum. Planta 1962, 59, 123–137. [Google Scholar] [CrossRef]
- Rau, W. Untersuchungen über die lichtabhängige Carotinoidsynthese. II. Ersatz der Lichtinduktion durch Mercuribenzoat. Planta 1967, 74, 263–277. [Google Scholar] [CrossRef] [PubMed]
- Rau, W. Untersuchungen über die lichtabhängige Carotinoidsynthese. VII. Reversible Unterbrechung der Reaktionskette durch Cycloheximid und anaerobe Bedingungen. Planta 1971, 101, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Rau, W.; Feuser, B.; Rau-Hund, A. Substitution of p-chloro- or p-hydroxymercuribenzoate for light in carotenoid synthesis by Fusarium aquaeductuum. Biochim. Biophys. Acta 1967, 136, 589–590. [Google Scholar] [CrossRef]
- Theimer, R.R.; Rau, W. Untersuchungen über die lichtabhängige Carotinoidsynthese V. Aufhebung der Lichtinduktion dutch Reduktionsmittel und Ersatz des Lichts durch Wasserstoffperoxid. Planta 1970, 92, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Theimer, R.R.; Rau, W. Untersuchungen über die lichtabhängige Carotinoidsynthese VIII. Die unterschiedlichen Wirkungsmechanismen von Licht und Mercuribenzoat. Planta 1972, 106, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.; Cerdá-Olmedo, E. Chemical modification of carotenogenesis in Gibberella fujikuroi. Phytochemistry 1986, 25, 1837–1841. [Google Scholar] [CrossRef]
- Rau, W. Untersuchungen über die lichtabhängige Carotinoidsynthese. I. Das Wirkungsspektrum von Fusarium aquaeductuum. Planta 1967, 72, 14–28. [Google Scholar] [CrossRef] [PubMed]
- Schrott, E.L.; Huber-Willer, A.; Rau, W. Is phytochrome involved in the light-mediated carotenogenesis in Fusarium aquaeductuum and Neurospora crassa? Photochem. Photobiol. 1982, 35, 213–216. [Google Scholar] [CrossRef]
- Lang-Feulner, J.; Rau, W. Redox dyes as artificial photoreceptors in light-dependent carotenoid synthesis. Photochem. Photobiol. 1975, 21, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Prado, M.M.; Prado-Cabrero, A.; Fernández-Martín, R.; Avalos, J. A gene of the opsin family in the carotenoid gene cluster of Fusarium fujikuroi. Curr. Genet. 2004, 46, 47–58. [Google Scholar] [CrossRef] [PubMed]
- De Fabo, E.C.; Harding, R.W.; Shropshire, W., Jr. Action spectrum between 260 and 800 nanometers for the photoinduction of carotenoid biosynthesis in Neurospora crassa. Plant Physiol. 1976, 57, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.; Aguirre, J.; Herrera-Estrella, A.; Corrochano, L.M. The complexity of fungal vision. Microbiol. Spectr. 2016, 4, 1–22. [Google Scholar]
- Estrada, A.F.; Avalos, J. The White Collar protein WcoA of Fusarium fujikuroi is not essential for photocarotenogenesis, but is involved in the regulation of secondary metabolism and conidiation. Fungal Genet. Biol. 2008, 45, 705–718. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Roldán, M.C.; Garre, V.; Guarro, J.; Mariné, M.; Roncero, M.I. Role of the white collar 1 photoreceptor in carotenogenesis, UV resistance, hydrophobicity, and virulence of Fusarium oxysporum. Eukaryot. Cell 2008, 7, 1227–1230. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Son, H.; Lee, Y.-W. Effects of light on secondary metabolism and fungal development of Fusarium graminearum. J. Appl. Microbiol. 2014, 116, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Castrillo, M.; Avalos, J. The flavoproteins CryD and VvdA cooperate with the white collar protein WcoA in the control of photocarotenogenesis in Fusarium fujikuroi. PLoS ONE 2015, 10, e0119785. [Google Scholar] [CrossRef] [PubMed]
- Castrillo, M.; Bernhardt, A.; Avalos, J.; Batschauer, A.; Pokorny, R. Biochemical characterization of the DASH-type cryptochrome CryD from Fusarium fujikuroi. Photochem. Photobiol. 2015, 91, 1356–1367. [Google Scholar] [CrossRef] [PubMed]
