Protein Intake, Protein Mealtime Distribution and Seafood Consumption in Elderly Norwegians: Associations with Physical Function and Strength
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Study Design
2.2. Data Collection
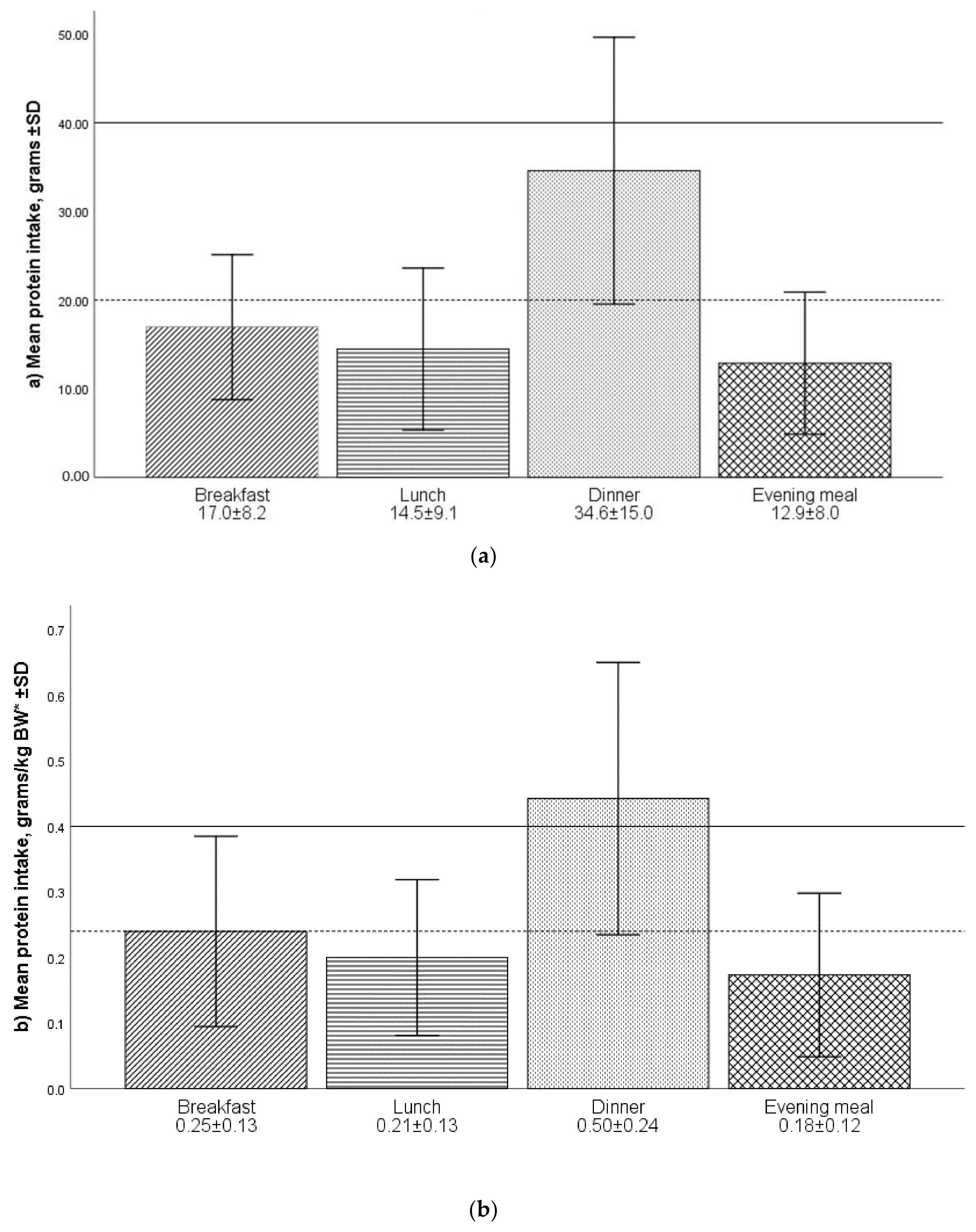
2.2.1. Protein Intake and Mealtime Distribution
2.2.2. Seafood Consumption
2.2.3. Short Physical Performance Battery (SPPB) and Gait Speed
2.2.4. Handgrip Strength
2.2.5. Other Variables
2.3. Statistical Analysis
2.4. Ethical Approval and Registration
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Kiesswetter, E.; Drey, M.; Sieber, C.C. Nutrition, frailty, and sarcopenia. Aging Clin. Exp. Res 2017, 29, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Cardiovascular Health Study Collaborative Research, G. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, S.; Travison, T.G.; Manini, T.M.; Patel, S.; Pencina, K.M.; Fielding, R.A.; Magaziner, J.M.; Newman, A.B.; Kiel, D.P.; Cooper, C.; et al. Sarcopenia Definition: The Position Statements of the Sarcopenia Definition and Outcomes Consortium. J. Am. Geriatr. Soc. 2020, 68, 1410–1418. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Writing Group for the European Working Group on Sarcopenia in Older, P.; the Extended Group for, E. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 601. [Google Scholar] [CrossRef]
- Damanti, S.; Azzolino, D.; Roncaglione, C.; Arosio, B.; Rossi, P.; Cesari, M. Efficacy of Nutritional Interventions as Stand-Alone or Synergistic Treatments with Exercise for the Management of Sarcopenia. Nutrients 2019, 11, 1991. [Google Scholar] [CrossRef]
- Traylor, D.A.; Gorissen, S.H.M.; Phillips, S.M. Perspective: Protein Requirements and Optimal Intakes in Aging: Are We Ready to Recommend More Than the Recommended Daily Allowance? Adv. Nutr. 2018, 9, 171–182. [Google Scholar] [CrossRef]
- Bloom, I.; Shand, C.; Cooper, C.; Robinson, S.; Baird, J. Diet Quality and Sarcopenia in Older Adults: A Systematic Review. Nutrients 2018, 10, 308. [Google Scholar] [CrossRef]
- Hernandez Morante, J.J.; Gomez Martinez, C.; Morillas-Ruiz, J.M. Dietary Factors Associated with Frailty in Old Adults: A Review of Nutritional Interventions to Prevent Frailty Development. Nutrients 2019, 11, 102. [Google Scholar] [CrossRef]
- Franzke, B.; Neubauer, O.; Cameron-Smith, D.; Wagner, K.H. Dietary Protein, Muscle and Physical Function in the Very Old. Nutrients 2018, 10, 935. [Google Scholar] [CrossRef]
- Robinson, S.M.; Reginster, J.Y.; Rizzoli, R.; Shaw, S.C.; Kanis, J.A.; Bautmans, I.; Bischoff-Ferrari, H.; Bruyere, O.; Cesari, M.; Dawson-Hughes, B.; et al. Does nutrition play a role in the prevention and management of sarcopenia? Clin. Nutr. 2018, 37, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- McDonald, C.K.; Ankarfeldt, M.Z.; Capra, S.; Bauer, J.; Raymond, K.; Heitmann, B.L. Lean body mass change over 6 years is associated with dietary leucine intake in an older Danish population. Br. J. Nutr. 2016, 115, 1556–1562. [Google Scholar] [CrossRef] [PubMed]
