New Vision of Cell Walls in Aspergillus fumigatus from Solid-State NMR Spectroscopy
Abstract
1. Introduction
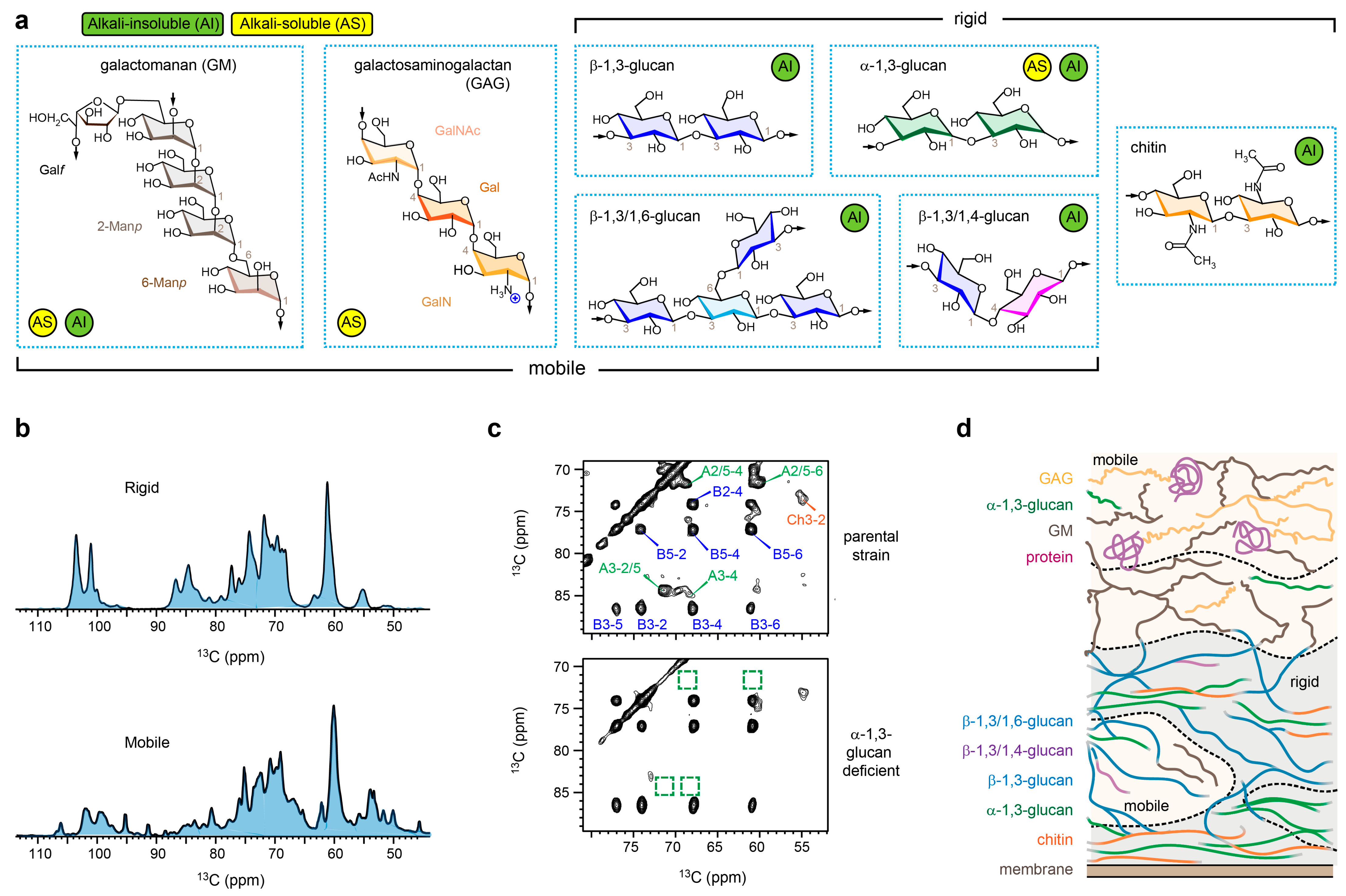
2. Indirect Assessment of Cell Wall Organization in A. fumigatus Mycelia
3. Morphotype Transition in A. fumigatus Conidia
4. Structural Complexity of Invisible Polysaccharides in Chemical Assays
5. Tracking Aspergillus’ Structural Responses to Stress
6. Ambiguity in Assessing Protein and Lipid Components
7. Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gow, N.A.R.; Lenardon, M.D. Architecture of the dynamic fungal cell wall. Nat. Rev. Microbiol. 2022, 21, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Gow, N.A.R.; Latge, J.P.; Munro, C.A. The Fungal Cell Wall: Structure, Biosynthesis, and Function. Microbiol. Spectr. 2017, 5, FUNK-0035-2016. [Google Scholar] [CrossRef]
- Latgé, J.P. Aspergillus fumigatus and aspergillosis. Clin. Microbiol. Rev. 1999, 12, 310–350. [Google Scholar] [CrossRef]
- Perlin, D.S. Current perspectives on echinocandin class drugs. Future Microbiol. 2011, 6, 441–457. [Google Scholar] [CrossRef]
- Ghannoum, M.A.; Arendrup, M.C.; Chaturvedi, V.P.; Lockhart, S.R.; McCormick, T.S.; Chaturvedi, S.; Berkow, E.L.; Juneja, D.; Tarai, B.; Azie, N.; et al. Ibrexafungerp: A Novel Oral Triterpenoid Antifungal in Development for the Treatment of Candida auris Infections. Antibiotics 2020, 9, 539. [Google Scholar] [CrossRef] [PubMed]
- Latgé, J.P. The cell wall: A carbohydrate armour for the fungal cell. Mol. Microbiol. 2007, 66, 279–290. [Google Scholar] [CrossRef]
- Bikmurzin, R.; Marsalka, A.; Kalediene, L. Solid-State 13C Nuclear Magnetic Resonance Study of Soluble and Insoluble β-Glucans Extracted from Candida lusitaniae. Molecules 2023, 28, 8066. [Google Scholar] [CrossRef]
- Stalhberger, T.; Simenel, C.; Clavaud, C.; Eijsink, V.G.; Jourdain, R.; Delepierre, M.; Latge, J.P.; Breton, L.; Fontaine, T. Chemical organization of the cell wall polysaccharide core of Malassezia restricta. J. Biol. Chem. 2014, 289, 12647–12656. [Google Scholar] [CrossRef]
- Fontaine, T.; Simenel, C.; Dubreucq, G.; Adam, O.; Delepierre, M.; Lemoine, J.; Vorgias, C.E.; Diaquin, M.; Latgé, J.P. Molecular organization of the alkali-insoluble fraction of Aspergillus fumigatus cell wall. J. Biol. Chem. 2000, 275, 27594–27607. [Google Scholar] [CrossRef]
- Chakraborty, A.; Fernando, L.D.; Fang, W.; Dickwella Widanage, M.C.; Wei, P.; Jin, C.; Fontaine, T.; Latgé, J.P.; Wang, T. A molecular vision of fungal cell wall organization by functional genomics and solid-state NMR. Nat. Commun. 2021, 12, 6346. [Google Scholar] [CrossRef]
- Latge, J.P.; Chamilos, G. Aspergillus fumigatus and Aspergillosis in 2019. Clin. Microbiol. Rev. 2019, 33, e00140-18. [Google Scholar] [CrossRef]
