Pilot Study: Assessing the Expression of Serum Lactate Dehydrogenase and Peripheral Leukocyte Ratios in Canine Oral Malignant Melanoma
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection Criteria
2.2. Evaluation of Serum LDH
2.3. Evaluation of NLR and LMR
2.4. Statistical Analysis
3. Results
3.1. Patient Population
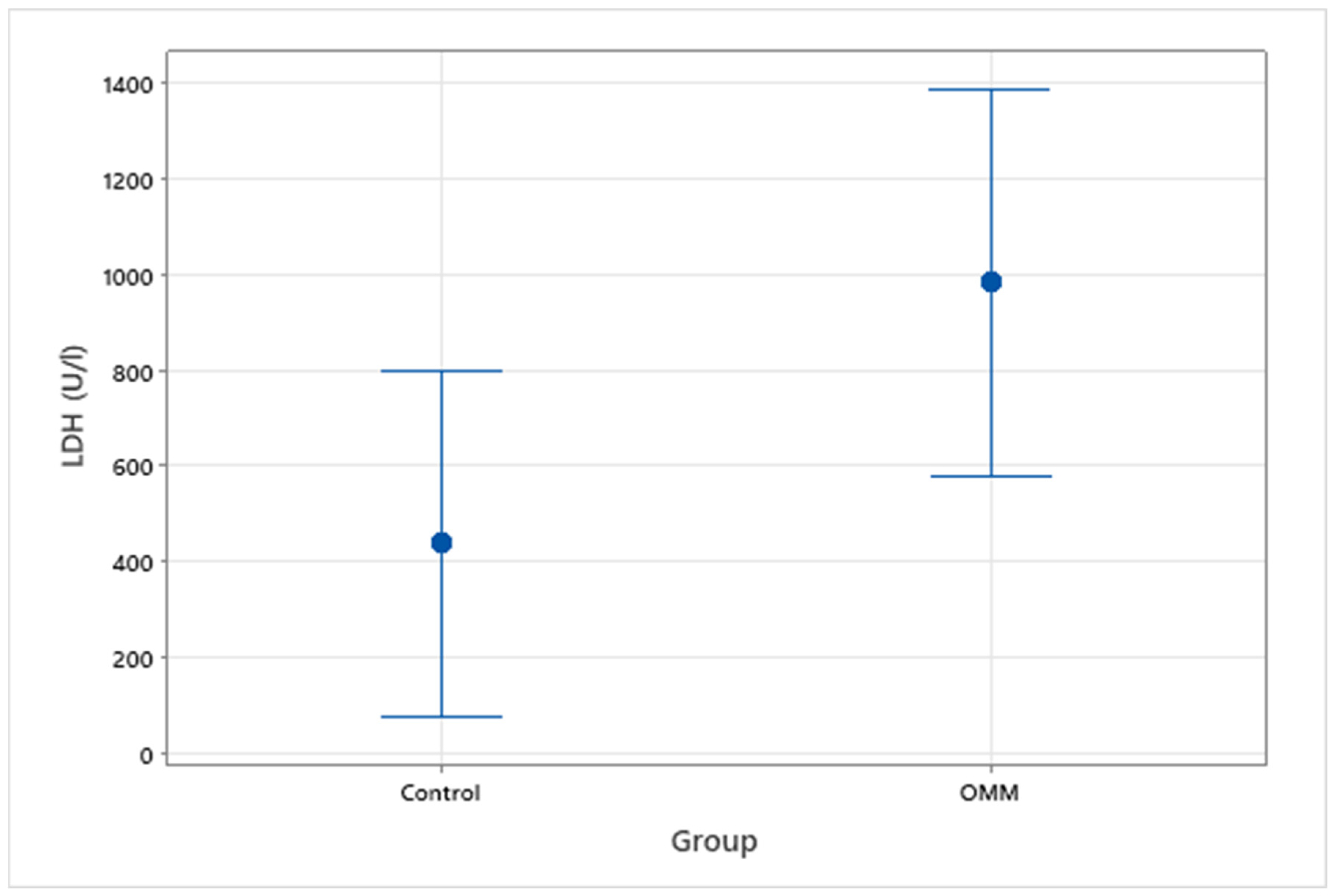
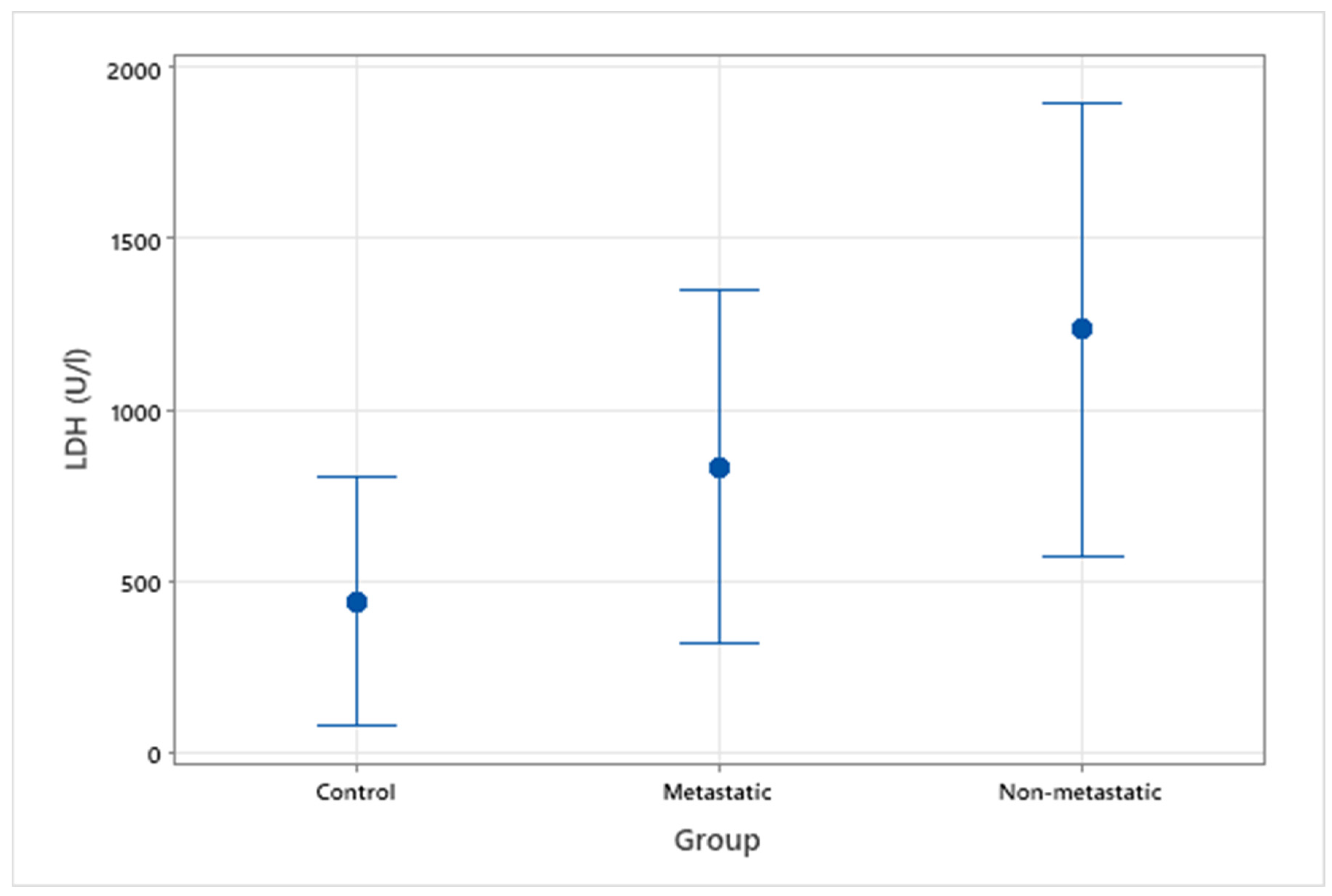
3.2. Serum LDH Values between Groups
3.3. NLR and LMR between Groups
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bergman, P.J. Canine Oral Melanoma. Clin. Tech. Small Anim. Pract. 2007, 22, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Wingo, K. Histopathologic Diagnoses From Biopsies of the Oral Cavity in 403 Dogs and 73 Cats. J. Veter Dent. 2018, 35, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Liptak, J.M. Oral tumors In Withrow and MacEwen’s Small Animal Clinical Oncology, 6th ed.; Vail, D.M., Thamm, D.H., Liptak, J.M., Eds.; Elsevier: St. Louis, MO, USA, 2020; pp. 432–448. [Google Scholar]
- Hahn, K.A.; DeNicola, D.B.; Richardson, R.C.; Hahn, E.A. Canine oral malignant melanoma: Prognostic utility of an alternative staging system. J. Small Anim. Pract. 1994, 35, 251–256. [Google Scholar] [CrossRef]
- Smedley, R.C.; Spangler, W.L.; Esplin, D.G.; Kitchell, B.E.; Bergman, P.J.; Ho, H.-Y.; Bergin, I.L.; Kiupel, M. Prognostic Markers for Canine Melanocytic Neoplasms. Veter Pathol. 2011, 48, 54–72. [Google Scholar] [CrossRef] [PubMed]
- Camerino, M.; Giacobino, D.; Iussich, S.; Ala, U.; Riccardo, F.; Cavallo, F.; Martano, M.; Morello, E.; Buracco, P. Evaluation of prognostic impact of pre-treatment neutrophil to lymphocyte and lymphocyte to monocyte ratios in dogs with oral malignant melanoma treated with surgery and adjuvant CSPG4 -antigen electrovaccination: An explorative study. Veter Comp. Oncol. 2021, 19, 353–361. [Google Scholar] [CrossRef]
- Bergin, I.L.; Smedley, R.C.; Esplin, D.G.; Spangler, W.L.; Kiupel, M. Prognostic Evaluation of Ki67 Threshold Value in Canine Oral Melanoma. Veter Pathol. 2011, 48, 41–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giacobino, D.; Camerino, M.; Riccardo, F.; Cavallo, F.; Tarone, L.; Martano, M.; Dentini, A.; Iussich, S.; Lardone, E.; Franci, P.; et al. Difference in outcome between curative intent vs marginal excision as a first treatment in dogs with oral malignant melanoma and the impact of adjuvant CSPG4-DNA electrovaccination: A retrospective study on 155 cases. Veter Comp. Oncol. 