- Granic, A.; Mendonca, N.; Hill, T.R.; Jagger, C.; Stevenson, E.J.; Mathers, J.C.; Sayer, A.A. Nutrition in the Very Old. Nutrients 2018, 10, 269. [Google Scholar] [CrossRef] [PubMed]
- Lundblad, M.W.; Andersen, L.F.; Jacobsen, B.K.; Carlsen, M.H.; Hjartaker, A.; Grimsgaard, S.; Hopstock, L.A. Energy and nutrient intakes in relation to National Nutrition Recommendations in a Norwegian population-based sample: The Tromso Study 2015–16. Food Nutr. Res. 2019, 63. [Google Scholar] [CrossRef] [PubMed]
- Hodson, N.; West, D.W.D.; Philp, A.; Burd, N.A.; Moore, D.R. Molecular regulation of human skeletal muscle protein synthesis in response to exercise and nutrients: A compass for overcoming age-related anabolic resistance. Am. J. Physiol. Cell Physiol. 2019, 317, C1061–C1078. [Google Scholar] [CrossRef]
- Breen, L.; Phillips, S.M. Skeletal muscle protein metabolism in the elderly: Interventions to counteract the ‘anabolic resistance’ of ageing. Nutr. Metab. (Lond.) 2011, 8, 68. [Google Scholar] [CrossRef]
- Drummond, M.J.; Dreyer, H.C.; Fry, C.S.; Glynn, E.L.; Rasmussen, B.B. Nutritional and contractile regulation of human skeletal muscle protein synthesis and mTORC1 signaling. J. Appl. Physiol. 2009, 106, 1374–1384. [Google Scholar] [CrossRef]
- Wall, B.T.; Gorissen, S.H.; Pennings, B.; Koopman, R.; Groen, B.B.; Verdijk, L.B.; van Loon, L.J. Aging Is Accompanied by a Blunted Muscle Protein Synthetic Response to Protein Ingestion. PLoS ONE 2015, 10, e0140903. [Google Scholar] [CrossRef]
- Phillips, S.M. Nutrition in the elderly: A recommendation for more (evenly distributed) protein? Am. J. Clin. Nutr. 2017, 106, 12–13. [Google Scholar] [CrossRef]
- Larsen, R.; Eilertsen, K.E.; Elvevoll, E.O. Health benefits of marine foods and ingredients. Biotechnol. Adv. 2011, 29, 508–518. [Google Scholar] [CrossRef]
- Aadland, E.K.; Lavigne, C.; Graff, I.E.; Eng, O.; Paquette, M.; Holthe, A.; Mellgren, G.; Jacques, H.; Liaset, B. Lean-seafood intake reduces cardiovascular lipid risk factors in healthy subjects: Results from a randomized controlled trial with a crossover design. Am. J. Clin. Nutr. 2015, 102, 582–592. [Google Scholar] [CrossRef] [PubMed]
- National Council for Nutrition. Dietary Advice to Promote Public Health and Prevent Chronic Diseases: Methodology and Scientific Knowledge Contribution (In Norwegian: Kostråd for å Fremme Folkehelsen og Forebygge Kroniske Sykdommer: Metodologi og Vitenskapelig Kunnskapsbidrag); The Norwegian Directorate of Health: Oslo, Norway, 2011. [Google Scholar]
- Eurostat People in the EU-Population Projections. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=People_in_the_EU_-_population_projections#Population_projections (accessed on 16 April 2020).
