Umbellaceae fam. nov. (Hymenochaetales, Basidiomycota) for Umbellus sinensis gen. et sp. nov. and Three New Combinations
Abstract
1. Introduction
2. Materials and Methods
2.1. Morphological Examination
2.2. Molecular Sequencing
2.3. Phylogenetic Analyses
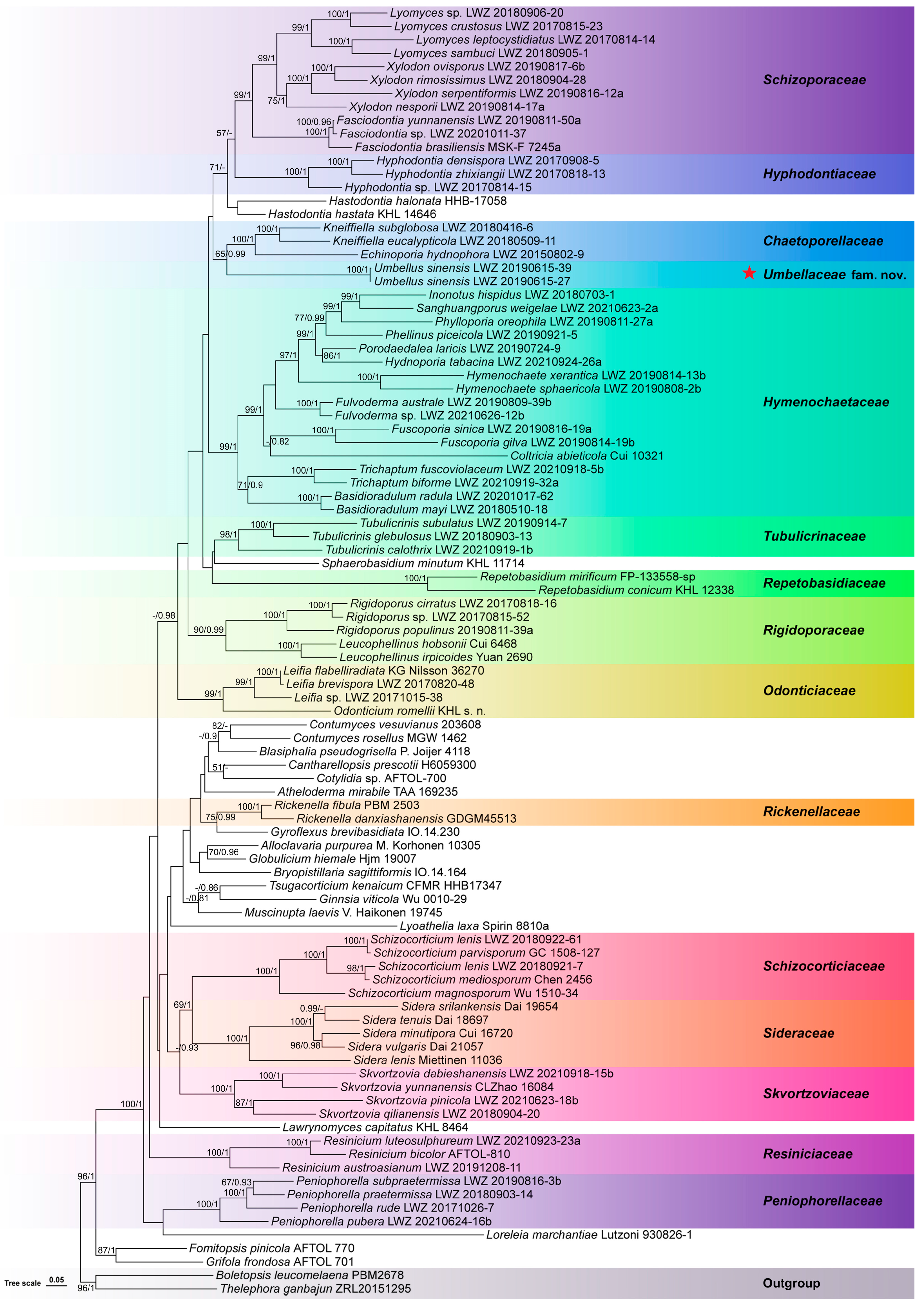
3. Results
3.1. Molecular Phylogeny
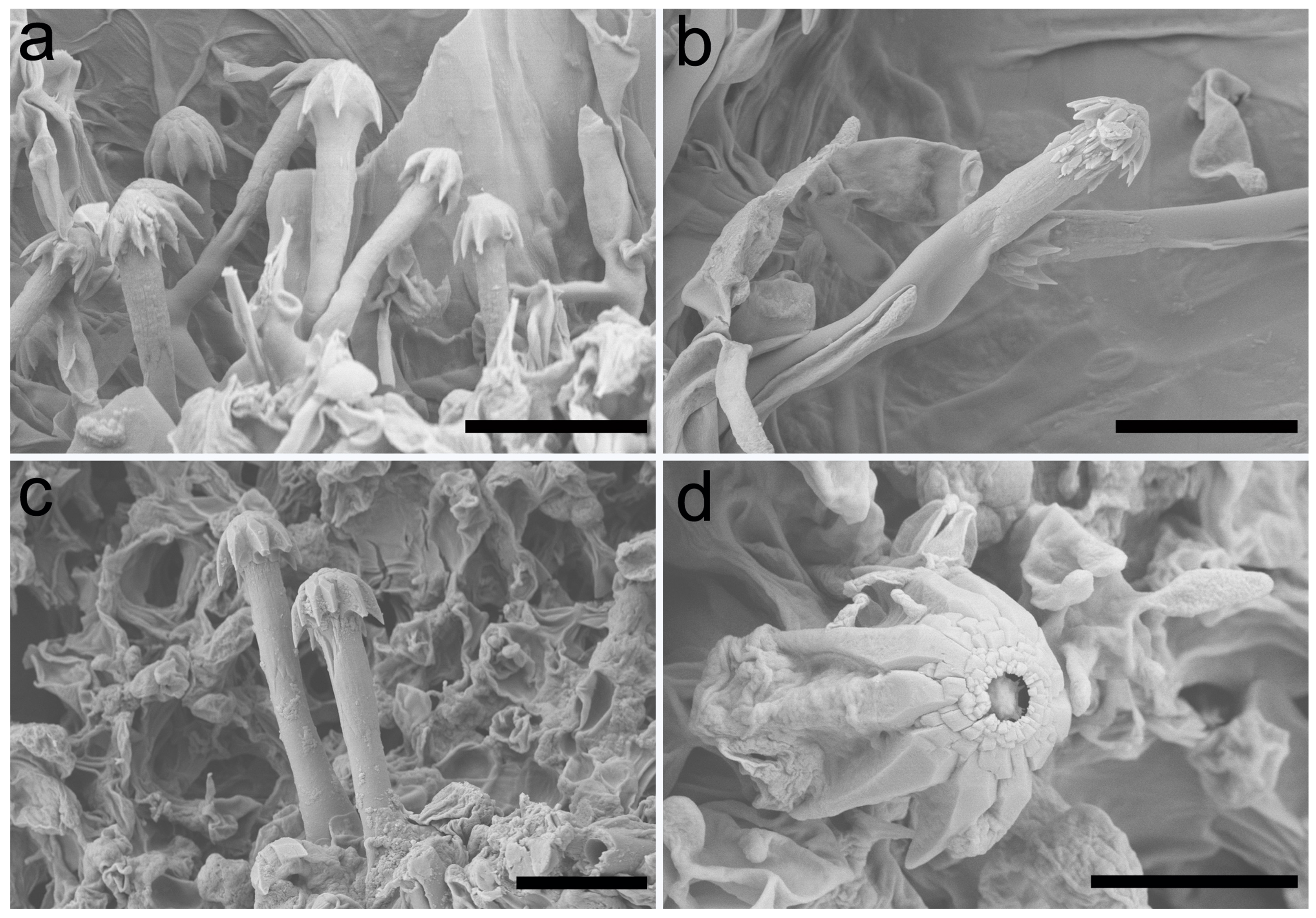
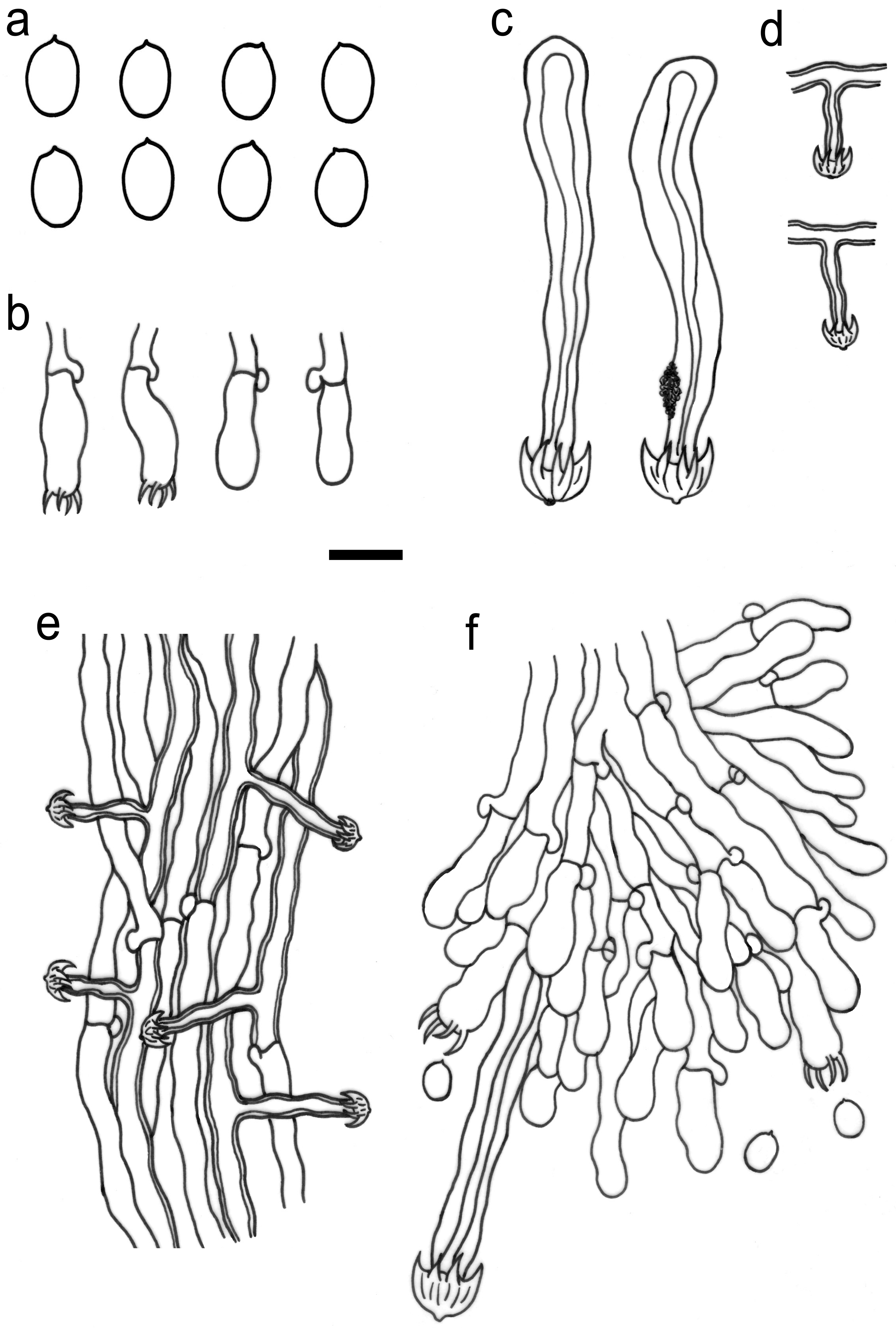
3.2. Taxonomy
- 1 Hymenophore odontioid or slightly hydnoid..........................................................U. corneri
- 1 Hymenophore smooth to grandinioid....................................................................................2
- 2 Basidiospores oblong-ellipsoid or obovate.......................................................U. umbraculus
- 2 Basidiospores broadly ellipsoid...............................................................................................3
- 3 Basidiospores flattened on one side, with a prominent lateral apiculus, 5.5–7.5 × 4–4.5 µm; on coniferous wood..............................................................................................U. hamatus
- 3 Basidiospores not flattened, with a tiny apiculus, 4.5–5 × 3.3–4.2 µm; on angiosperm wood...............................................................................................................................U. sinensis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frey, W.; Hurka, K.; Oberwinkler, F. Beiträge zur Biologie der Niederen Pflanzen. Systematik, Stammesgeschichte, Ökologie; Gustav Fischer Verlag: Stuttgart, Germany; New York, NY, USA, 1977. [Google Scholar]
- James, T.Y.; Stajich, J.E.; Hittinger, C.T.; Rokas, A. Toward a fully resolved fungal tree of life. Annu. Rev. Microbiol. 2020, 74, 29–313. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.W.; Liu, S.L.; Zhou, L.W. An updated taxonomic framework of Hymenochaetales (Agaricomycetes, Basidiomycota). Mycosphere 2023, 14, 452–496. [Google Scholar] [CrossRef]
- Zhou, L.W.; Ghobad-Nejhad, M.; Tian, X.M.; Wang, Y.F.; Wu, F. Current status of ‘Sanghuang’ as a group of medicinal mushrooms and their perspective in industry development. Food Rev. Int. 2022, 38, 589–607. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhou, L.J.; Jiang, J.H.; Tian, X.M.; Zhou, L.W. Phylloporia (Hymenochaetales, Basidiomycota), a medicinal wood-inhabiting fungal genus with much potential for commercial development. Food Rev. Int. 2023, 39, 2776–2789. [Google Scholar] [CrossRef]
