New Trends in Paracoccidioidomycosis Epidemiology
Abstract
:1. Introduction
2. Prevalence, Incidence, Lethality, and Mortality
3. Predisposing and Modulating Factors
3.1. Gender
3.2. Age
3.3. Ethnicity
3.4. Genetic Variability
3.5. Exposure in Rural Areas
3.6. Smoking, Alcohol Drinking, and Life Style
4. Relationship with Cancer, AIDS, and Chronic Infections
5. Paracoccidioidomycosis-Infection
6. Consolidated Endemic Areas and Expansion of the Endemy
7. Additional Causes of Epidemiological Modification
Acknowledgments
Conflicts of Interest
References
- Lacaz, C.S. Historical Evolution of the knowledge on paracoccidioidomycosis and its etiologic agent, Paracoccidioides brasiliensis. In Paracoccidioidomycosis, 1st ed.; Franco, M., da Lacaz, C.S., Restrepo-Moreno, A., Del Negro, G., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 1–11. [Google Scholar]
- Teixeira, M.M.; Theodoro, R.C.; Nino-Vega, G.; Bagagli, E.; Felipe, M.S. Paracoccidioides species complex: Ecology, phylogeny sexual reproduction and virulence. PLoS Pathog. 2014, 10, e1004397. [Google Scholar] [CrossRef] [PubMed]
- Arantes, T.D.; Theodoro, R.C.; de Melo Teixeira, M.; de Moraes Gimenes Bosco, S.; Bagagli, E. Environmental mapping of Paracoccidioides spp. in Brazil reveals new clues into genetic diversity, biogeography and wild host association. PLoS Negl. Trop. Dis. 2016, 10, e0004606. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.; Bagagli, E.; Scapolio, S.; da Silva Lacaz, C. A critical analysis of isolation of Paracoccidioides brasiliensis from soil. Med. Mycol. 2000, 38, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, A.; McEwen, J.G.; Castañeda, E. The habitat of Paracoccidioides brasiliensis: How far from solving the riddle? Med. Mycol. 2001, 39, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Bellíssimo-Rodrigues, F.; Machado, A.A.; Martinez, R. Paracoccidioidomycosis epidemiological features of a 1000-cases series from a hyperendemic area on the Southeast of Brazil. Am. J. Trop. Med. Hyg. 2011, 85, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Cadavid, D.; Restrepo, A. Factors associated with Paracoccidioides brasiliensis infection among permanent residents of three endemic areas in Colombia. Epidemiol. Infect. 1993, 111, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Bagagli, E.; Franco, M.; Bosco Sde, M.; Hebeler-Barbosa, F.; Trinca, L.A.; Montenegro, M.R. High frequency of Paracoccidioides brasiliensis infection in armadillos: An ecological study. Med. Mycol. 2003, 41, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Corredor, G.G.; Castaño, J.H.; Peralta, L.A.; Diéz, S.; Arango, M.; McEwen, J.; Restrepo, A. Isolation of Paracoccidioides brasiliensis from nine-banded armadillo Dasypus novemcinctus, in a endemic area for paracoccidioidomycosis in Colombia. Rev. Iberoam. Micol. 1999, 16, 216–220. [Google Scholar] [PubMed]
