Antibiotic Resistance and Species Profile of Enterococcus Species in Dogs with Chronic Otitis Externa
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Enterococcus Isolation and Growth Conditions
2.2. Biochemical Test for Enterococcus Species Confirmation
2.3. Antibiotic Susceptibility Test
2.4. Statistical Analysis
3. Results
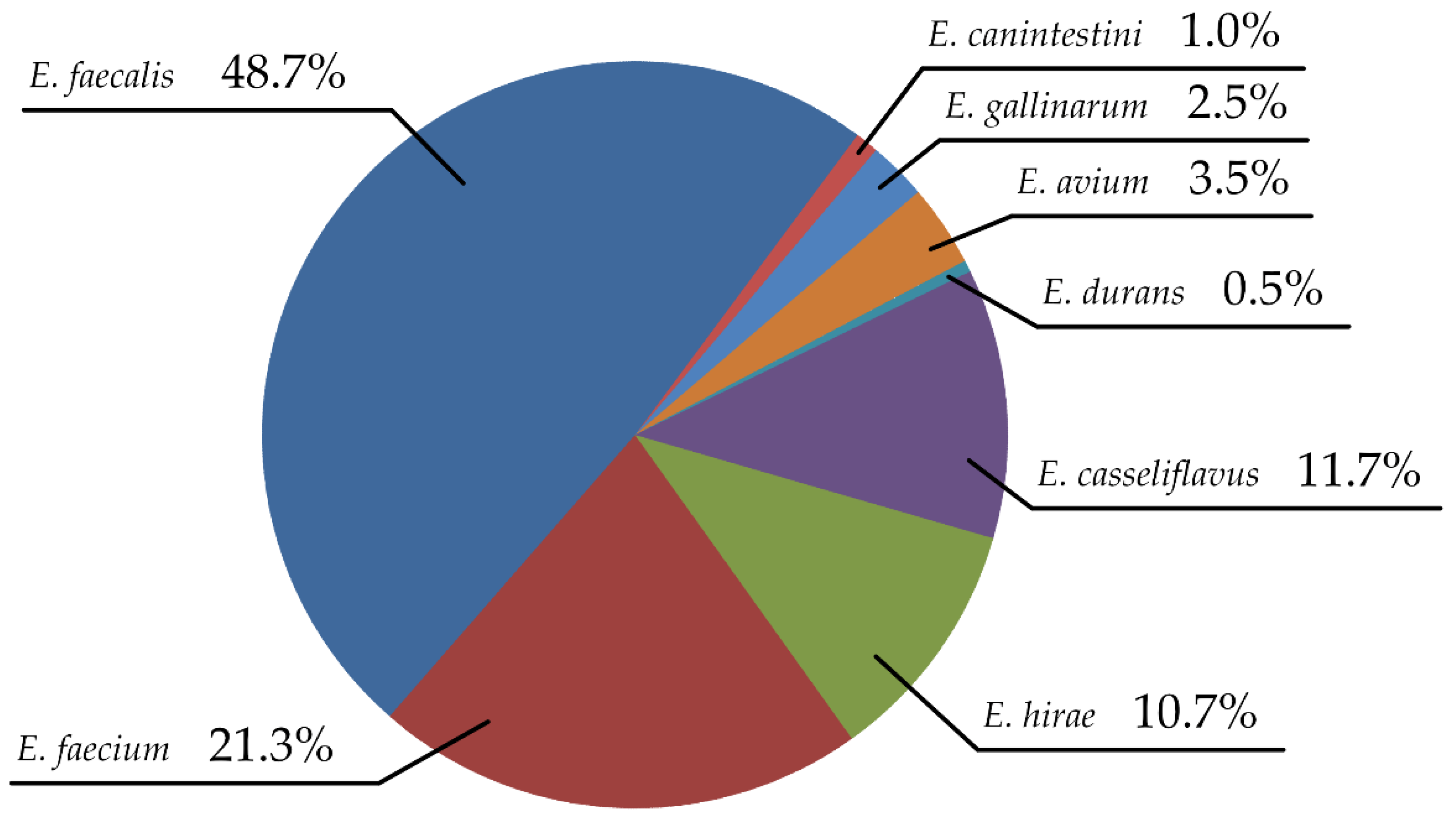
3.1. Enterococcus Species Distribution
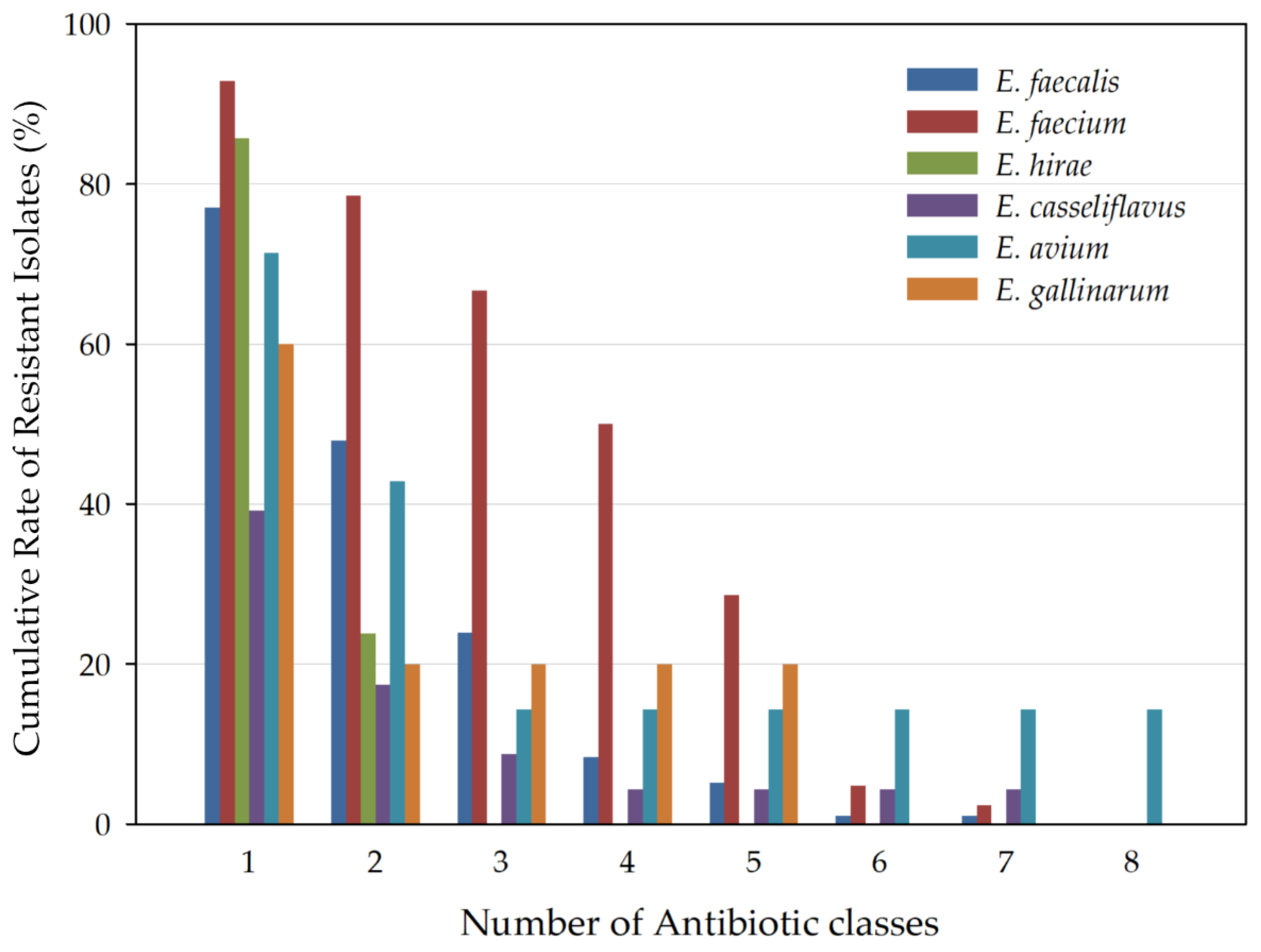
3.2. Antibiotic Resistance Profile
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Harvey, R.G. Ear Diseases of the Dog and Cat; CRC Press: London, UK, 2005; ISBN 9781840765274. [Google Scholar]
- Rosser, E.J. Causes of otitis externa. Vet. Clin. N. Am. Small Anim. Pract. 2004, 34, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Quinn, P.J.; Markey, B.K.; Leonard, F.C.; Hartigan, P.; Fanning, S.; Fitzpatrick, E.S. Veterinary Microbiology and Microbial Disease, 2nd ed.; Wiley-Blackwell: Chichester, UK, 2011; ISBN 9781405158237. [Google Scholar]
- Vergis, E.N.; Hayden, M.K.; Chow, J.W.; Snydman, D.R.; Zervos, M.J.; Linden, P.K.; Wagener, M.M.; Schmitt, B.; Murder, R.R. Determinants of vancomycin resistance and mortality rates in enterococcal bacteremia: A prospective multicenter study. Ann. Intern. Med. 2001, 135, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Van den Bogaard, A.E.; Stobberingh, E.E. Epidemiology of resistance to antibiotics: Links between animals and humans. Int. J. Antimicrob. Agents 2000, 14, 327–335. [Google Scholar] [CrossRef]