- Bai, M.; Zhou, L.W.; Tong, Y.J.; Yin, F. Risk assessment and warning system for strategic biological resources in China. Innov. Life 2023, 1, 100004. [Google Scholar] [CrossRef]
- Parmasto, E. Hymenochaetoid fungi (Basidiomycota) of North America. Mycotaxon 2001, 81, 107–176. [Google Scholar]
- Arras, L.; Piga, A.; Bernicchia, A.; Gorjon, S.P. Fibricium gloeocystidiatum (Polyporales, Basidiomycetes), new to Europe. Mycotaxon 2007, 100, 343–347. [Google Scholar]
- Telleria, M.T.; Melo, I.; Dueñas, M. Resinicium aculeatum, a new species of corticiaceous fungi from Equatorial Guinea. Nova Hedwig. 2008, 87, 195–200. [Google Scholar] [CrossRef]
- Parmasto, E.; Saar, I.; Larsson, E.; Rummo, S. Phylogenetic taxonomy of Hymenochaete and related genera (Hymenochaetales). Mycol. Prog. 2014, 13, 55–64. [Google Scholar] [CrossRef]
- Miettinen, O.; Larsson, K.H.; Spirin, V. Hydnoporia, an older name for Pseudochaete and Hymenochaetopsis, and typification of the genus Hymenochaete (Hymenochaetales, Basidiomycota). Fungal Syst. Evol. 2019, 4, 77–96. [Google Scholar] [CrossRef]
- Kaur, N.; Singh, A.P.; Dhingra, G.S. Hymenochaete longisterigmata sp. nov. from India. Mycotaxon 2020, 135, 631–635. [Google Scholar] [CrossRef]
- Maubet, Y.; Campi, M.; Robledo, G. Hymenochaetaceae from Paraguay: Revision of the family and new records. Curr. Res. Environ. Appl. Mycol. (J. Fungal Biol.) 2020, 10, 242–261. [Google Scholar] [CrossRef]
- Yurchenko, E.; Riebesehl, J.; Langer, E. Fasciodontia gen. nov. (Hymenochaetales, Basidiomycota) and the taxonomic status of Deviodontia. Mycol. Prog. 2020, 19, 171–184. [Google Scholar] [CrossRef]
- Kossmann, T.; Costa-Rezende, D.H.; Góes-Neto, A.; Drechsler-Santos, E.R. A new and threatened species of Trichaptum (Basidiomycota, Hymenochaetales) from urban mangroves of Santa Catarina Island, Southern Brazil. Phytotaxa 2021, 482, 197–207. [Google Scholar] [CrossRef]
- Viner, I.; Bortnikov, F.; Ryvarden, L.; Miettinen, O. On six African species of Lyomyces and Xylodon. Fungal Syst. Evol. 2021, 8, 163–178. [Google Scholar] [CrossRef] [PubMed]
- Lima, V.X.; de Oliveira, V.R.T.; de Lima-Junior, N.C.; de Oliveira-Filho, J.R.C.; Santos, C.; Lima, N.; Gibertoni, T.B. Taxonomy and phylogenetic analysis reveal one new genus and three new species in Inonotus s.l. (Hymenochaetaceae) from Brazil. Cryptogam. Mycol. 2022, 43, 1–21. [Google Scholar]