- Speth, C.; Rambach, G.; Lass-Florl, C.; Howell, P.L.; Sheppard, D.C. Galactosaminogalactan (GAG) and its multiple roles in Aspergillus pathogenesis. Virulence 2019, 10, 976–983. [Google Scholar] [CrossRef]
- Ghassemi, N.; Poulhazan, A.; Deligey, F.; Mentink-Vigier, F.; Marcotte, I.; Wang, T. Solid-State NMR Investigations of Extracellular Matrixes and Cell Walls of Algae, Bacteria, Fungi, and Plants. Chem. Rev. 2022, 122, 10036–10086. [Google Scholar] [CrossRef]
- Latgé, J.P.; Wang, T. Modern Biophysics Redefines Our Understanding of Fungal Cell Wall Structure, Complexity, and Dynamics. mBio 2022, 13, e0114522. [Google Scholar] [CrossRef] [PubMed]
- Fernando, L.D.; Zhao, W.; Gautam, I.; Ankur, A.; Wang, T. Polysacchride Assemblies in Fungal and Plant Cell Walls Explored by Solid-State NMR. Structure 2023, 31, 1375–1385. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Prados-Rosales, R.; Frases, S.; Itin, B.; Casadevall, A.; Stark, R.E. Using solid-state NMR to monitor the molecular consequences of Cryptococcus neoformans melanization with different catecholamine precursors. Biochemistry 2012, 51, 6080–6088. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Frases, S.; Wang, H.; Casadevall, A.; Stark, R.E. Following Fungal Melanin Biosynthesis with Solid-State NMR: Biopolymer Molecular Structures and Possible Connections to Cell-Wall Polysaccharides. Biochemistry 2009, 47, 4701–4710. [Google Scholar] [CrossRef] [PubMed]
- Camacho, E.; Vij, R.; Chrissian, C.; Prados-Rosales, R.; Gil, D.; O’Meally, R.N.; Cordero, R.J.B.; Cole, R.N.; McCaffery, J.M.; Stark, R.E.; et al. The structural unit of melanin in the cell wall of the fungal pathogen Cryptococcus neoformans. J. Biol. Chem. 2019, 27, 10471–10489. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Hong, M. Solid-state NMR investigations of cellulose structure and interactions with matrix polysaccharides in plant primary cell walls. J. Exp. Bot. 2016, 67, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Perez-Garcia, M.; Zhang, Y.; Haynes, J.; Salaza, A.; OZabotina, O.A.; Hong, M. Structure and interactions of plant cell-wall polysaccharides by two-and three-dimensional magic-angle-spinning solid-state NMR. Biochemistry 2011, 50, 989–1000. [Google Scholar] [CrossRef] [PubMed]
- Romaniuk, J.A.H.; Cegelski, L. Bacterial cell wall composition and the influence of antibiotics by cell-wall and whole-cell NMR. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20150024. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Ayala, I.; Bardet, M.; De Paëpe, G.; Simorre, J.P.; Hediger, S. Solid-state NMR on bacterial cells: Selective cell wall signal enhancement and resolution improvement using dynamic nuclear polarization. J. Am. Chem. Soc. 2013, 135, 5105–5110. [Google Scholar] [CrossRef] [PubMed]
- Renault, S.; Boxtel, R.T.v.; Bos, M.P.; Post, J.A.; Tommassen, J.; Baldus, M. Cellular solid-state nuclear magnetic resonance spectroscopy. Proc. Natl. Acad. Sci. USA 2012, 109, 4863–4868. [Google Scholar] [CrossRef] [PubMed]