2021, 19, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Grosenbaugh, D.A.; Leard, A.T.; Bergman, P.J.; Klein, M.K.; Meleo, K.; Susaneck, S.; Hess, P.R.; Jankowski, M.K.; Jones, P.D.; Leibman, N.F.; et al. Safety and efficacy of a xenogeneic DNA vaccine encoding for human tyrosinase as adjunctive treatment for oral malignant melanoma in dogs following surgical excision of the primary tumor. Am. J. Veter Res. 2011, 72, 1631–1638. [Google Scholar] [CrossRef]
- Treggiari, E.; Grant, J.P.; North, S.M. A retrospective review of outcome and survival following surgery and adjuvant xenogeneic DNA vaccination in 32 dogs with oral malignant melanoma. J. Veter Med Sci. 2016, 78, 845–850. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.Y.; Lee, J.; Diefenbach, R.; Kefford, R.; Rizos, H. Liquid biomarkers in melanoma: Detection and discovery. Mol. Cancer 2018, 17, 1–14. [Google Scholar] [CrossRef]
- Weinstein, D.; Leininger, J.; Hamby, C.; Safai, B. Diagnostic and prognostic biomarkers in melanoma. J. Clin. Aesthetic Dermatol. 2014, 7, 13–24. [Google Scholar]
- Campos, L.; Lavalle, G.; Estrela-Lima, A.; de Faria, J.M.; Guimarães, J.; Dutra; Ferreira, E.; de Sousa, L.; Rabelo; da Costa, A.V.; et al. CA15.3, CEA and LDH in Dogs with Malignant Mammary Tumors. J. Veter Intern. Med. 2012, 26, 1383–1388. [Google Scholar] [CrossRef]
- Marconato, L.; Crispino, G.; Finotello, R.; Mazzotti, S.; Salerni, F.; Zini, E. Serum lactate dehydrogenase activity in canine malignancies. Veter Comp. Oncol. 2009, 7, 236–243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keung, E.Z.; Gershenwald, J.E. The eighth edition American Joint Committee on Cancer (AJCC) melanoma staging system: Implications for melanoma treatment and care. Expert Rev. Anticancer Ther. 2018, 18, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Blank, C.U.; Kelderman, S.; Van Tinteren, H.; Heemskerk, B.; Brom, R.V.D.; Hospers, G.A.; Eertwegh, A.J.V.D.; Kapiteijn, E.; De Groot, J.W.; Jansen, R.L.; et al. Serum lactate dehydrogenase (LDH) as a prognostic selection criterion for ipilimumab treatment in metastatic melanoma. J. Clin. Oncol. 2013, 31, 3036. [Google Scholar] [CrossRef]
- Agarwala, S.S.; Keilholz, U.; Gilles, E.; Bedikian, A.Y.; Wu, J.; Kay, R.; Stein, C.A.; Itri, L.M.; Suciu, S.; Eggermont, A.M. LDH correlation with survival in advanced melanoma from two large, randomised trials (Oblimersen GM301 and EORTC 18951). Eur. J. Cancer 2009, 45, 1807–1814. [Google Scholar] [CrossRef] [PubMed]
