Comparison of the Clinical Characteristics of Histiocytic Sarcoma in Bernese Mountain Dogs and Flat-Coated Retrievers
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Case Selection
2.2. Diagnosis
2.3. Statistics
3. Results
3.1. Demographics
3.2. Presenting Signs
3.3. Blood Results
3.4. Imaging
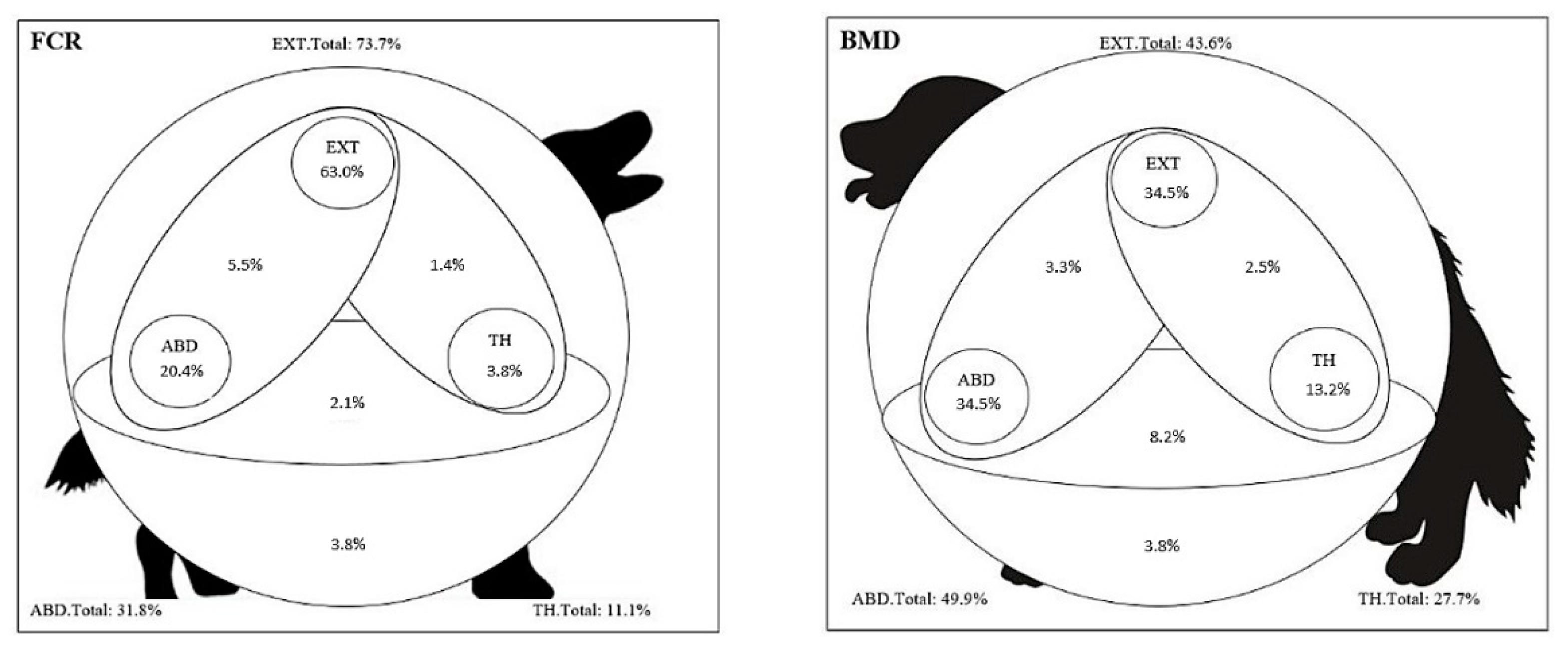
3.5. Location of Tumors
3.5.1. Abdomen
3.5.2. Thorax
3.5.3. External
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Affolter, V.K.; Moore, P.F. Localized and disseminated histiocytic sarcoma of dendritic cell origin in dogs. Vet. Pathol. 2002, 39, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.F.; Rosin, A. Malignant histiocytosis of Bernese Mountain dogs. Vet. Pathol. 1986, 23, l–10. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.E.; Thrall, M.A.; Getzy, D.M.; Weiser, M.G.; Ogilvie, G.K. Cytology of canine malignant histiocytosis. Vet. Clin. Pathol. 1994, 23, 118–122. [Google Scholar] [CrossRef]
- Fidel, J.; Schiller, I.; Hauser, B.; Jausi, Y.; Rohrer-Bley, C.; Roos, M.; Kaser-Hotz, B. Histiocytic sarcomas in flat-coated retrievers: A summary of 37 cases (November 1998–March 2005). Vet. Comp. Oncol. 2006, 4, 63–74. [Google Scholar] [CrossRef]
- Gamlem, H.; Nordstoga, K.; Glattre, E. Canine neoplasia—Introductory paper. APMIS 2008, 125, 5–18. [Google Scholar] [CrossRef]
- Dobson, J.; Hoather, T.; McKinley, T.J.; Wood, J.L.N. Mortality in a cohort of flat-coated retrievers in the UK. Vet. Comp. Oncol. 2009, 7, 115–121. [Google Scholar] [CrossRef]
- Morris, J.S.; Bostock, D.E.; McInnes, E.F.; Hoather, T.M.; Dobson, J.M. Histopathological survey of neoplasms in flat-coated retrievers, 1990 to 1998. Vet. Rec. 2000, 147, 291–295. [Google Scholar] [CrossRef]
- Shearin, A.L.; Hedan, B.; Cadieu, E.; Erich, S.A.; Schmidt, E.V.; Faden, D.L.; Cullen, J.; Abadie, J.; Kwon, E.M.; Gröne, A.; et al. The MTAP-CDKN2A locus confers susceptibility to a naturally occurring canine cancer. Cancer Epidemiol. Biomark. Prev. 2012, 21, 1019–1027. [Google Scholar] [CrossRef]
- Evans, J.M.; Parker, H.G.; Rutteman, G.R.; Plassais, J.; Grinwis, G.C.M.; Harris, A.C.; Lana, S.E.; Ostrander, E.A. Multi-omics approach identifies germline regulatory variants associated with hematopoietic malignancies in retriever dog breeds. PLoS Genet. 2021, 17, e1009543. [Google Scholar] [CrossRef]
- Hédan, B.; Cadieu, E.; Rimbault, M.; Vaysse, A.; Dufaure de Citres, C.; Devauchelle, P.; Botherel, N.; Abadie, J.; Quignon, P.; Derrien, T.; et al. Identification of common predisposing loci to hematopoietic cancers in four dog breeds. PLoS Genet. 2021, 17, e1009395. [Google Scholar] [CrossRef]