- Nygard, L.A.K.; Mundal, I.; Dahl, L.; Saltyte Benth, J.; Rokstad, A.M.M. Nutrition and physical performance in older people-effects of marine protein hydrolysates to prevent decline in physical performance: A randomised controlled trial protocol. BMJ Open 2018, 8, e023845. [Google Scholar] [CrossRef] [PubMed]
- Moshfegh, A.J.; Rhodes, D.G.; Baer, D.J.; Murayi, T.; Clemens, J.C.; Rumpler, W.V.; Paul, D.R.; Sebastian, R.S.; Kuczynski, K.J.; Ingwersen, L.A.; et al. The US Department of Agriculture Automated Multiple-Pass Method reduces bias in the collection of energy intakes. Am. J. Clin. Nutr. 2008, 88, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Øverby, N.C.; Frost Andersen, L. Ungkost-2000: Landsomfattende Kostholdsundersøkelse Blant Elever i 4.-og 8; Klasse i Norge. Sosial- og Helsedirektoratet: Oslo, Norway, 2002. [Google Scholar]
- Guttormsen, A.B.; Hensrud, A.; Irtun, Ø.; Mowé, M.; Sørbye, L.W.; Thoresen, L.; Øien, H.; Alhaug, J.; Smedshaug, G.B. The National guidelines for prevention and treatment of malnutrition (In Norwegian: Nasjonale Faglige Retningslinjer for Forebygging og Behandling av Underernæring); The Norwegian Directorate of Health: Oslo, Norway, 2010. [Google Scholar]
- Berner, L.A.; Becker, G.; Wise, M.; Doi, J. Characterization of dietary protein among older adults in the United States: Amount, animal sources, and meal patterns. J. Acad. Nutr. Diet 2013, 113, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Wijnhoven, H.A.H.; Elstgeest, L.E.M.; de Vet, H.C.W.; Nicolaou, M.; Snijder, M.B.; Visser, M. Development and validation of a short food questionnaire to screen for low protein intake in community-dwelling older adults: The Protein Screener 55+ (Pro55+). PLoS ONE 2018, 13, e0196406. [Google Scholar] [CrossRef] [PubMed]
- Bollwein, J.; Diekmann, R.; Kaiser, M.J.; Bauer, J.M.; Uter, W.; Sieber, C.C.; Volkert, D. Distribution but not amount of protein intake is associated with frailty: A cross-sectional investigation in the region of Nurnberg. Nutr. J. 2013, 12, 109. [Google Scholar] [CrossRef]
- Dahl, L.; Maeland, C.A.; Bjorkkjaer, T. A short food frequency questionnaire to assess intake of seafood and n-3 supplements: Validation with biomarkers. Nutr. J. 2011, 10, 127. [Google Scholar] [CrossRef]
- Markhus, M.W.; Graff, I.E.; Dahl, L.; Seldal, C.F.; Skotheim, S.; Braarud, H.C.; Stormark, K.M.; Malde, M.K. Establishment of a seafood index to assess the seafood consumption in pregnant women. Food Nutr. Res. 2013, 57. [Google Scholar] [CrossRef]
- Bergh, S.; Lyshol, H.; Selbæk, G.; Strand, B.H.; Taraldsen, K.; Thingstad, P. Short Physical Performance Battery (SPPB). Available online: https://stolav.no/PublishingImages/Sider/Bevegelsesvansker-og-fall-hos-eldre/SPPB%20Norsk%20versjon%2006.05.13.pdf (accessed on 1 October 2020).