- Cetinkaya, Y.; Falk, P.; Mayhall, C.G. Vancomycin-resistant enterococci. Clin. Microbiol. Rev. 2000, 13, 686–707. [Google Scholar] [CrossRef]
- McVey, D.S.; Kennedy, M.; Chengappa, M. Veterinary Microbiology; Wiley-Blackwell: Hoboken, NJ, USA, 2013; ISBN 9780470959497. [Google Scholar]
- García Martínez de Artola, D.; Castro, B.; Ramos, M.J.; Díaz Cuevas, Z.; Lakhwani, S.; Lecuona, M. Outbreak of vancomycin-resistant enterococcus on a haematology ward: Management and control. J. Infect. Prev. 2017, 18, 149–153. [Google Scholar] [CrossRef]
- Zhou, X.; Willems, R.J.; Friedrich, A.W.; Rossen, J.W.; Bathoorn, E. Enterococcus faecium: From microbiological insights to practical recommendations for infection control and diagnostics. Antimicrob. Resist. Infect. Control 2020, 9, 130. [Google Scholar] [CrossRef]
- Said, M.S.; Tirthani, E.; Lesho, E. Enterococcus Infections; StatPearls Publishing: Treasure Isalnd, FL, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK567759/ (accessed on 20 October 2022).
- Dolka, B.; Gołębiewska-Kosakowska, M.; Krajewski, K.; Kwieciński, P.; Nowak, T.; Szubstarski, J.; Wilczyński, J.; Szeleszczuk, P. Occurrence of Enterococcus spp. in poultry in Poland based on 2014–2015 data. Med. Weter. 2017, 73, 220–224. [Google Scholar] [CrossRef]