- Zanatta, R.; Abate, O.; D’Angelo, A.; Miniscalco, B.; Mannelli, A. Diagnostic and prognostic value of serum lactate dehydrogenase (LDH) and LDH isoenzymes in canine lymphoma. Veter Res. Commun. 2003, 27 (Suppl. 1), 449–452. [Google Scholar] [CrossRef] [PubMed]
- Choisunirachon, N.; Klansnoh, U.; Phoomvuthisarn, P.; Pisamai, S.; Thanaboonnipat, C.; Rungsi, A. The expression of serum lactate dehydrogenase in canine oral tumors. Thai J. Vet. Med. 2019, 49, 283–288. [Google Scholar]
- Paramanathan, A.; Saxena, A.; Morris, D.L. A systematic review and meta-analysis on the impact of pre-operative neutrophil lymphocyte ratio on long term outcomes after curative intent resection of solid tumours. Surg. Oncol. 2014, 23, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Zaragoza, J.; Caille, A.; Beneton, N.; Bens, G.; Christiann, F.; Maillard, H.; Machet, L. High neutrophil to lymphocyte ratio measured before starting ipilimumab treatment is associated with reduced overall survival in patients with melanoma. Br. J. Dermatol. 2016, 174, 146–151. [Google Scholar] [CrossRef]
- Nishijima, T.F.; Muss, H.B.; Shachar, S.S.; Tamura, K.; Takamatsu, Y. Prognostic value of lymphocyte-to-monocyte ratio in patients with solid tumors: A systematic review and meta-analysis. Cancer Treat. Rev. 2015, 41, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Chiti, L.E.; Martano, M.; Ferrari, R.; Boracchi, P.; Giordano, A.; Grieco, V.; Buracco, P.; Iussich, S.; Giudice, C.; Miniscalco, B.; et al. Evaluation of leukocyte counts and neutrophil-to-lymphocyte ratio as predictors of local recurrence of feline injection site sarcoma after curative intent surgery. Veter Comp. Oncol. 2020, 18, 105–116. [Google Scholar] [CrossRef]
- Sottnik, J.; Rao, S.; Lafferty, M.; Thamm, D.; Morley, P.; Withrow, S.; Dow, S. Association of Blood Monocyte and Lymphocyte Count and Disease-Free Interval in Dogs with Osteosarcoma. J. Veter Intern. Med. 2010, 24, 1439–1444. [Google Scholar] [CrossRef] [PubMed]
- Marconato, L.; Martini, V.; Stefanello, D.; Moretti, P.; Ferrari, R.; Comazzi, S.; Laganga, P.; Riondato, F.; Aresu, L. Peripheral blood lymphocyte/monocyte ratio as a useful prognostic factor in dogs with diffuse large B-cell lymphoma receiving chemoimmunotherapy. Veter J. 2015, 206, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, M.J.; Macfarlane, L.L.; Scase, T.; Parkin, T.; Morris, J.S. Use of neutrophil to lymphocyte ratio for predicting histopathological grade of canine mast cell tumours. Veter Rec. 2016, 179, 491. [Google Scholar] [CrossRef]