- Cannon, C.; Borgatti, A.; Henson, M.; Husbands, B. Evaluation of a combination chemotherapy protocol including lomustine and doxorubicin in canine histiocytic sarcoma. J. Small Anim. Pract. 2015, 56, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Mason, S.L.; Finotello, R.; Blackwood, L. Epirubicin in the treatment of canine histiocytic sarcoma: Sequential, alternating and rescue chemotherapy. Vet. Comp. Oncol. 2018, 16, E30–E37. [Google Scholar] [CrossRef] [PubMed]
- Skorupski, K.A.; Rodriguez, C.O.; Krick, E.L.; Clifford, C.A.; Ward, R.; Kent, M.S. Long-term survival in dogs with localized histiocytic sarcoma treated with CCNU as an adjuvant to local therapy. Vet. Comp. Oncol. 2009, 7, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Erich, S.A.; Rutteman, G.R.; Teske, E. Causes of death and the impact of histiocytic sarcoma on the life expectancy of the Dutch population of Bernese Mountain dogs and Flat-coated retrievers. Vet. J. 2013, 198, 678–683. [Google Scholar] [CrossRef]
- Rosin, A.; Moore, P.; Dubielzig, R. Malignant histiocytosis in Bernese Mountain dogs. J. Am. Vet. Med. Assoc. 1986, 188, 1041–1045. [Google Scholar] [PubMed]
- Ramsey, I.K.; McKay, J.S.; Rudorf, H.; Dobson, J.M. Malignant histiocytosis in three Bernese Mountain dogs. Vet. Rec. 1996, 138, 440–444. [Google Scholar] [CrossRef]
- Takahashi, M.; Tomiyasu, H.; Hotta, E.; Asada, H.; Fukushima, K.; Kanemoto, H.; Fujino, Y.; Ohno, K.; Uchida, K.; Nakayama, H.; et al. Clinical characteristics and prognostic factors in dogs with histiocytic sarcomas in Japan. J. Vet. Med. Sci. 2014, 76, 661–666. [Google Scholar] [CrossRef][Green Version]
- Dervisis, N.G.; Kiupel, M.; Qin, Q.; Cesario, L. Clinical prognostic factors in canine histiocytic sarcoma. Vet. Comp. Oncol. 2017, 15, 1171–1180. [Google Scholar] [CrossRef]
- Erich, S.A.; Constantino-Casas, F.; Dobson, J.M.; Teske, E. Morphological distinction of histiocytic sarcoma from other tumor yypes in Bernese Mountain dogs and Flatcoated retrievers. In Vivo 2018, 32, 7–17. [Google Scholar]
- Heller, D.A.; Clifford, C.A.; Goldschmidt, M.H.; Holt, D.E.; Manfredi, M.J.; Sorenmo, K.U. Assessment of cyclooxygenase-2 expression in canine hemangiosarcoma, histiocytic sarcoma, and mast cell tumor. Vet. Pathol. 2005, 42, 350–353. [Google Scholar] [CrossRef]
- Lenz, J.A.; Furrow, E.; Craig, L.E.; Cannon, C.M. Histiocytic sarcoma in 14 miniature schnauzers—A new breed predisposition? J. Small Anim. Pract. 2017, 58, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Mariani, C.L.; Jennings, M.K.; Olby, N.J.; Borst, L.B.; Brown, J.C., Jr.; Robertson, I.D.; Seiler, G.S.; MacKillop, E. Histiocytic sarcoma with central nervous system involvement in dogs: 19 cases (2006–2012). J. Vet. Intern. Med. 2015, 29, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Rassnick, K.M.; Moore, A.S.; Russell, D.S.; Northrup, N.C.; Kristal, O.; Bailey, D.B.; Flory, A.B.; Kiselow, M.A.; Intile, J.L. Phase II, open-label trial of single-agent CCNU in dogs with previously untreated histiocytic sarcoma. J. Vet. Intern. Med. 2010, 24, 1528–1531. [Google Scholar] [CrossRef] [PubMed]