- Zmitrovich, I.V.; Malysheva, V.F. Studies on Oxyporus. I. Segregation of Emmia and general topology of phylogenetic tree. Mikol. I Fitopatol. 2014, 48, 161–171. [Google Scholar]
- Ariyawansa, H.A.; Hyde, K.D.; Jayasiri, S.C.; Buyck, B.; Chethana, K.W.T.; Dai, D.Q.; Dai, Y.C.; Daranagama, D.A.; Jayawardena, R.S.; Lücking, R.; et al. Fungal Diversity notes 111–252–taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers. 2015, 75, 27–274. [Google Scholar] [CrossRef]
- Larsson, K.H. Re-thinking the classification of corticioid fungi. Mycol. Res. 2007, 111, 1040–1063. [Google Scholar] [CrossRef]
- Jülich, W. Studies in resupinate basidiomycetes-VI. Persoonia 1979, 10, 325–336. [Google Scholar]
- Cunningham, G.H. Thelephoraceae of New Zealand. Part VI. The genus Peniophora. Trans. R. Soc. N. Z. 1955, 83, 247–293. [Google Scholar]
- Jackson, H.S. Studies of Canadian Thelephoraceae. I. Some new species of Peniophora. Can. J. Res. 1948, 26, 128–139. [Google Scholar] [CrossRef]
- Petersen, J.H. Farvekort. In The Danish Mycological Society’s Colour-Chart; Foreningen til Svampekundskabens Fremme: Greve, Denmark, 1996. [Google Scholar]
- Wang, X.W.; Jiang, J.H.; Zhou, L.W. Basidioradulum mayi and B. tasmanicum spp. nov. (Hymenochaetales, Basidiomycota) from both sides of Bass Strait, Australia. Sci. Rep. 2020, 10, 102. [Google Scholar] [CrossRef] [PubMed]
- Hibbett, D.S. Phylogenetic evidence for horizontal transmission of group I introns in the nuclear ribosomal DNA of mushroom-forming fungi. Mol. Biol. Evol. 1996, 13, 903–917. [Google Scholar] [CrossRef] [PubMed]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specifity for Basidiomycetes: Application to identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.D.; Lee, S.B.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Liu, Y.J.; Whelen, S.; Hall, B.D. Phylogenetic relationships among Ascomycetes: Evidence from an RNA polymerse II subunit. Mol. Biol. Evol. 1999, 16, 1799–1808. [Google Scholar] [CrossRef]
- Matheny, P.B. Improving phylogenetic inference of mushrooms with RPB1 and RPB2 nucleotide sequences (Inocybe; Agaricales). Mol. Phylogenet. Evol. 2005, 35, 1–20. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Katoh, K.; Kuma, K.; Toh, H.; Miyata, T. MAFFT version 5: Improvement in accuracy of multiple sequence alignment. Nucleic Acids Res. 2005, 33, 511–518. [Google Scholar] [CrossRef]
