The Italian Network of Laboratories for Veterinary Oncology (NILOV) 2.0: Improving Knowledge on Canine Tumours
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection and Handling
2.2. Data Analysis
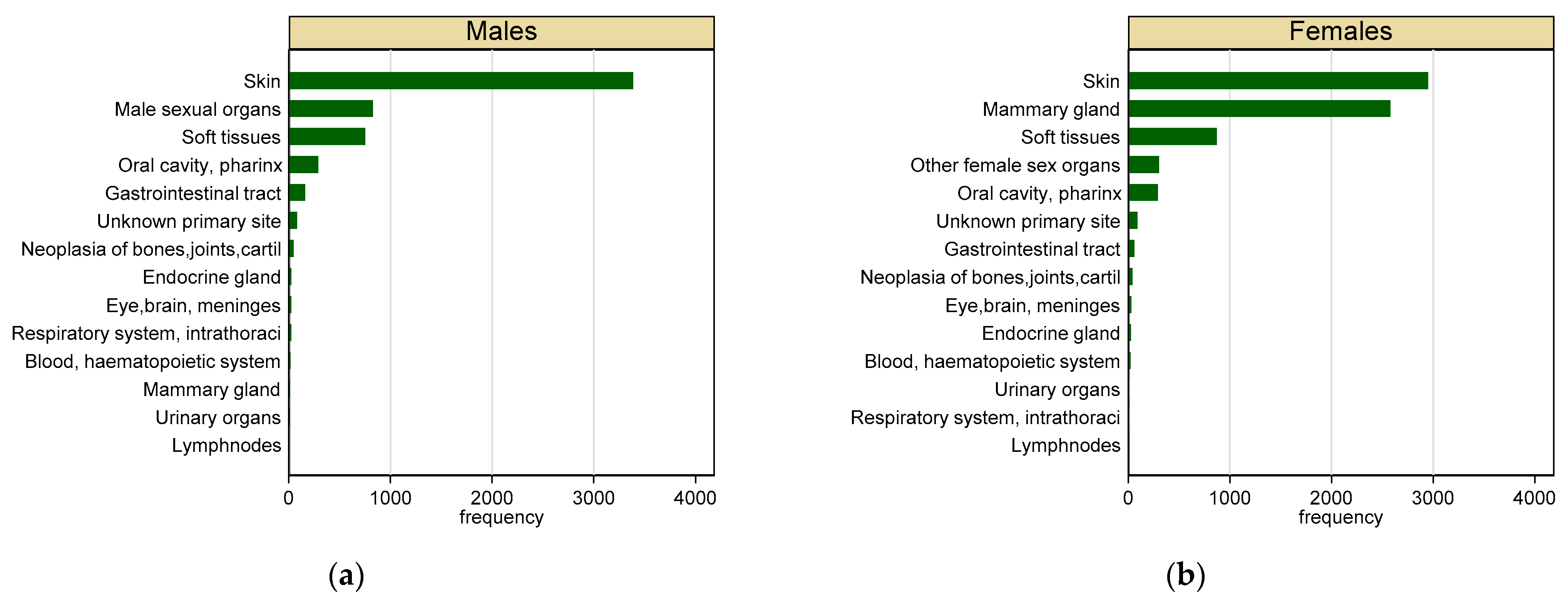
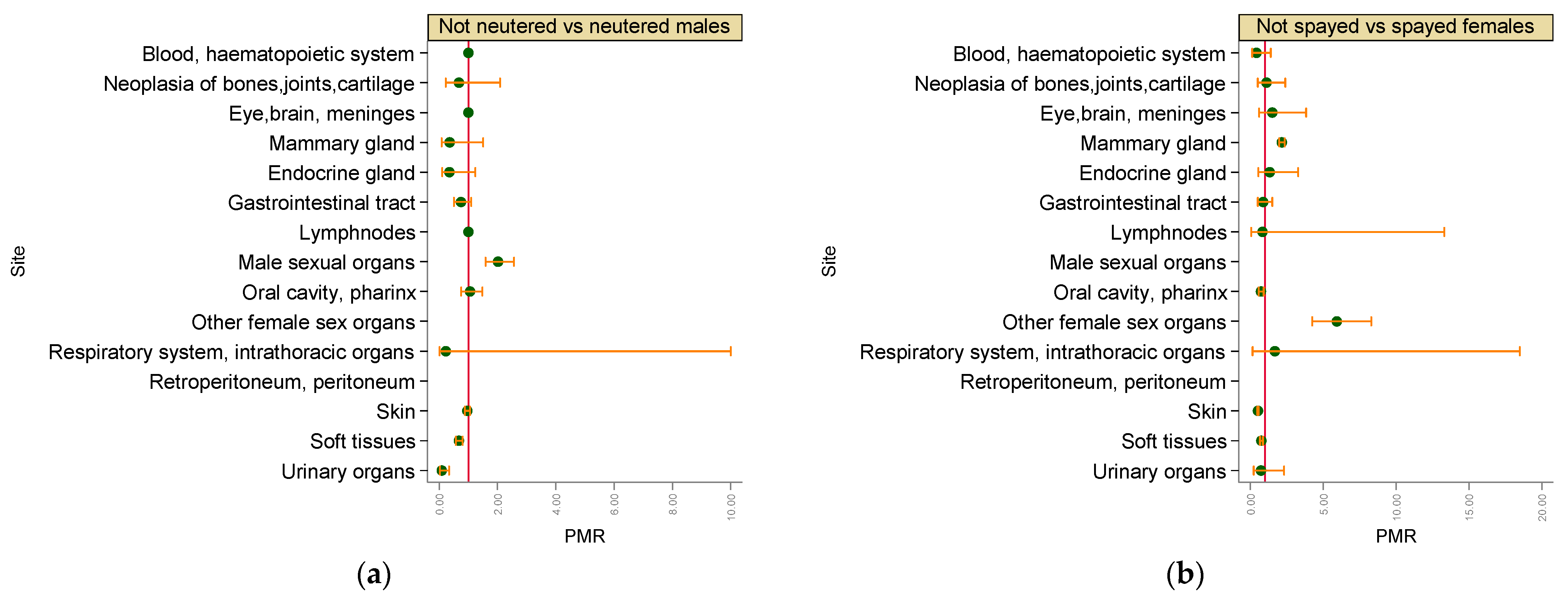
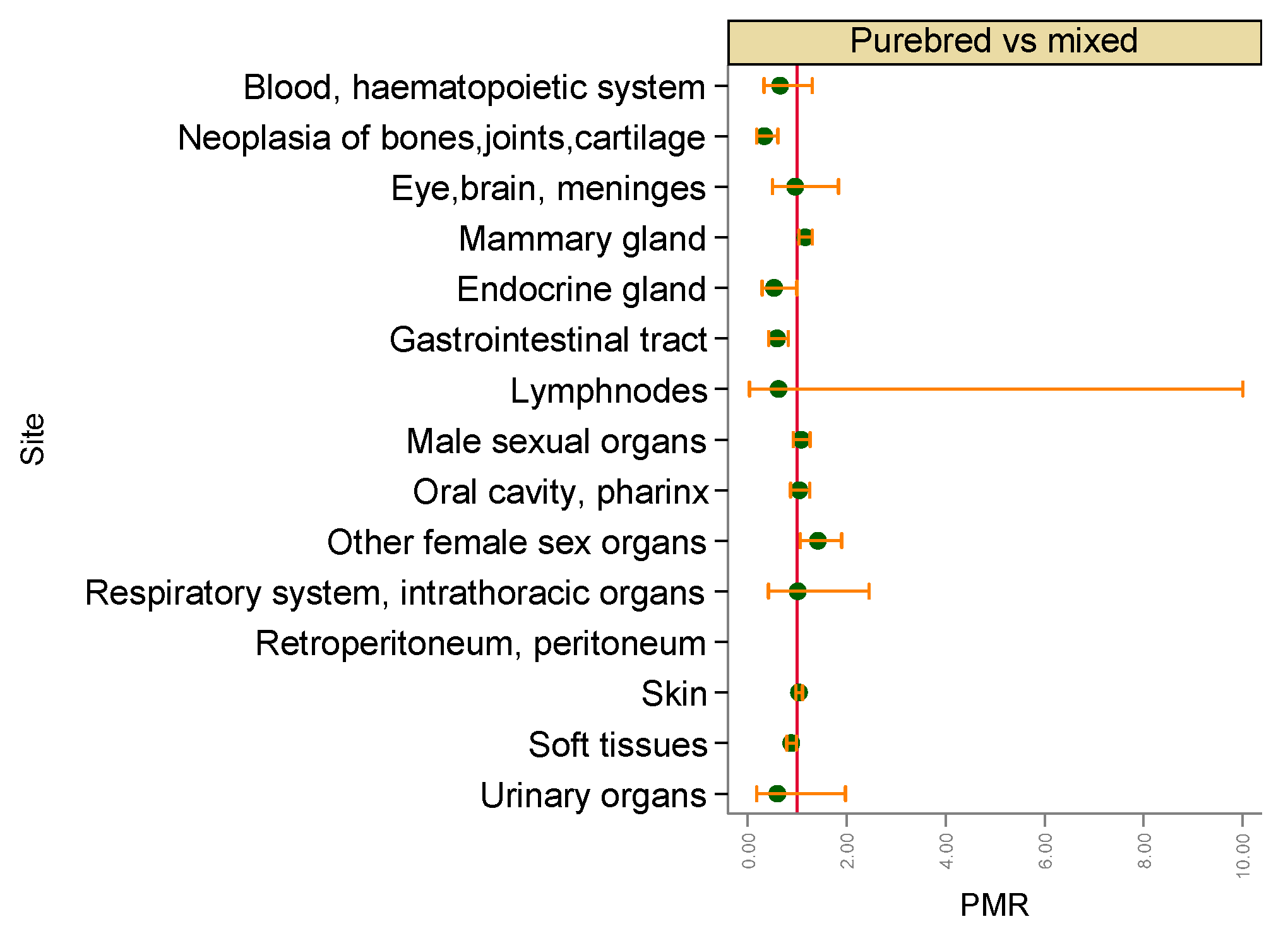
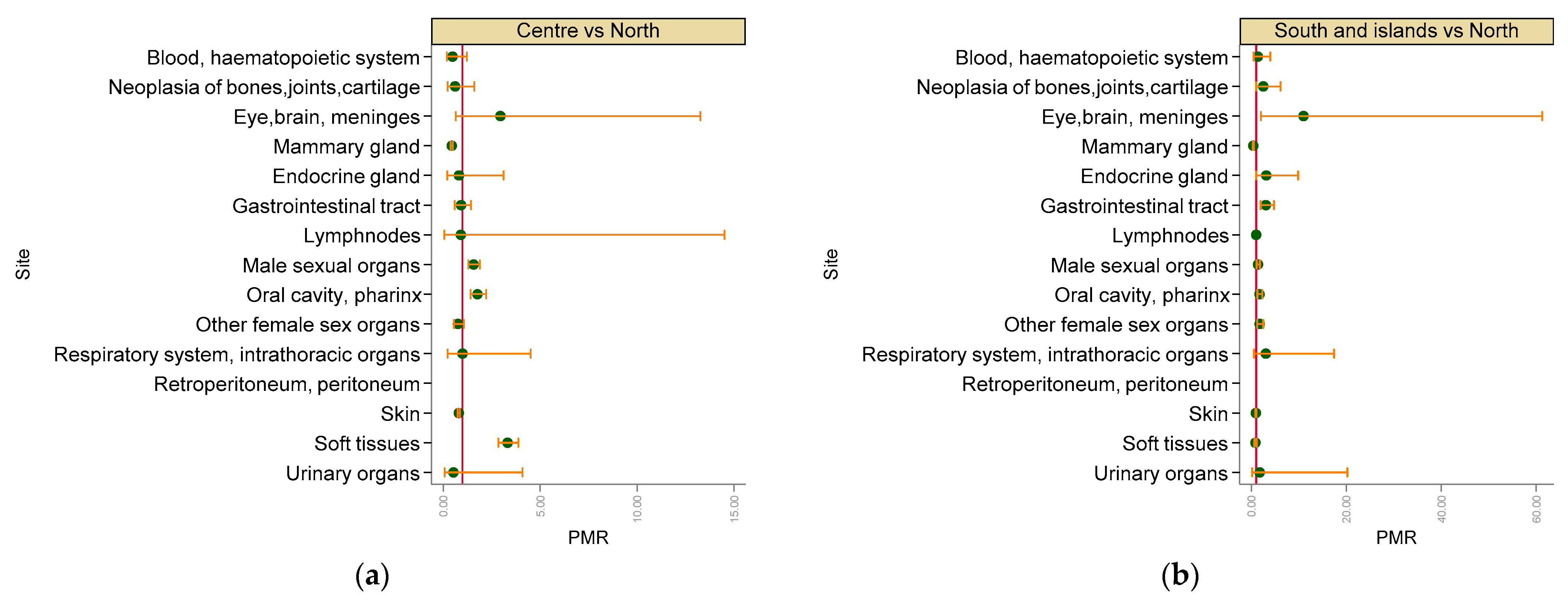
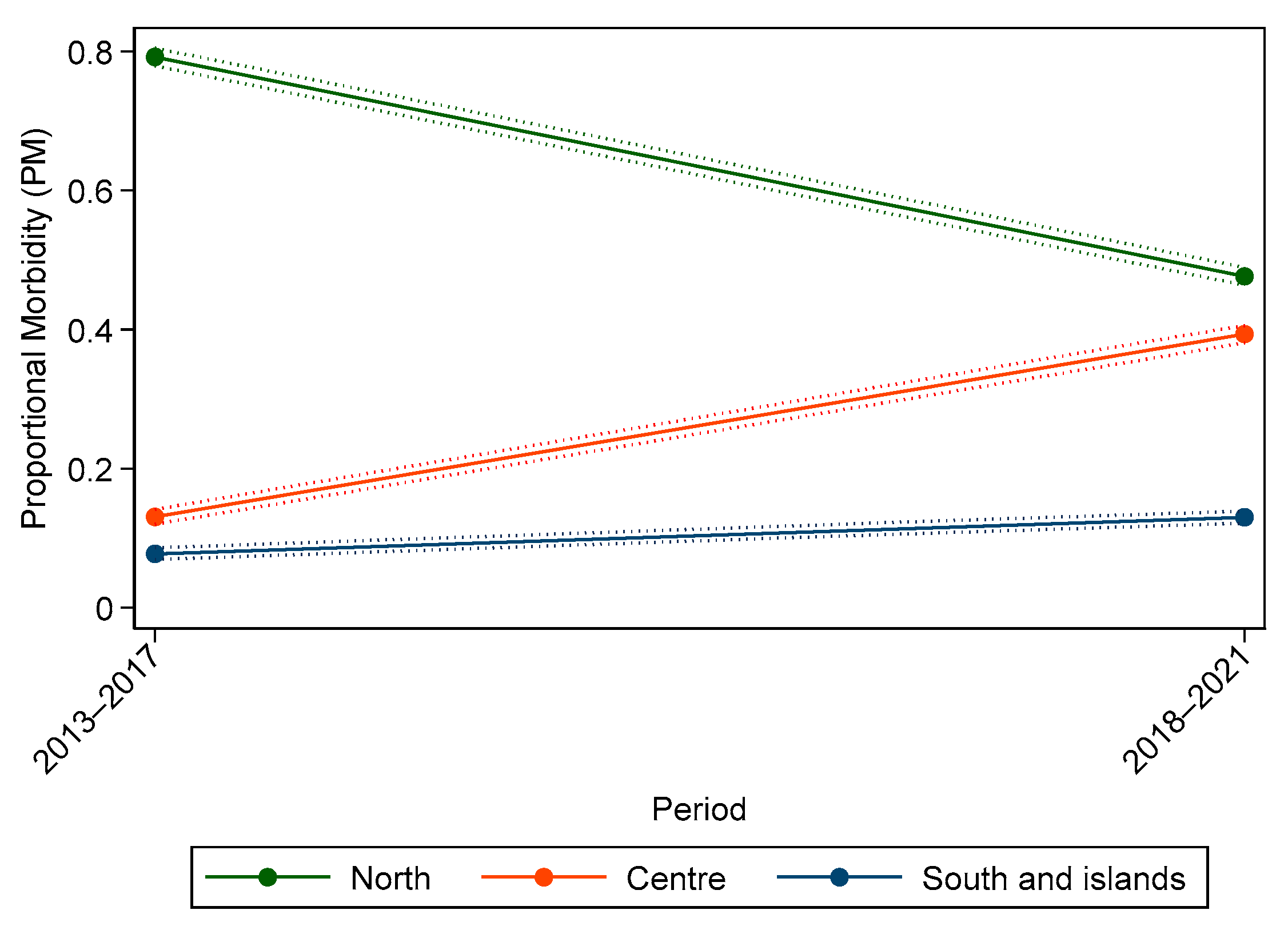
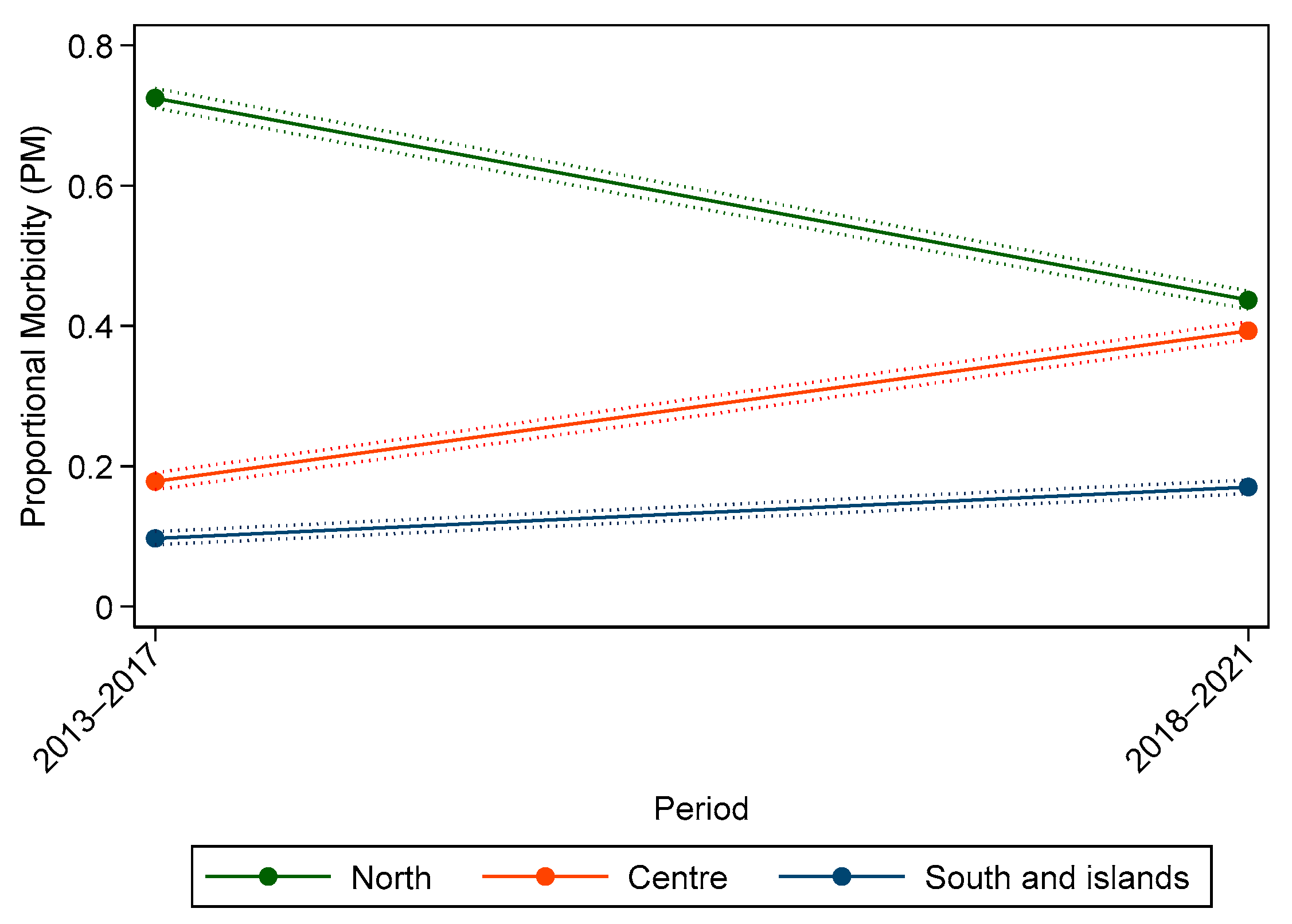
3. Results
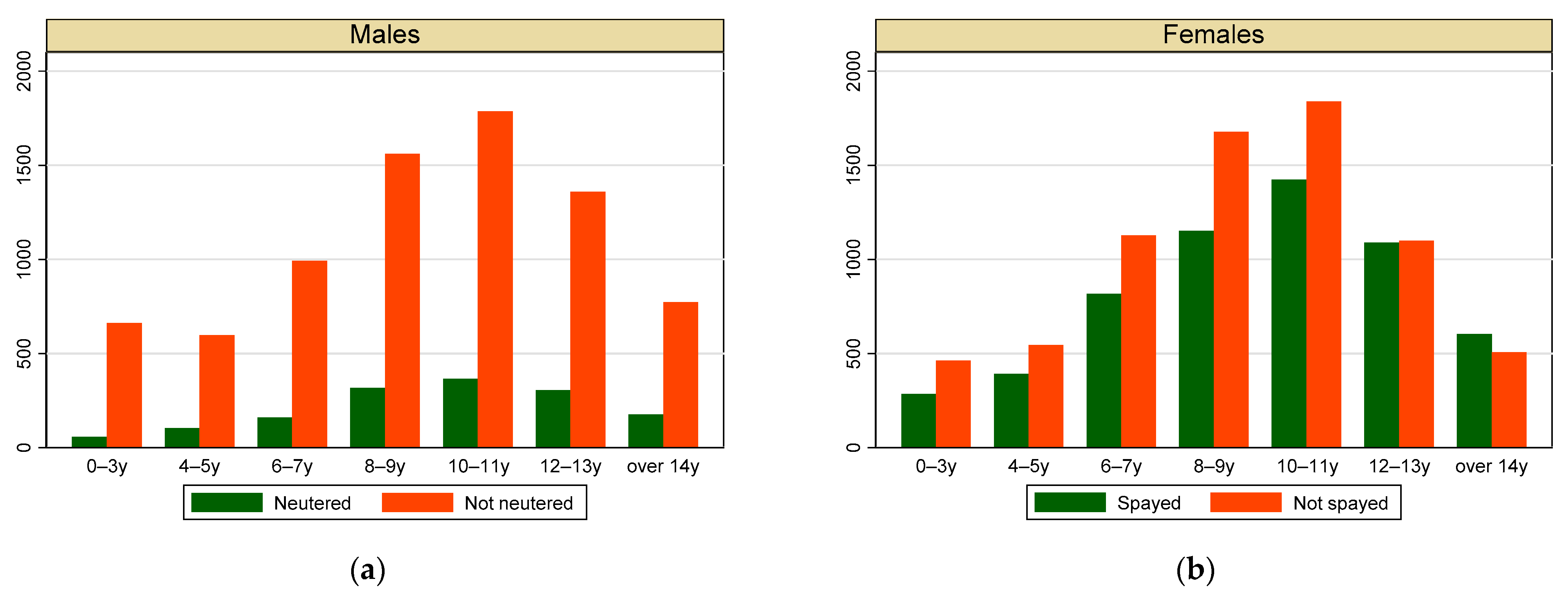
3.1. Individual Data
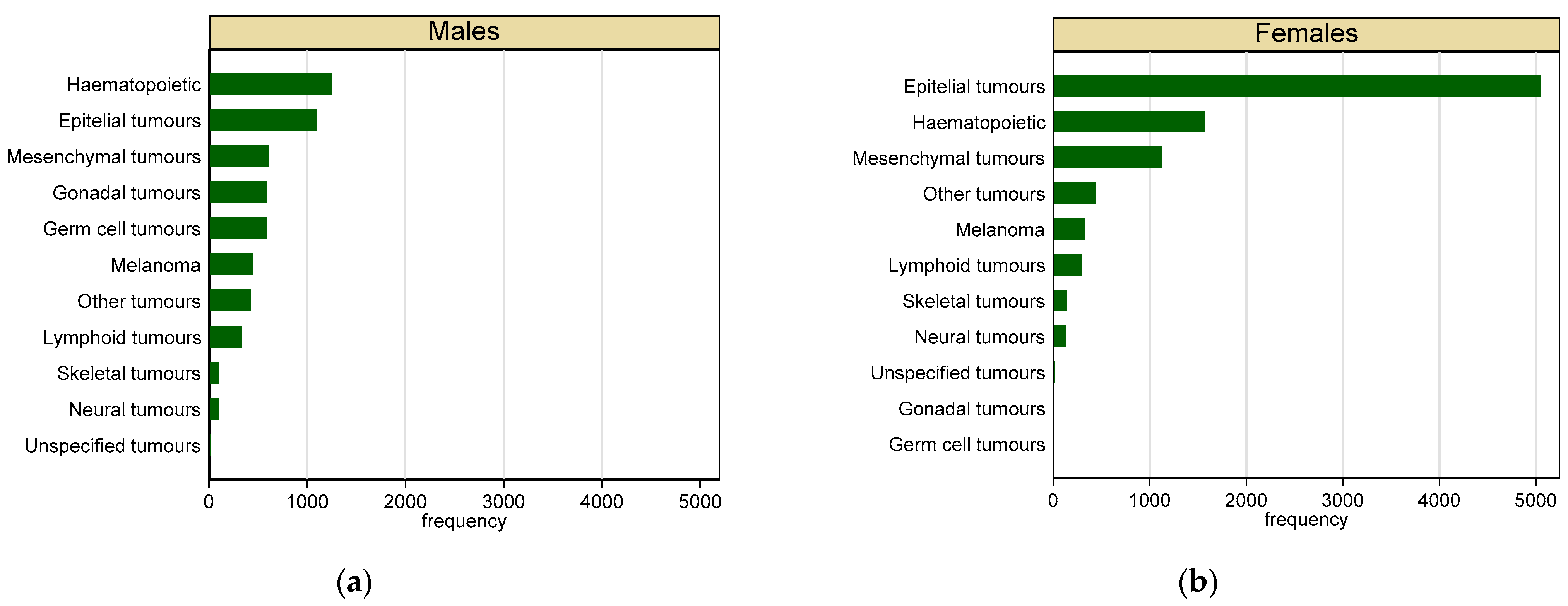
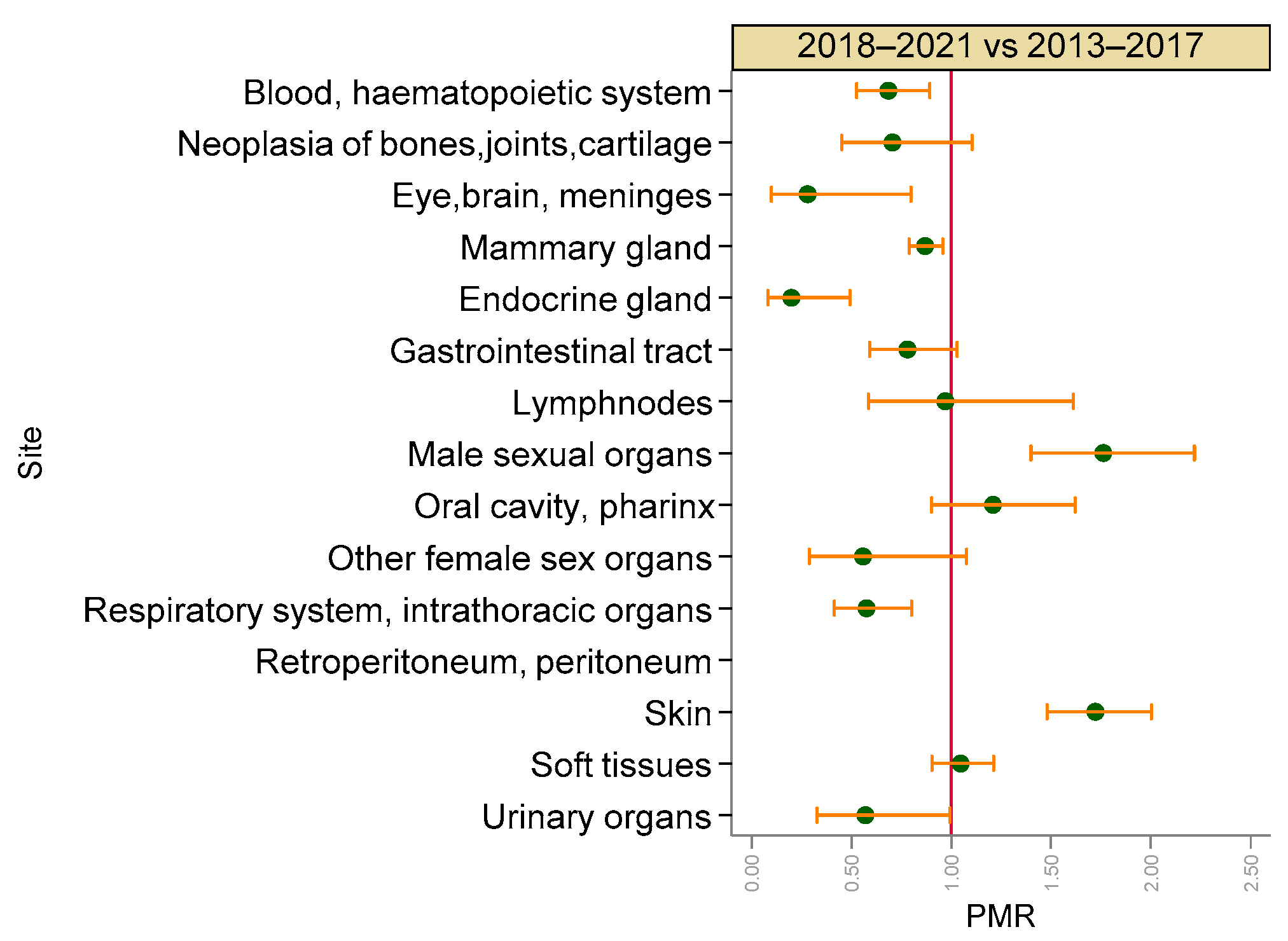
3.2. Malignant Tumours
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A. NILOV Contributors
Appendix B
| Location | ICD-9 | ICD-10 |
|---|---|---|
| Blood, haemopoietic system | T 169.0–169.9 | C 42 |
| Neoplasia of bones, joints, cartilage | T 170.0–170.9 | C 40–41 |
| Brain, meninges, other parts of CNS | T 190.0–192.9 | C 70–72 |
| Mammary gland | T 174.0–175.9 | C 50 |
| Endocrine gland | T 193.0–194.9 | C 73–75 |
| Gastrointestinal tract | T 150.0–159.9 (158 excluded) | C 16–26.8 |
| Lymph nodes | T 196.0–196.9 | C 77 |
| Male sexual organs | T 185.0–186.9; T 187.1–187.9 | C 60–63.2 |
| Oral cavity, pharynx | T 140.0–149.9 | C 2.9–11 |
| Other female sex organs | T 179.0 -184.9 | C 51–58 |
| Respiratory system, intrathoracic organs | T 160.0–165.9 | C 30–39 |
| Retroperitoneum, peritoneum | T 158 | C 48 |
| Skin | T 173.0–173.9 | C 44 |
| Soft tissues | T 171.0–171.9 | C 49; C47 |
| Urinary organs | T 188.0–189.9 | C 67–68 |
Appendix C
Results Regarding Benign Tumours


References