- Thaiwong, T.; Sirivisoot, S.; Takada, M.; Yuzbasiyan-Gurkan, V.; Kiupel, M. Gain-of-function mutation in PTPN11 in histiocytic sarcomas of Bernese Mountain dogs. Vet. Comp. Oncol. 2018, 16, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Thongtharb, A.; Uchida, K.; Chambers, J.K.; Kagawa, Y.; Nakayama, H. Histological and immunohistochemical studies on primary intracranial canine histiocytic sarcomas. J. Vet. Med. Sci. 2016, 78, 593–599. [Google Scholar] [CrossRef]
- Wouda, R.M.; Miller, M.E.; Chon, E.; Stein, T.J. Clinical effects of vinorelbine administration in the management of various malignant tumor types in dogs: 58 cases (1997–2012). J. Am. Vet. Med. Assoc. 2015, 246, 1230–1237. [Google Scholar] [CrossRef] [PubMed]
- Abadie, J.; Hédan, B.; Cadieu, E.; De Brito, C.; Devauchelle, P.; Bourgain, C.; Parker, H.G.; Vaysse, A.; Margaritte-Jeannin, P.; Galibert, F.; et al. Epidemiology, pathology, and genetics of histiocytic sarcoma in the Bernese Mountain Dog Breed. J. Hered. 2009, 100, S19–S27. [Google Scholar] [CrossRef]
- Hostettler, F.C.; Wiener, D.J.; Welle, M.M.; Posthaus, H.; Geissbühler, U. Post mortem computed tomography and core needle biopsy in comparison to autopsy in eleven bernese mountain dogs with histiocytic sarcoma. BMC Vet. Res. 2015, 11, 1–11. [Google Scholar] [CrossRef]
- Constantino-Casas, F.; Mayhew, D.; Hoather, T.M.; Dobson, J.M. The clinical presentation and histopathologic immunohistochemical classification of histiocytic sarcomas in the Flat Coated Retriever. Vet. Pathol. 2011, 48, 764–771. [Google Scholar] [CrossRef]
- Moore, P.F.; Affolter, V.K.; Vernau, W. Canine hemophagocytic histiocytic sarcoma: A proliferative disorder of CD11d+ macrophages. Vet. Pathol. 2006, 43, 632–645. [Google Scholar] [CrossRef]
- Kato, Y.; Murakami, M.; Hoshino, Y.; Mori, T.; Maruo, K.; Hirata, A.; Nakagawa, T.L.D.R.; Yanai, T.; Sakai, H. The class A macrophage scavenger receptor CD204 is a useful immunohistochemical marker of canine histiocytic sarcoma. J. Comp. Pathol. 2012, 148, 188–196. [Google Scholar] [CrossRef]
- Wada, M.; Hasegawa, D.; Hamamoto, Y.; Yu, Y.; Fujiwara-Igarashi, A.; Fujita, M. Comparisons among MRI signs, apparent diffusion coefficient, and fractional anisotropy in dogs with a solitary intracranial meningioma or histiocytic sarcoma. Vet. Radiol. Ultrasound 2017, 58, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Gelain, M.E.; Comazzi, S. Disseminated histiocytic sarcoma with peripheral blood involvement in a Bernese Mountain dog. Vet. Clin. Pathol. 2009, 38, 126–130. [Google Scholar] [CrossRef]