- Buccheri, R.; Khouri, Z.; Barata, L.C.; Benard, G. Incubation period and early natural history events of the acute form of paracoccidioidomycosis: Lessons from patients with single Paracoccidioides spp. exposure. Mycopathologia 2016, 181, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Bellíssimo-Rodrigues, F.; Bollela, V.R.; da Fonseca, B.A.L.; Martinez, R. Endemic paracoccidioidomycosis: Relationship between clinical presentation and patients’ demographic features. Med. Mycol. 2013, 51, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Talhari, S.; Cunha, M.G.; Sehettini, A.P.; Talhari, A.C. Deep mycoses in Amazon region. Int. J. Dermatol. 1988, 27, 481–484. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, E.R.; Pardal, P.P.; Severo, L.C. Paracoccidioidomicose em crianças em Belém do Pará. Rev. Soc. Bras. Med. Trop. 1999, 32, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Vieira, G.D.; Alves, T.C.; Lima, S.M.D.; Souza, C.M. Paracoccidioidomycosis in a western Brazilian Amazon State: Clinical-epidemiologic profile and spatial distribution of the disease. Rev. Soc. Bras. Med. Trop. 2014, 47, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Matos, W.B.; Dos Santos, G.M.C.; Silva, V.E.B.; Rosário Gonçalves, E.G.; Silva, A.R. Paracoccidioidomycosis in the state of Maranhão, Brazil: Geographic and clinical aspects. Rev. Soc. Bras. Med. Trop. 2012, 45, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Barrozo, L.V.; Mendes, R.P.; Marques, S.A.; Benard, G.; Siqueira Silva, M.E.; Bagagli, E. Climate and acute/subacute paracoccidioidomycosis in a hyperendemic area in Brazil. Int. J. Epidemiol. 2009, 38, 1642–1649. [Google Scholar] [CrossRef] [PubMed]
- Blotta, M.H.; Mamoni, R.I.; Oliveira, S.J.; Nouer, S.A.; Papaiordanou, P.M.; Goveia, A.; Camargo, Z.P. Endemic regions of paracoccidioidomycosis in Brazil: A clinical and epidemiologic study of 584 cases in the southeast region. Am. J. Trop. Med. Hyg. 1999, 61, 390–394. [Google Scholar] [PubMed]
- Lacaz, C.S. South American blastomycosis. An. Fac. Med. Univ. Sao Paulo 1956, 29, 9–120. [Google Scholar]
- Neves, F.F.; Gerolin, G.P.; Tavares, M.G.; Castro-Silva, M.H.; Lopes, G.P.; Michelin, M.A.; Silva-Vergara, M.L. Epidemiological and clinical profile of 137 patients with paracoccidioidomycosis in Uberaba, Minas Gerais, Brazil. In Proceedings of the X International Congress on Paracoccidioidomycosis, Medellin, Colombia, 7–10 August 2008; Biomédica: Medellin, Colombia; Volume 28, p. 144.
- Valle, A.C.F.; Wanke, B.; Fernandes, N.C.; Peixoto, T.C.; Perez, M. Tratamento da paracoccidioidomicose: Estudo retrospectivo de 500 casos. I. Análise clínica, laboratorial e epidemiológica. An. Bras. Dermatol. 1992, 67, 251–254. [Google Scholar]
- Londero, A.T.; Ramos, C.D. Paracoccidioidomicose. Estudo clínico e micológico de 260 casos observados no interior do Estado do Rio Grande do Sul. J. Pneumol. 1990, 16, 129–132. [Google Scholar]
