Spotted Fever Group Rickettsia spp. Diversity in Ticks and the First Report of Rickettsia hoogstraalii in Romania
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Tick Collection and Identification
2.2. DNA Extraction
2.3. Polymerase Chain Reaction (PCR)
2.4. DNA Sequencing
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ginsberg, H.S.; Faulde, M.K. Ticks; World Health Organization, Regional Office for Europe: Copenhagen, Denmark, 2008; pp. 303–345. ISBN 9789289071888.
- Dantas-Torres, F.; Chomel, B.B.; Otranto, D. Ticks and tick-borne diseases: A One Health perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Tagliapietra, V.; Rosà, R.; Arnoldi, D.; Cagnacci, F.; Capelli, G.; Montarsi, F.; Hauffe, H.C.; Rizzoli, A. Saturation deficit and deer density affect questing activity and local abundance of Ixodes ricinus (Acari, Ixodidae) in Italy. Vet. Parasitol. 2011, 183, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Hofmeester, T.R.; Sprong, H.; Jansen, P.A.; Prins, H.H.; Van Wieren, S.E. Deer presence rather than abundance determines the population density of the sheep tick, Ixodes ricinus, in Dutch forests. Parasites Vectors 2017, 10, 433. [Google Scholar] [CrossRef] [PubMed]
- Ogden, N.H.; Lindsay, L.R. Effects of climate and climate change on vectors and vector-borne diseases: Ticks are different. Trends Parasitol. 2016, 32, 646–656. [Google Scholar] [CrossRef]
- Titcomb, G.; Allan, B.F.; Ainsworth, T.; Henson, L.; Hedlund, T.; Pringle, R.M.; Palmer, T.M.; Njoroge, L.; Campana, M.G.; Fleischer, R.C.; et al. Interacting effects of wildlife loss and climate on ticks and tick-borne disease. Proc. R. Soc. B Biol. Sci. 2017, 284, 20170475. [Google Scholar] [CrossRef]
- Allen, T.; Murray, K.A.; Zambrana-Torrelio, C.; Morse, S.S.; Rondinini, C.; Di Marco, M.; Breit, N.; Olival, K.J.; Daszak, P. Global Hotspots and Correlates of Emerging Zoonotic Diseases. Nat. Commun. 2017, 8, 1124. [Google Scholar] [CrossRef]
- Rosà, R.; Andreo, V.; Tagliapietra, V.; Baráková, I.; Arnoldi, D.; Hauffe, H.C.; Manica, M.; Rosso, F.; Blaňarová, L.; Bona, M.; et al. Effect of climate and land use on the spatio-temporal variability of tick-borne bacteria in Europe. Int. J. Environ. Res. Public Health 2018, 15, 732. [Google Scholar] [CrossRef]
- Boulanger, N.; Boyer, P.; Talagrand-Reboul, E.; Hansmann, Y. Ticks and tick-borne diseases. Med. Mal. Infect. 2019, 49, 87–97. [Google Scholar] [CrossRef]
- Moonga, L.C.; Hayashida, K.; Mulunda, N.R.; Nakamura, Y.; Chipeta, J.; Moonga, H.B.; Namangala, B.; Sugimoto, C.; Mtonga, Z.; Mutengo, M.; et al. Molecular Detection and Characterization of Rickettsia asembonensis in Human Blood, Zambia. Emerg. Infect. Dis. 2021, 27, 2237. [Google Scholar] [CrossRef]
- Souza, U.A.; Fagundes-Moreira, R.; Costa, F.B.; Alievi, M.M.; Labruna, M.B.; Soare, J.F. Rickettsia amblyommatis-infected Amblyomma coelebs parasitizing a human traveler in Rio Grande do Sul, southern Brazil, after returning from the Amazon. Travel Med. Infect. Dis. 2022, 10, 102328. [Google Scholar] [CrossRef]
- Stothard, D.R.; Clark, J.B.; Fuerst, P.A. Ancestral Divergence of Rickettsia bellii from the Spotted Fever and Typhus Groups of Rickettsia and Antiquity of the Genus Rickettsia. Int. J. Syst. Bacteriol. 1994, 44, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Ciuofo, S.; Domrachev, M.; Hotton, C.; Kannan, S.; Khovanskaya, R.; Leipe, D.; Mcveigh, R.; O’Neill, K.; Robbertse, B.; et al. NCBI Taxonomy: A Comprehensive Update on Curation, Resources and Tools. Database (Oxford). 2020. Available online: https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Tree&id=780&lvl=3&keep=1&srchmode=1&unlock (accessed on 10 April 2022).