- Schmidt, M.L.; Rutteman, G.R.; van Niel, M.H.F. Clinical and radiographic manifestations of canine malignant histiocytosis. Vet. Quart. 1993, 15, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Skorupski, K.A.; Clifford, C.A.; Paoloni, M.C.; Lara-Garcia, A.; Barber, L.; Kent, M.S.; LeBlanc, A.K.; Sabhlok, A.; Mauldin, E.A.; Shofer, F.S.; et al. CCNU for the treatment of dogs with histiocytic sarcoma. J. Vet. Intern. Med. 2007, 21, 121–126. [Google Scholar] [CrossRef]
- VanderHart, D.J.; Reese, D.J.; Greenberg, S.M. What is your diagnosis? J. Am. Vet. Med. Assoc. 2014, 244, 899–901. [Google Scholar] [CrossRef]
- Soare, T.; Noble, P.-J.; Hetzel, U.; Fonfara, S.; Kipar, A. Paraneoplastic syndrome in haemophagocytic histiocytic sarcoma in a dog. J. Comp. Pathol. 2012, 146, 168–174. [Google Scholar] [CrossRef]
- Manor, E.K.; Craig, L.E.; Sun, X.; Cannon, C.M. Prior joint disease is associated with increased risk of periarticular histiocytic sarcoma in dogs. Vet. Comp. Oncol. 2018, 16, E83–E88. [Google Scholar] [CrossRef]
- Kuijk, V.L.; Ginkel, V.K.; Vos, D.J.P.; Brearly, M.J.; Butinar, J.; Gielen, I.; Garderen, V.E.; Chiers, K.; Verhoeven, P.S. Peri-articular histiocytic sarcoma and previous joint disease in Bernese Mountain dogs. J. Vet. Intern. Med. 2013, 27, 293–299. [Google Scholar] [CrossRef]
- Ide, T.; Uchida, K.; Kagawa, Y.; Suzuki, K.; Nakayama, H. Pathological and immunohistochemical features of subdural histiocytic sarcomas in 15 dogs. J. Vet. Diagn. Investig. 2011, 23, 127–132. [Google Scholar] [CrossRef]
- Hayden, D.W.; Waters, D.J.; Burke, B.A.; Manivel, J.C. Disseminated malignant histiocytosis in a Golden Retriever: Clinicopathologic, ultrastructural, and immunohistochemical findings. Vet. Pathol. 1993, 30, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Stowe, D.M.; Escobar, C.; Neel, J.A. What is your diagnosis? Cerebrospinal fluid from a dog. Vet. Clin. Pathol. 2012, 41, 429–430. [Google Scholar] [CrossRef] [PubMed]
- Repellin, R.; Barnhart, M.D.; Watson, A.T.; Hans, E.C. What is your neurologic diagnosis? J. Am. Vet. Med. Assoc. 2017, 251, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.; Eichelberger, B.; Hodo, C.; Cooper, J.; Porter, B. Imaging diagnosis—Spinal cord histiocytic sarcoma in a dog. Vet. Radiol. Ultrasound 2015, 56, 17–21. [Google Scholar] [CrossRef]
- Tamura, S.; Tamura, Y.; Nakamoto, Y.; Ozawa, T.; Uchida, K. MR imaging of histiocytic sarcoma of the canine brain. Vet. Radiol. Ultrasound 2009, 50, 178–181. [Google Scholar] [CrossRef]