- Poulhazan, A.; Dickwella Widanage, M.C.; Muszyński, A.; Arnold, A.A.; Warschawski, D.E.; Azadi, P.; Marcotte, I.; Wang, T. Identification and Quantification of Glycans in Whole Cells: Architecture of Microalgal Polysaccharides Described by Solid-State Nuclear Magnetic Resonance. J. Am. Chem. Soc. 2021, 143, 19374–19388. [Google Scholar] [CrossRef] [PubMed]
- Arnold, A.A.; Genard, B.; Zito, F.; Tremblay, R.; Warschawski, D.E.; Marcotte, I. Identification of lipid and saccharide constituents of whole microalgal cells by ¹³C solid-state NMR. Biochim. Biophys. Acta 2015, 1848, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.; Kirui, A.; Muszyński, A.; Widanage, M.C.D.; Chen, A.; Azadi, P.; Wang, P.; Mentink-Vigier, F.; Wang, T. Molecular architecture of fungal cell walls revealed by solid-state NMR. Nat. Commun. 2018, 9, 2747. [Google Scholar] [CrossRef] [PubMed]
- Lamon, G.; Lends, A.; Valsecchi, I.; Wong, S.S.W.; Dupres, V.; Lafont, F.; Tolchard, J.; Schmitt, C.; Mallet, A.; Grelard, A.; et al. Solid-state NMR molecular snapshots of Aspergillus fumigatus cell wall architecture during a conidial morphotype transition. Proc. Natl. Acad. Sci. USA 2023, 120, e2212003120. [Google Scholar] [CrossRef]
- Fernando, L.D.; Perez-Llano, Y.; Dickwella Widanage, M.C.; Jacob, A.; Martinez-Avila, L.; Lipton, A.S.; Gunde-Cimerman, N.; Latge, J.P.; Batista-Garcia, R.A.; Wang, T. Structural adaptation of fungal cell wall in hypersaline environment. Nat. Commun. 2023, 14, 7082. [Google Scholar] [CrossRef]
- Fernando, L.D.; Dickwella Widanage, M.C.; Penfield, J.; Lipton, A.S.; Washton, N.; Latgé, J.P.; Wang, P.; Zhang, L.; Wang, T. Structural polymorphism of chitin and chitosan in fungal cell walls from solid-state NMR and principal component analysis. Front. Mol. Biosci. 2021, 8, 727053. [Google Scholar] [CrossRef]
- Fernando, L.D.; Dickwella Widanage, M.C.; Shekar, S.C.; Mentink-Vigier, F.; Wang, P.; Wi, S.; Wang, T. Solid-state NMR analysis of unlabeled fungal cell walls from Aspergillus and Candida species. J. Struct. Biol. X 2022, 6, 100070. [Google Scholar] [CrossRef]
- Reichhardt, C.; Ferreira, J.A.G.; Joubert, L.M.; Clemons, K.V.; Stevens, D.A.; Cegelski, L. Analysis of the Aspergillus fumigatus Biofilm Extracellular Matrix by Solid-State Nuclear Magnetic Resonance Spectroscopy. Eukaryot. Cell 2015, 14, 1064–1072. [Google Scholar] [CrossRef]
- Reichhardt, C.; Joubert, L.M.; Clemons, K.V.; Stevens, D.A.; Cegelski, L. Integration of electron microscopy and solid-state NMR analysis for new views and compositional parameters of Aspergillus fumigatus biofilms. Med. Mycol. 2019, 57, S239–S244. [Google Scholar] [CrossRef] [PubMed]