- Global Burden of Disease Tumour Collaboration. Global, Regional, and National Tumour Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-Years for 29 Tumour Groups, 1990 to 2017: A Systematic Analysis for the Global Burden of Disease. JAMA Oncol. 2019, 5, 1749–1768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knapp, D.W.; Ramos-Vara, J.A.; Moore, G.E.; Dhawan, D.; Bonney, P.L.; Young, K.E. Urinary bladder tumour in dogs, a naturally occurring model for tumour biology and drug development. ILAR J. 2014, 55, 100–118. [Google Scholar] [CrossRef]
- Fenger, J.M.; London, C.A.; Kisseberth, W.C. Canine osteosarcoma: A naturally occurring disease to inform pediatric oncology. ILAR J. 2014, 55, 69–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bushell, K.R.; Kim, Y.; Chan, F.C.; Ben-Neriah, S.; Jenks, A.; Alcaide, M.; Fornika, D.; Grande, B.M.; Arthur, S.; Gascoyne, R.D.; et al. Genetic inactivation of TRAF3 in canine and human B-cell lymphoma. Blood 2015, 125, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Ito, D.; Frantz, A.M.; Modiano, J.F. Canine lymphoma as a comparative model for human non-Hodgkin lymphoma: Recent progress and applications. Vet. Immunol. Immunopathol. 2014, 159, 192–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knapp, D.W.; Dhawan, D.; Ostrander, E. “Lassie,” “Toto,” and fellow pet dogs: Poised to lead the way for advances in tumour prevention. Am. Soc. Clin. Oncol. Educ. Book 2015, 35, e667–e672. [Google Scholar] [CrossRef]
- Parker, H.G.; Shearin, A.L.; Ostrander, E.A. Man’s best friend becomes biology’s best in show: Genome analyses in the domestic dog. Annu. Rev. Genet. 2010, 44, 309–336. [Google Scholar] [CrossRef] [Green Version]
- Ranieri, G.; Gadaleta, C.D.; Patruno, R.; Zizzo, N.; Daidone, M.G.; Hansson, M.G.; Paradiso, A.; Ribatti, D. A model of study for human tumour: Spontaneous occurring tumours in dogs. Biological features and translation for new antitumour therapies. Crit. Rev. Oncol. Hematol. 2013, 88, 187–197. [Google Scholar] [CrossRef]
