Postoperative Analgesic Effects of Laserpuncture and Meloxicam in Bitches Submitted to Ovariohysterectomy
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Procedures
2.3. Outcome Measures
2.4. Statistical Analysis
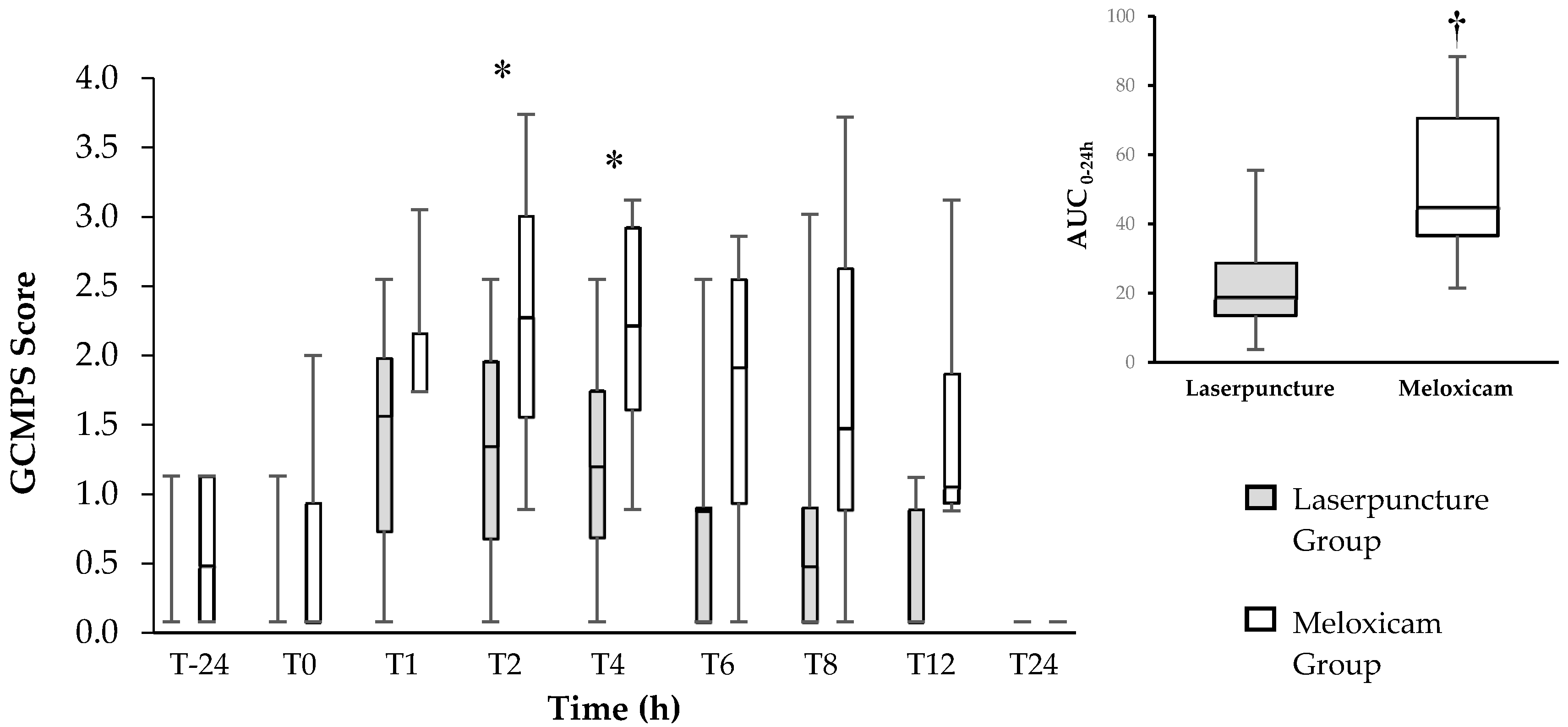
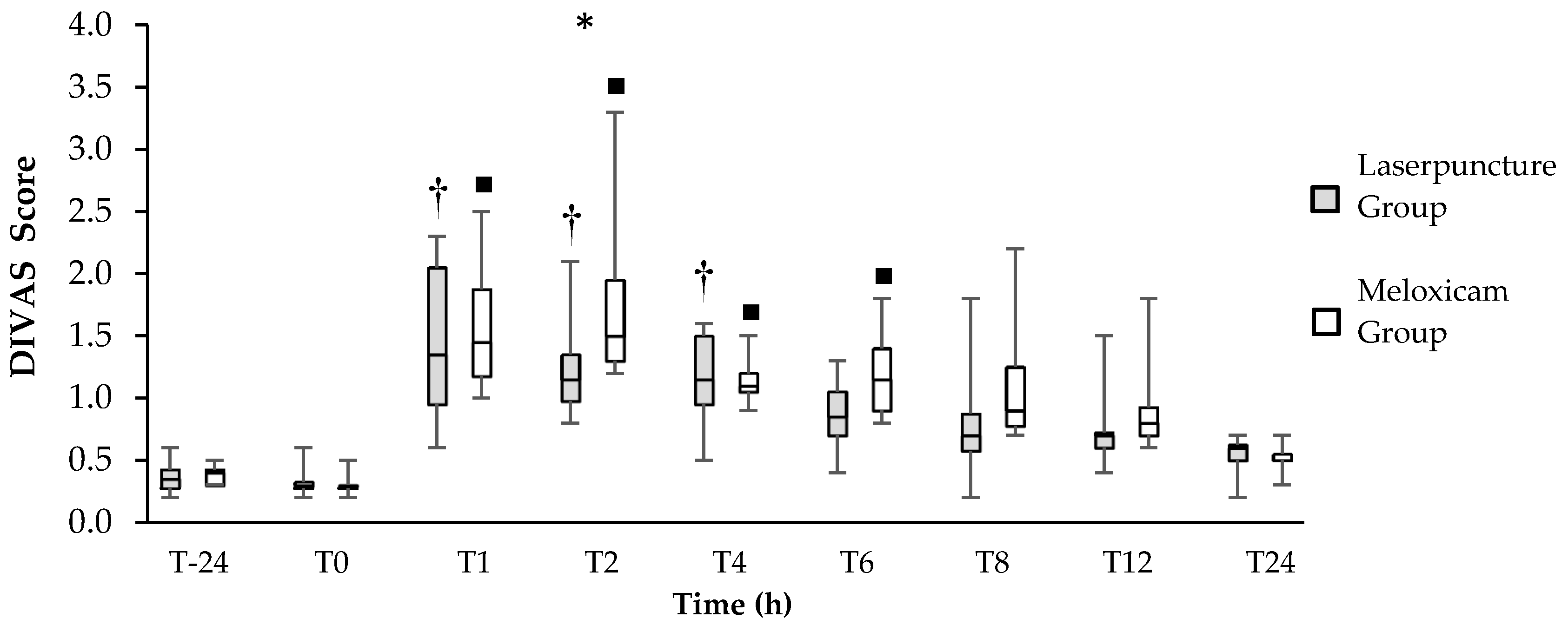
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Epstein, M.; Rodan, I.; Griffenhagen, G.; Kadrlik, J.; Petty, M.; Robertson, S.; Simpson, W. 2015 AAHA/AAFP Pain Management Guidelines for Dogs and Cats*. J. Am. Anim. Hosp. Assoc. 2015, 51, 67–84. [Google Scholar] [CrossRef] [PubMed]
- Luna, S.P.L.; Basílio, A.C.; Steagall, P.V.M.; Machado, L.P.; Moutinho, F.Q.; Takahira, R.K.; Brandão, C.V.S. Evaluation of adverse effects of long-term oral administration of carprofen, etodolac, flunixin meglumine, ketoprofen, and meloxicam in dogs. Am. J. Vet. Res. 2007, 68, 258–264. [Google Scholar] [CrossRef]
- Monteiro-Steagall, B.P.; Steagall, P.V.M.; Lascelles, B.D.X. Systematic review of nonsteroidal anti-inflammatory drug-induced adverse effects in dogs. J. Vet. Intern. Med. 2013, 27, 1011–1019. [Google Scholar] [CrossRef]
- Lorena, S.E.; Luna, S.P.; Lascelles, B.D.X.; Corrente, J.E. Current attitudes regarding the use of perioperative analgesics in dogs and cats by Brazilian veterinarians. Vet. Anaesth. Analg. 2014, 41, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Gakiya, H.H.; Silva, D.A.; Gomes, J.; Stevanin, H.; Cassu, R.N. Electroacupuncture versus morphine for the postoperative control pain in dogs. Acta Cir. Bras. 2011, 26, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Cassu, R.N.; Silva, D.A.D.; Genari Filho, T.; Stevanin, H. Electroanalgesia for the postoperative control pain in dogs. Acta Cir. Bras. 2012, 27, 43–48. [Google Scholar] [CrossRef]
- Luna, S.P.L.; Martino, I.D.; Lorena, S.E.R.D.S.; Capua, M.L.B.D.; Lima, A.F.D.M.; Santos, B.P.C.R.D.; Brondani, J.T.; Vesce, G.; Luna, S.P.L.; Martino, I.D.; et al. Acupuncture and pharmacopuncture are as effective as morphine or carprofen for postoperative analgesia in bitches undergoing ovariohysterectomy. Acta Cir. Bras. 2015, 30, 831–837. [Google Scholar] [CrossRef]