- El Nokab, M.E.H.; Lasorsa, A.; Sebakhy, K.O.; Picchioni, F.; van der Wel, P.C.A. Solid-state NMR spectroscopy insights for resolving different water pools in alginate hydrogels. Food Hydrocol. 2022, 127, 107500. [Google Scholar] [CrossRef]
- Hong, M.; Schmidt-Rohr, K. Magic-Angle-Spinning NMR Techniques for Measuring Long-Range Distances in Biological Macromolecules. Acc. Chem. Res. 2013, 9, 2154–2163. [Google Scholar] [CrossRef] [PubMed]
- Ader, C.; Schneider, R.; Seidel, K.; Etzkorn, M.; Becker, S.; Baldus, M. Structural rearrangements of membrane proteins probed by water-edited solid-state NMR spectroscopy. J. Am. Chem. Soc. 2009, 131, 170–176. [Google Scholar] [CrossRef]
- Rief, B.; Ashbrook, S.E.; Emsley, L.; Hong, M. Solid-state NMR spectroscopy. Nat. Rev. Methods Primers 2021, 1, 2. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.E.; Chrissian, C.; Stark, R.E. Tailoring NMR experiments for structural characterization of amorphous biological solids: A practical guide. Solid State Nucl. Magn. Reson. 2020, 109, 101686. [Google Scholar] [CrossRef]
- Aimanianda, V.; Simenel, C.; Garnaud, C.; Clavaud, C.; Tada, R.; Barbin, L.; Mouyna, I.; Heddergott, C.; Popolo, L.; Ohya, Y.; et al. The Dual Activity Responsible for the Elongation and Branching of β-(1,3)-Glucan in the Fungal Cell Wall. mBio 2017, 8, 00619-00617. [Google Scholar] [CrossRef]
- Phyo, P.; Hong, M. Fast MAS 1H–13C correlation NMR for structural investigations of plant cell walls. J. Biomol. NMR 2019, 73, 661–674. [Google Scholar] [CrossRef]
- Simmons, T.J.; Mortimer, J.C.; Bernardinelli, O.D.; Poppler, A.C.; Brown, S.P.; deAzevedo, E.R.; Dupree, R.; Dupree, P. Folding of xylan onto cellulose fibrils in plant cell walls revealed by solid-state NMR. Nat. Commun. 2016, 7, 13902. [Google Scholar] [CrossRef]
- Wang, T.; Yang, H.; Kubichi, J.D.; Hong, M. Cellulose Structural Polymorphism in Plant Primary Cell Walls Investigated by High-Field 2D Solid-State NMR Spectroscopy and Density Functional Theory Calculations. Biomacromolecules 2016, 17, 2210–2222. [Google Scholar] [CrossRef]
- Zhao, W.; Deligey, F.; Shekar, S.C.; Mentink-Vigier, F.; Wang, T. Current limitations of solid-state NMR in carbohydrate and cell wall research. J. Magn. Reson. 2022, 341, 107263. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Debnath, D.; Gautam, I.; Fernando, L.D.; Wang, T. Charting the solid-state NMR signals of polysaccharides: A database-driven roadmap. Magn. Reson. Chem. 2023, in press. [CrossRef] [PubMed]
- Muszkieta, L.; Aimanianda, V.; Mellado, E.; Gribaldo, S.; Alcazar-Fuoli, L.; Szewczyk, E.; Prevost, M.C.; Latge, J.P. Deciphering the role of the chitin synthase families 1 and 2 in the in vivo and in vitro growth of Aspergillus fumigatus by multiple gene targeting deletion. Cell. Microbiol. 2014, 66, 1164–1173. [Google Scholar]