- Loth, E.A.; Castro, S.V.; Silva, J.R.; Gandra, R.F. Ocurrence of 102 cases of paracoccidioidomycosis in 18 months in the Itaipu Lake region, Western Paraná. Rev. Soc. Bras. Med. Trop. 2011, 44, 636–637. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues Gda, S.; Severo, C.B.; Oliveira Fde, M.; Moreira Jda, S.; Prolla, J.C.; Severo, L.C. Association between paracoccidioidomycosis and cancer. J. Bras. Pneumol. 2010, 36, 356–362. [Google Scholar] [PubMed]
- de Souza, S.P.; Jorge, V.M.; Xavier, M.O. Paracoccidioidomycosis in southern Rio Grande do Sul: A retrospective study of histopathologically diagnosed cases. Braz. J. Microbiol. 2014, 45, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Andrade, A.L.S.S. Paracoccidioidomicose linfático-abdominal. Contribuição ao seu estudo. Rev. Patol. Trop. 1983, 12, 165–256. [Google Scholar]
- Campos, M.V.; Penna, G.O.; Castro, C.N.; Moraes, M.A.; Ferreira, M.S.; Santos, J.B. Paracoccidioidomicose no hospital universitário de Brasília. Rev. Soc. Bras. Med. Trop. 2008, 41, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Fabris, L.R.; Andrade, U.V.; Ferreira dos Santos, A.; Marques, A.P.; Oliveira, S.M.; Mendes, R.P.; Paniago, A.M. Decreasing prevalence of the acute/subacute clinical form of paracoccidioidomycosis in Mato Grosso do Sul State, Brazil. Rev. Inst. Med. Trop. Sao Paulo 2014, 56, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Davel, C.; Canteros, C.E. Situación de lãs micosis em La República Argentina. Rev. Argent. Microbiol. 2007, 39, 28–33. [Google Scholar] [PubMed]
- Calle, D.; Rosero, D.S.; Orozco, L.C.; Camargo, D.; Castañeda, E.; Restrepo, A. Paracoccidioidomycosis in Colombia: An ecological study. Epidemiol. Infect. 2001, 126, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Martínez Mendéz, D.; Hernández Valles, R.; Alvarado, P.; Mendoza, M. Las micosis en Venezuela: Casuística de lós grupos de trabajos em micologia (1984–2010). Rev. Iberoam. Micol. 2013, 30, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Lazo, R.F.; Fernández, T.; Mera, R. Prevalencia en la paracoccidioidomycosis e histoplasmosis em la cuenca Del Rio Guayas. Rev. Ecuat. Hyg. Med. Trop. 1987, 37, 15–35. [Google Scholar]
- Rolón, P.A. Paracoccidioidomycosis: Epidemiologia en la República Del Paraguay, centro Sud América. Mycopathologia 1976, 59, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Burstein, Z. Aspectos clínicos de la Blastomicosis sudamericana (Paracoccidioidomicosis) en el Perú. Rev. Peru. Exp. Salud Publica 2002, 19, 43–47. [Google Scholar]