- Kopanke, J.H.; Chen, A.V.; Brune, J.E.; Brenna, A.C.; Thomovsky, S.A. Reference intervals for the activity of lactate dehydrogenase and its isoenzymes in the serum and cerebrospinal fluid of healthy canines. Veter Clin. Pathol. 2018, 47, 267–274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forkasiewicz, A.; Dorociak, M.; Stach, K.; Szelachowski, P.; Tabola, R.; Augoff, K. The usefulness of lactate dehydrogenase measurements in current oncological practice. Cell. Mol. Biol. Lett. 2020, 25, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Grecian, R.; Whyte, M.K.B.; Walmsley, S. The role of neutrophils in cancer. Br. Med Bull. 2018, 128, 5–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faria, S.S.; Fernandes, P.C., Jr.; Silva, M.J.B.; Lima, V.C.; Fontes, W.; Freitas-Junior, R.; Eterovic, A.K.; Forget, P. The neutrophil-to-lymphocyte ratio: A narrative review. Ecancermedicalscience 2016, 10, 702. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Haematological Parameter | Reference Values |
|---|---|
| White blood cell count (×109/L) | 6.0–15.0 |
| Neutrophil count (×109/L) | 3.6–12.0 |
| Lymphocyte count (×109/L) | 0.7–4.8 |
| Monocyte count (×109/L) | 0–1.5 |
| Patient | Age (Years) | Breed | Sex | Group | Tumour Location | LN Metastasis | Lung Metastasis | LN Assessment | Chest Assessment | LDH (U/L) | NLR | LMR |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 11 | Cross breed | FE | Metastatic | Gingival | Yes | No | FNA | 3-view chest X-ray | 442 | 2.46 | 4.00 |
| 2 | 12 | Labrador | ME | Metastatic | Gingival | Yes | No | FNA | CT | 611 | 7.73 | 3.67 |
| 3 | 9 | Labrador | FN | Metastatic | Lingual | Yes | Yes | FNA | CT | 198 | 9.11 | 1.50 |
| 4 | 10 | Cocker spaniel | FN | Metastatic | Gingival | Yes | No | FNA | CT | 2032 | 5.37 | 4.67 |
| 5 | 10 | Cocker spaniel | FN | Metastatic | Gingival | Yes | No | Histopathology | CT | 885 | 7.50 | 1.46 |
| 6 | 13 | Tibetan terrier | ME | Metastatic | Gingival | Yes | No | FNA | CT | 1.94 | 5.33 | |
| 7 | 8 | Golden retriever | ME | Metastatic | Gingival | Yes | No | Histopathology | CT | 3.43 | 4.20 | |
| 8 | 10 | Golden retriever | FN | Metastatic | Gingival | Yes | No | Histopathology | CT | 3.04 | 7.67 | |