- Glickman, L.T.; Domanski, L.M.; Maguire, T.G.; Dubielzig, R.R.; Churg, A. Mesothelioma in pet dogs associated with exposure of their owners to asbestos. Environ. Res. 1983, 32, 305–313. [Google Scholar] [CrossRef]
- Hayes, H.M.; Tarone, R.E.; Casey, H.W. A cohort study of the effects of Vietnam service on testicular pathology of U.S. military working dogs. Mil. Med. 1995, 160, 248–255. [Google Scholar] [CrossRef]
- Glickman, L.T.; Raghavan, M.; Knapp, D.W.; Bonney, P.L.; Dawson, M.H. Herbicide exposure and the risk of transitional cell carcinoma of the urinary bladder in Scottish Terriers. J. Am. Vet. Med. Assoc. 2004, 224, 1290–1297. [Google Scholar] [CrossRef] [Green Version]
- Glickman, L.T.; Schofer, F.S.; McKee, L.J.; Reif, J.S.; Goldschmidt, M.H. Epidemiologic study of insecticide exposures, obesity, and risk of bladder tumour in household dogs. J. Toxicol. Environ. Health 1989, 28, 407–414. [Google Scholar] [CrossRef]
- Dorn, C.R.; Taylor, D.O.; Frye, F.L.; Hibbard, H.H. Survey of animal neoplasms in Alameda and Contra Costa Counties, California. I. Methodology and description of cases. J. Natl. Tumour Inst. 1968, 40, 295–305. [Google Scholar]
- Dorn, C.R.; Taylor, D.O.; Schneider, R.; Hibbard, H.H.; Klauber, M.R. Survey of animal neoplasms in Alameda and Contra Costa Counties, California. II. Tumour morbidity in dogs and cats from Alameda County. J. Natl. Tumour Inst. 1968, 40, 307–318. [Google Scholar]
- Nødtvedt, A.; Berke, O.; Bonnett, B.N.; Brønden, L. Current status of canine tumour registration-report from an international workshop. Vet. Comp. Oncol. 2012, 10, 95–101. [Google Scholar] [CrossRef]
- Merlo, D.F.; Rossi, L.; Pellegrino, C.; Ceppi, M.; Cardellino, U.; Capurro, C.; Ratto, A.; Sambucco, P.L.; Sestito, V.; Tanara, G.; et al. Tumour incidence in pet dogs: Findings of the Animal Tumour Registry of Genoa, Italy. J. Vet. Intern. Med. 2008, 22, 976–984. [Google Scholar] [CrossRef]