- Marques, V.I.; Cassu, R.N.; Nascimento, F.F.; Tavares, R.C.P.; Crociolli, G.C.; Guilhen, R.C.; Nicácio, G.M. Laser acupuncture for postoperative pain management in cats. Evidence-Based Complement. Altern. Med. 2015, 2015, 1–6. [Google Scholar] [CrossRef]
- Baratto, L.; Calzà, L.; Capra, R.; Gallamini, M.; Giardino, L.; Giuliani, A.; Lorenzini, L.; Traverso, S. Ultra-low-level laser therapy. Lasers Med. Sci. 2011, 26, 103–112. [Google Scholar] [CrossRef]
- Fabre, H.S.C.; Navarro, R.L.; Oltramari-Navarro, P.V.P.; Oliveira, R.F.; Pires-Oliveira, D.A.A.; Andraus, R.A.C.; Fuirini, N.; Fernandes, K.B.P. Anti-Inflammatory and analgesic effects of low-level laser therapy on the postoperative healing process. J. Phys. Ther. Sci. 2015, 27, 1645–1648. [Google Scholar] [CrossRef]
- Nascimento, F.F.; Marques, V.I.; Crociolli, G.C.; Nicácio, G.M.; Nicácio, I.P.A.G.; Cassu, R.N. Analgesic efficacy of laser acupuncture and electroacupuncture in cats undergoing ovariohysterectomy. J. Vet. Med. Sci. 2019, 81, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Leung, W.-W.; Jones, A.Y.M.; Ng, S.S.M.; Wong, C.Y.N.; Lee, J.F.Y. Electroacupuncture in reduction of discomfort associated with barostat-induced rectal distension—A randomized controlled study. J. Gastrointest. Surg. 2011, 15, 660–666. [Google Scholar] [CrossRef]
- Groppetti, D.; Pecile, A.M.; Sacerdote, P.; Bronzo, V.; Ravasio, G. Effectiveness of electroacupuncture analgesia compared with opioid administration in a dog model: A pilot study. Br. J. Anaesth. 2011, 107, 612–618. [Google Scholar] [CrossRef] [PubMed]
- Streiner, D.L.; Norman, G.R.; Cairney, J. Health Measurement Scales: A Practical Guide to Their Development and Use; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Gaynor, J.S.; Muir, W.W. Handbook of Veterinary Pain Management, 3rd ed.; Mosby Elsevier: Maryland Heights, MO, USA, 2014. [Google Scholar] [CrossRef]
- Murrell, J.C.; Psatha, E.P.; Scott, E.M.; Reid, J.; Hellebrekers, L.J. Application of a modified form of the glasgow pain scale in a veterinary teaching centre in the Netherlands. Vet. Rec. 2008, 162, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.C. Essentials of Veterinary Hematology; Lea and Febiger: Philadelphia, PA, USA, 1993. [Google Scholar]