- Kwon, S.; Perera, S.; Pahor, M.; Katula, J.A.; King, A.C.; Groessl, E.J.; Studenski, S.A. What is a meaningful change in physical performance? Findings from a clinical trial in older adults (the LIFE-P study). J. Nutr. Health Aging 2009, 13, 538–544. [Google Scholar] [CrossRef]
- Freiberger, E.; de Vreede, P.; Schoene, D.; Rydwik, E.; Mueller, V.; Frandin, K.; Hopman-Rock, M. Performance-based physical function in older community-dwelling persons: A systematic review of instruments. Age Ageing 2012, 41, 712–721. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef] [PubMed]
- Guralnik, J.M.; Ferrucci, L.; Pieper, C.F.; Leveille, S.G.; Markides, K.S.; Ostir, G.V.; Studenski, S.; Berkman, L.F.; Wallace, R.B. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J. Gerontol. A Biol. Sci. Med. Sci. 2000, 55, M221–M231. [Google Scholar] [CrossRef] [PubMed]
- Dodds, R.M.; Syddall, H.E.; Cooper, R.; Benzeval, M.; Deary, I.J.; Dennison, E.M.; Der, G.; Gale, C.R.; Inskip, H.M.; Jagger, C.; et al. Grip strength across the life course: Normative data from twelve British studies. PLoS ONE 2014, 9, e113637. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Argiles, J.M.; Evans, W.J.; Bhasin, S.; Cella, D.; Deutz, N.E.; Doehner, W.; Fearon, K.C.; Ferrucci, L.; Hellerstein, M.K.; et al. Nutritional recommendations for the management of sarcopenia. J. Am. Med. Dir. Assoc. 2010, 11, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznaric, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr.(Edinb. Scotl.) 2014, 33, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-Based Recommendations for Optimal Dietary Protein Intake in Older People: A Position Paper From the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Totland, T.H. Helsedirektoratet, Norkost 3: En Landsomfattende Kostholdsundersøkelse Blant Menn og Kvinner i Norge i Alderen 18–70 år, 2010–2011; Helsedirektoratet: Oslo, Norway, 2012. [Google Scholar]
- Gingrich, A.; Spiegel, A.; Kob, R.; Schoene, D.; Skurk, T.; Hauner, H.; Sieber, C.C.; Volkert, D.; Kiesswetter, E. Amount, Distribution, and Quality of Protein Intake Are Not Associated with Muscle Mass, Strength, and Power in Healthy Older Adults without Functional Limitations-An enable Study. Nutrients 2017, 9, 1358. [Google Scholar] [CrossRef]
- Farsijani, S.; Morais, J.A.; Payette, H.; Gaudreau, P.; Shatenstein, B.; Gray-Donald, K.; Chevalier, S. Relation between mealtime distribution of protein intake and lean mass loss in free-living older adults of the NuAge study. Am. J. Clin. Nutr. 2016, 104, 694–703. [Google Scholar] [CrossRef]
- Nordic Council of Ministers. Nordic Nutrition Recommendations 2012: Integrating Nutrition and Physical Activity, 5th ed.; Nordic Council of Ministers: Copenhagen, Denmark, 2014; Available online: https://www.norden.org/no/node/7832 (accessed on 30 October 2020).