- Ruzauskas, M.; Virgailis, M.; Šiugždinienė, R.; Sužiedėlienė, E.; Šeputienė, V.; Daugelavičius, R.; Zienius, D.; Šengaut, J.; Pavilonis, A. Antimicrobial resistance of Enterococcus spp. isolated from livestock in Lithuania. Vet. Arhiv. 2009, 79, 439–449. [Google Scholar]
- Hamilton, E.; Kaneene, J.B.; May, K.J.; Kruger, J.M.; Schall, W.; Beal, M.W.; Hauptman, J.G.; DeCamp, C.E. Prevalence and antimicrobial resistance of Enterococcus spp and Staphylococcus spp isolated from surfaces in a veterinary teaching hospital. J. Am. Vet. Med. Assoc. 2012, 240, 1463–1473. [Google Scholar] [CrossRef]
- Murray, B.E. Problems and dilemmas of antimicrobial resistance. Pharmacotherapy 1992, 12, 86S–93S. [Google Scholar]
- Martel, J.L.; Tardy, F.; Sanders, P.; Boisseau, J. New trends in regulatory rules and surveillance of antimicrobial resistance in bacteria of animal origin. Vet. Res. 2001, 32, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Herrero, I.A.; Fernández-Garayzábal, J.F.; Moreno, M.A.; Domínguez, L. Dogs should be included in surveillance programs for vancomycin-resistant enterococci. J. Clin. Microbiol. 2004, 42, 1384–1385. [Google Scholar] [CrossRef] [PubMed]
- Performance Standards for Antimicrobial Susceptibility Testing, 32nd ed.; Clinical and Laboratory Standard Institute: Wayne, PA, USA, 2022.