- Thakkar, P.; Pease, A.P.; Guiot, L.P.; Fitzgerald, S.D.; Addai, B.A.; Smedley, R.C. What is your diagnosis? J. Am. Vet. Med. Assoc. 2013, 243, 1255–1257. [Google Scholar] [CrossRef]
- Tzipory, L.; Vernau, K.M.; Sturges, B.K.; Zabka, T.S.; Highland, M.A.; Petersen, S.A.; Wisner, E.R.; Moore, P.F.; Vernau, W. Antemortem diagnosis of localized central nervous system histiocytic sarcoma in 2 dogs. J. Vet. Intern. Med. 2009, 23, 369–374. [Google Scholar] [CrossRef]
- Marcinowska, A.; Constantino-Casas, F.; Dobson, J. Histiocytic sarcoma in a flat-coated retriever with central and peripheral nervous system sarcomatosis. Vet. Rec. Case Rep. 2014, 2, e000029. [Google Scholar]
- Hicks, J.; Barber, R.; Childs, B.; Kirejczyk, S.G.M.; Uhl, E.W. Canine histiocytic sarcoma presenting as a target lesion on brain magnetic resonance imaging and as a solitary pulmonary mass. Vet. Radiol. Ultrasound 2018, 59, E66–E70. [Google Scholar] [CrossRef]
- Tamura, K.; Ishigaki, K.; Iizuka, K.; Nagumo, T.; Yoshida, O.; Asano, K. Neutrophilic leucocytosis induced by granulocyte colony-stimulating factor and interleukin-6 in canine primary lung adenocarcinoma. Vet. Med. Sci. 2022, 8, 483–491. [Google Scholar] [CrossRef]
- Coussens, L.M.; Zitvogel, L.; Palucka, A.K. Neutralizing tumor-promoting chronic inflammation: A magic bullet? Science 2013, 339, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Carioto, L. Malignant histiocytosis in a Bernese Mountain dog presenting as a mandibular mass. Can. Vet. J. 1997, 38, 105–107. [Google Scholar] [PubMed]
- Castellanos, V.C.; O’Neill, S.; Seiler, G.S. What is your diagnosis? J. Am. Vet. Med. Assoc. 2010, 236, 1293–1294. [Google Scholar] [CrossRef] [PubMed]
- Peaston, A.E.; Munn, R.J.; Madewell, B.R. Clinical vignette. J. Vet. Intern. Med. 1993, 7, 101–103. [Google Scholar] [CrossRef] [PubMed]
- Dobson, J.; Villiers, E.; Roulois, A.; Gould, S.; Mellor, P.; Hoather, T.; Watson, P. Histiocytic sarcoma of the spleen in flat-coated retrievers with regenerative anaemia and hypoproteinaemia. Vet. Rec. 2006, 158, 825–829. [Google Scholar] [CrossRef]
- Cruz-Arámbulo, R.; Wrigley, R.; Powers, B. Sonographic features of histiocytic neoplasms in the canine abdomen. Vet. Radiol. Ultrasound 2004, 45, 554–558. [Google Scholar] [CrossRef]
- Kezer, K.A.; Barber, L.G.; Jennings, S.H. Efficacy of dacarbazine as a rescue agent for histiocytic sarcoma in dogs. Vet. Comp. Oncol. 2017, 16, 77–80. [Google Scholar] [CrossRef]
- Yamazaki, H.; Takagi, S.; Oh, N.; Hoshino, Y.; Hosoya, K.; Okumura, M. Comparative analysis of mRNA expression of surface antigens between histiocytic and nonhistiocytic sarcoma in dogs. J. Vet. Intern. Med. 2014, 28, 204–210. [Google Scholar] [CrossRef]