- Van Til, A.J.; Naumann, E.; Cox-Claessens, I.J.; Kremer, S.; Boelsma, E.; de van der Schueren, M.A. Effects of the daily consumption of protein enriched bread and protein enriched drinking yoghurt on the total protein intake in older adults in a rehabilitation centre: A single blind randomised controlled trial. J. Nutr. Health Aging 2015, 19, 525–530. [Google Scholar] [CrossRef]
- Murphy, C.H.; Oikawa, S.Y.; Phillips, S.M. Dietary Protein to Maintain Muscle Mass in Aging: A Case for Per-meal Protein Recommendations. J. Frailty Aging 2016, 5, 49–58. [Google Scholar]
- Tieland, M.; Borgonjen-van den Berg, K.J.; van Loon, L.J.; de Groot, L.C. Dietary protein intake in community-dwelling, frail, and institutionalized elderly people: Scope for improvement. Eur. J. Nutr. 2012, 51, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Witard, O.C.; McGlory, C.; Hamilton, D.L.; Phillips, S.M. Growing older with health and vitality: A nexus of physical activity, exercise and nutrition. Biogerontology 2016, 17, 529–546. [Google Scholar] [CrossRef] [PubMed]
- Bergland, A.; Strand, B.H. Norwegian reference values for the Short Physical Performance Battery (SPPB): The Tromsø Study. BMC Geriatr. 2019, 19, 216. [Google Scholar] [CrossRef] [PubMed]
- Rosendahl-Riise, H.; Sulo, G.; Karlsson, T.; Drevon, C.A.; Dierkes, J.; Tell, G.S. The Limited Benefit of Fish Consumption on Risk of Hip Fracture among Men in the Community-Based Hordaland Health Study. Nutrients 2018, 10, 873. [Google Scholar] [CrossRef] [PubMed]
- Rosendahl-Riise, H.; Karlsson, T.; Drevon, C.A.; Apalset, E.M.; Nygard, O.K.; Tell, G.S.; Dierkes, J. Total and lean fish intake is positively associated with bone mineral density in older women in the community-based Hordaland Health Study. Eur. J. Nutr. 2019, 58, 1403–1413. [Google Scholar] [CrossRef]
- Dalane, J.Ø.; Bergvatn, T.A.M.; Kielland, E.; Carlsen, M.H. Mål, vekt og porsjonsstørrelser for matvarer = Weights, measures and portion sizes for foods. In Weights, Measures and Portion Sizes for Foods; Mattilsynet Universitetet i Oslo Helsedirektoratet: Oslo, Norway, 2015. [Google Scholar]
- Nyberg, M.; Olsson, V.; Pajalic, Z.; Örtman, G.; Andersson, H.S.; Blücher, A.; Wendin, K.; Westergren, A.; Blucher, A. Eating difficulties, nutrition, meal preferences and experiences among elderly-a literature overview from a Scandinavian context. J. Food Res. 2015, 4, 22–37. [Google Scholar] [CrossRef]
- Wennberg, M. Tornevi, A. Johansson, I. Diet and lifestyle factors associated with fish consumption in men and women: A study of whether gender differences can result in gender-specific confounding. Nutr. J. 2012, 11, 101. [Google Scholar] [CrossRef]