- Parola, P.; Paddock, C.D.; Socolovschi, C.; Labruna, M.B.; Mediannikov, O.; Kernif, T.; Abdad, M.Y.; Stenos, J.; Bitam, I.; Fournier, P.E.; et al. Update on tick-borne rickettsioses around the world: A geographic approach. Clin. Microbiol. Rev. 2013, 26, 657–702. [Google Scholar] [CrossRef] [PubMed]
- Portillo, A.; Santibáñez, S.; García-Álvarez, L.; Palomar, A.M.; Oteo, J.A. Rickettsioses in Europe. Microbes Infect. 2015, 17, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Otranto, D.; Dantas-Torres, F.; Santos-Silvia, M.M. Ixodes ricinus (Linnaeus, 1758). In Ticks of Europe and North Africa, a Guide to Species Identification; Estrada-Peña, A., Mihalca, A.D., Petney, T., Eds.; Springer: Cham, Switzeland, 2017; pp. 189–197. [Google Scholar]
- Borawski, K.; Dunaj, J.; Pancewicz, S.; Król, M.; Czupryna, P.; Moniuszko-Malinowska, A. Tick-borne rickettsioses in Europe–A review. Prz. Epidemiol. 2019, 73, 523–530. [Google Scholar]
- Klitgaard, K.; Kjær, L.J.; Isbrand, A.; Hansen, M.F.; Bødker, R. Multiple infections in questing nymphs and adult female Ixodes ricinus ticks collected in a recreational forest in Denmark. Ticks Tick Borne Dis. 2019, 10, 1060–1065. [Google Scholar] [CrossRef]
- Wallménius, K.; Pettersson, J.H.O.; Jaenson, T.G.; Nilsson, K. Prevalence of Rickettsia spp., Anaplasma phagocytophilum, and Coxiella burnetii in adult Ixodes ricinus ticks from 29 study areas in central and southern Sweden. Ticks Tick Borne Dis. 2012, 3, 100–106. [Google Scholar] [CrossRef]
- Hajduskova, E.; Literak, I.; Papousek, I.; Costa, F.B.; Novakova, M.; Labruna, M.B.; Zdrazilova-Dubska, L. ‘Candidatus Rickettsia mendelii’, a novel basal group rickettsia detected in Ixodes ricinus ticks in the Czech Republic. Ticks Tick Borne Dis. 2016, 7, 482–486. [Google Scholar] [CrossRef]
- Stańczak, J.; Biernat, B.; Racewicz, M.; Zalewska, M.; Matyjasek, A. Prevalence of different Rickettsia spp. in Ixodes ricinus and Dermacentor reticulatus ticks (Acari: Ixodidae) in north-eastern Poland. Ticks Tick Borne Dis. 2018, 9, 427–434. [Google Scholar] [CrossRef]
- Hornok, S.; Csörgő, T.; Fuente, J.; Gyuranecz, M.; Privigyei, C.; Meli, M.L.; Kreizinger, Z.; Gönczi, E.; Fernandez de Mera, I.G.; Hofmann-Lehmann, R. Synanthropic birds associated with high prevalence of tick-borne rickettsiae and with the first detection of Rickettsia aeschlimannii in Hungary. Vector-Borne Zoonotic Dis. 2013, 13, 77–83. [Google Scholar] [CrossRef]
- Vikentjeva, M.; Geller, J.; Remm, J.; Golovljova, I. Rickettsia spp. in rodent-attached ticks in Estonia and first evidence of spotted fever group Rickettsia species Candidatus Rickettsia uralica in Europe. Parasites Vectors 2021, 14, 65. [Google Scholar] [CrossRef]
- National Environment Protection Agency. Annual Report 2020. Available online: http://www.anpm.ro/habitate-si-specii (accessed on 10 April 2022).
- Mihalca, A.D.; Dumitrache, M.O.; Magdaş, C.; Gherman, C.M.; Domşa, C.; Mircean, V.; Ghira, I.V.; Pocora, V.; Ionescu, D.T.; Barabási, S.S.; et al. Synopsis of the hard ticks (Acari: Ixodidae) of Romania with update on host associations and geographical distribution. Exp. Appl. Acarol. 2012, 58, 183–206. [Google Scholar] [CrossRef] [PubMed]
- Mihalca, A.D.; Gherman, C.M.; Magdaş, C.; Dumitrache, M.O.; Györke, A.; Sándor, A.D.; Domşa, C.; Oltean, M.; Mircean, V.; Mărcuţan, D.I.; et al. Ixodes ricinus is the dominant questing tick in forest habitats in Romania: The results from a countrywide dragging campaign. Exp. Appl. Acarol. 2012, 58, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Matei, I.; Mihalca, A.; Nadăş, G.; Fiţ, N. Testing two PCR protocols targeting different genes to detect SFG Rickettsia DNA in ticks samples. Vet. Med. 2018, 49, 146–149. [Google Scholar]