- Magiorakos, A.P.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, C.G.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Liljequist, B.; et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.J.; Chae, H.S.; Kim, H.J.; Kim, M.J.; Park, G.N.; Kim, S.H.; Chang, K.S. High prevalence of Enterococcus spp. From dogs with otitis externa. Korean J. Vet. Serv. 2012, 35, 99–104. [Google Scholar] [CrossRef]
- Klare, I.; Konstabel, C.; Badstübner, D.; Werner, G.; Witte, W. Occurrence and spread of antibiotic resistances in Enterococcus faecium. Int. J. Food Microbiol. 2003, 88, 269–290. [Google Scholar] [CrossRef]
- Golob, M.; Pate, M.; Kušar, D.; Dermota, U.; Avberšek, J.; Papić, B.; Zdovc, I. Antimicrobial resistance and virulence genes in Enterococcus faecium and Enterococcus faecalis from humans and retail red meat. Biomed Res. Int. 2019, 2019, 2815279. [Google Scholar] [CrossRef]
- Jamet, E.; Akary, E.; Poisson, M.A.; Chamba, J.F.; Bertrand, X.; Serror, P. Prevalence and characterization of antibiotic resistant Enterococcus faecalis in French cheeses. Food Microbiol. 2012, 31, 191–198. [Google Scholar] [CrossRef]
- Kim, Y.B.; Seo, K.W.; Shim, J.B.; Hyun Son, S.; Noh, E.B.; Lee, Y.J. Molecular characterization of antimicrobial-resistant Enterococcus faecalis and Enterococcus faecium isolated from layer parent stock. Poult. Sci. 2019, 98, 5892–5899. [Google Scholar] [CrossRef]
- De Graef, E.M.; Devriese, L.A.; Baele, M.; Vancanneyt, M.; Swings, J.; Haesebrouck, F.; Decostere, A. Identification of enterococcal, streptococcal and Weissella species in the faecal flora of individually owned dogs. J. Appl. Microbiol. 2005, 99, 348–353. [Google Scholar] [CrossRef]
- Hariharan, H.; Coles, M.; Poole, D.; Lund, L.; Page, R. Update on antimicrobial susceptibilities of bacterial isolates from canine and feline otitis externa. Can. Vet. J. 2006, 47, 253. [Google Scholar]
- Oh, J.H.; Cho, A.Y.; Kim, Y.S.; Lee, K.Y.; Sun, I.O. A rare case of urinary tract infection caused by Enterococcus hirae in an elderly man with benign prostate hyperplasia. Chonnam Med. J. 2019, 55, 177–178. [Google Scholar] [CrossRef] [PubMed]
- Dicpinigaitis, P.V.; De Aguirre, M.; Divito, J. Enterococcus hirae bacteremia associated with acute pancreatitis and septic shock. Case. Rep. Infect. Dis. 2015, 123852. [Google Scholar]