- Chrisman, C.; Xie, H. Canine Transpositional Acupoints. In Xie’s Veterinary Acupuncture; Blackwell Publishing: Oxford, UK, 2007; pp. 129–215. [Google Scholar] [CrossRef]
- Smalle, T.M.; Hartman, M.J.; Bester, L.; Buck, R.K.; Fosgate, G.T.; Zeiler, G.E. Effects of thiopentone, propofol and alfaxalone on laryngeal motion during oral laryngoscopy in healthy dogs. Vet. Anaesth. Analg. 2017, 44, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Morton, C.M.; Reid, J.; Scott, E.M.; Holton, L.L.; Nolan, A.M. Application of a scaling model to establish and validate an interval level pain scale for assessment of acute pain in dogs. Am. J. Vet. Res. 2005, 66, 2154–2166. [Google Scholar] [CrossRef]
- Hellyer, P.; Robertson, A.; Fails, A. Pain and Its Management. In Lumb & Jones’ Veterinary Anesthesia Analgesia, 4th ed.; Tranquilli, W., Thurmon, J., Grimm, K., Eds.; Blackwell Publishing: Danvers, MA, USA, 2007; pp. 31–57. [Google Scholar]
- Quarterone, C.; Luna, S.P.L.; Crosignani, N.; de Oliveira, F.A.; Lopes, C.; da Maia Lima, A.F.; de Araújo Aguiar, A.J. Ovariohysterectomy requires more post-operative analgesia than orchiectomy in dogs and cats. Can. Vet. J. La Rev. Vet. Can. 2017, 58, 1191–1194. [Google Scholar]
- Teixeira, L.R.; Luna, S.P.L.; Taffarel, M.O.; Lima, A.F.M.; Sousa, N.R.; Joaquim, J.G.F.; Freitas, P.M.C. Comparison of intrarectal ozone, ozone administered in acupoints and meloxicam for postoperative analgesia in bitches undergoing ovariohysterectomy. Vet. J. 2013, 197, 794–799. [Google Scholar] [CrossRef]
- Leece, E.A.; Brearley, J.C.; Harding, E.F. Comparison of carprofen and meloxicam for 72 hours following ovariohysterectomy in dogs. Vet. Anaesth. Analg. 2005, 32, 184–192. [Google Scholar] [CrossRef]
- Shih, A.C.; Robertson, S.; Isaza, N.; Pablo, L.; Davies, W. Comparison between analgesic effects of buprenorphine, carprofen, and buprenorphine with carprofen for canine ovariohysterectomy. Vet. Anaesth. Analg. 2008, 35, 69–79. [Google Scholar] [CrossRef]
- Caulkett, N.; Read, M.; Fowler, D.; Waldner, C. A Comparison of the analgesic effects of butorphanol with those of meloxicam after elective ovariohysterectomy in dogs. Can. Vet. J. La Rev. Vet. Can. 2003, 44, 565–570. [Google Scholar]