- Borşan, S.D.; Ionică, A.M.; Galon, C.; Toma-Naic, A.; Peştean, C.; Sándor, A.D.; Moutailler, S.; Mihalca, A.D. High Diversity, Prevalence, and Co-infection Rates of Tick-Borne Pathogens in Ticks and Wildlife Hosts in an Urban Area in Romania. Front. Microbiol. 2021, 12, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Raileanu, C.; Moutailler, S.; Porea, D.; Oslobanu, L.; Anita, D.; Anita, A.; Vayssier-Taussat, M.; Savuta, G. Molecular evidence of Rickettsia spp., Anaplasma phagocytophilum, and “Candidatus Neoehrlichia mikurensis” in ticks from natural and urban habitats in Eastern Romania. Vector-Borne Zoonotic Dis. 2018, 18, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Ioniţă, M.; Mitrea, I.L.; Pfister, K.; Hamel, D.; Silaghi, C. Molecular evidence for bacterial and protozoan pathogens in hard ticks from Romania. Vet. Parasitol. 2013, 196, 71–76. [Google Scholar] [CrossRef]
- Mărcuţan, I.D.; Kalmár, Z.; Ionică, A.M.; D’Amico, G.; Mihalca, A.D.; Vasile, C.; Sándor, A.D. Spotted fever group rickettsiae in ticks of migratory birds in Romania. Parasites Vectors 2016, 9, 294–301. [Google Scholar] [CrossRef]
- Matei, I.A.; Kalmár, Z.; Lupşe, M.; D’Amico, G.; Ionică, A.M.; Dumitrache, M.O.; Gherman, C.M.; Mihalca, A.D. The risk of exposure to rickettsial infections and human granulocytic anaplasmosis associated with Ixodes ricinus tick bites in humans in Romania: A multiannual study. Ticks Tick Borne Dis. 2017, 8, 375–378. [Google Scholar] [CrossRef]
- Andersson, M.O.; Marga, G.; Banu, T.; Dobler, G.; Chitimia-Dobler, L. Tick-borne pathogens in tick species infesting humans in Sibiu County, central Romania. Parasitol. Res. 2018, 117, 1591–1597. [Google Scholar] [CrossRef]
- Ioniţă, M.; Silaghi, C.; Mitrea, I.L.; Edouard, S.; Parola, P.; Pfister, K. Molecular detection of Rickettsia conorii and other zoonotic spotted fever group rickettsiae in ticks, Romania. Ticks Tick Borne Dis. 2016, 7, 150–153. [Google Scholar] [CrossRef]
- Andersson, M.O.; Tolf, C.; Tamba, P.; Stefanache, M.; Radbea, G.; Frangoulidis, D.; Tomaso, H.; Waldenström, J.; Dobler, G.; Chitimia-Dobler, L. Molecular survey of neglected bacterial pathogens reveals an abundant diversity of species and genotypes in ticks collected from animal hosts across Romania. Parasites Vectors 2018, 11, 144. [Google Scholar] [CrossRef] [PubMed]
- Matei, I.A.; Corduneanu, A.; Sándor, A.D.; Ionică, A.M.; Panait, L.; Kalmár, Z.; Ivan, T.; Papuc, I.; Bouari, C.; Fit, N.; et al. Rickettsia spp. in bats of Romania: High prevalence of Rickettsia monacensis in two insectivorous bat species. Parasites Vectors 2021, 14, 1–8. [Google Scholar] [CrossRef]
- Zaharia, M.; Popescu, C.P.; Florescu, S.A.; Ceausu, E.; Raoult, D.; Parola, P.; Socolovschi, C. Rickettsia massiliae infection and SENLAT syndrome in Romania. Ticks Tick Borne Dis. 2016, 7, 759–762. [Google Scholar] [CrossRef] [PubMed]
- Pitigoi, D.; Olaru, I.D.; Badescu, D.; Rafila, A.; Arama, V.; Hristea, A. Mediterranean spotted fever in southeastern Romania. Biomed. Res. Int. 2013, 2013, 395806. [Google Scholar] [CrossRef]
- Serban, R.; Pistol, A.; Neguţ, M.; Cucuiu, R. Rickettsia conorii infection in Romania, 2000–2008. Bacteriol. Virusol. Parazitol. Epidemiol. 2009, 54, 177–183. [Google Scholar]
- Estrada-Peña, A.; Mihalca, A.D.; Petney, T.N. Ticks of Europe and North Africa: A Guide to Species Identification; Springer: Cham, Switzeland, 2017; pp. 1–403. [Google Scholar]
- Mihalca, A.D.; Kalmár, Z.; Dumitrache, M.O. Rhipicephalus rossicus, a neglected tick at the margin of Europe: A review of its distribution, ecology and medical importance. Med. Vet. Entomol. 2015, 29, 215–224. [Google Scholar] [CrossRef]
- Sándor, A.D.; Dumitrache, M.O.; D’Amico, G.; Kiss, B.J.; Mihalca, A.D. Rhipicephalus rossicus and not R. sanguineus is the dominant tick species of dogs in the wetlands of the Danube Delta, Romania. Vet. Parasitol. 2014, 204, 430–432. [Google Scholar] [CrossRef]
- Regnery, R.L.; Spruill, C.L.; Plikaytis, B. Genotypic identification of rickettsiae and estimation of intraspecies sequence divergence for portions of two rickettsial genes. J. Bacteriol. 1991, 173, 1576–1589. [Google Scholar] [CrossRef]
- Leitner, M.; Yitzhaki, S.; Rzotkiewicz, S.; Keysary, A. Polymerase chain reaction-based diagnosis of Mediterranean spotted fever in serum and tissue samples. Am. J. Trop. Med. Hyg. 2002, 67, 166–169. [Google Scholar] [CrossRef]