- Chan, T.S.; Wu, M.S.; Suk, F.M.; Chen, C.N.; Chen, Y.F.; Hou, Y.H.; Lien, G.S. Enterococcus hirae-related acute pyelonephritis and cholangitis with bacteremia: An unusual infection in humans. Kaohsiung J. Med. Sci. 2012, 28, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.H.; Lee, H.W.; Lee, Y.J.; Park, B.S.; Kim, Y.W.; Park, S. Acute pyelonephritis with Enterococcus hirae and literature review. Urogenit. Tract Infect. 2017, 12, 49–53. [Google Scholar] [CrossRef]
- Brayer, S.; Linn, A.; Holt, S.; Ellery, K.; Mitchell, S.; Williams, J. Enterococcus hirae bacteremia in an infant: Case report and review of the literature. J. Pediatr. Infect. Dis. 2019, 8, 571–573. [Google Scholar] [CrossRef]
- Chadfield, M.S.; Christensen, J.P.; Juhl-Hansen, J.; Christensen, H.; Bisgaard, M. Characterization of Enterococcus hirae outbreaks in broiler flocks demonstrating increased mortality because of septicemia and endocarditis and/or altered production parameters. Avian Dis. 2005, 49, 16–23. [Google Scholar] [CrossRef]
- Devriese, L.A.; Chiers, K.; De Herdt, P.; Vanrompay, D.; Desmidt, M.; Ducatelle, R.; Haesebrouck, F. Enterococcus hirae infections in psittacine birds: Epidemiological, pathological and bacteriological observations. Avian Pathol. 1995, 24, 523–531. [Google Scholar] [CrossRef]
- Toye, B.; Shymanski, J.; Bobrowska, M.; Woods, W.; Ramotar, K. Clinical and Epidemiologic Significance of Enterococci Intrinsically Resistant to Vancomycin (Possessing the van C Genotype). J. Clin. Microbiol. 1998, 36, 1469. [Google Scholar] [CrossRef]
- Leclercq, R.; Dutka-Malen, S.; Duval, J.; Courvalin, P. Vancomycin resistance gene vanC is specific to Enterococcus gallinarum. Antimicrob. Agents Chemother. 1992, 36, 2005–2008. [Google Scholar] [CrossRef]
- Schouten, M.A.; Hoogkamp-Korstanje, J.A.A.; Meis, J.F.G.; Voss, A. Prevalence of vancomycin-resistant enterococci in Europe. Eur. J. Clin. Microbiol. 2000, 19, 816–822. [Google Scholar] [CrossRef]
- Choi, S.H.; Lee, S.O.; Kim, T.H.; Chung, J.W.; Choo, E.J.; Kwak, Y.G.; Kim, M.N.; Kim, Y.S.; Woo, J.H.; Ryu, J.; et al. Clinical features and outcomes of bacteremia caused by Enterococcus casseliflavus and Enterococcus gallinarum: Analysis of 56 cases. Clin. Infect. Dis. 2004, 38, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Monticelli, J.; Knezevich, A.; Luzzati, R.; Di Bella, S. Clinical management of non-faecium non-faecalis vancomycin-resistant enterococci infection. Focus on Enterococcus gallinarum and Enterococcus casseliflavus/flavescens. J. Infect. Chemother. 2018, 24, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Huycke, M.M.; Sahm, D.F.; Gilmore, M.S. Multiple-drug resistant enterococci: The nature of the problem and an agenda for the future. Emerg. Infect. Dis. 1998, 4, 239. [Google Scholar] [CrossRef] [PubMed]