- Roncero, C. The genetic complexity of chitin synthesis in fungi. Curr. Genet. 2002, 41, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Lendardon, M.D.; Whitton, R.K.; Munro, C.A.; Marshall, D.; Gow, N.A.R. Individual chitin synthase enzymes synthesize microfibrils of differing structure at specific locations in the Candida albicans cell wall. Mol. Microbiol. 2007, 66, 1164–1173. [Google Scholar] [CrossRef] [PubMed]
- Dickwella Widanage, M.C.; Gautam, I.; Sarkar, D.; Mentink-Vigier, F.; Vermaas, J.V.; Fontaine, T.; Latge, J.P.; Wang, P.; Wang, T. Structural Remodeling of Fungal Cell Wall Promotes Resistance to Echinocandins. bioRxiv 2024. [Google Scholar] [CrossRef]
- Shekar, S.C.; Zhao, W.; Fernando, L.D.; Hung, I.; Wang, T. A 13C three-dimensional DQ-SQ-SQ correlation experiment for high-resolution analysis of complex carbohydrates using solid-state NMR. J. Magn. Reson. 2022, 336, 107148. [Google Scholar] [CrossRef]
- Beauvais, A.; Bozza, S.; Kniemeyer, O.; Formosa, C.; Balloy, V.; Henry, C.; Roberson, R.W.; Dague, E.; Chignard, M.; Brakhage, A.A.; et al. Deletion of the α-(1,3)-Glucan Synthase Genes Induces a Restructuring of the Conidial Cell Wall Responsible for the Avirulence of Aspergillus fumigatus. PLOS Pathog. 2013, 9, e1003716. [Google Scholar] [CrossRef]
- Henry, C.; Latgé, J.P.; Beauvais, A. Alpha-1,3-glucans are dispensable in Aspergillus fumigatus. Eukaryot. Cell 2012, 11, 26–29. [Google Scholar] [CrossRef]
- Maubon, D.; Park, S.; Tanguy, M.; Huerre, M.; Schmitt, C.; Prevost, M.C.; Perlin, D.S.; Latgé, J.P.; Beauvais, A. AGS3, an α(1–3)glucan synthase gene family member of Aspergillus fumigatus, modulates mycelium growth in the lung of experimentally infected mice. Fungal Genet. Biol. 2006, 43, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Stevens, D.A.; Espiritu, M.; Parmar, R. Paradoxical effect of caspofungin: Reduced activity against Candida albicans at high drug concentrations. Antimicrob. Agents Chemother. 2004, 48, 3407–3411. [Google Scholar] [CrossRef] [PubMed]
- Loiko, V.; Wagener, J. The paradoxical effect of echinocandins in Aspergillus fumigatus relies on recovery of the β-1, 3-glucan synthase Fks1. Antimicrob. Agents Chemother. 2017, 61, e01690-16. [Google Scholar] [CrossRef] [PubMed]
- Ries, L.N.A.; Rocha, M.C.; De Castro, P.A.; Silva-Rocha, R.; Silva, R.N.; Freitas, F.Z.; De Assis, L.J.; Bertolini, M.C.; Malavazi, I.; Goldman, G.H. The Aspergillus fumigatus CrzA transcription factor activates chitin synthase gene expression during the caspofungin paradoxical effect. mBio 2017, 8, e00705–e00717. [Google Scholar] [CrossRef] [PubMed]
- Gow, N.A.R.; Casadevall, A.; Fang, W. Top five unanswered questions in fungal cell surface research. Cell Surf. 2023, 10, 100114. [Google Scholar] [CrossRef] [PubMed]
- Maji, A.; Soutar, C.P.; Zhang, J.; Lewandowska, A.; Uno, B.E.; Yan, S.; Shelke, Y.; Murhade, G.; Nimerovsky, E.; Borcik, C.G.; et al. Tuning sterol extraction kinetics yields a renal-sparing polyene antifungal. Nature 2023, 623, 1079–1085. [Google Scholar] [CrossRef]