- López-Martínez, R.; Hernandez-Hernandez, F.; Mendez-Toiar, L.J.; Manzano-Gayosso, P.; Bonilfaz, A.; Arenas, R.; Padilla-Desgarennes Mdel, C.; Estrada, R.; Chávez, G. Paracoccidioidomycosis in Mexico: Clinical and epidemiological data from 93 new cases (1972–2012). Mycoses 2014, 57, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Conti-Diáz, I.A.; Calegari, L.F. Paracoccidioidomycosis en Uruguay: Su estado y problemática actuales. Bol. Oficina Sanit. Panam. 1979, 86, 219–229. [Google Scholar] [PubMed]
- Prado, M.; Silva, M.B.; Laurenti, R.; Travassos, L.R.; Taborda, C.P. Mortality due to systemic mycosis as a primary cause of death or in association with AIDS in Brazil: Review from 1996 to 2006. Mem. Inst. Oswaldo Cruz 2009, 104, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, Z.F.; Wanke, B.; Travassos, C.; Oliveira, R.M.; Xavier, D.R.; Coimbra, C.E., Jr. Hospital morbidity due to paracoccidioidomycosis in Brazil (1998–2006). Trop. Med. Int. Health 2015, 20, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Greer, D.L.; Restrepo, A.M. La epidemiologia de la paracoccidioidomycosis. Bol. Oficina Sanit. Panam. 1977, 82, 428–445. [Google Scholar]
- Paniago, A.M.M.; Aguiar, J.I.A.; Aguiar, E.S.; da Cunha, R.V.; Pereira, G.R.; Londero, A.T.; Wanke, B. Paracoccidioidomicose: Estudo clínico e epidemiológico de 422 casos observados no Estado de Mato Grosso do Sul. Rev. Soc. Bras. Med. Trop. 2003, 36, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Queiroz-Telles, F.; Goldani, L.Z.; Schlamm, H.T.; Goodrich, J.M.; Espinel-Ingroff, A.; Shikanai-Yasuda, M.A. An open label comparative study of oral voriconazole and itraconazole for long-term treatment of paracoccidioidomycosis. Clin. Infect. Dis. 2007, 45, 1462–1469. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, Z.F.; da Silva, D.; Lazera, M.; Petri, V.; Oliveira, R.M.; Sabroza, P.C.; Wanke, B. Paracoccidioidomycosis mortality in Brazil (1980–1995). Cad. Saude Publica 2002, 18, 1441–1454. [Google Scholar] [CrossRef] [PubMed]
- Shankar, J.; Restrepo, A.; Clemons, K.V.; Stevens, D.A. Hormones and the resistance of women to paracoccidioidomycosis. Clin. Microbiol. Rev. 2011, 24, 296–313. [Google Scholar] [CrossRef] [PubMed]
- Loth, E.A.; Cecatto, V.; Biazim, S.K.; Ferreira, J.H.; Danielli, C.; Genske, R.D.; Gandra, R.F.; Franco, M.F. Experimental paracoccidioidomycosis in pregnant rats. Rev. Inst. Med. Trop. Sao Paulo 2015, 57, 515–518. [Google Scholar] [CrossRef] [PubMed]
- De Messias, I.J.; Reis, A.; Brenden, M.; Queiroz-Telles, F.; Mauff, G. Association of major histocompatibility complex class III complement components C2, BF, and C4 with Brazilian paracoccidioidomycosis. Complement. Inflamm. 1991, 8, 288–293. [Google Scholar] [PubMed]