- Bang, K.; An, J.U.; Kim, W.; Dong, H.J.; Kim, J.; Cho, S. Antibiotic resistance patterns and genetic relatedness of Enterococcus faecalis and Enterococcus faecium isolated from military working dogs in Korea. J. Vet. Sci. 2017, 18, 229–236. [Google Scholar] [CrossRef]
- Hwang, I.Y.; Ku, H.O.; Lim, S.K.; Park, C.K.; Jung, G.S.; Jung, S.C.; Nam, H.M. Species distribution and resistance patterns to growth-promoting antimicrobials of enterococci isolated from pigs and chickens in Korea. J. Vet. Diagn. Investig. 2009, 21, 858–862. [Google Scholar] [CrossRef]
- Herman, D.J.; Gerding, D.N. Antimicrobial resistance among enterococci. Antimicrob. Agents Chemother. 1991, 35, 1–4. [Google Scholar] [CrossRef]
- Bush, L.M.; Calmon, J.; Cherney, C.L.; Wendeler, M.; Pitsakis, P.; Poupard, J.; Levison, M.E.; Johnson, C.C. High-level penicillin resistance among isolates of enterococci: Implications for treatment of enterococcal infections. Ann. Intern. Med. 1989, 110, 515–520. [Google Scholar] [CrossRef]
- Werner, G.; Coque, T.M.; Hammerum, A.M.; Hope, R.; Hryniewicz, W.; Johnson, A.; Klare, I.; Kristinsson, K.G.; Leclercq, R.; Lester, C.H.; et al. Emergence and spread of vancomycin resistance among enterococci in Europe. Euro Surveill. 2008, 13, 19046. [Google Scholar] [CrossRef]
- Nilsson, O. Vancomycin resistant enterococci in farm animals–occurrence and importance. Infect. Ecol. Epidemiol. 2012, 2, 16959. [Google Scholar]
- Hombach, M.; Böttger, E.C.; Roos, M. The critical influence of the intermediate category on interpretation errors in revised EUCAST and CLSI antimicrobial susceptibility testing guidelines. Clin. Microbiol. Infect. 2013, 19, E59–E71. [Google Scholar] [CrossRef]
- Molechan, C.; Amoako, D.G.; Abia, A.L.K.; Somboro, A.M.; Bester, L.A.; Essack, S.Y. Molecular epidemiology of antibiotic-resistant Enterococcus spp. from the farm-to-fork continuum in intensive poultry production in KwaZulu-Natal, South Africa. Sci. Total Environ. 2019, 692, 868–878. [Google Scholar] [CrossRef] [PubMed]
- Lester, C.H.; Sandvang, D.; Olsen, S.S.; Schønheyder, H.C.; Jarløv, J.O.; Bangsborg, J.; Hansen, D.S.; Jensen, T.G.; Frimodt-Møller, N.; Hammerum, A.M.; et al. Emergence of ampicillin-resistant Enterococcus faecium in Danish hospitals. J. Antimicrob. Chemother. 2008, 62, 1203–1206. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, G.; Hachem, R.; Jiang, Y.; Chemaly, R.F.; Raad, I. Outcomes for and risk factors associated with vancomycin-resistant Enterococcus faecalis and vancomycin-resistant Enterococcus faecium bacteremia in cancer patients. Infect. Control Hosp. Epidemiol. 2007, 28, 1054–1059. [Google Scholar] [CrossRef] [PubMed]