- Dessimoz, C.; Gil, M. Phylogenetic assessment of alignments reveals neglected tree signal in gaps. Genome Biol. 2010, 11, R37. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Gascuel, O. A simple, fast and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. jModelTest 2: More models, new heuristics and parallel computing. Nat. Methods 2012, 9, 772. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Pattengale, N.D.; Alipour, M.; Bininda-Emonds, O.R.P.; Moret, B.M.E.; Stamatakis, A. How many bootstrap replicates are necessary? J. Comput. Biol. 2010, 17, 337–354. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2 efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior summarisation in Bayesian phylogenetics using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef]
- Xie, J.M.; Chen, Y.R.; Cai, G.J.; Cai, R.L.; Hu, Z.; Wang, H. Tree Visualization By One Table (tvBOT): A web application for visualizing, modifying and annotating phylogenetic trees. Nucleic Acids Res. 2023, 51, 587–592. [Google Scholar] [CrossRef]
- Shen, S.; Liu, S.L.; Zhou, L.W. Taxonomy of Hyphodermella: A case study to show that simple phylogenies cannot always accurately place species in appropriate genera. IMA Fungus 2023, 14, 11. [Google Scholar] [CrossRef]
- Redhead, S.A.; Moncalvo, J.M.; Vilgalys, R.; Lutzoni, F. Phylogeny of agarics: Partial systematics solutions for bryophilous omphalinoid agarics outside of the Agaricales (euagarics). Mycotaxon 2002, 82, 151–168. [Google Scholar]
- Nakasone, K.K., Jr.; Burdsall, H.H. Tsugacorticium kenaicum (Hymenochaetales, Basidiomycota), a new corticioid genus and species from Alaska. N. Am. Fungi 2012, 7, 1–9. [Google Scholar] [CrossRef][Green Version]
- Zhou, L.W.; May, T.W. Fungal taxonomy: Current status and research agendas for the interdisciplinary and globalisation era. Mycology 2023, 14, 52–59. [Google Scholar] [CrossRef] [PubMed]

| Order/Family | Species | Voucher | nrSSU | ITS | nrLSU | mt-SSU | RPB2 |
|---|---|---|---|---|---|---|---|
| Hymenochaetales/Chaetoporellaceae | Echinoporia hydnophora | LWZ 20150802-9 | ON063768 | ON063639 | ON063838 | ON063707 | |
| Kneiffiella eucalypticola | LWZ 20180509-11 | MT319410 | MT319142 | MT326421 | |||
| Kneiffiella subglobosa | LWZ 20180416-6 | MT319413 | MT319145 | MT326422 | |||
