Geographic Distribution of Ehrlichia canis TRP Genotypes in Brazil
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zweygarth, E.; Cabezas-Cruz, A.; Josemans, A.I.; Oosthuizen, M.C.; Matjila, P.T.; Lis, K.; Broniszewska, M.; Schöl, H.; Ferrolho, J.; Grubhoffer, L.; et al. In vitro culture and structural differences in the major immunoreactive protein gp36 of geographically distant Ehrlichia canis isolates. Ticks Tick Borne Dis. 2014, 5, 423–431. [Google Scholar] [CrossRef]
- Cárdenas, A.M.; Doyle, K.; Zhang, X.; Nethery, K.; Corstvet, R.E.; Walker, D.H.; McBride, J.W. Enzyme-Linked Immunosorbent Assay with Conserved Immunoreactive Glycoproteins gp36 and gp19 Has Enhanced Sensitivity and Provides Species-Specific Immunodiagnosis of Ehrlichia canis Infection. Clin. Vaccine Immunol. 2007, 14, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Doyle, C.K.; Nethery, K.A.; Popov, V.L.; McBride, J.W. Differentially Expressed and Secreted Major Immunoreactive Protein Orthologs of Ehrlichia canis and E. chaffeensis Elicit Early Antibody Responses to Epitopes on Glycosylated Tandem Repeats. Infect. Immun. 2006, 74, 711–720. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, D.M.; Zhang, X.; Braga, I.A.; Taques, I.I.G.G.; McBride, J.W. Detection of genotype-specific Ehrlichia canis exposure in Brazilian dogs by TRP36 peptide ELISA. Ticks Tick-Borne Dis. 2016, 7, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Taques, I.I.G.G.; Koiyama, M.F.G.; Campos, A.N.S.; Costa, J.S.; Hongyu, K.; Aguiar, D.M. Canonical correlation analysis between the ELISA assay using synthetic peptides, the indirect fluorescent antibody test and hematology values in dogs infected with Ehrlichia canis. Vet. Clin. Pathol 2020, in press. [Google Scholar] [CrossRef]
- Zhang, X.; Luo, T.; Keysary, A.; Baneth, G.; Miyashiro, S.; Strenger, C.; Waner, T.; McBride, J.W. Genetic and Antigenic Diversities of Major Immunoreactive Proteins in Globally Distributed Ehrlichia canis Strains. Clin. Vaccine Immunol. 2008, 15, 1080–1088. [Google Scholar] [CrossRef] [PubMed]
- McBride, J.W.; Doyle, C.K.; Zhang, X.; Cárdenas, A.M.; Popov, V.L.; Nethery, K.A.; Woods, M.E. Identification of a Glycosylated Ehrlichia canis 19-Kilodalton Major Immunoreactive Protein with a Species-Specific Serine-Rich Glycopeptide Epitope. Infect. Immun. 2007, 75, 74–82. [Google Scholar] [CrossRef]