- Vascellari, M.; Baioni, E.; Ru, G.; Carminato, A.; Mutinelli, F. Animal tumour registry of two provinces in northern Italy: Incidence of spontaneous tumours in dogs and cats. BMC Vet. Res. 2009, 5, 39. [Google Scholar] [CrossRef] [Green Version]
- Baioni, E.; Scanziani, E.; Vincenti, M.C.; Leschiera, M.; Bozzetta, E.; Pezzolato, M.; Desiato, R.; Bertolini, S.; Maurella, C.; Ru, G. Estimating canine tumour incidence: Findings from a population-based tumour registry in northwestern Italy. BMC Vet. Res. 2017, 13, 203. [Google Scholar] [CrossRef]
- Manuali, E.; Morgante, R.A.; Maresca, C.; Leonardi, L.; Purificato, I.; Giaimo, M.D.; Giovannini, G. A web-based tumour registration system for a regional Canine Tumour Registry in Umbria, central Italy. Ann. Ist. Super. Sanita 2019, 55, 357–362. [Google Scholar] [CrossRef]
- Dittmer, K.E.; Roccabianca, P.; Bell, C.; Murphy, B.G.; Foster, R.A.; Scruggs, J.; Schulman, F.Y.; Thompson, J.J.; Avallone, G.; Kiupel, M. Tumours of Bone, Cartilage, And Other Hard Tissues. In Surgical Pathology of Tumours of Domestic Animals; Kiupel, M., Ed.; Davis-Thompson DVM Foundation: Formoor Ln Gurnee, IL, USA, 2021; Volume 4. [Google Scholar]
- Roccabianca, P.; Schulman, F.Y.; Avallone, G.; Foster, R.A.; Scruggs, J.L.; Dittmer, K.E.; Kiupel, M. Tumours of Soft Tissue. In Surgical Pathology of Tumours of Domestic Animals; Kiupel, M., Ed.; Davis-Thompson DVM Foundation: Formoor Ln Gurnee, IL, USA, 2020; Volume 3, p. 307. [Google Scholar]
- Goldschmidt, M.H.; Goldschmidt, K. Epithelial and Melanocytic Tumours of the Skin. In Tumours of Domestic Animals, 5th ed.; Meuten, D.J., Ed.; Wiley: Hoboken, NJ, USA, 2017. [Google Scholar]
- Goldschmidt, M.H.; Kiupel, M.; Klopfleisch, R.; Munday, J.S.; Scruggs, J.L. Epithelial Tumours of the Skin; Kiupel, M., Ed.; Davis-Thompson DVM Foundation: Formoor Ln Gurnee, IL, USA, 2018; Volume 1. [Google Scholar]