| -/Hymenochaetaceae | Basidioradulum mayi | LWZ 20180510-18 | ON427363 | MN017785 | MN017792 | ON463756 | ON456070 |
| Basidioradulum radula | LWZ 20201017-62 | ON063814 | ON063684 | ON063884 | ON063747 | ON100713 | |
| Coltricia abieticola | Cui 10321 | KY693761 | KX364785 | KX364804 | KY693823 | KX364876 | |
| Fulvoderma australe | LWZ 20190809-39b | ON063771 | ON063644 | ON063843 | ON063712 | ON100686 | |
| Fulvoderma sp. | LWZ 20210626-12b | ON063772 | ON063646 | ON063845 | ON063714 | ON100687 | |
| Fuscoporia gilva | LWZ 20190814-19b | ON063775 | ON063648 | ON063848 | ON063717 | ON100734 | |
| Fuscoporia sinica | LWZ 20190816-19a | ON063776 | ON063649 | ON427358 | ON063719 | ON100691 | |
| Hydnoporia tabacina | LWZ 20210924-26a | ON063778 | ON063651 | ON063851 | ON063720 | ON100685 | |
| Hymenochaete sphaericola | LWZ 20190808-2b | ON063783 | ON063656 | ON063855 | ON063725 | ON100700 | |
| Hymenochaete xerantica | LWZ 20190814-13b | ON063784 | ON063657 | ON063856 | ON063726 | ON100699 | |
| Inonotus hispidus | LWZ 20180703-1 | ON063785 | ON063659 | ON063858 | ON063727 | ON100692 | |
| Phellinus piceicola | LWZ 20190921-5 | ON063790 | ON063662 | ON063862 | ON063731 | ON100695 | |
| Phylloporia oreophila | LWZ 20190811-27a | ON063793 | ON063665 | ON063865 | ON063733 | ON100694 | |
| Porodaedalea laricis | LWZ 20190724-9 | ON063796 | ON063668 | ON063868 | ON063735 | ON100693 | |
| Sanghuangporus weigelae | LWZ 20210623-2a | ON063799 | ON063671 | ON063870 | ON063736 | ON100697 | |
| Trichaptum biforme | LWZ 20210919-32a | ON063832 | ON063701 | ON063901 | ON063764 | ON100730 | |
| Trichaptum fuscoviolaceum | LWZ 20210918-5b | ON063834 | ON063703 | ON063903 | ON063765 | ON100732 | |
| -/Hyphodontiaceae | Hyphodontia pachyspora | LWZ 20170908-5 | MT319426 | MT319160 | MT326431 | MT326261 | |
| Hyphodontia zhixiangii | LWZ 20170818-13 | MT319420 | MT319151 | MT326424 | MT326270 | ||
| Hyphodontia sp. | LWZ 20170814-15 | MT319417 | MT319148 | MT326423 | MT326269 | ||
| -/Odonticiaceae | Leifia brevispora | LWZ 20170820-48 | ON427367 | MK343470 | MK343474 | ON463759 | |
| Leifia flabelliradiata | KG Nilsson 36270 | DQ873635 | DQ873635 | ||||
| Leifia sp. | LWZ 20171015-38 | ON427368 | ON427471 | ON427354 | ON463760 | ||
| Odonticium romellii | KHL s. n. | DQ873639 | DQ873639 | ||||