- Aguiar, D.M.; Zhang, X.; Melo, A.L.T.; Pacheco, T.A.; Meneses, A.M.C.; Zanutto, M.S.; Horta, M.C.; Santarém, V.A.; Camargo, L.M.A.; McBride, J.W.; et al. Genetic diversity of Ehrlichia canis in Brazil. Vet. Microbiol. 2013, 164, 315–321. [Google Scholar] [CrossRef]
- Bouza-Mora, L.; Dolza, G.; Solórzano-Morales, A.; Romero-Zuñigaa, J.J.; Salazar-Sánchez, L.; Labruna, M.B.; Aguiar, D.M. Novel genotype of Ehrlichia canis detected in samples of human blood bank donors in Costa Rica. Ticks Tick Borne Dis. 2017, 8, 36–40. [Google Scholar] [CrossRef]
- Doyle, C.K.; Cárdenas, A.M.; Aguiar, D.M.; Labruna, M.B.; Ndip, L.M.; Yu, X.; Mcbride, J.W. Molecular Characterization of E. canis gp36 and E. chaffeensis gp47 Tandem Repeats among Isolates from Different Geographic Locations. Ann. N. Y. Acad. Sci. 2005, 1063, 433–435. [Google Scholar] [CrossRef]
- Arroyave, E.; Rodas-González, J.D.; Zhang, X.; Labruna, M.B.; González, M.S.; Fernández-Silva, J.A.; McBride, J.W. Ehrlichia canis TRP36 diversity in naturally infected dogs from an urban area of Colombia. Ticks Tick Borne Dis. 2020, 11, 101367. [Google Scholar] [CrossRef]
- Aguiar, D.M.; Saito, T.B.; Hagiwara, M.K.; Machado, R.Z.; Labruna, M.B. Diagnóstico sorológico de erliquiose canina com antígeno brasileiro de Ehrlichia canis. Ciênc. Rural 2007, 37, 796–802. [Google Scholar] [CrossRef]
- Saito, T.B.; Cunha-Filho, N.A.; Pacheco, R.C.; Ferreira, F.; Pappen, F.G.; Farias, N.A.R.; Larsson, C.E.; Labruna, M.B. Canine Infection by Rickettsiae and Ehrlichiae in Southern Brazil. Am. J. Trop. Med. Hyg. 2008, 70, 102–108. [Google Scholar] [CrossRef]
- Vieira, R.F.C.; Biondo, A.W.; Guimarães, A.M.S.; Santos, A.P.; Santos, R.P.; Leonardo Hermes Dutra, L.H.; Diniz, P.P.V.P.; Morais, H.A.; Joanne Belle Messick, J.B.; Labruna, M.B.; et al. Ehrlichiosis in Brazil. Braz. J. Vet. Parasitol. 2011, 20, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Palmer, G.H.; Bankhead, T.; Seifert, H.S. Antigenic Variation in Bacterial Pathogens. Microbiol. Spectr. 2016, 4. [Google Scholar] [CrossRef]
- Allsopp, M.T.E.P.; Allsopp, B.A. Extensive genetic recombination occurs in the field between different genotypes of Ehrlichia ruminantium. Vet. Microbiol. 2007, 124, 58–65. [Google Scholar] [CrossRef]