- Carvalho, F.M.; Busser, F.D.; Freitas, V.L.; Furucho, C.R.; Sadahiro, A.; Kono, A.S.; Criado, P.R.; Moretti, M.L.; Sato, P.K.; Shikanai-Yasuda, M.A. Polymorphisms on IFNG, IL12B and IL12RB1 genes and paracoccidioidomycosis in the Brazilian population. Infect. Genet. Evol. 2016, 43, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Coimbra Junior, C.F.; Wanke, B.; Santos, R.V.; do Valle, A.C.; Costa, R.L.; Zancopé-Oliveira, R.M. Paracoccidioidin and histoplasmin sensitivy in Tupi-Mondé Ameridian populations from Brazilian Amazonia. Ann. Trop. Med. Parasitol. 1994, 88, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, W.A.; da Silva, B.M.; Passos, E.D.; Zandonade, E.; Falquete, A. Associação entre tabagismo e paracoccidioidomicose: Um estudo de caso-controle no Estado do Espírito Santo, Brasil. Cad. Saude Publica 2003, 19, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Quagliato Junior, R.; Granjeia Tde, A.; Massucia, R.A.; De Capitani, E.M.; Rezende Sde, M.; Balthazar, A.B. Association between paracoccidioidomycosis and tuberculosis: Reality and misdiagnosis. J. Bras. Pneumol. 2007, 33, 295–300. [Google Scholar] [PubMed]
- Shikanai-Yasuda, M.A.; Conceição, Y.M.; Kono, A.; Rivitti, E.; Campos, A.F.; Campos, S.V. Neoplasia and paracoccidioidomycosis. Mycopathologia 2008, 165, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Pontes, A.M.; Borborema, J.; Correia, C.R.; de Almeida, W.L.; Maciel, R.F. A rare paracoccidioidomycosis diagnosis in a kidney transplant receptor: Case report. Transpl. Proc. 2015, 47, 1048–1050. [Google Scholar] [CrossRef] [PubMed]
- Moraes Vasconcelos, D.; Grumach, A.S.; Yamaguti, A.; Andrade, M.F.; Fieschi, C.; de Beaucoudrey, L.; Casanova, J.L.; Duarte, A.J. Paracoccidioides brasiliensis disseminated disease in a patient with inherited deficiency in the beta 1 subunit of the interleukin (IL)-12/IL-23 receptor. Clin. Infect. Dis. 2005, 41, e31–e37. [Google Scholar] [CrossRef] [PubMed]
- Morejón, K.M.; Machado, A.A.; Martinez, R. Paracoccidioidomycosis in patients infected with and not infected with human immunodeficiency vírus: A case-control study. Am. J. Trop. Med. Hyg. 2009, 80, 359–366. [Google Scholar] [PubMed]
- Paniago, A.M.; de Freitas, A.C.; Aguiar, E.S.; Aguiar, J.I.; da Cunha, R.V.; Castro, A.R.; Wanke, B. Paracoccidioidomycosis in patients with human immunodeficiency virus: Review of 12 cases observed in an endemic region on Brazil. J. Infect. 2005, 51, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.; Moya, M.J. Complexo primário da paracoccidioidomicose e hipereosinofilia. J. Bras. Pneumol. 2009, 35, 1259–1262. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, J.W.; Michel, G.T.; Londero, A.T. Paracoccidioidoma: Case report and review. Mycopathologia 1997, 137, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Fava, S.; di, C.; Netto, C.F. Epidemiologic surveys of histoplasmin and paracoccidioidin sensitivity in Brazil. Rev. Inst. Med. Trop. Sao Paulo 1998, 40, 155–164. [Google Scholar] [CrossRef]
- Pereira, A.J.C.S. Inquérito intradérmico para paracoccidioidomicose em Goiânia. Rev. Patol. Trop. 1988, 17, 157–186. [Google Scholar]