| -/Peniophorellaceae | Peniophorella praetermissa | LWZ 20180903-14 | ON063816 | ON063686 | ON063886 | ON063749 | ON100714 |
| Peniophorella pubera | LWZ 20210624-16b | ON063817 | ON063687 | ON063887 | ON063750 | ON100715 | |
| Peniophorella rude | LWZ 20171026-7 | ON063818 | ON063688 | ON063888 | ON063751 | ON100716 | |
| Peniophorella subpraetermissa | LWZ 20190816-3b | ON063819 | ON063689 | ON063889 | ON063752 | ON100717 | |
| -/Repetobasidiaceae | Repetobasidium conicum | KHL 12338 | DQ873646 | DQ873647 | DQ873647 | ||
| Repetobasidium mirificum | FP-133558-sp | AY293155 | AY293208 | AY293243 | |||
| -/Resiniciaceae | Resinicium austroasianum | LWZ 20191208-11 | ON063821 | ON063691 | ON063891 | ON063753 | ON100720 |
| Resinicium bicolor | AFTOL-810 | DQ218310 | AF393061 | DQ457635 | |||
| Resinicium friabile | LWZ 20210923-23a | ON063822 | ON063692 | ON427362 | ON063754 | ON100719 | |
| -/Rickenellaceae | Rickenella danxiashanensis | GDGM45513 | ON063823 | MF326424 | ON063755 | ON100721 | |
| Rickenella fibula | PBM 2503 | MF319021 | DQ241782 | MF318953 | DQ408115 | ||
| -/Rigidoporaceae | Leucophellinus hobsonii | Cui 6468 | KT203288 | KT203309 | KT203330 | KT210365 | |
| Leucophellinus irpicoides | Yuan 2690 | KT203289 | KT203310 | KT203331 | KT210366 | ||
| Rigidoporus cirratus | LWZ 20170818-16 | ON427369 | ON427472 | ON427355 | ON463761 | ON456073 | |
| Rigidoporus populinus | LWZ 20190811-39a | ON063803 | ON063674 | ON063874 | ON063740 | ON100702 | |
| Rigidoporus sp. | LWZ 20170815-52 | ON427370 | ON427473 | ON427356 | ON463762 | ON456074 | |
| -/Schizocorticiaceae | Schizocorticium lenis | LWZ 20180921-7 | ON063827 | ON063696 | ON063896 | ON063760 | ON100726 |
| Schizocorticium lenis | LWZ 20180922-61 | ON063829 | ON063698 | ON063898 | ON063762 | ON100728 | |
| Schizocorticium magnosporum | Wu 1510-34 | MK405351 | MK405337 | ||||
| Schizocorticium mediosporum | Chen 2456 | MK405359 | MK405345 | ||||
| Schizocorticium parvisporum | GC 1508-127 | MK405361 | MK405347 | ||||
| -/Schizoporaceae | Fasciodontia brasiliensis | MSK-F 7245a | MK575201 | MK598734 | |||
| Fasciodontia yunnanensis | LWZ 20190811-50a | ON063804 | ON063675 | ON427360 | ON063741 | ON100704 | |
| Fasciodontia sp. | LWZ 20201011-37 | ON063805 | ON063676 | ON427361 | ON063742 | ON100705 | |
| Lyomyces crustosus | LWZ 20170815-23 | MT319465 | MT319201 | MT326446 | MT326275 | ||