| n | Min–Max | ||
|---|---|---|---|
| Age, mean years ± SD | 92 | 73.8 ± 8.9 | 65–93 |
| Male gender, n (%) | 92 | 31 (33.7) | |
| Have higher education, n (%) | 92 | 41 (44.6) | |
| Living alone, n (%) | 92 | 34 (37.0) | |
| Strength training ≥1/week, n (%) | 92 | 43 (46.7) | |
| Body mass index (BMI), mean kg/m2 ± SD | 91 | 25.9 ± 4.5 | 16.6–39.9 |
| Energy Intake (kcal) | Protein Intake (g) | Protein Intake (g/kg) 1 | CV Meals 2 | Seafood Index 3 | ||
|---|---|---|---|---|---|---|
| All | All, mean ± SD | 1804 ± 529 | 75.4 ± 22.6 | 1.1 ± 0.4 | 0.6 ± 0.3 | 3.3 ± 1.3 |
| Gender difference | Women (n = 60), mean ± SD | 1734 ± 535 | 71.3 ± 23.4 | 1.1 ± 0.4 | 0.6 ± 0.3 | 3.3 ± 1.4 |
| Men (n = 30), mean ± SD | 1943 ± 496 | 83.7 ± 18.7 | 1.1 ± 0.2 | 0.6 ± 0.3 | 3.2 ± 1.1 | |
| Mean difference | −208.9 | −12.4 | 0.04 | 0.05 | 0.08 | |
| 95% CI | −441.1; 23.4 | −22.2; −2.6 | −0.1; 0.2 | −0.1; 0.2 | −0.5; 0.7 | |
| p-value | 0.08 | 0.01 | 0.57 | 0.40 | 0.78 | |
| Age difference | Age 65–84 (n = 74), mean ±SD | 1814 ± 563 | 76.8 ± 24.0 | 1.1 ± 0.4 | 0.6 ± 0.3 | 3.4 ± 1.3 |
| Age ≥85 (n = 16), mean ± SD | 1759 ± 342 | 69.0 ± 13.7 | 1.1 ± 0.3 | 0.6 ± 0.3 | 2.7 ± 1.0 | |
| Mean difference | 54.4 | 7.8 | −0.05 | −0.008 | 0.7 | |
| 95% CI | −236.9; 345.7 | 4.4; −1.2 | −0.2; 0.2 | −0.2; 0.2 | 0.1; 1.4 | |
| p-value | 0.71 | 0.09 | 0.65 | 0.92 | 0.03 |
| Mean ± SD | Min–Max | ||
|---|---|---|---|
| SPPB 1, score | All participants | 10.3 ± 2.7 | 2–12 |
| Men | 10.7 ± 2.1 | 5–12 | |
| Women | 10.0 ± 2.9 | 2–12 | |
| Grip strength, kg | All participants | 32.3 ± 11.6 | 7.8–57.6 |
| Men | 44.4 ± 9.4 * | 20.9–57.6 | |
| Women | 26.2 ± 6.8 * | 7.8–40.3 | |
| Gait speed, m/s | All participants | 1.0 ± 0.3 | 0.2–1.9 |
| Men | 1.0 ± 0.3 | 0.4–1.9 | |
| Women | 1.0 ± 0.3 | 0.2–1.6 |
| SPPB 1 | Grip Strength | Gait Speed | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bivariate models | Multiple Model | Bivariate Models | Multiple Model | Bivariate Models | Multiple Model | |||||||
| Regr. Coeff. (95% CI) | p-value | Regr. Coeff. (95% CI) | p-value | Regr. Coeff. (95% CI) | p-value | Regr. Coeff. (95% CI) | p-value | Regr. Coeff. (95% CI) | p-value | Regr. Coeff. (95% CI) | p-value | |
| Age | −0.2 (−0.3; −0.2) | <0.01 | −0.2 (−0.3; −0.2) | <0.01 | −0.5 (−0.8; −0.3) | <0.01 | −0.5 (−0.7; −0.4) | <0.01 | −0.02 (−0.03; −0.02) | <0.01 | −0.02 (−0.03; −0.02) | <0.01 |
| Male gender | 0.7 (−0.5; 1.9) | 0.27 | 1.1 (0.4; 1.9) | 0.01 | 18.2 (14.6; 21.7) | <0.01 | 18.9 (15.9; 21.9) | <0.01 | 0.02 (−0.1; 0.2) | 0.73 | 0.3 (0.1) | 0.02 |