| 9 | 9 | Red setter | ME | Metastatic | Gingival | Yes | No | Histopathology | 3-view chest X-ray | 6.15 | 2.17 | |
| 10 | 5 | Labrador | MN | Non-metastatic | Gingival | No | No | Histopathology | 3-view chest X-ray | 1612 | 3.40 | 5.00 |
| 11 | 8 | Labrador | ME | Non-metastatic | Gingival | No | No | Histopathology | 3-view chest X-ray | 1762 | ||
| 12 | 11 | Labrador | FN | Non-metastatic | Gingival | No | No | Histopathology | CT | 327 | 4.29 | 2.43 |
| 13 | 13 | Border terrier | FN | Non-metastatic | Gingival | No | No | Histopathology | CT | 7.90 | 1.11 | |
| 14 | 12 | Bearded collie | MN | Non-metastatic | Gingival | No | No | Histopathology | 3-view chest X-ray | 11.57 | 1.17 | |
| 15 | 12 | Cocker spaniel | FN | Non-metastatic | Gingival | No | No | Histopathology | 3-view chest X-ray | 1.14 | 5.25 | |
| 16 | 1 | Hungarian vizsla | MN | Control | 505 | |||||||
| 17 | 10 | Cross breed | FE | Control | 294 | |||||||
| 18 | 11 | Labrador | MN | Control | 488 | |||||||
| 19 | 8 | Spaniel (unspecified) | FE | Control | 228 | |||||||
| 20 | 2 | Golden retriever | FN | Control | 278 | |||||||
| 21 | 4 | Goldenretriever | ME | Control | 405 | |||||||
| 22 | 3 | Cocker spaniel | FN | Control | 303 | |||||||
| 23 | 11 | Labrador | FN | Control | 1368 | |||||||
| 24 | 1 | Cocker spaniel | FN | Control | 235 | |||||||
| 25 | 2 | Golden retriever | MN | Control | 290 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murray, B.; Blacklock, K.L.B. Pilot Study: Assessing the Expression of Serum Lactate Dehydrogenase and Peripheral Leukocyte Ratios in Canine Oral Malignant Melanoma. Vet. Sci. 2022, 9, 421. https://doi.org/10.3390/vetsci9080421
Murray B, Blacklock KLB. Pilot Study: Assessing the Expression of Serum Lactate Dehydrogenase and Peripheral Leukocyte Ratios in Canine Oral Malignant Melanoma. Veterinary Sciences. 2022; 9(8):421. https://doi.org/10.3390/vetsci9080421
Chicago/Turabian StyleMurray, Ben, and Kelly L. Bowlt Blacklock. 2022. "Pilot Study: Assessing the Expression of Serum Lactate Dehydrogenase and Peripheral Leukocyte Ratios in Canine Oral Malignant Melanoma" Veterinary Sciences 9, no. 8: 421. https://doi.org/10.3390/vetsci9080421
APA StyleMurray, B., & Blacklock, K. L. B. (2022). Pilot Study: Assessing the Expression of Serum Lactate Dehydrogenase and Peripheral Leukocyte Ratios in Canine Oral Malignant Melanoma. Veterinary Sciences, 9(8), 421. https://doi.org/10.3390/vetsci9080421