| Lyomyces leptocystidiatus | LWZ 20170814-14 | MT319429 | MT319163 | MT326512 | MT326256 | ||
| Lyomyces sambuci | LWZ 20180905-1 | ON063807 | MT319444 | MT319178 | MT326438 | MT326291 | |
| Lyomyces sp. | LWZ 20180906-20 | ON063808 | ON063678 | ON063878 | ON063743 | ON100707 | |
| Xylodon nesporii | LWZ 20190814-17a | ON063809 | ON063679 | ON063879 | ON100708 | ||
| Xylodon ovisporus | LWZ 20190817-6b | ON063810 | ON063680 | ON063880 | ON063744 | ON100709 | |
| Xylodon rimosissimus | LWZ 20180904-28 | ON063812 | ON063682 | ON063882 | ON063745 | ON100711 | |
| Xylodon serpentiformis | LWZ 20190816-12a | ON063813 | ON063683 | ON063883 | ON063746 | ON100712 | |
| -/Sideraceae | Sidera lenis | Miettinen 11036 | FN907914 | FN907914 | |||
| Sidera minutipora | Cui 16720 | MW418078 | MN621349 | MN621348 | MW424986 | MW505865 | |
| Sidera srilankensis | Dai 19654 | MW418087 | MN621344 | MN621346 | MW424989 | MW505868 | |
| Sidera tenuis | Dai 18697 | MW418083 | MK331865 | MK331867 | MW424988 | MW505866 | |
| Sidera vulgaris | Dai 21057 | MW418090 | MW198484 | MW192009 | MW424987 | MW505869 | |
| -/Skvortzoviaceae | Skvortzovia dabieshanensis | LWZ 20210918-15b | ON063825 | ON063694 | ON063894 | ON063757 | ON100723 |
| Skvortzovia pinicola | LWZ 20210623-18b | ON063826 | ON063695 | ON063895 | ON063758 | ON100724 | |
| Skvortzovia qilianensis | LWZ 20180904-20 | ON063824 | ON063693 | ON063893 | ON063756 | ON100722 | |
| Skvortzovia yunnanensis | CLZhao 16084 | MW472754 | MW473473 | ON063759 | ON100725 | ||
| -/Tubulicrinaceae | Tubulicrinis calothrix | LWZ 20210919-1b | ON063835 | ON063704 | ON063904 | ON063766 | ON100733 |
| Tubulicrinis glebulosus | LWZ 20180903-13 | ON063836 | ON063705 | ON063905 | |||
| Tubulicrinis subulatus | LWZ 20190914-7 | ON063837 | ON063706 | ON063906 | ON063767 | ||
| -/Umbellaceae | Umbellus sinensis | LWZ 20190615-27 | OR240268 | OR242616 | OR236212 | OR250300 | OR242518 |
| Umbellus sinensis | LWZ 20190615-39 | OR240269 | OR242617 | OR236213 | OR242519 | ||
| -/Incertae sedis | Alloclavaria purpurea | M. Korhonen 10305 | MF318986 | MF319044 | MF318895 | ||
| Atheloderma mirabile | TAA 169235 | DQ873592 | DQ873592 | ||||
| Blasiphalia pseudogrisella | P. Joijer 4118 | MF318989 | MF319047 | MF318898 | |||
| Bryopistillaria sagittiformis | IO.14.164 | MT232349 | MT232303 | MT242333 | |||
| Cantharellopsis prescotii | H6059300 | MF318993 | MF319051 | MF318903 | MF288855 | ||