- Kalmar, E.M.; Alencar, F.E.; Alves, F.P.; Pang, L.W.; Del Negro, G.M.; Camargo, Z.P.; Shikanai-Yasuda, M.A. Paracoccidioidomycosis: An epidemiologic survey in a pediatric population from the Brazilian Amazon using skin tests. Am. J. Trop. Med. Hyg. 2004, 71, 82–86. [Google Scholar] [PubMed]
- Marques, A.P.; Oliveira, S.M.; Rezende, G.R.; Melo, D.A.; Fernandes-Fitts, S.M.; Pontes, E.R.; Bonecini-Almeida Mda, G.; Camargo, Z.P.; Paniago, A.M. Evaluation of Paracoccidioides brasiliensis infection by the gp43 intradermal test in rural settlements in Central-West Brazil. Mycopathologia 2013, 176, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Cermeño, J.; Cermeño, J.; Godoy, G.; Hernández, I.; Orellán, Y.; Blanco, Y.; Penna, S.; García, L.; Mender, T.; Gonsálvez, M.; et al. Epidemiological study of paracoccidioidomycosis and histoplasmosis in a suburb of San Felix city, Bolivar State, Venezuela. Investg. Clin. 2009, 50, 213–220. [Google Scholar]
- De Martin, M.C.; Suaréz, M. Infection caused by Paracoccidioides brasiliensis in people living in Cocle and Veraguas, Republic of Panama. Rev. Med. Panama 1989, 14, 112–115. [Google Scholar] [PubMed]
- Van Gelderen de Komaid, A.; Durán, E.; Borges de Kestelman, I. Histoplasmosis and paracoccidioidomycosis in northwestern Argentina. III. Epidemiological survey in Vipos, La Toma, and Choromoro-Trancas, Tucumán, Argentina. Eur. J. Epidemiol. 1999, 15, 383–388. [Google Scholar] [PubMed]
- Mangiaterra, M.L.; Giusiano, G.E.; Alonso, J.M.; Gorodner, J.O. Paracoccidioidomycosis infection in a subtropical region with important environmental changes. Bull. Soc. Pathol. Exot. 1999, 92, 173–176. [Google Scholar] [PubMed]
- Maluf, M.L.; Pereira, S.R.; Takahachi, G.; Svidzinski, T.I. Prevalência da paracoccidioidomicose-infecção determinada através de teste sorológico em doadores de sangue na região noroeste do Paraná, Brasil. Rev. Soc. Bras. Med. Trop. 2003, 36, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.; Vitali, L.H.; Henriques, J.H.; Machado, A.A.; Albernaz, A.; Lima, A.A. Inquérito soroepidemiológico para infecções por fungos causadores de micoses sistêmicas na Reserva Indígena Xacriabá, Estado de Minas Gerais. Rev. Soc. Bras. Med. Trop. 2002, 35, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Belitardo, D.R.; Calefi, A.S.; Sbeghen, M.R.; de Oliveira, G.G.; Watanabe, M.A.; de Camargo, Z.P. Paracoccidioides brasiliensis infection in domestic rabbits (Oryctolagus cuniculus). Mycoses 2014, 57, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Albano, A.P.; Klafke, G.B.; Brandolt, T.M.; da Hora, V.P.; Nogueira, C.E.; Xavier, M.O.; Meireles, M.C. Seroepidemiology of Paracoccidioides brasiliensis infection in horses from Rio Grande do Sul, Brazil. Braz. J. Microbiol. 2015, 46, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Sbeghen, M.R.; Zanata, T.B.; Macagnan, R.; de Abreu, K.C.; da Cunha, W.L.; Watanabe, M.A.; de Camargo, Z.P.; Ono, M.A. Paracoccidioides brasiliensis infection in small wild mamals. Mycopathologia 2015, 180, 435–470. [Google Scholar] [CrossRef] [PubMed]