- Parola, P.; Raoult, D. Ticks and tick borne bacterial diseases in humans: An emerging infectious threat. Clin. Infect. Dis. 2001, 32, 897–928. [Google Scholar] [CrossRef]
- De Vito, A.; Geremia, N.; Mameli, S.M.; Fiore, V.; Serra, P.A.; Rocchitta, G.; Nuvoli, S.; Spanu, A.; Lobrano, R.; Cossu, A.; et al. Epidemiology, clinical aspects, laboratory diagnosis and treatment of rickettsial diseases in the mediterranean area during COVID-19 pandemic: A review of the literature. Mediterr. J. Hematol. 2020, 12, e2020056. [Google Scholar] [CrossRef] [PubMed]
- Sprong, H.; Wielinga, P.R.; Fonville, M.; Reusken, C.; Brandenburg, A.H.; Borgsteede, F.; Gaasenbeek, C.; van der Giessen, J.W. Ixodes ricinus ticks are reservoir hosts for Rickettsia helvetica and potentially carry flea-borne Rickettsia species. Parasites Vectors 2009, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tomassone, L.; Ceballos, L.A.; Ragagli, C.; Martello, E.; De Sousa, R.; Stella, M.C.; Mannelli, A. Importance of common wall lizards in the transmission dynamics of tick-borne pathogens in the northern Apennine Mountains, Italy. Microb. Ecol. 2017, 74, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.F.; Magnarelli, L.A. Biology of ticks. Infect. Dis. Clin. N. Am. 2008, 22, 195–215. [Google Scholar] [CrossRef] [PubMed]
- Briciu, V.T.; Titilincu, A.; Tatulescu, D.F.; Carstina, D.; Lefkaditis, M.; Mihalca, A.D. First survey on hard ticks Ixodidae collected from humans in Romania:possible risks for tick-borne diseases. Exp. Appl. Acarol. 2011, 54, 199–204. [Google Scholar] [CrossRef]
- Levin, M.L.; Killmaster, L.F.; Zemtsova, G.E. Domestic dogs (Canis familiaris) as reservoir hosts for Rickettsia conorii. Vector-Borne Zoonotic Dis. 2012, 12, 28–33. [Google Scholar] [CrossRef]
- Kidd, L.; Maggi, R.; Diniz, P.P.V.P.; Hegarty, B.; Tucker, M.; Breitschwerdt, E. Evaluation of conventional and real-time PCR assays for detection and differentiation of spotted fever group Rickettsia in dog blood. Vet. Microbiol. 2008, 129, 294–303. [Google Scholar] [CrossRef]
- Tomassone, L.; Conte, V.; Parrilla, G.; De Meneghi, D. Rickettsia infection in dogs and Rickettsia parkeri in Amblyomma tigrinum ticks, Cochabamba Department, Bolivia. Vector-Borne Zoonotic Dis. 2010, 10, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Satoh, H.; Motoi, Y.; Camer, G.A.; Inokuma, H.; Izawa, M.; Kiyuuna, T.; Kumazawa, N.; Muramatsu, Y.; Ueno, H.; Morita, C. Characterization of spotted fever group rickettsiae detected in dogs and ticks in Okinawa, Japan. Microbiol. Immunol. 2002, 46, 257–263. [Google Scholar] [CrossRef][Green Version]
- Heyman, P.; Cochez, C.; Hofhuis, A.; Van Der Giessen, J.; Sprong, H.; Porter, S.R.; Losson, B.; Saegerman, C.; Donoso-Mantke, O.; Niedrig, M.; et al. A clear and present danger: Tick-borne diseases in Europe. Expert Rev. Anti-Infect. Ther. 2010, 8, 33–50. [Google Scholar] [CrossRef]
- Gargili, A.; Palomar, A.M.; Midilli, K.; Portillo, A.; Kar, S.; Oteo, J.A. Rickettsia species in ticks removed from humans in Istanbul, Turkey. Vector-Borne Zoonotic Dis. 2012, 12, 938–941. [Google Scholar] [CrossRef] [PubMed]
- Márquez, F.J. Spotted fever group Rickettsia in ticks from southeastern Spain natural parks. Exp. Appl. Acarol. 2008, 45, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Tijsse-Klasen, E.; Jacobs, J.J.; Swart, A.; Fonville, M.; Reimerink, J.H.; Brandenburg, A.H.; van der Giessen, J.W.; Hofhuis, A.; Sprong, H. Small risk of developing symptomatic tick-borne diseases following a tick bite in The Netherland. Parasites Vectors 2011, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Radulović, Ž.; Chochlakis, D.; Tomanović, S.; Milutinović, M.; Tselentis, Y.; Psaroulaki, A. First detection of spotted fever group Rickettsiae in ticks in Serbia. Vector-Borne Zoonotic Dis. 2011, 11, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Franke, J.; Fritzsch, J.; Tomaso, H.; Straube, E.; Dorn, W.; Hildebrandt, A. Coexistence of pathogens in host-seeking and feeding ticks within a single natural habitat in Central Germany. Appl. Environ. Microbiol. 2010, 76, 6829–6836. [Google Scholar] [CrossRef]