| Education, higher | 1.6 (0.5; 2.7) | 0.01 | 0.9 (0.2; 1.6) | 0.02 | 0.7 (−4.3; 5.6) | 0.78 | 0.4 (−2.4; 3.1) | 0.8 | 0.1 (−0.002; 0.3) | 0.05 | 0.04 (−0.06; 0.1) | 0.42 |
| Living alone | −1.7 (−2.8; −0.6) | <0.01 | 0.04 (−0.8; 0.9) | 0.93 | −9.9 (−14.5; −5.2) | <0.01 | 0.1 (−3.2; 3.3) | 0.97 | −0.2 (−0.3; −0.03) | 0.02 | 0.02 (−0.09; 0.1) | 0.77 |
| Strength training 2 | 1.2 (0.1; 2.3) | 0.04 | 0.6 (−0.2; 1.4) | 0.11 | −2.4 (−7.3; 2.5) | 0.34 | 0.1 (−2.8; 3.0) | 0.96 | 0.2 (0.1; 0.3) | < 0.01 | 0.1 (0.02; 0.2) | 0.02 |
| BMI | 0.1 (−0.1; 0.2) | 0.4 | 0.1 (−0.04; 0.2) | 0.22 | 0.3 (−0.3; 0.8) | 0.29 | 0.2 (−0.2; 0.5) | 0.32 | −0.003 (−0.02; 0.01) | 0.63 | −0.005 (−0.02; 0.01) | 0.44 |
| CV meals 3 | 0.7 (−1.4; 2.7) | 0.52 | 1.0 (−0.3; 2.2) | 0.14 | −2.3 (−11.0; 6.5) | 0.61 | 2.2 (−2.7; 7.0) | 0.38 | 0.1 (−0.1; 0.3) | 0.34 | 0.08 (−0.09; 0.2) | 0.34 |
| Seafood index 4 | 0.4 (−0.1; 0.8) | 0.1 | 0.1 (−0.2; 0.4) | 0.38 | 1.3 (−0.6; 3.2) | 0.19 | 1.0 (−0.1; 2.0) | 0.07 | 0.05 (−0.003; 0.09) | 0.07 | 0.1 (0.06) | 0.03 |
| Protein intake 5 | 0.3 (−1.3; 1.2) | 0.73 | 0.5 (−0.6; 0.4) | 0.4 | −1.0 (−7.9; 5.9) | 0.78 | 0.8 (−3.3; 4.9) | 0.7 | −0.04 (−0.2; 0.1) | 0.67 | −0.09 (−0.2; 0.05) | 0.2 |
| Gender × seafood index interaction | −0.08 (0.04) | 0.07 | ||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nygård, L.K.; Dahl, L.; Mundal, I.; Šaltytė Benth, J.; Rokstad, A.M.M. Protein Intake, Protein Mealtime Distribution and Seafood Consumption in Elderly Norwegians: Associations with Physical Function and Strength. Geriatrics 2020, 5, 100. https://doi.org/10.3390/geriatrics5040100
Nygård LK, Dahl L, Mundal I, Šaltytė Benth J, Rokstad AMM. Protein Intake, Protein Mealtime Distribution and Seafood Consumption in Elderly Norwegians: Associations with Physical Function and Strength. Geriatrics. 2020; 5(4):100. https://doi.org/10.3390/geriatrics5040100
Chicago/Turabian StyleNygård, Linda Kornstad, Lisbeth Dahl, Ingunn Mundal, Jūratė Šaltytė Benth, and Anne Marie Mork Rokstad. 2020. "Protein Intake, Protein Mealtime Distribution and Seafood Consumption in Elderly Norwegians: Associations with Physical Function and Strength" Geriatrics 5, no. 4: 100. https://doi.org/10.3390/geriatrics5040100
APA StyleNygård, L. K., Dahl, L., Mundal, I., Šaltytė Benth, J., & Rokstad, A. M. M. (2020). Protein Intake, Protein Mealtime Distribution and Seafood Consumption in Elderly Norwegians: Associations with Physical Function and Strength. Geriatrics, 5(4), 100. https://doi.org/10.3390/geriatrics5040100