- Fontana, F.F.; dos Santos, S.T.; Esteves, F.M.; Rocha, A.; Fernandes, G.F.; do Amaral, C.C.; Domingues, M.A.; de Camargo, Z.P.; Silva-Vergara, M.L. Seroepidemiological survey of paracoccidioidomycosis infection among urban and rural dogs from Uberaba, Minas Gerais, Brazil. Mycopathologia 2010, 169, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Ricci, G.; Motta, F.T.; Wakamatsu, A.; Serafim, R.C.; Borra, R.C.; Franco, M. Canine paracoccidioidomycosis. Med. Mycol. 2004, 42, 379–383. [Google Scholar] [CrossRef] [PubMed]
- de Farias, M.R.; Condas, L.A.Z.; Ribeiro, M.G.; Bosco Sde, M.; Muro, M.D.; Werner, J.; Theodoro, R.C.; Bagagli, E.; Marques, S.A.; Franco, M. Paracoccidioidomycosis in a dog: Case report of generalized lymphadenomegaly. Mycopathologia 2011, 172, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.D.; Lange, C.M. Paracoccidoidomycosis (South American Blastomycosis) in a squirrel monkey (Saimiri sciureus). Vet. Pathol. 1977, 14, 368–371. [Google Scholar] [PubMed]
- Trejo-Cháves, A.; Ramírez-Romero, R.; Ancer-Rodríguez, J.; Nevárez-Garza, A.M.; Rodríguez-Trovar, L.E. Disseminated paracoccidioidomycosis in a Souther two-toed sloth (Choloepus didactylus). J. Comp. Pathol. 2011, 144, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Faiolla, R.C.; Coelho, M.C.; Santana, R.; de, C.; Martinez, R. Histoplasmosis in immunocompetent individuals living in an endemic area in the Brazilian Southeast. Rev. Soc. Bras. Med. Trop. 2013, 46, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Ajello, L.; Polonelli, L. Imported paracoccidioidomycosis: A public health problem in non-endemic areas. Eur. J. Epidemiol. 1985, 1, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Buitrago, M.J.; Cuenca-Estrella, M. Current epidemiology and laboratory diagnosis of endemic mycosis in Spain. Enferm. Infecc. Microbiol. Clin. 2012, 30, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Kamei, K.; Sano, A.; Kikuchi, K.; Makimura, K.; Niimi, M.; Suzuki, K.; Uehara, Y.; Okabe, N.; Nishimura, K.; Miyaji, M. The trends of imported mycosis in Japan. J. Infect. Chemother. 2003, 9, 16–20. [Google Scholar] [CrossRef] [PubMed]
- McGreevy, P.B.; Dietze, R.; Prata, A.; Hembree, S.C. Effects of imigration on the prevalence of malaria in rural areas of the Amazon basin of Brazil. Mem. Inst. Oswaldo Cruz 1989, 84, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Valera, C.A.; Valle Júnior, R.F.; Varandas, S.G.; Sanches Fernandes, L.F.; Pacheco, F.A. The role of environmental land use conflicts in soil fertility: A study in the Uberaba River basin, Brazil. Sci. Total Environ. 2016, 562, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, M.A. The natural habitat of the fungus Paracoccidioides brasiliensis, how to draw the limit between the rural and the urban environment? Biomedica 2014, 34, 5–6. [Google Scholar] [CrossRef] [PubMed]