- Cabezas-Cruz, A.; Valdés, J.J.; de la Fuente, J. The glycoprotein TRP36 of Ehrlichia sp. UFMG-EV and related cattle pathogen Ehrlichia sp. UFMT-BV evolved from a highly variable clade of E. canis under adaptive diversifying selection. Parasites Vectors 2014, 7, 584. [Google Scholar] [CrossRef]
- Ferreira, R.F.; Cerqueira, A.M.F.; Castro, T.X.; Ferreira, E.O.; Neves, F.P.G.; Barbosa, A.V.; Macieira, D.B.; Almosny, N.R.P. Genetic diversity of Ehrlichia canis strains from naturally infected dogs in Rio de Janeiro, Brazil. Braz. J. Vet. Parasitol. 2014, 23, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Geiger, J.; Morton, B.A.; Vasconcelos, E.J.R.; Tngrian, M.; Kachani, M.; Barrón, E.A.; Gavidia, C.M.; Gilman, R.H.; Angulo, N.P.; Lerner, R.; et al. Molecular Characterization of Tandem Repeat Protein 36 Gene of Ehrlichia canis Detected in Naturally Infected Dogs from Peru. Am. J. Trop. Med. Hyg. 2018, 99, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Harrus, S.; Waner, T. Diagnosis of canine monocytotropic ehrlichiosis (Ehrlichia canis): An overview. Vet. J. 2011, 187, 292–296. [Google Scholar] [CrossRef]
- Moraes-Filho, J.; Marcili, A.; Nieri-Bastos, F.A.; Richtzenhain, L.J.; Marcelo, B.; Labruna, M.B. Genetic analysis of ticks belonging to the Rhipicephalus sanguineus group in Latin America. Acta Trop. 2011, 117, 51–55. [Google Scholar] [CrossRef] [PubMed]
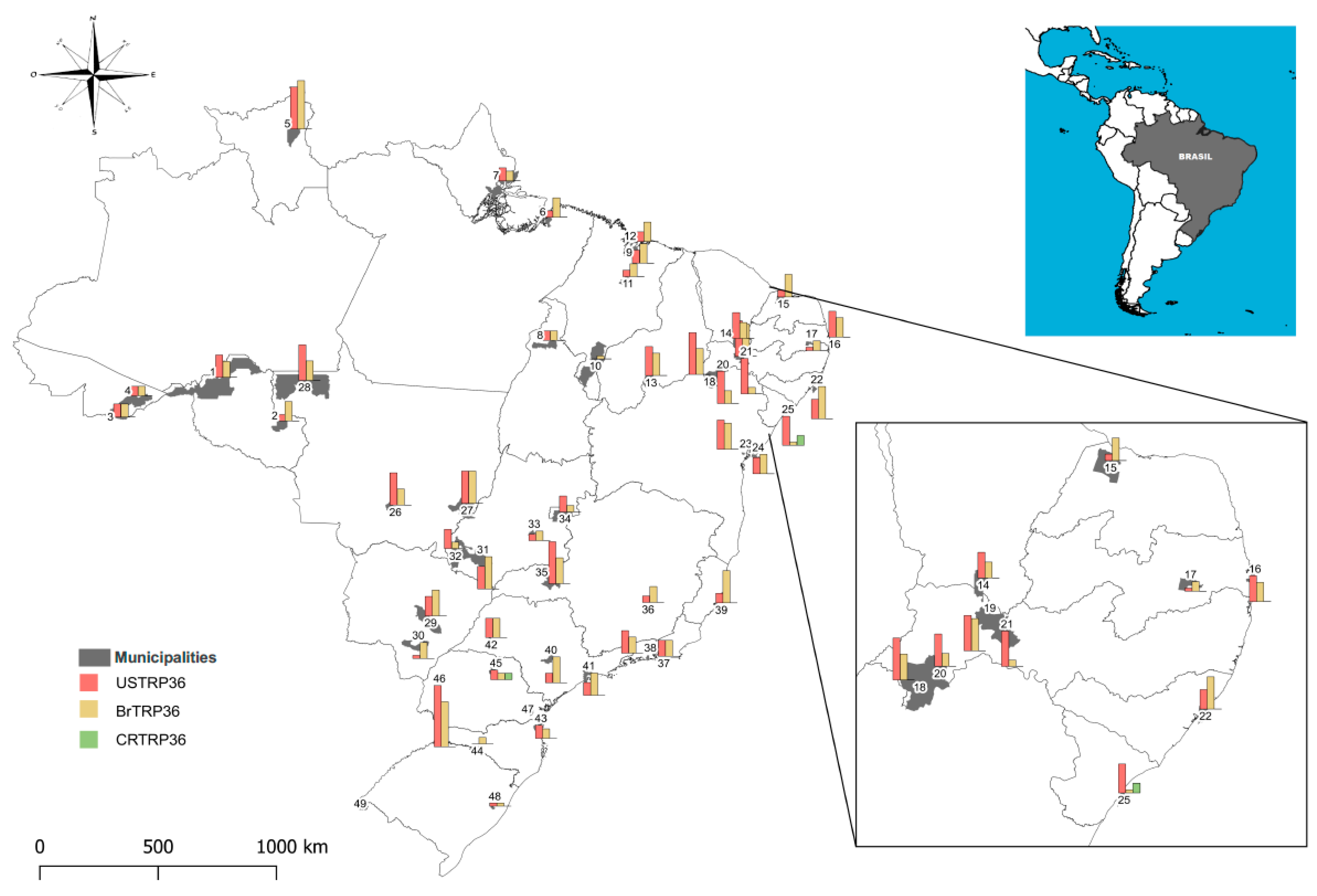
| Location * | Map Reference | No. of Dogs | ELISA (%) | IFA (Immunofluorescence Assay) (%) | |||
|---|---|---|---|---|---|---|---|
| TRP19 | USTRP36 | BrTRP36 | CRTRP36 | ||||
| North | 122 | 65 (53.2) | 38 (31.1) | 45 (36.8) | 0 | 58 (47.5) | |
| Porto Velho, RO | 1 | 15 | 11 | 7 | 5 | 0 | 9 |
| Cacoal, RO | 2 | 15 | 8 | 2 | 6 | 0 | 8 |
| Brasiléia, AC | 3 | 13 | 8 | 4 | 4 | 0 | 6 |
| Rio Branco, AC | 4 | 16 | 9 | 3 | 3 | 0 | 10 |
| Boa Vista, RR | 5 | 18 | 14 | 13 | 15 | 0 | 12 |
| Belém, PA | 6 | 16 | 5 | 2 | 6 | 0 | 4 |
| Macapá, AP | 7 | 15 | 4 | 4 | 3 | 0 | 3 |
| Araguaína, TO | 8 | 14 | 6 | 3 | 3 | 0 | 6 |
| Northeast | 236 | 114 (48.3) | 111 (47.0) | 94 (39.8) | 3 (1.27) | 124 (52.5) | |
| São Luís, MA | 9 | 18 | 7 | 4 | 6 | 0 | 4 |