- Movila, A.; Reye, A.L.; Dubinina, H.V.; Tolstenkov, O.O.; Toderas, I.; Hübschen, J.M.; Muller, C.P.; Alekseev, A.N. Detection of Babesia sp. EU1 and members of spotted fever group rickettsiae in ticks collected from migratory birds at Curonian Spit, North-Western Russia. Vector-Borne Zoonotic Dis. 2011, 11, 89–91. [Google Scholar] [CrossRef]
- Floris, R.; Yurtman, A.N.; Margoni, E.F.; Mignozzi, K.; Boemo, B.; Altobelli, A.; Cinco, M. Detection and identification of Rickettsia species in the northeast of Italy. Vector-Borne Zoonotic Dis. 2008, 8, 777–782. [Google Scholar] [CrossRef]
- Elfving, K.; Olsen, B.; Bergström, S.; Waldenström, J.; Lundkvist, Å.; Sjöstedt, A.; Mejlon, H.; Nilsson, K. Dissemination of spotted fever rickettsia agents in Europe by migrating birds. PLoS ONE 2010, 5, 8572. [Google Scholar] [CrossRef]
- Sgroi, G.; Iatta, R.; Lia, R.P.; Napoli, E.; Buono, F.; Bezerra-Santos, M.A.; Veneziano, V.; Otranto, D. Tick exposure and risk of tick-borne pathogens infection in hunters and hunting dogs: A citizen science approach. Transbound. Emerg. Dis. 2021. [Google Scholar] [CrossRef]
- Oteo, J.A.; Portillo, A. Tick-borne rickettsioses in Europe. Ticks Tick Borne Dis. 2012, 3, 271–278. [Google Scholar] [CrossRef]
- Fournier, P.E.; Allombert, C.; Supputamongkol, Y.; Caruso, G.; Brouqui, P.; Raoult, D. Aneruptive fever associated with antibodies to Rickettsia helvetica in Europe and Thailand. J. Clin. Microbiol. 2004, 42, 816–818. [Google Scholar] [CrossRef] [PubMed]
- Boretti, F.S.; Perreten, A.; Meli, M.L.; Cattori, V.; Willi, B.; Wengi, N.; Hornok, S.; Honegger, H.; Hegglin, D.; Woelfel, R.; et al. Molecular Investigations of Rickettsia helvetica infection in dogs, foxes, humans, and Ixodes ticks. Appl. Environ. Microbiol. 2009, 75, 3230–3237. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, K.; Lindquist, O.; Påhlson, C. Association of Rickettsia helvetica with chronic perimyocarditis in sudden cardiac death. Lancet 1999, 354, 1169–1173. [Google Scholar] [CrossRef]
- Brouqui, P.; Parola, P.; Fournier, P.E.; Raoult, D. Spotted fever rickettsioses in southern and eastern Europe. FEMS Microbiol. Immunol. 2007, 49, 2–12. [Google Scholar] [CrossRef]
- Angelakis, E.; Pulcini, C.; Waton, J.; Imbert, P.; Socolovschi, C.; Edouard, S.; Dellamonica, P.; Raoult, D. Scalp eschar and neck lymphadenopathy caused by Bartonella henselae after tick bite. Clin. Infect. Dis. 2010, 50, 549–551. [Google Scholar] [CrossRef]
- Raoult, D.; Berbis, P.H.; Roux, V.; Xu, W.; Maurin, M. A new tick-transmitted disease due to Rickettsia slovaca. Lancet 1997, 350, 112–113. [Google Scholar] [CrossRef]
- Chmielewski, T.; Podsiadly, E.; Karbowiak, G.; Tylewska-Wierzbanowska, S. Rickettsia spp. in ticks, Poland. Emerg. Infect. Dis. 2009, 15, 486–488. [Google Scholar] [CrossRef]
- Kho, K.L.; Tan, P.E.; Tay, S.T. Diversity of rickettsiae in feeding and questing ticks collected from a Malaysian forest reserve area. J. Med. Entomol. 2019, 56, 547–552. [Google Scholar] [CrossRef]
- Boldiš, V.; Štrus, J.; Kocianová, E.; Tušek-Žnidarič, M.; Štefanidesová, K.; Schwarzová, K.; Kúdelová, M.; Sekeyová, Z.; Špitalská, E. Life cycle of Rickettsia slovaca in L929 cell line studied by quantitative real-time PCR and transmission electron microscopy. FEMS Microbiol. Lett. 2009, 293, 102–106. [Google Scholar] [CrossRef][Green Version]
- Revuelta, J.A.O.; Cucalón, V.I.; Ramos, J.R.B. Eritema, necrosis y linfedenopatía: Una nueva enfermedad transmitida por “Dermacentor marginatus” Sulzer, 1776. Zubía 2000, 12, 49–58. [Google Scholar]
- Lakos, A. Tick-borne lymphadenopathy—A new rickettsial disease? Lancet 1997, 350, 1006. [Google Scholar] [CrossRef]
- Selmi, M.; Bertolotti, L.; Tomassone, L.; Mannelli, A. Rickettsia slovaca in Dermacentor marginatus and tick-borne lymphadenopathy, Tuscany, Italy. Emerg. Infect. Dis. 2008, 14, 817–820. [Google Scholar] [CrossRef] [PubMed]