- Klahn, S.L.; Kitchell, B.E.; Dervisis, N.G. Evaluation and comparison of outcomes in dogs with periarticular and nonperiarticular histiocytic sarcoma. J. Am. Vet. Med. Assoc. 2011, 239, 90–96. [Google Scholar] [CrossRef]
- Eckersall, P.D.; Bell, R. Acute phase proteins: Biomarkers of infection and inflammation in veterinary medicine. Vet. J. 2010, 185, 23–27. [Google Scholar] [CrossRef]
- Uehlinger, P.; Glaus, T.; Hauser, B.; Reusch, C. Differential diagnosis for hypercalcemia—A retrospective study of 46 dogs. Schweiz. Arch. Tierheilkd. 1998, 140, 188–197. [Google Scholar] [PubMed]
- Barrett, L.E.; Pollard, R.E.; Zwingenberger, A.; Zierenberg-Ripoll, A.; Skorupski, K.A. Radiographic characterization of primary lung tumors in 74 dogs. Vet. Radiol. Ultrasound 2014, 55, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Haers, H.; Vignoli, M.; Paes, G.; Rossi, F.; Taeymans, O.; Daminet, S.; Saunders, J.H. Contrast harmonic ultrasonographic appearance of focal space-occupying renal lesions. Vet. Radiol. Ultrasound 2010, 51, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Ballegeer, E.A.; Forrest, L.J.; Dickinson, R.M.; Schutten, M.M.; Delaney, F.A.; Young, K.M. Correlation of ultrasonographic appearance of lesions and cytologic and histologic diagnoses in splenic aspirates from dogs and cats: 32 cases (2002–2005). J. Am. Vet. Med. Assoc. 2007, 230, 690–696. [Google Scholar] [CrossRef]
- Cuccovillo, A.; Lamb, C.R. Cellular features of sonographic target lesions of the liver and spleen in 21 dogs and a cat. Vet. Radiol. Ultrasound 2002, 43, 275–278. [Google Scholar] [CrossRef]
- Weiss, D.J. A retrospective study of the incidence and the classification of bone marrow disorders in the dog at a veterinary teaching hospital (1996–2004). J. Vet. Intern. Med. 2006, 20, 955–961. [Google Scholar]
- Weiss, D.J. Flow cytometric evaluation of hemophagocytic disorders in canine bone marrow. Vet. Clin. Pathol. 2002, 31, 36–41. [Google Scholar] [CrossRef]
- Ramirez, S.; Douglass, J.P.; Robertson, I.D. Ultrasonographic features of canine abdominal malignant histiocytosis. Vet. Radiol. Ultrasound 2002, 43, 167–170. [Google Scholar] [CrossRef]
- Labelle, P.; DeCock, H.E.V. Metastatic tumors to the adrenal glands in domestic animals. Vet. Pathol. 2005, 42, 52–58. [Google Scholar] [CrossRef]
- Bass, M.; Gardelle, O.; Grest, P.; Bernasconi, C. Localized histiocytic sarcoma in a dog: An uncommon diagnosis in forelimb lameness. Vet. Comp. Orthop. Traumatol. 2004, 17, 48–52. [Google Scholar]