| Contumyces rosellus | MGW 1462 | MF319001 | MF319059 | MF318912 | MF288859 | ||
| Contumyces vesuvianus | 203608 | MF319002 | MF318913 | MF288860 | |||
| Cotylidia sp. | AFTOL-700 | AY705958 | AY854079 | AY629317 | FJ436111 | AY883422 | |
| Ginnsia viticola | Wu 0010-29 | MN123802 | GQ470670 | ||||
| Globulicium hiemale | Hjm 19007 | DQ873595 | DQ873595 | ||||
| Gyroflexus brevibasidiata | IO.14.230 | MT232351 | MT232305 | MT242335 | |||
| Hastodontia halonata | HHB-17058 | MK575207 | MK598738 | ||||
| Hastodontia hastata | KHL 14646 | MH638232 | MH638232 | ||||
| Lawrynomyces capitatus | KHL 8464 | DQ677491 | DQ677491 | ||||
| Loreleia marchantiae | Lutzoni 930826-1 | U66432 | U66432 | ||||
| Lyoathelia laxa | Spirin 8810a | MT305998 | MT305998 | ||||
| Muscinupta laevis | V. Haikonen 19745 | MF319004 | MF319066 | MF318921 | MF288861 | ||
| Sphaerobasidium minutum | KHL 11714 | DQ873652 | DQ873653 | ||||
| Tsugacorticium kenaicum | CFMR HHB17347 | JN368234 | JN368221 | JN368203 | |||
| Polyporales/Fomitopsidaceae | Fomitopsis pinicola | AFTOL 770 | AY705967 | AY854083 | AY684164 | AY786056 | |
| -/Grifolaceae | Grifola frondosa | AFTOL 701 | AY705960 | AY854084 | AY629318 | AY786057 | |
| Thelephorales/Bankeraceae | Boletopsis leucomelaena | PBM2678 | DQ435797 | DQ484064 | DQ154112 | GU187820 | |
| -/Thelephoraceae | Thelephora ganbajun | ZRL20151295 | KY418962 | LT716082 | KY418908 | KY419043 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.-W.; Zhou, L.-W. Umbellaceae fam. nov. (Hymenochaetales, Basidiomycota) for Umbellus sinensis gen. et sp. nov. and Three New Combinations. J. Fungi 2024, 10, 22. https://doi.org/10.3390/jof10010022
Wang X-W, Zhou L-W. Umbellaceae fam. nov. (Hymenochaetales, Basidiomycota) for Umbellus sinensis gen. et sp. nov. and Three New Combinations. Journal of Fungi. 2024; 10(1):22. https://doi.org/10.3390/jof10010022
Chicago/Turabian StyleWang, Xue-Wei, and Li-Wei Zhou. 2024. "Umbellaceae fam. nov. (Hymenochaetales, Basidiomycota) for Umbellus sinensis gen. et sp. nov. and Three New Combinations" Journal of Fungi 10, no. 1: 22. https://doi.org/10.3390/jof10010022
APA StyleWang, X.-W., & Zhou, L.-W. (2024). Umbellaceae fam. nov. (Hymenochaetales, Basidiomycota) for Umbellus sinensis gen. et sp. nov. and Three New Combinations. Journal of Fungi, 10(1), 22. https://doi.org/10.3390/jof10010022