- Kennedy, P.C.; Cullen, J.M.; Edwards, J.F.; Goldschmidt, M.H.; Larsen, S.; Munson, L.; Nielsen, S. Histological Classification of Tumours of the Genital System of Domestic Animals; Armed Forces Institute of Pathology and American Registry of Pathology: Washington, DC, USA, 2007; Volume IV. [Google Scholar]
- Dungworth, D.L.; Hauser, B.; Hahn, F.F.; Wilson, D.W.; Haenichen, T.; Harkemaa, J.R. Histological Classification of Tumours of the Respiratory System of Domestic Animals; Armed Forces Institute of Pathology and American Registry of Pathology: Washington, DC, USA, 2007; Volume VI. [Google Scholar]
- Wilcock, B.; Richard, R.D.; Render, J.A. Histological Classification of Ocular and Otic Tumours of Domestic Animals; Armed Forces Institute of Pathology and American Registry of Pathology: Washington, DC, USA, 2007; Volume VIII. [Google Scholar]
- Head, K.W.; Cullen, J.M.; Dubielzig, R.R. Histological Classification of Tumours of the Alimentary System of Domestic Animals; Armed Forces Institute of Pathology and American Registry of Pathology: Washington, DC, USA, 2007; Volume IX. [Google Scholar]
- Meuten, D.J.; Everitt, J.; Inskeep, W. Histological Classification of Tumours of the Urinary System of Domestic Animals; Armed Forces Institute of Pathology: Washington, DC, USA, 2007; Volume XI, p. 70. [Google Scholar]
- Misdorp, W.; Else, R.W.; Hellmén, E.; Lipscomb, T.P. Histological Classification of the Mammary Tumours of the Dog and Cat; Armed Force Institute of Pathology: Washington, DC, USA, 1999; Volume XI. [Google Scholar]
- Kiupel, M.; Capen, C.; Miller, M.; Smedley, R. Histological Classification of Tumours of the Endocrine System of Domestic Animals; Armed Forces Institute of Pathology and American Registry of Pathology: Washington, DC, USA, 2008; Volume XII. [Google Scholar]