- Anderson, T.M.; Clay, M.C.; Cioffi, A.G.; Diaz, K.A.; Hisao, G.S.; Tuttle, M.D.; Nieuwkoop, A.J.; Comellas, G.; Maryum, N.; Wang, S.; et al. Amphotericin forms an extramembranous and fungicidal sterol sponge. Nat. Chem. Biol. 2014, 10, 400–406. [Google Scholar] [CrossRef]
- Bruneau, J.M.; Magnin, T.; Tagat, E.; Legrand, R.; Bernard, M.; Diaquin, M.; Fudali, C.; Latgé, J.P. Proteome analysis of Aspergillus fumigatus identifies glycosylphosphatidylinositol-anchored proteins associated to the cell wall biosynthesis. Electrophoresis 2001, 22, 2812–2823. [Google Scholar] [CrossRef]
- Samalova, M.; Carr, P.; Bromley, M.; Blatzer, M.; Moya-Nilges, M.; Latgé, J.P.; Mouyna, I. GPI Anchored Proteins in Aspergillus fumigatus and Cell Wall Morphogenesis. Curr. Top. Microbiol. Immunol. 2020, 425, 167–186. [Google Scholar]
- Ball, S.R.; Kwan, A.H.; Sunde, M. Hydrophobin Rodlets on the Fungal Cell Wall. Curr. Top. Microbiol. Immunol. 2020, 425, 29–51. [Google Scholar]
- Valsecchi, I.; Lai, J.I.; Stephen-Victor, E.; Pille, A.; Beaussart, A.; Lo, V.; Pham, C.L.L.; Aimanianda, V.; Kwan, A.H.; Duchateau, M.; et al. Assembly and disassembly of Aspergillus fumigatus conidial rodlets. Cell. Surf. 2019, 5, 100023. [Google Scholar] [CrossRef] [PubMed]
- Chrissian, C.; Camacho, E.; Fu, M.S.; Prados-Rosales, R.; Chatterjee, S.; Cordero, R.J.B.X.; Lodge, J.K.X.; Casadevall, A.; Stark, R.E. Melanin deposition in two Cryptococcus species depends on cell-wall composition and flexibility. J. Biol. Chem. 2020, 295, 1815–1828. [Google Scholar] [CrossRef] [PubMed]
- Chrissian, C.; Camacho, E.; Kelly, J.E.; Wang, H.; Casadevall, A.; Stark, R.E. Solid-state NMR spectroscopy identifies three classes of lipids in Cryptococcus neoformans melanized cell walls and whole fungal cells. J. Biol. Chem. 2020, 295, 15083–15096. [Google Scholar] [CrossRef] [PubMed]
- Latge, J.P.; Kobayashi, H.; Debeaupuis, J.P.; Diaquin, M.; Sarfati, J.; Wieruszeski, J.M.; Parra, E.; Bouchara, J.P.; Fournet, B. Chemical and immunological characterization of the extracellular galactomannan of Aspergillus fumigatus. Infect. Immun. 1994, 62, 5424–5433. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, T.; Latge, J.P. Galactomannan Produced by Aspergillus fumigatus: An Update on the Structure, Biosynthesis and Biological Functions of an Emblematic Fungal Biomarker. J. Fungi 2020, 6, 283. [Google Scholar] [CrossRef] [PubMed]
- Perez-Llano, Y.; Rodriguez-Pupo, E.C.; Druzhinina, I.S.; Chenthamara, K.; Cai, F.; Gunde-Cimerman, N.; Zalar, P.; Gostincar, C.; Kostanjsek, R.; Folch-Mallol, J.L.; et al. Stress reshapes the physiological response of halophile fungi to salinity. Cells 2020, 9, 525. [Google Scholar] [CrossRef] [PubMed]