- Komitova, R.; Lakos, A.; Aleksandrov, A.; Christova, I.; Murdjeva, M. A case of tick-transmitted lymphadenopathy in Bulgaria associated with Rickettsia slovaca. Scand. J. Infect. Dis. 2003, 35, 213. [Google Scholar] [CrossRef]
- Switaj, K.; Chmielewski, T.; Borkowski, P.; Tylewska-Wierzbanowska, S.; Olszynska-Krowicka, M. Spotted fever rickettsiosis caused by Rickettsia raoultii–case report. Prz. Epidemiol. 2012, 66, 347–350. [Google Scholar]
- Duh, D.; Punda-Polic, V.; Avsic-Zupanc, T.; Bouyer, D.; Walker, D.H.; Popov, V.L.; Jelovsek, M.; Gracner, M.; Trilar, T.; Bradaric, N.; et al. Rickettsia hoogstraalii sp. nov., isolated from hard-and soft-bodied ticks. Int. J. Syst. Evol. Microbiol. 2010, 60, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Mattila, J.T.; Burkhardt, N.Y.; Hutcheson, H.J.; Munderloh, U.G.; Kurtti, T.J. Isolation of cell lines and a rickettsial endosymbiont from the soft tick Carios capensis (Acari: Argasidae: Ornithodorinae). J. Med. Entomol. 2007, 44, 1091–1101. [Google Scholar] [CrossRef]
- Chochlakis, D.; Ioannou, I.; Sandalakis, V.; Dimitriou, T.; Kassinis, N.; Papadopoulos, B.; Tselentis, Y.; Psaroulaki, A. Spotted fever group Rickettsiae in ticks in Cyprus. Microb. Ecol. 2012, 63, 314–323. [Google Scholar] [CrossRef]
- Chisu, V.; Leulmi, H.; Masala, G.; Piredda, M.; Foxi, C.; Parola, P. Detection of Rickettsia hoogstraalii, Rickettsia helvetica, Rickettsia massiliae, Rickettsia slovaca and Rickettsia aeschlimannii in ticks from Sardinia, Italy. Ticks Tick Borne Dis. 2017, 8, 347–352. [Google Scholar] [CrossRef]
- Orkun, Ö.; Karaer, Z.; Çakmak, A.; Nalbantoğlu, S. Spotted fever group rickettsiae in ticks in Turkey. Ticks Tick Borne Dis. 2014, 5, 213–218. [Google Scholar] [CrossRef]
- Moraga-Fernández, A.; Chaligiannis, Ι.; Cabezas-Cruz, A.; Papa, A.; Sotiraki, S.; de la Fuente, J.; G Fernández de Mera, I. Molecular identification of spotted fever group Rickettsia in ticks collected from dogs and small ruminants in Greece. Exp. Appl. Acarol. 2019, 78, 421–430. [Google Scholar] [CrossRef]
- Sukhiashvili, R.; Zhgenti, E.; Khmaladze, E.; Burjanadze, I.; Imnadze, P.; Jiang, J.; John, H.S.; Farris, C.M.; Gallagher, T.; Obiso, R.J.; et al. Identification and distribution of nine tick-borne spotted fever group rickettsiae in the country of Georgia. Ticks Tick Borne Dis. 2020, 11, 101470. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, H.; Ando, S.; Kishimoto, T.; Kurane, I.; Takano, A.; Nogami, S.; Fujita, H.; Tsurumi, M.; Nakamura, N.; Sato, F.; et al. First detection of Rickettsia in soft-bodied ticks associated with seabirds, Japan. Microbiol. Immunol. 2006, 50, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Pader, V.; Buniak, J.N.; Abdissa, A.; Adamu, H.; Tolosa, T.; Gashaw, A.; Cutler, R.R.; Cutler, S.J. Candidatus Rickettsia hoogstraalii in Ethiopian Argas persicus ticks. Ticks Tick Borne Dis. 2012, 3, 338–345. [Google Scholar] [CrossRef]
- Dietrich, M.; Lebarbenchon, C.; Jaeger, A.; Le Rouzic, C.; Bastien, M.; Lagadec, E.; McCoy, K.D.; Pascalis, H.; Le Corre, M.; Dellagi, K.; et al. Rickettsia spp. in seabird ticks from western Indian Ocean islands, 2011–2012. Emerg. Infect. Dis. 2010, 20, 838. [Google Scholar] [CrossRef] [PubMed]
- Kooshki, H.; Goudarzi, G.; Faghihi, F.; Telmadarraiy, Z.; Edalat, H.; Hosseini-Chegeni, A. The first record of Rickettsia hoogstraalii (Rickettsiales: Rickettsiaceae) from Argas persicus (Acari: Argasidae) in Iran. Syst. Appl. Acarol. 2020, 25, 1611–1617. [Google Scholar] [CrossRef]
- Reeves, W.K.; Mans, B.J.; Durden, L.A.; Miller, M.M.; Gratton, E.M.; Laverty, T.M. Rickettsia hoogstraalii and a Rickettsiella from the Bat Tick Argas transgariepinus, in Namibia. J. Parasitol. Res. 2020, 106, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Simuunza, M.; Kajihara, M.; Chambaro, H.; Harima, H.; Eto, Y.; Simulundu, E.; Squarre, D.; Torii, S.; Takada, A.; et al. Screening of tick-borne pathogens in argasid ticks in Zambia: Expansion of the geographic distribution of Rickettsia lusitaniae and Rickettsia hoogstraalii and detection of putative novel Anaplasma species. Ticks Tick Borne Dis. 2021, 12, 101720. [Google Scholar] [CrossRef]