- Hendrick, M.J.; Mahaffrey, E.A.; Moore, F.M. Histological Classification of Mesenchymal Tumours of Skin and Soft Tissues of Domestic Animals; Armed Force Institute of Pathology: Washington, DC, USA, 1998; Volume II. [Google Scholar]
- Koestner, A.; Bilzer, T.; Fatzer, R.; Schulman, F.Y.; Summers, B.A.; Van Winkle, T.J. Histological Classification of Tumours of the Nervous System of Domestic Animals; Armed Force Institute of Pathology: Washington, DC, USA, 1999; Volume V. [Google Scholar]
- Zappulli, V.; Pena, L.; Rasotto, R.; Goldschmidt, M.; Gama, A.; Scruggs, J.; Kiupel, M. Mammary Tumours; Davis Thompson Foundation: Formoor Ln Gurnee, IL, USA, 2020; Volume 2. [Google Scholar]
- WHO. International Classification of Diseases for Oncology2 (ICD-O-2), 2nd ed.; Percy, C., Van Holten, V., Muir, C.G., Eds.; World Health Organization Press: Geneva, Switzerland, 1990. [Google Scholar]
- Grüntzig, K.; Graf, R.; Boo, G.; Guscetti, F.; Hässig, M.; Axhausen, K.W.; Fabrikant, S.; Welle, M.; Meier, D.; Folkers, G.; et al. Swiss Canine Cancer Registry 1955–2008: Occurrence of the Most Common Tumour Diagnoses and Influence of Age, Breed, Body Size, Sex and Neutering Status on Tumour Development. J. Comp. Pathol. 2016, 155, 156–170. [Google Scholar] [CrossRef] [Green Version]
- Dos Santos Silva, I. Cancer Epidemiology: Principles and Methods; International Agency for Research on Cancer: Lyon, France, 1999; pp. 77–80. [Google Scholar]
- Nødtvedt, A.; Gamlem, H.; Gunnes, G.; Grotmol, T.; Indrebø, A.; Moe, L. Breed differences in the proportional morbidity of testicular tumours and distribution of histopathologic types in a population-based canine cancer registry. Vet. Comp. Oncol. 2011, 9, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.H.; Dawson, S.; Gaskell, R.M.; Coyne, K.P.; Tierney, A.; Setzkorn, C.; Radford, A.D.; Noble, P.-J.M. Surveillance of diarrhoea in small animal practice through the Small Animal Veterinary Surveillance Network (SAVSNET). Vet. J. 2014, 201, 412–418. [Google Scholar] [CrossRef]