- Seiler, S.M.F.; Baumgarter, C.; Hirschberger, J.; Beer, A.J.; Brühschwein, A.; Kreutzmann, N.; Laberke, S.; Wergin, M.C.; Meyer-Lindenberg, A.; Brandl, J.; et al. Comparative oncology: Evaluation of 2-deoxy-2 [18F]fluoro-d-glucose (FDG) positron emission tomography/computed tomography (PET/CT) for the staging of dogs with malignant tumors. PLoS ONE 2015, 10, e0127800. [Google Scholar] [CrossRef] [PubMed]
| Parameter | FCR | BMD | p-Value |
|---|---|---|---|
| Number of dogs | 289 | 365 | |
| Age (years; mean, range) | 8.2 (0.6–13.7) | 6.6 (0.6–12.2) | <0.001 |
| Sex | 0.981 | ||
| Male (number, %) | 142 (49.1%) | 181 (49.6%) | |
| Female (number, %) | 147 (50.9%) | 184 (50.4%) | |
| Neutered (all sexes) | 109 (37.7%) | 189 (51.8%) | <0.001 |
| Male | 39 (27.6%) | 75 (41.4%) | 0.010 |
| Female | 70 (47.6%) | 114 (61.9%) | 0.013 |
| HS-type | <0.001 | ||
| Localized | 175 (60.6%) | 143 (39.2%) | |
| Disseminated | 114 (39.4%) | 222 (60.8%) | |
| TimeToDx (mean, range) a | 35.6 (0–500) | 27.9 (0–377) | 0.103 |
| FUperiod (mean, range) b | 105.6 (0–2507) | 102.5 (0–2570) | 0.888 |
| Parameter | FCR | BMD | p-Value |
|---|---|---|---|
| Number of dogs | 289 | 365 | |
| Pale mucous membranes | 67 (23.3%) | 151 (41.4%) | <0.001 |
| Icterus | 9 (3.1%) | 26 (7.1%) | 0.023 |
| Pyrexia | 35 (12.2%) | 92 (25.3%) | <0.001 |
| Lameness | 114 (39.5%) | 87 (23.8%) | <0.001 |
| Front leg | 89 (78.1%) | 38 (43.7%) | <0.001 |
| Hind leg | 25 (21.9%) | 44 (56.3%) | |
| Neurologic Symptoms | 13 (4.5%) | 14 (3.8%) | 0.776 |
| Ataxia | 2 | 7 | |
| Epilepsy | 2 | 2 | |
| Paresis/paralysis | 10 | 10 | |
| Weight loss | 46 (15.9%) | 132 (36.2%) | <0.001 |
| Ascites | 5 (1.7%) | 15 (4.1%) | 0.078 |
| Anorexia | 75 (26.0%) | 207 (56.7%) | <0.001 |
| Vomiting | 19 (6.6%) | 29 (7.9%) | 0.504 |
| Diarrhea | 5 (1.7%) | 10 (2.7%) | 0.389 |
| Polydipsia | 21 (7.3%) | 30 (8.2%) | 0.652 |
| Dyspnoea | 19 (6.6%) | 38 (10.4%) | 0.080 |
| Coughing | 13 (4.5%) | 41 (11.2%) | 0.001 |
| Lethargy | 98 (33.9%) | 215 (58.9%) | <0.001 |
| Parameter | FCR | BMD | p-Value | LHS | DHS | p-Value |
|---|---|---|---|---|---|---|
| Anemia | ||||||
| PCV < ref.value | 58/78 (74.4%) | 124/160 (77.0%) | 0.650 | 46/67 (68.7%) | 136/171 (79.5%) | 0.075 |
| PCV< 0.30 l/l | 24/78 (30.8%) | 63/160 (39.1%) | 0.384 | 9/67 (13.4%) | 78/171 (45.6%) | <0.001 |
| Leukocytosis | 15/44 (34.1%) | 72/108 (66.7%) | <0.001 | 20/39 (51.3%) | 67/113 (59.3%) | 0.383 |
| Thrombocytopenia | 14/41 (34.1%) | 52/94 (55.3%) | 0.027 | 7/31 (22.6%) | 59/104 (56.7%) | 0.003 |
| Elevated AP | 34/43 (79.1%) | 60/87 (69.0%) | 0.316 | 21/33 (63.6%) | 70/93 (75.2%) | 0.200 |
| Hypoalbuminemia | 26/41 (63.4%) | 61/94 (64.9%) | 0.869 | 14/36 (38.9%) | 65/99 (65.7%) | 0.626 |
| Hypercalcemia | 0/23 (0%) | 15/53 (28.3%) | <0.001 | 4/23 (17.4%) | 11/53 (20.8%) | 0.735 |
| Abn. coagulation | 4/5 | 8/17 | 0.179 | 1/3 | 11/19 | 0.427 |