- Duan, D.Y.; Liu, Y.K.; Liu, L.; Liu, G.H.; Cheng, T.Y. Microbiome analysis of the midguts of different developmental stages of Argas persicus in China. Ticks Tick Borne Dis. 2022, 13, 101868. [Google Scholar] [CrossRef]
- Hornok, S.; Kontschán, J.; Takács, N.; Chaber, A.L.; Halajian, A.; Szekeres, S.; Sándor, A.D.; Plantard, O. Rickettsiaceae in two reptile-associated tick species, Amblyomma exornatum and Africaniella transversale: First evidence of Occidentia massiliensis in hard ticks (Acari: Ixodidae). Ticks Tick Borne Dis. 2022, 13, 101830. [Google Scholar] [CrossRef]
- Pascucci, I.; Antognini, E.; Canonico, C.; Montalbano, M.G.; Necci, A.; di Donato, A.; Moriconi, M.; Morandi, B.; Morganti, G.; Crotti, S.; et al. One Health Approach to Rickettsiosis: A Five-Year Study on Spotted Fever Group Rickettsiae in Ticks Collected from Humans, Animals and Environment. Microorganisms 2021, 10, 35. [Google Scholar] [CrossRef]
| Rickettsia spp. | Tick Species | Host Species | County | Reference |
|---|---|---|---|---|
| Questing ticks | ||||
| Rickettsia spp. 1 | D. marginatus, D. reticulatus | - | Cluj | [27,28] |
| R. helvetica 1 | I. ricinus, Hae. punctata | - | Cluj | [28] |
| I. ricinus | - | Iași, Tulcea | [29] | |
| R. monacensis 1 | I. ricinus | - | Iași, Tulcea | [29] |
| I. ricinus, Hae. punctata | - | Cluj | [28] | |
| R. raoultii 1 | D. reticulatus | - | Iași | [29] |
| R. conorii 1 | Hae. punctata | - | Cluj | [28] |
| Ticks collected from hosts | ||||
| R. helvetica 1 | I. ricinus | Bos taurus, Equus caballus | Ilfov, Prahova | [30] |
| I. ricinus, I. arboricola, I. redikorzevi | Erithacus rubecula, Panurus biarmicus, Turdus merula, T. philomelos | Constanța | [31] | |
| I. ricinus | Homo sapiens | Cluj | [32] | |
| I. ricinus | H. sapiens | Sibiu | [33] | |
| I. ricinus, I. hexagonus | Erinaceus roumanicus | Cluj | [28] | |
| I. ricinus | E. rubecula, T. merula | Cluj | [28] | |
| I. ricinus | Talpa europea | Cluj | [28] | |
| R. monacensis 1 | I. ricinus | Canis familiaris | Ilfov | [34] |
| I. ricinus, I. arboricola, Hae. concinna | E. rubecula, T. merula, T. philomelos | Constanța | [31] | |
| I. ricinus | H. sapiens | Cluj | [32] | |
| I. ricinus | H. sapiens | Sibiu | [33] | |
| I. ricinus, D. reticulatus, R. sanguineus | C. familiaris, Felis catus, Ovis aries, Vulpes vulpes | Satu-Mare, Călărași, Ilfov, Timiș, Dâmbovița, Mehedinți | [35] | |
| I. ricinus, I. hexagonus, Hae. punctata | E. roumanicus | Cluj | [28] | |
| Hae. concina | Sturnus vulgaris | Cluj | [28] | |
| R. raoultii 1 | D. marginatus | B. taurus, O. aries | Dâmbovița, Satu-Mare, Vâlcea | [30] |
| D. reticulatus | C. familiaris | Ilfov | [34] | |
| D. marginatus | H. sapiens | Sibiu | [33] | |
| D. marginatus, D. reticulatus, R. sanguineus | C. familiaris, Capra hircus, O. aries | Ilfov, Călărași, Covasna, Dâmbovița, Bistrița-Năsăud, Mehedinți, Vâlcea | [35] | |
| R. slovaca 1 | D. marginatus | B. taurus | Dâmbovița | [30] |
| D. reticulatus | C. familiaris | Ilfov | [34] | |
| I. ricinus | T. merula | Constanța | [31] | |
| I. ricinus, D. marginatus, R. sanguineus | C. familiaris, Capra hircus, O. aries, V. vulpes | Călărași, lfov, Covasna, Timiș, Mehedinți, Vâlcea | [35] | |
| R. aeschlimannii 1 | Hy. marginatum | B. taurus | Bistrița-Năsăud | [35] |
| Hae. concina | S. vulgaris | Cluj | [28] | |
| R. conorii 1 | R. sanguineus | C. familiaris | Ilfov | [34] |
| R. felis 1 | I. ricinus | T. merula | Cluj | [28] |
| R. massiliae 1 | I. ricinus, I. arboricola | T. philomelos | Constanța | [31] |
| Vertebrate host tissues | ||||
| R. helvetica 1 | - - | Parus major, Corvus frugilegus | Cluj | [28] |
| E. roumanicus | Cluj | [28] | ||
| R. monacensis 1 | - | T. merula | Cluj | [28] |
| - | Apodemus agrarius, A. sylvaticus, Mus musculus | Cluj | [28] | |