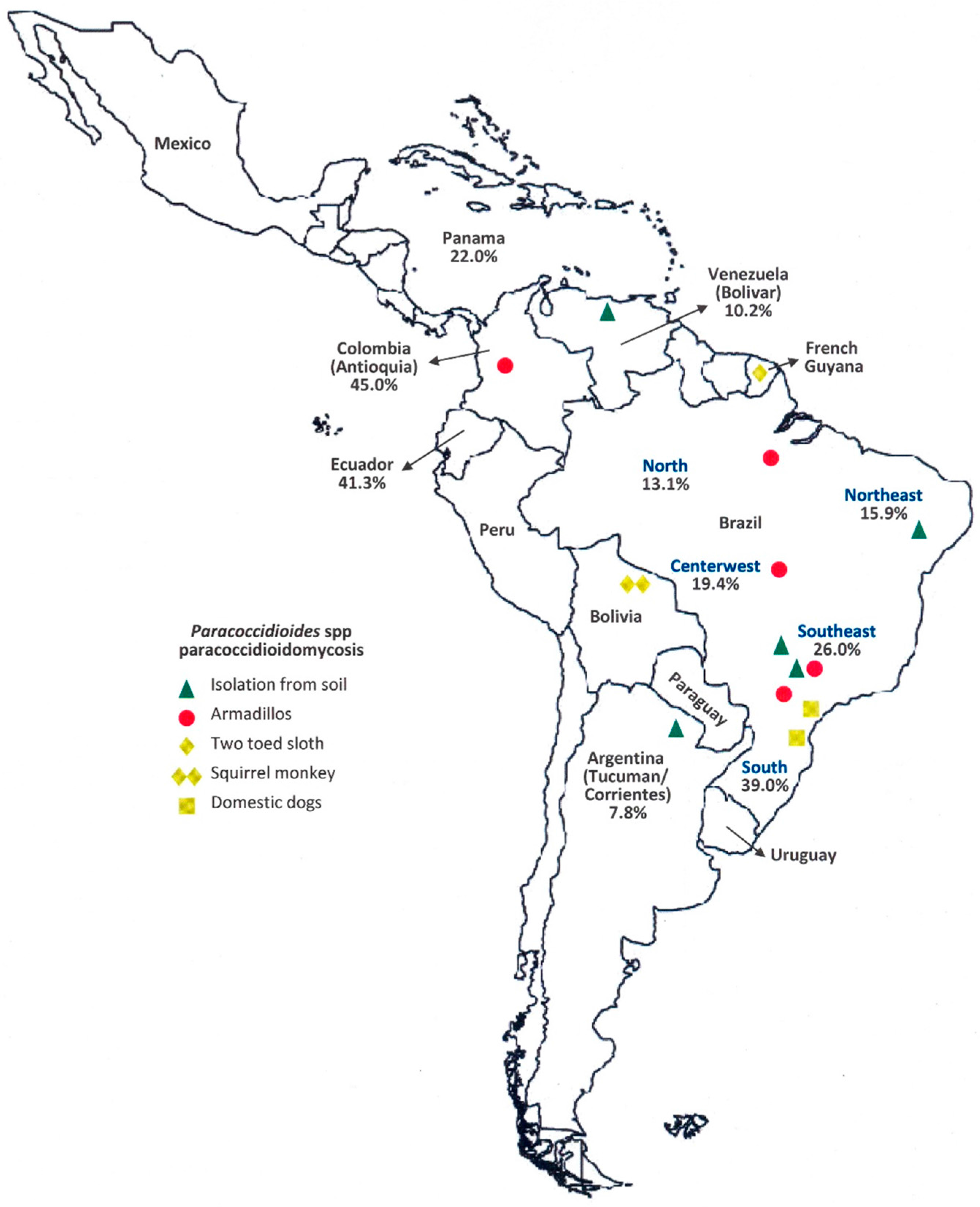
 ) First recognized areas of high endemicity; (
) First recognized areas of high endemicity; (  ) high endemicity observed since the last decades of the 20th century; (
) high endemicity observed since the last decades of the 20th century; (  ) area with some recent evidence of increasing endemicity; (
) area with some recent evidence of increasing endemicity; (  ) areas of moderate endemicity; (
) areas of moderate endemicity; (  ) low endemicity; (
) low endemicity; (  ) no areas or rare cases of paracoccidioidomycosis reported in these countries or regions.
) no areas or rare cases of paracoccidioidomycosis reported in these countries or regions.
 ) First recognized areas of high endemicity; (
) First recognized areas of high endemicity; (  ) high endemicity observed since the last decades of the 20th century; (
) high endemicity observed since the last decades of the 20th century; (  ) area with some recent evidence of increasing endemicity; (
) area with some recent evidence of increasing endemicity; (  ) areas of moderate endemicity; (
) areas of moderate endemicity; (  ) low endemicity; (
) low endemicity; (  ) no areas or rare cases of paracoccidioidomycosis reported in these countries or regions.
) no areas or rare cases of paracoccidioidomycosis reported in these countries or regions.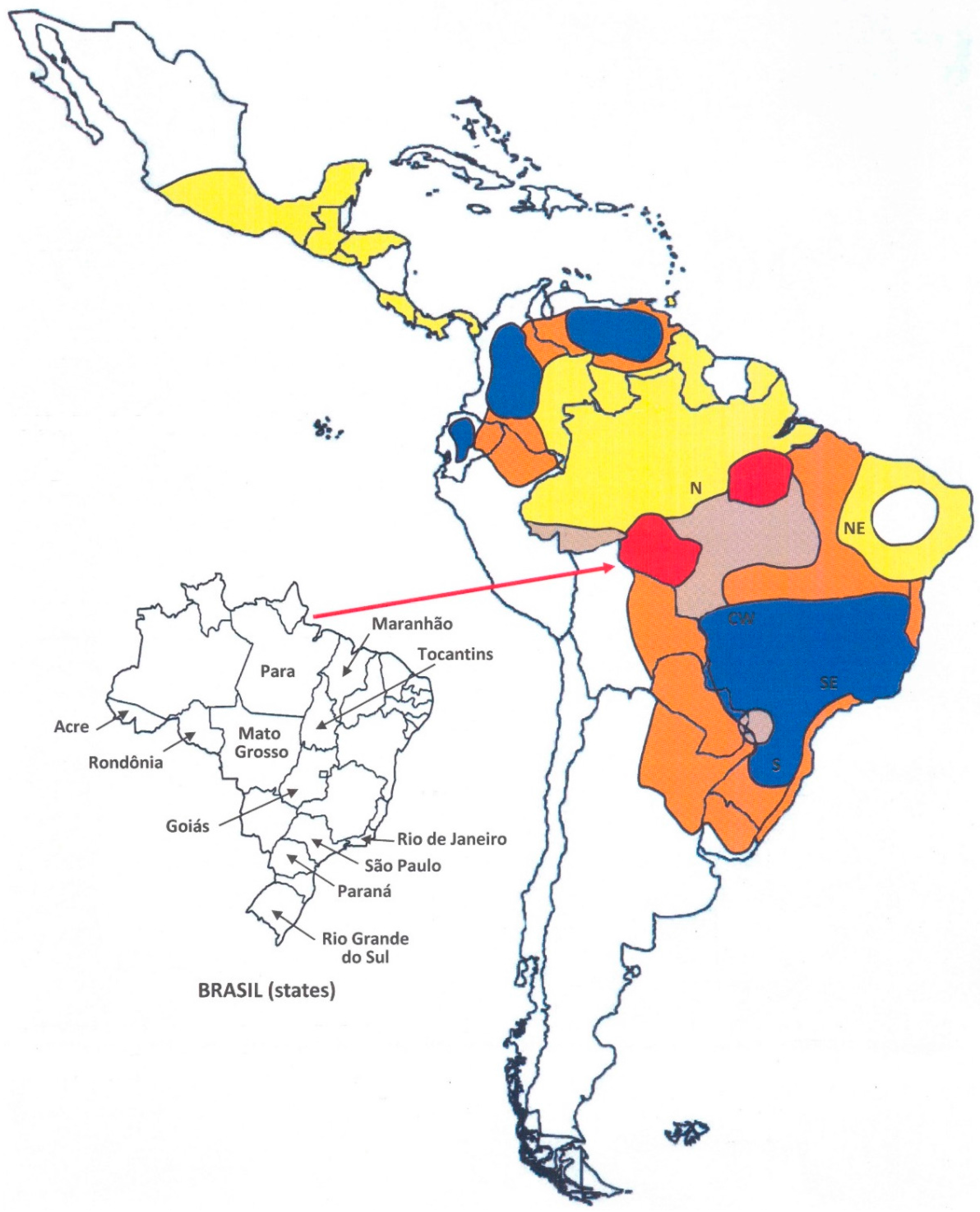
| Country/Geographic Area | Mortality Rate [36] a | Hospitalization Rate [37] b | Reported Cases (No.) | Annual Mean Number of Cases c | Case Series References |
|---|---|---|---|---|---|
| Brazil | 1.0 | 4.3 | 12,508 | ||
| North * | 1.8 | 6.1 | 2375 | 159.9 | [12,13,14] |
| Northeast | 0.2 | 1.6 | 278 | 21.6 | [15] |
| Southeast | 1.0 | 5.7 | 6784 | 207.8 | [11,16,17,18,19,20] |
| South | 1.5 | 2.4 | 2169 | 140.6 | [21,22,23,24] |
| Midwest | 2.1 | 8.3 | 902 | 49.2 | [25,26,27] |
| Argentina | 110 | 110.0 | [28] | ||
| Colombia | 940 | 32.4 | [29] | ||
| Venezuela | 674 | 25.9 | [30] | ||
| Ecuador | 333 | 15.1 | [31] | ||
| Paraguay | 50 | 5.0 | [32] | ||
| Peru | 111 | 3.3 | [33] | ||
| Mexico | 93 | 3.1 | [34] | ||
| Uruguay | 48 | 1.1 | [35] |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinez, R. New Trends in Paracoccidioidomycosis Epidemiology. J. Fungi 2017, 3, 1. https://doi.org/10.3390/jof3010001
Martinez R. New Trends in Paracoccidioidomycosis Epidemiology. Journal of Fungi. 2017; 3(1):1. https://doi.org/10.3390/jof3010001
Chicago/Turabian StyleMartinez, Roberto. 2017. "New Trends in Paracoccidioidomycosis Epidemiology" Journal of Fungi 3, no. 1: 1. https://doi.org/10.3390/jof3010001
APA StyleMartinez, R. (2017). New Trends in Paracoccidioidomycosis Epidemiology. Journal of Fungi, 3(1), 1. https://doi.org/10.3390/jof3010001