- Castrillo, M.; García-Martínez, J.; Avalos, J. Light-dependent functions of the Fusarium fujikuroi CryD DASH cryptochrome in development and secondary metabolism. Appl. Environ. Microbiol. 2013, 79, 2777–2788. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ortiz, R.; Michielse, C.; Rep, M.; Limón, M.C.; Avalos, J. Genetic basis of carotenoid overproduction in Fusarium oxysporum. Fungal Genet. Biol. 2012, 49, 684–696. [Google Scholar] [CrossRef] [PubMed]
- Ádám, A.L.; García-Martínez, J.; Szücs, E.P.; Avalos, J.; Hornok, L. The MAT1–2-1 mating-type gene upregulates photo-inducible carotenoid biosynthesis in Fusarium verticillioides. FEMS Microbiol. Lett. 2011, 318, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.; Corrochano, L.M. Carotenoid biosynthesis in Neurospora. In Neurospora: Genomics and Molecular Biology; Kasbekar, D.P., McCluskey, K., Eds.; Caister Academic Press: Norfolk, UK, 2013; pp. 227–241. [Google Scholar]
- Ruger-Herreros, M. Participación de la Proteína CarS en la Regulación de la Carotenogénesis y el Estrés en Fusarium fujikuroi. Ph.D. Thesis, Universidad de Sevilla, Seville, Spain, 2016. [Google Scholar]
- Castrillo, M.; Avalos, J. Light-mediated participation of the VIVID-like protein of Fusarium fujikuroi VvdA in pigmentation and development. Fungal Genet. Biol. 2014, 71, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.; Casadesús, J.; Cerdá-Olmedo, E. Gibberella fujikuroi mutants obtained with UV radiation and N-methyl-N’-nitro-N-nitrosoguanidine. Appl. Environ. Microbiol. 1985, 49, 187–191. [Google Scholar] [PubMed]
- Avalos, J.; Mackenzie, A.; Nelki, D.S.; Bramley, P.M. Terpenoid biosynthesis in cell-extracts of wild type and mutant strains of Gibberella fujikuroi. Biochim. Biophys. Acta 1988, 966, 257–265. [Google Scholar] [CrossRef]
- Delgado-Pelayo, R.; Hornero-Méndez, D. Identification and quantitative analysis of carotenoids and their esters from sarsaparrilla (Smilax aspera L.) berries. J. Agric. Food Chem. 2012, 60, 8225–8232. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ortiz, R.; Limón, M.C.; Avalos, J. Functional analysis of the carS gene of Fusarium fujikuroi. Mol. Genet. Genom. 2013, 288, 157–173. [Google Scholar] [CrossRef] [PubMed]
- Navarro, E.; Lorca-Pascual, J.M.; Quiles-Rosillo, M.D.; Nicolás, F.E.; Garre, V.; Torres-Martínez, S.; Ruiz-Vázquez, R.M. A negative regulator of light-inducible carotenogenesis in Mucor circinelloides. Mol. Genet. Genom. 2001, 266, 463–470. [Google Scholar]
- Lorca-Pascual, J.M.; Murcia-Flores, L.; Garre, V.; Torres-Martínez, S.; Ruiz-Vázquez, R.M. The RING-finger domain of the fungal repressor crgA is essential for accurate light regulation of carotenogenesis. Mol. Microbiol. 2004, 52, 1463–1474. [Google Scholar] [CrossRef] [PubMed]
- Navarro, E.; Ruiz-Pérez, V.L.; Torres-Martínez, S. Overexpression of the crgA gene abolishes light requirement for carotenoid biosynthesis in Mucor circinelloides. Eur. J. Biochem. 2000, 267, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.; Navarro, E.; Peñaranda, A.; Murcia-Flores, L.; Torres-Martínez, S.; Garre, V. A RING-finger protein regulates carotenogenesis via proteolysis-independent ubiquitylation of a White Collar-1-like activator. Mol. Microbiol. 2008, 70, 1026–1036. [Google Scholar] [CrossRef] [PubMed]
- Quiles-Rosillo, M.D.; Torres-Martínez, S.; Garre, V. Ciga, a light-inducible gene involved in vegetative growth in Mucor circinelloides is regulated by the carotenogenic repressor crgA. Fungal Genet. Biol. 2003, 38, 122–132. [Google Scholar] [CrossRef]
- Murcia-Flores, L.; Lorca-Pascual, J.M.; Garre, V.; Torres-Martínez, S.; Ruiz-Vázquez, R.M. Non-AUG translation initiation of a fungal RING finger repressor involved in photocarotenogenesis. J. Biol. Chem. 2007, 282, 15394–15403. [Google Scholar] [CrossRef] [PubMed]
- Candau, R.; Avalos, J.; Cerdá-Olmedo, E. Gibberellins and carotenoids in the wild type and mutants of Gibberella fujikuroi. Appl. Environ. Microbiol. 1991, 57, 3378–3382. [Google Scholar] [PubMed]