| Balsas, MA | 10 | 9 | 1 | 0 | 1 | 0 | 1 |
| Bacabal, MA | 11 | 10 | 4 | 2 | 4 | 0 | 4 |
| Raposa, MA | 12 | 12 | 5 | 3 | 6 | 0 | 7 |
| Guaribas, PI | 13 | 14 | 8 | 9 | 7 | 0 | 9 |
| Crato, CE | 14 | 15 | 9 | 8 | 5 | 0 | 10 |
| Mossoró, RN | 15 | 14 | 7 | 2 | 7 | 0 | 8 |
| João Pessoa, PB | 16 | 14 | 5 | 8 | 6 | 0 | 7 |
| Campina Grande, PB | 17 | 10 | 4 | 1 | 3 | 0 | 3 |
| Petrolina, PE | 18 | 15 | 11 | 13 | 8 | 0 | 15 |
| Serrita, PE | 19 | 15 | 6 | 11 | 10 | 0 | 11 |
| Lagoa Grande, PE | 20 | 15 | 8 | 10 | 4 | 0 | 8 |
| Salgueiro, PE | 21 | 20 | 3 | 11 | 2 | 0 | 2 |
| Maceió, AL | 22 | 15 | 13 | 6 | 10 | 0 | 10 |
| Cruz das Almas, BA | 23 | 10 | 6 | 9 | 8 | 0 | 9 |
| Salvador, BA | 24 | 15 | 9 | 5 | 6 | 0 | 8 |
| Aracaju, SE | 25 | 15 | 8 | 9 | 1 | 3 | 8 |
| Central West | 131 | 75 (57.2) | 58 (44.2) | 51 (38.9) | 0 | 87 (66.4) | |
| Cuiabá, MT | 26 | 15 | 15 | 10 | 5 | 0 | 15 |
| Barra do Garças, MT | 27 | 15 | 13 | 10 | 10 | 0 | 13 |
| Colniza, MT | 28 | 15 | 11 | 11 | 6 | 0 | 11 |
| Campo Grande, MS | 29 | 15 | 9 | 6 | 8 | 0 | 9 |
| Dourados, MS | 30 | 15 | 7 | 1 | 5 | 0 | 12 |
| Jataí, GO | 31 | 15 | 8 | 7 | 10 | 0 | 12 |
| Mineiros, GO | 32 | 15 | 4 | 6 | 2 | 0 | 7 |
| Goiânia GO | 33 | 11 | 5 | 2 | 3 | 0 | 5 |
| Brasília, DF | 34 | 15 | 3 | 5 | 2 | 0 | 3 |
| Southeast | 123 | 62 (50.4) | 43 (34.9) | 54 (43.9) | 0 | 66 (53.6) | |
| Uberlândia, MG | 35 | 17 | 11 | 13 | 8 | 0 | 13 |
| Itabirito, MG | 36 | 16 | 4 | 2 | 5 | 0 | 4 |
| Niterói, RJ | 37 | 15 | 6 | 5 | 5 | 0 | 6 |
| Seropédica, RJ | 38 | 15 | 9 | 7 | 5 | 0 | 8 |
| Vitória, ES | 39 | 19 | 7 | 3 | 10 | 0 | 9 |
| Botucatu, SP | 40 | 15 | 12 | 3 | 8 | 0 | 15 |
| São Paulo, SP | 41 | 15 | 7 | 4 | 7 | 0 | 4 |
| Pres. Prudente, SP | 42 | 11 | 6 | 6 | 6 | 0 | 7 |
| South | 202 | 33 (16.3) | 27 (13.3) | 22 (10.8) | 2 (0.9) | 40 (19.8) | |
| Joinville, SC | 43 | 30 | 1 | 4 | 3 | 0 | 4 |
| Concórdia, SC | 44 | 40 | 2 | 0 | 2 | 0 | 1 |
| Londrina, PR | 45 | 14 | 5 | 3 | 2 | 2 | 6 |
| Palotina, PR | 46 | 30 | 25 | 19 | 14 | 0 | 27 |
| Curitiba, PR | 47 | 28 | 0 | 0 | 0 | 0 | 0 |
| Porto Alegre, RS | 48 | 30 | 0 | 1 | 1 | 0 | 2 |
| Barra do Quaraí, RS | 49 | 30 | 0 | 0 | 0 | 0 | 0 |
| Total | 814 | 349 (42.8) | 277 (34) | 266 (32.6) | 5 (0.61) | 375 (46) | |