| Parameter | FCR | BMD | p-Value |
|---|---|---|---|
| Clinical parameters | n = 289 | n = 365 | |
| External lymph nodes | 71 (24.6%) | 60 (16.4%) | 0.010 |
| Limb | <0.001 | ||
| Front leg | 102 (35.2%) | 25 (6.8%) | |
| Shoulder | 33 | 3 | |
| Elbow | 30 | 18 | |
| Carpal joint | 2 | 1 | |
| Other | 37 | 3 | |
| Hind leg | 31 (10.7%) | 36 (9.8%) | |
| Knee | 15 | 28 | |
| Tarsal joint | 2 | 1 | |
| Other | 16 | 7 | |
| Eye | 1 (0.3%) | 6 (1.6%) | 0.087 |
| CNS a | 5 (1.7%) | 3 (0.8%) | 0.312 |
| Soft tissue head/neck/trunk | 48 (16.6%) | 58 (15.9%) | 0.804 |
| Abdominal organs (based on imaging, expl laporatomy, and p.m. examination) | n = 88 | n = 185 | |
| Spleen | <0.001 | ||
| Single mass | 14 (15.9%) | 10 (5.4%) | |
| Multiple masses | 49 (55.6%) | 49 (26.5%) | |
| Diffuse | 1 (1.1%) | 43 (23.2%) | |
| Diffuse + masses | 1 (1.1%) | 11 (5.9%) | |
| Liver | 0.003 | ||
| Single mass | 2 (2.3%) | 9 (4.9%) | |
| Multiple masses | 29 (32.9%) | 57 (30.8%) | |
| Diffuse | 3 (3.4%) | 37 (20.0%) | |
| Diffuse + masses | 0 | 3 (1.6%) | |
| Lymph nodes | 0.939 | ||
| Abdominal | 21 (23.9%) | 45 (24.3%) | |
| Pelvic | 4 (4.5%) | 11 (5.9%) | |
| Abd + pelvic | 0 | 4 (2.2%) | |
| Kidney | 0.231 | ||
| Unilateral | 3 (3.4%) | 6 (3.2%) | |
| Bilateral | 7 (2.4%) | 6 (1.6%) | |
| Gastrointestinal tract | 2 (2.3%) | 3 (1.6%) | 0.169 |
| Thoracic organs (based on imaging and p.m. examination) | n = 73 | n = 167 | |
| Lungs | 0.058 | ||
| Single mass | 10 (13.7%) | 33 (19.8%) | |
| Multiple masses | 15 (20.5%) | 52 (31.1%) | |
| Thor. lymph nodes | 15 (20.5%) | 40 (23.9%) | 0.564 |
| Heart | 0.572 | ||
| Pericard | 2 (2.7%) | 2 (1.2%) | |
| Myocard | 1 (1.4%) | 1 (0.6%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erich, S.A.; Dobson, J.M.; Teske, E. Comparison of the Clinical Characteristics of Histiocytic Sarcoma in Bernese Mountain Dogs and Flat-Coated Retrievers. Vet. Sci. 2022, 9, 498. https://doi.org/10.3390/vetsci9090498
Erich SA, Dobson JM, Teske E. Comparison of the Clinical Characteristics of Histiocytic Sarcoma in Bernese Mountain Dogs and Flat-Coated Retrievers. Veterinary Sciences. 2022; 9(9):498. https://doi.org/10.3390/vetsci9090498
Chicago/Turabian StyleErich, Suzanne A., Jane M. Dobson, and Erik Teske. 2022. "Comparison of the Clinical Characteristics of Histiocytic Sarcoma in Bernese Mountain Dogs and Flat-Coated Retrievers" Veterinary Sciences 9, no. 9: 498. https://doi.org/10.3390/vetsci9090498
APA StyleErich, S. A., Dobson, J. M., & Teske, E. (2022). Comparison of the Clinical Characteristics of Histiocytic Sarcoma in Bernese Mountain Dogs and Flat-Coated Retrievers. Veterinary Sciences, 9(9), 498. https://doi.org/10.3390/vetsci9090498