- Rodríguez-Ortiz, R.; Limón, M.C.; Avalos, J. Regulation of carotenogenesis and secondary metabolism by nitrogen in wild-type Fusarium fujikuroi and carotenoid-overproducing mutants. Appl. Environ. Microbiol. 2009, 75, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Studt, L.; Tudzynski, B. Gibberellins and the red pigments bikaverin and fusarubin. In Biosynthesis and Molecular Genetics of Fungal Secondary Metabolites; Martín, J.-F., García-Estrada, C., Zeilinger, S., Eds.; Springer: New York, NY, USA, 2014; pp. 209–238. ISBN 978-1-4939-1190-5. [Google Scholar]
- Niehaus, E.-M.; Díaz-Sánchez, V.; von Bargen, K.W.; Kleigrewe, K.; Humpf, H.-U.; Limón, M.C.; Tudzynski, B. Fusarins and Fusaric Acid in Fusaria. In Biosynthesis and Molecular Genetics of Fungal Secondary Metabolites; Martín, J.-F., García-Estrada, C., Zeilinger, S., Eds.; Springer: New York, NY, USA, 2014; pp. 239–262. ISBN 978-1-4939-1190-5. [Google Scholar]
- Garbayo, I.; Vílchez, C.; Nava-Saucedo, J.E.; Barbotin, J.N. Nitrogen, carbon and light-mediated regulation studies of carotenoid biosynthesis in immobilized mycelia of Gibberella fujikuroi. Enzyme Microb. Technol. 2003, 33, 629–634. [Google Scholar] [CrossRef]
- Sokolovsky, V.Y.; Lauter, F.R.; Müller-Röber, B.; Ricci, M.; Schmidhauser, T.J.; Russo, V.E.A. Nitrogen regulation of blue light-inducible genes in Neurospora crassa. J. Gen. Microbiol. 1992, 138, 2045–2049. [Google Scholar] [CrossRef]
- Connolly, L.R.; Smith, K.M.; Freitag, M. The Fusarium graminearum histone H3 K27 methyltransferase KMT6 regulates development and expression of secondary metabolite gene clusters. PLoS Genet. 2013, 9, e1003916. [Google Scholar] [CrossRef] [PubMed]
- Wiemann, P.; Sieber, C.M.; von Bargen, K.W.; Studt, L.; Niehaus, E.M.; Espino, J.J.; Huss, K.; Michielse, C.B.; Albermann, S.; Wagner, D.; et al. Deciphering the cryptic genome: Genome-wide analyses of the rice pathogen Fusarium fujikuroi reveal complex regulation of secondary metabolism and novel metabolites. PLoS Pathog. 2013, 9, e1003475. [Google Scholar] [CrossRef] [PubMed]
- García-Martínez, J.; Ádám, A.L.; Avalos, J. Adenylyl cyclase plays a regulatory role in development, stress resistance and secondary metabolism in Fusarium fujikuroi. PLoS ONE 2012, 7, e28849. [Google Scholar] [CrossRef] [PubMed]
- Studt, L.; Humpf, H.-U.; Tudzynski, B. Signaling governed by G proteins and cAMP is crucial for growth, secondary metabolism and sexual development in Fusarium fujikuroi. PLoS ONE 2013, 8, e58185. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ortiz, R. Análisis Genético y Molecular del Fenotipo CarS en Fusarium. Ph.D. Thesis, Universidad de Sevilla, Seville, Spain, 2012. [Google Scholar]
- Domenech, C.E.; Giordano, W.; Avalos, J.; Cerdá-Olmedo, E. Separate compartments for the production of sterols, carotenoids and gibberellins in Gibberella fujikuroi. Eur. J. Biochem. 1996, 239, 720–725. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avalos, J.; Pardo-Medina, J.; Parra-Rivero, O.; Ruger-Herreros, M.; Rodríguez-Ortiz, R.; Hornero-Méndez, D.; Limón, M.C. Carotenoid Biosynthesis in Fusarium. J. Fungi 2017, 3, 39. https://doi.org/10.3390/jof3030039
Avalos J, Pardo-Medina J, Parra-Rivero O, Ruger-Herreros M, Rodríguez-Ortiz R, Hornero-Méndez D, Limón MC. Carotenoid Biosynthesis in Fusarium. Journal of Fungi. 2017; 3(3):39. https://doi.org/10.3390/jof3030039
Chicago/Turabian StyleAvalos, Javier, Javier Pardo-Medina, Obdulia Parra-Rivero, Macarena Ruger-Herreros, Roberto Rodríguez-Ortiz, Dámaso Hornero-Méndez, and María Carmen Limón. 2017. "Carotenoid Biosynthesis in Fusarium" Journal of Fungi 3, no. 3: 39. https://doi.org/10.3390/jof3030039
APA StyleAvalos, J., Pardo-Medina, J., Parra-Rivero, O., Ruger-Herreros, M., Rodríguez-Ortiz, R., Hornero-Méndez, D., & Limón, M. C. (2017). Carotenoid Biosynthesis in Fusarium. Journal of Fungi, 3(3), 39. https://doi.org/10.3390/jof3030039