- Pinello, K.; Baldassarre, V.; Steiger, K.; Paciello, O.; Pires, I.; Laufer-Amorim, R.; Oevermann, A.; Niza-Ribeiro, J.; Aresu, L.; Rous, B.; et al. Vet-ICD-O-Canine-1, a System for Coding Canine Neoplasms Based on the Human ICD-O-3.2. Cancers 2022, 14, 1529. [Google Scholar] [CrossRef] [PubMed]
- Pinello, K.; Pires, I.; Castro, A.F.; Carvalho, P.T.; Santos, A.; de Matos, A.; Queiroga, F.; Canadas-Sousa, A.; Dias-Pereira, P.; Catarino, J.; et al. Cross Species Analysis and Comparison of Tumors in Dogs and Cats, by Age, Sex, Topography and Main Morphologies. Data from Vet-OncoNet. Vet Sci. 2022, 9, 167. [Google Scholar] [CrossRef] [PubMed]
- Brønden, L.B.; Nielsen, S.S.; Toft, N.; Kristensen, A.T. Data from the Danish veterinary cancer registry on the occurrence and distribution of neoplasms in dogs in Denmark. Vet Rec. 2010, 166, 586–590. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.N. The role of neutering in cancer development. Vet. Clin. N. Am. Small Anim. Pract. 2014, 44, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Torres de la Riva, G.; Hart, B.L.; Farver, T.B.; Oberbauer, A.M.; Messam, L.L.; Willits, N.; Hart, L.A. Neutering dogs: Effects on joint disorders and cancers in golden retrievers. PLoS ONE 2013, 8, e55937. [Google Scholar] [CrossRef] [PubMed]
- Dobson, J.M.; Samuel, S.; Milstein, H.; Rogers, K.; Wood, J.L. Canine neoplasia in the UK: Estimates of incidence rates from a population of insured dogs. J. Small Anim. Pract. 2002, 43, 240–246. [Google Scholar] [CrossRef]
- Vascellari, M.; Capello, K.; Carminato, A.; Zanardello, C.; Baioni, E.; Mutinelli, F. Incidence of mammary tumors in the canine population living in the Veneto region (Northeastern Italy): Risk factors and similarities to human breast cancer. Prev. Vet. Med. 2016, 126, 183–189. [Google Scholar] [CrossRef]