| Region | No. of Dogs | Number of Positive Dogs (Frequency) (Optical Density Average; Range) | IFA (Frequency) (Titer Ranging) | |||
|---|---|---|---|---|---|---|
| TRP19 | USTRP36 * | BrTRP36 * | CRTRP36 | |||
| North | 122 | 65 (53.2%) a (2.25; 0.42–3.77) | 38 (31.1%) A b (1.21; 0.33–3.58) | 45 (36.8%) A a (1.8; 0.33–3.8) | 0 | 58 (47.5%) c b (40 to 10,240) |
| Northeast | 236 | 114 (48.3%) a (2.05; 0.39–3.80) | 111 (47.0%) A a (1.48; 0.33–3.47) | 94 (39.8%) A a (2.06; 0.33–4.02) | 3 (1.27%) (1.6; 1.04–2.13) | 124 (52.5%) b (40 to 10,240) |
| Center West | 131 | 75 (57.2%) a (2.22; 0.34–3.9) | 58 (44.2%) A a (1.63; 0.33–3.24) | 51 (38.9%) A a (1.82; 0.33–4.02) | 0 (0%) | 87 (66.4%) a (40 to 10,240) |
| Southeast | 123 | 62 (50.4%) a (2.01; 0.35–3.74) | 43 (34.9%) A b (1.45; 0.37–3.3) | 54 (43.9%) A a (1.72; 0.36–3.96) | 0 | 66 (53.6%) b (40 to 10,240) |
| South | 202 | 33 (16.3%) b (2.33; 0.37–3.95) | 27 (13.3%) A c (0.78; 0.33–2.51) | 22 (10.8%) A b (1.6; 0.33–3.65) | 2 (0.9%) (0.5; 0.35–0.66) | 40 (19.8%) d (40 to 10,240) |
| TOTAL | 814 | 349 (42.8%) (2.17; 0.34–3.95) | 277 (34%) A (1.51; 0.33–3.58) | 266 (32.6%) A (1.8; 0.33–4.02) | 5 (0.61%) (1.05; 0.35–2.13) | 375 (46%) (40 to 10,240) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taques, I.I.G.G.; Campos, A.N.S.; Kavasaki, M.L.; de Almeida, S.L.H.; de Aguiar, D.M. Geographic Distribution of Ehrlichia canis TRP Genotypes in Brazil. Vet. Sci. 2020, 7, 165. https://doi.org/10.3390/vetsci7040165
Taques IIGG, Campos ANS, Kavasaki ML, de Almeida SLH, de Aguiar DM. Geographic Distribution of Ehrlichia canis TRP Genotypes in Brazil. Veterinary Sciences. 2020; 7(4):165. https://doi.org/10.3390/vetsci7040165
Chicago/Turabian StyleTaques, Isis Indaiara Gonçalves Granjeiro, Amanda Noeli Silva Campos, Mayara Lima Kavasaki, Sayanne Luns Hatum de Almeida, and Daniel Moura de Aguiar. 2020. "Geographic Distribution of Ehrlichia canis TRP Genotypes in Brazil" Veterinary Sciences 7, no. 4: 165. https://doi.org/10.3390/vetsci7040165
APA StyleTaques, I. I. G. G., Campos, A. N. S., Kavasaki, M. L., de Almeida, S. L. H., & de Aguiar, D. M. (2020). Geographic Distribution of Ehrlichia canis TRP Genotypes in Brazil. Veterinary Sciences, 7(4), 165. https://doi.org/10.3390/vetsci7040165