- Damborg, P.; Top, J.; Hendrickx, A.P.; Dawson, S.; Willems, R.J.; Guardabassi, L. Dogs are a reservoir of ampicillin-resistant Enterococcus faecium lineages associated with human infections. Appl. Environ. Microbiol. 2009, 75, 2360–2365. [Google Scholar] [CrossRef]
- Tremblay, C.L.; Charlebois, A.; Masson, L.; Archambault, M. Characterization of hospital-associated lineages of ampicillin-resistant Enterococcus faecium from clinical cases in dogs and humans. Front. Microbiol. 2013, 4, 245. [Google Scholar] [CrossRef]
- Van den Bunt, G.; Top, J.; Hordijk, J.; De Greeff, S.C.; Mughini-Gras, L.; Corander, J.; van Pelt, W.; Bonten, M.J.M.; Fluit, A.C.; Willems, R.J.L. Intestinal carriage of ampicillin-and vancomycin-resistant Enterococcus faecium in humans, dogs and cats in The Netherlands. J. Antimicrob. Chemother. 2018, 73, 607–614. [Google Scholar] [CrossRef]
- Lyskova, P.; Vydrzalova, M.; Mazurova, J. Identification and antimicrobial susceptibility of bacteria and yeasts isolated from healthy dogs and dogs with otitis externa. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2007, 54, 559–563. [Google Scholar] [CrossRef]
- Park, S.; Bae, S.; Kim, J.; Oh, T. Identification and antimicrobial susceptibility of bacteria isolated from dogs with chronic otitis externa. J. Vet. Clin. 2017, 34, 23–26. [Google Scholar] [CrossRef]
- Nelson, R.E.; Hyun, D.; Jezek, A.; Samore, M.H. Mortality, Length of Stay, and Healthcare Costs Associated with Multidrug-Resistant Bacterial Infections among Elderly Hospitalized Patients in the United States. Clin. Infect. Dis. 2022, 74, 1070–1080. [Google Scholar] [CrossRef]
- Loeffler, A.; Lloyd, D.H. Companion animals: A reservoir for methicillin-resistant Staphylococcus aureus in the community? Epidemiol. Infect. 2010, 138, 595–605. [Google Scholar] [CrossRef]
- Overgaauw, P.A.; Vinke, C.M.; van Hagen, M.A.; Lipman, L.J. A one health perspective on the human–companion animal relationship with emphasis on zoonotic aspects. Int. J. Environ. Health Res. 2020, 17, 3789. [Google Scholar] [CrossRef] [PubMed]
- Day, M.J. One health: The importance of companion animal vector-borne diseases. Parasit. Vectors 2011, 4, 49. [Google Scholar] [CrossRef] [PubMed]