- Marchand, T.L.; Schubeis, T.; Bonaccorsi, M.; Paluch, P.; Lalli, D.; Pell, A.J.; Andreas, L.B.; Jaudzems, K.; Stanek, J.; Pintacuda, G. 1H-Detected Biomolecular NMR under Fast Magic-Angle Spinning. Chem. Rev. 2022, 122, 9943–10018. [Google Scholar] [CrossRef]
- Zhou, D.; Shea, J.J.; Nieuwkoop, A.J.; Franks, W.T.; Wylie, B.J.; Mullen, C.; Sandoz, D.; Rienstra, C.M. Solid-state protein-structure determination with proton-detected triple-resonance 3D magic-angle-spinning NMR spectroscopy. Angew. Chem. Int. Ed. 2009, 46, 8380–8383. [Google Scholar] [CrossRef]
- Safeer, A.; Kleiburg, F.; Bahri, S.; Beriashvili, D.; Veldhuizen, E.J.A.; van Neer, J.; Tegelaar, M.; de Cock, H.; Wosten, H.A.B.; Baldus, M. Probing Cell-Surface Interactions in Fungal Cell Walls by High-Resolution 1H-Detected Solid-State NMR Spectroscopy. Chem. Euro. J. 2022, 29, e202202616. [Google Scholar] [CrossRef]
- Kleijburg, F.E.L.; Safeer, A.; Baldus, M.; Wosten, H.A.B. Binding of micro-nutrients to the cell wall of the fungus Schizophyllum commune. Cell Surf. 2023, 10, 100108. [Google Scholar] [CrossRef]
- Bahri, S.; Safeer, A.; Adler, A.; Smedes, H.; van Ingen, H.; Baldus, M. 1H-detected characterization of carbon–carbon networks in highly flexible protonated biomolecules using MAS NMR. J. Biomol. NMR 2023, 77, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Hediger, S.; De Paëpe, G. Matrix-free dynamic nuclear polarization enables solid-state NMR 13C–13C correlation spectroscopy of proteins at natural isotopic abundance. Chem. Commun. 2013, 49, 9479–9481. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.N.; Marker, K.; Piretra, T.; Boatz, J.C.; Matlahov, I.; Kodali, R.; Hediger, S.; van der Wel, P.C.A.; De Paëpe, G. Structural fingerprinting of protein aggregates by dynamic nuclear polarization-enhanced solid-state NMR at natural isotopic abundance. J. Am. Chem. Soc. 2018, 140, 14576–14580. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gautam, I.; Singh, K.; Dickwella Widanage, M.C.; Yarava, J.R.; Wang, T. New Vision of Cell Walls in Aspergillus fumigatus from Solid-State NMR Spectroscopy. J. Fungi 2024, 10, 219. https://doi.org/10.3390/jof10030219
Gautam I, Singh K, Dickwella Widanage MC, Yarava JR, Wang T. New Vision of Cell Walls in Aspergillus fumigatus from Solid-State NMR Spectroscopy. Journal of Fungi. 2024; 10(3):219. https://doi.org/10.3390/jof10030219
Chicago/Turabian StyleGautam, Isha, Kalpana Singh, Malitha C. Dickwella Widanage, Jayasubba Reddy Yarava, and Tuo Wang. 2024. "New Vision of Cell Walls in Aspergillus fumigatus from Solid-State NMR Spectroscopy" Journal of Fungi 10, no. 3: 219. https://doi.org/10.3390/jof10030219
APA StyleGautam, I., Singh, K., Dickwella Widanage, M. C., Yarava, J. R., & Wang, T. (2024). New Vision of Cell Walls in Aspergillus fumigatus from Solid-State NMR Spectroscopy. Journal of Fungi, 10(3), 219. https://doi.org/10.3390/jof10030219