| Sex and Neutering Status | Mean Age at Diagnosis (Years) Benign | Mean Age at Diagnosis (Years) Malignant |
|---|---|---|
| Male, neutered | 9.6 | 10.1 |
| Male, not neutered | 8.8 | 9.6 |
| Female, spayed | 9.2 | 9.9 |
| Female, not spayed | 8.6 | 9.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crescio, M.I.; Ru, G.; Aresu, L.; Bozzetta, E.; Cancedda, M.G.; Capello, K.; Castagnaro, M.; Carnio, A.; Cocumelli, C.; Degli Uberti, B.; et al. The Italian Network of Laboratories for Veterinary Oncology (NILOV) 2.0: Improving Knowledge on Canine Tumours. Vet. Sci. 2022, 9, 394. https://doi.org/10.3390/vetsci9080394
Crescio MI, Ru G, Aresu L, Bozzetta E, Cancedda MG, Capello K, Castagnaro M, Carnio A, Cocumelli C, Degli Uberti B, et al. The Italian Network of Laboratories for Veterinary Oncology (NILOV) 2.0: Improving Knowledge on Canine Tumours. Veterinary Sciences. 2022; 9(8):394. https://doi.org/10.3390/vetsci9080394
Chicago/Turabian StyleCrescio, Maria Ines, Giuseppe Ru, Luca Aresu, Elena Bozzetta, Maria Giovanna Cancedda, Katia Capello, Massimo Castagnaro, Azzurra Carnio, Cristiano Cocumelli, Barbara Degli Uberti, and et al. 2022. "The Italian Network of Laboratories for Veterinary Oncology (NILOV) 2.0: Improving Knowledge on Canine Tumours" Veterinary Sciences 9, no. 8: 394. https://doi.org/10.3390/vetsci9080394
APA StyleCrescio, M. I., Ru, G., Aresu, L., Bozzetta, E., Cancedda, M. G., Capello, K., Castagnaro, M., Carnio, A., Cocumelli, C., Degli Uberti, B., Eleni, C., Foiani, G., Fonti, N., Gibelli, L. R., Maniscalco, L., Manuali, E., Moccia, V., Paciello, O., Petrella, A., ... on behalf of NILOV. (2022). The Italian Network of Laboratories for Veterinary Oncology (NILOV) 2.0: Improving Knowledge on Canine Tumours. Veterinary Sciences, 9(8), 394. https://doi.org/10.3390/vetsci9080394