- Buma, R.; Maeda, T.; Kamei, M.; Kourai, H. Pathogenic bacteria carried by companion animals and their susceptibility to antibacterial agents. Biocontrol Sci. 2006, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pomba, C.; Rantala, M.; Greko, C.; Baptiste, K.E.; Catry, B.; Van Duijkeren, E.; Mateus, A.; Moreno, A.M.; Pyörälä, S.; Ružauskas, M.; et al. Public health risk of antimicrobial resistance transfer from companion animals. J. Antimicrob. Chemother. 2017, 72, 957–968. [Google Scholar] [CrossRef] [PubMed]
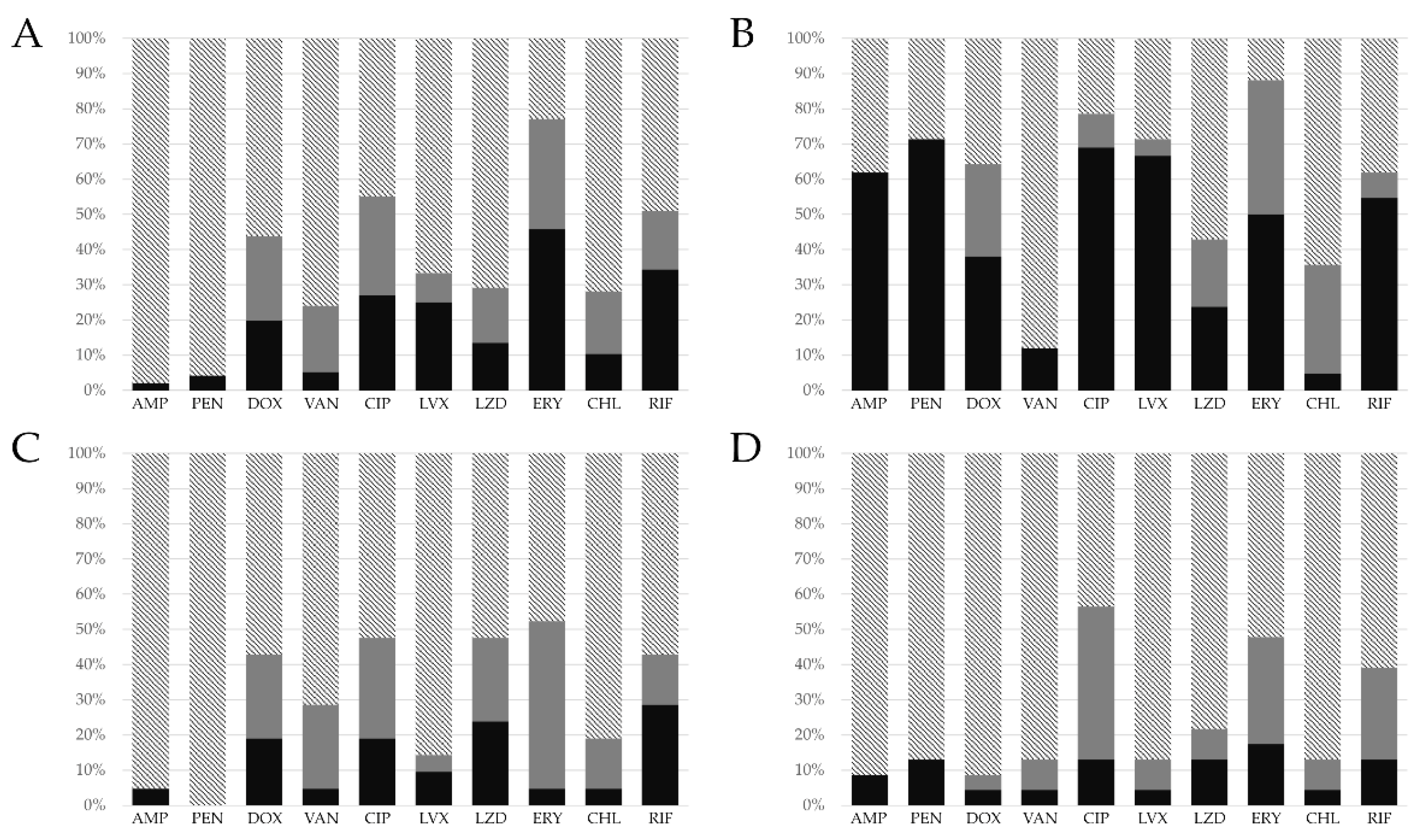
| Antibiotics | E. faecalis | E. faecium | E. casseliflavus | E. hirae | E. avium | E. gallinarum | E. canintestini | E. durans |
|---|---|---|---|---|---|---|---|---|
| n = 96 | n = 42 | n = 23 | n = 21 | n = 7 | n = 5 | n = 2 | n = 1 | |
| AMP | 2 (2.1%) | 26 (61.9%) | 2 (8.7%) | 1 (4.8%) | 2 (28.5%) | 1 (20%) | 1 (50%) | - |
| PEN | 4 (4.2%) | 30 (71.4%) | 3 (13%) | - | 2 (28.5%) | 1 (20%) | - | - |
| DOX | 19 (19.8%) | 16 (38.1%) | 1 (4.3%) | 4 (19%) | 1 (14.3%) | - | - | - |
| VAN | 5 (5.2%) | 5 (11.9%) | 1 (4.3%) | 1 (4.8%) | 2 (28.5%) | 2 (40%) | - | - |
| CIP | 26 (27.1%) | 29 (69%) | 3 (13%) | 4 (19%) | 1 (14.3%) | 1 (20%) | 1 (50%) | - |
| LVX | 24 (25%) | 28 (66.7%) | 1 (4.3%) | 2 (9.5%) | 1 (14.3%) | 1 (20%) | 1 (50%) | - |
| LZD | 13 (13.5%) | 10 (23.8%) | 3 (13%) | 5 (23.8%) | 2 (28.5%) | - | - | - |
| ERY | 44 (45.8%) | 21 (50%) | 4 (17.4%) | 1 (4.8%) | 3 (42.8%) | 1 (20%) | - | - |
| CHL | 10 (10.4%) | 2 (4.7%) | 1 (4.3%) | 1 (4.8%) | 1 (14.3%) | - | 1 (50%) | - |
| RIF | 33 (34.4%) | 23 (54.7%) | 3 (13%) | 6 (28.6%) | 2 (28.5%) | 2 (40%) | 1 (50%) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, J.; Ko, H.J.; Yang, M.H.; Park, C.; Park, S.C. Antibiotic Resistance and Species Profile of Enterococcus Species in Dogs with Chronic Otitis Externa. Vet. Sci. 2022, 9, 592. https://doi.org/10.3390/vetsci9110592
Kwon J, Ko HJ, Yang MH, Park C, Park SC. Antibiotic Resistance and Species Profile of Enterococcus Species in Dogs with Chronic Otitis Externa. Veterinary Sciences. 2022; 9(11):592. https://doi.org/10.3390/vetsci9110592
Chicago/Turabian StyleKwon, Jun, Hyoung Joon Ko, Myoung Hwan Yang, Chul Park, and Se Chang Park. 2022. "Antibiotic Resistance and Species Profile of Enterococcus Species in Dogs with Chronic Otitis Externa" Veterinary Sciences 9, no. 11: 592. https://doi.org/10.3390/vetsci9110592
APA StyleKwon, J., Ko, H. J., Yang, M. H., Park, C., & Park, S. C. (2022). Antibiotic Resistance and Species Profile of Enterococcus Species in Dogs with Chronic Otitis Externa. Veterinary Sciences, 9(11), 592. https://doi.org/10.3390/vetsci9110592