| - | Pipistrellus pipistrellus, Nyctalus noctula | Alba, Neamț | [36] | |
| R. slovaca 2 | - | H. sapiens | Ilfov | [37] |
| R. massiliae2 | - | H. sapiens | Ilfov | [37] |
| R. slovaca/R. raoultii 2 | - | H. sapiens | Ilfov | [37] |
| R. conorii 3 | - | H. sapiens | Unspecified | [38] |
| R. conorii 3 | - | H. sapiens | Unspecified | [39] |
| Fragments of Genes | Names of Gene | Citations |
|---|---|---|
| Rsfg877: GGGGGCCTGCTCACGGCGG | gltA | [43] |
| Rsfg1258: ATTGCAAAAAGTACAGTGAACA | ||
| rickP3: GGAACACTTCTTGGCGGTG | 17-kDa | [44] |
| rickP2: CATTGTCCGTCAGGTTGGCG | ||
| rickP5: GCATTACTTGGTTCTCAATTCGG | ||
| rickP4: AACCGTAATTGCCGTTATCCGG |
| Tick sp. | Origin | Developmental Stage | Sex | Prevalence % (n/Total) | 95% CI |
|---|---|---|---|---|---|
| Haemaphysalis punctata | Questing | AD | F | 7.14% (1/14) | 0.18–33.87 |
| M | 7.14% (1/14) | 0.18–33.87 | |||
| N | 6.98% (3/43) | 1.46–19.06 | |||
| L | 6.9% (2/29) | 3.99–10.04 | |||
| Total | 7.00% (7/100) | 5.02–7.87 | |||
| Ixodes ricinus | Questing | AD | M | 6.84% (27/395) | 4.74–9.76 |
| F | 6.55% (19/290) | 3.99–10.04 | |||
| N | 5.83% (25/429) | 3.98–8.46 | |||
| L | 0% (0/14) | NA | |||
| Total | 6.29% (71/1128) | 2.86–13.89% | |||
| Dermacentor marginatus | Questing | AD | F | 1.98% (5/253) | 0.64–4.55% |
| M | 1.57% (4/254) | 0.43–3.98% | |||
| Total | 1.78% (9/507) | 0.94–3.34% | |||
| Dermacentor reticulatus | Questing | AD | F | 0% (0/94) | NA |
| M | 0% (0/71) | NA | |||
| Total | 0% (0/128) | NA | |||
| Rhipicephalus rossicus | Engorged | AD | F | 33.33% (23/69) | 22.44–45.71% |
| M | 13.56% (8/59) | 6.04–24.98% | |||
| Total | 24.22% (31/128) | 17.09–32.58% | |||
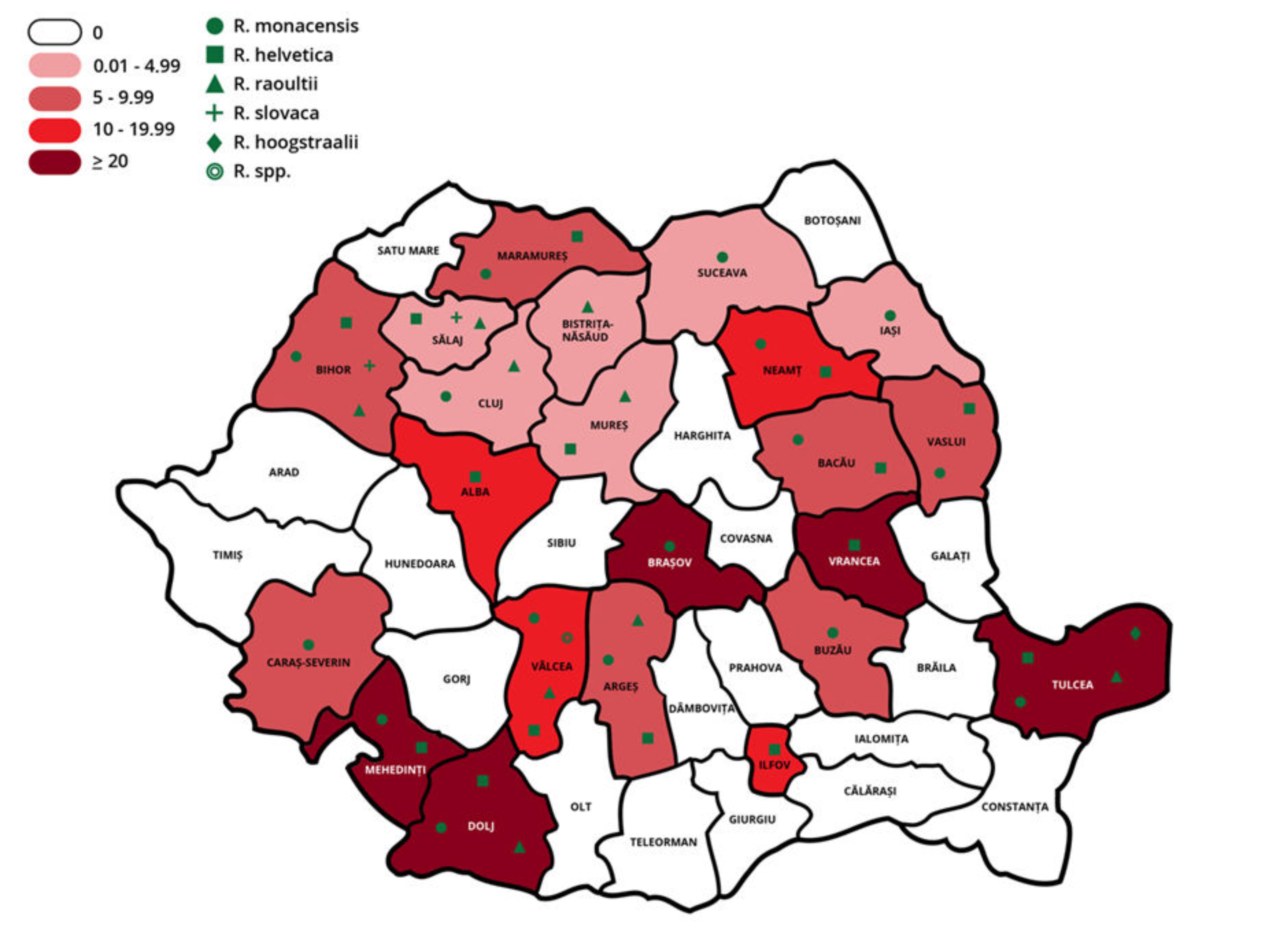
| County | Prevalence % (n/Total) | 95% CI |
|---|---|---|
| Alba | 18.18 (2/11) | 2.28–51.78 |
| Argeș | 6 (6/100) | 2.23–12.60 |
| Bacău | 5.63 (4/71) | 1.56–13.8 |
| Bihor | 6.1 (10/164) | 2.96–10.93 |
| Bistrița-Năsăud | 2.50 (1/40) | 0.06–13.16 |
| Brașov | 20 (1/5) | 0.51–71.64 |
| Buzău | 7.5 (3/40) | 1.57–20.39 |
| Cluj | 3.5 (7/200) | 1.42–7.08 |
| Covasna | 7.5 (3/40) | 1.57–20.39 |
| Dolj | 50 (5/10) | 18.71–81.29 |
| Ilfov | 10 (2/20) | 1.23–31.70 |
| Iași | 4.12 (4/97) | 1.13–10.22 |
| Mehedinți | 30 (3/10) | 6.67–65.25 |
| Maramureș | 8 (6/75) | 2.99–16.6 |
| Mureș | 3.64 (4/110) | 1.00–9.05 |
| Neamț | 10 (5/50) | 3.33–21.81 |
| Sălaj | 1.35 (4/297) | 0.37–3.41 |
| Suceava | 1.67 (1/60) | 0.04–8.94 |
| Tulcea | 23.87 (37/155) | 17.4–31.37 |
| Vâlcea | 15 (4/40) | 5.71–29.84 |
| Vrancea | 20 (2/10) | 2.52–55.61 |
| Vaslui | 9.09 (2/22) | 1.12–29.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivan, T.; Matei, I.A.; Novac, C.Ș.; Kalmár, Z.; Borșan, S.-D.; Panait, L.-C.; Gherman, C.M.; Ionică, A.M.; Papuc, I.; Mihalca, A.D. Spotted Fever Group Rickettsia spp. Diversity in Ticks and the First Report of Rickettsia hoogstraalii in Romania. Vet. Sci. 2022, 9, 343. https://doi.org/10.3390/vetsci9070343
Ivan T, Matei IA, Novac CȘ, Kalmár Z, Borșan S-D, Panait L-C, Gherman CM, Ionică AM, Papuc I, Mihalca AD. Spotted Fever Group Rickettsia spp. Diversity in Ticks and the First Report of Rickettsia hoogstraalii in Romania. Veterinary Sciences. 2022; 9(7):343. https://doi.org/10.3390/vetsci9070343
Chicago/Turabian StyleIvan, Talida, Ioana Adriana Matei, Cristiana Ștefania Novac, Zsuzsa Kalmár, Silvia-Diana Borșan, Luciana-Cătălina Panait, Călin Mircea Gherman, Angela Monica Ionică, Ionel Papuc, and Andrei Daniel Mihalca. 2022. "Spotted Fever Group Rickettsia spp. Diversity in Ticks and the First Report of Rickettsia hoogstraalii in Romania" Veterinary Sciences 9, no. 7: 343. https://doi.org/10.3390/vetsci9070343
APA StyleIvan, T., Matei, I. A., Novac, C. Ș., Kalmár, Z., Borșan, S.-D., Panait, L.-C., Gherman, C. M., Ionică, A. M., Papuc, I., & Mihalca, A. D. (2022). Spotted Fever Group Rickettsia spp. Diversity in Ticks and the First Report of Rickettsia hoogstraalii in Romania. Veterinary Sciences, 9(7), 343. https://doi.org/10.3390/vetsci9070343