- Hunt, J.R.; Dean, R.S.; Davis, G.N.D.; Murrell, J.C. An analysis of the relative frequencies of reported adverse events associated with NSAID administration in dogs and cats in the United Kingdom. Vet. J. 2015, 206, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Pogatzki-Zahn, E.M.; Segelcke, D.; Schug, S.A. Postoperative pain-from mechanisms to treatment. Pain Rep. 2017, 2, e588. [Google Scholar] [CrossRef] [PubMed]
- Erthal, V.; da Silva, M.D.; Cidral-Filho, F.J.; Santos, A.R.S.; Nohama, P. ST36 Laser acupuncture reduces pain-related behavior in rats: Involvement of the opioidergic and serotonergic systems. Lasers Med. Sci. 2013, 28, 1345–1351. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.T.; Lim, K.S.; Chung, K.; Ju, H.; Chung, J.M. Electroacupuncture-induced analgesia in a rat model of ankle sprain pain is mediated by spinal alpha-adrenoceptors. Pain 2008, 135, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Han, J.-S. Acupuncture and endorphins. Neurosci. Lett. 2004, 361, 258–261. [Google Scholar] [CrossRef]
- Han, J.S.; Tang, J.; Ren, M.F.; Zhou, Z.F.; Fan, S.G.; Qiu, X.C. Central neurotransmitters and acupuncture analgesia. Am. J. Chin. Med. 1980, 8, 331–348. [Google Scholar] [CrossRef]
- Hofmeister, E.H.; Barletta, M.; Shepard, M.; Brainard, B.M.; Trim, C.M.; Quandt, J. Agreement among anesthesiologists regarding postoperative pain assessment in dogs. Vet. Anaesth. Analg. 2018, 45, 695–702. [Google Scholar] [CrossRef]
- Comassetto, F.; Rosa, L.; Ronchi, S.J.; Fuchs, K.; Regalin, B.D.; Regalin, D.; Padiha, V.; Oleskovicz, N. Correlação entre as escalas analógica visual, de glasgow, colorado e melbourne na avaliação de dor pós-operatória em cadelas submetidas à mastectomia total unilateral. Arq. Bras. Med. Veterinária e Zootec. 2017, 69, 355–363. [Google Scholar] [CrossRef][Green Version]
- Zanuzzo, F.S.; Teixeira-Neto, F.J.; Teixeira, L.R.; Diniz, M.S.; Souza, V.L.; Thomazini, C.M.; Steagall, P.V.M. Analgesic and antihyperalgesic effects of dipyrone, meloxicam or a dipyrone–meloxicam combination in bitches undergoing ovariohysterectomy. Vet. J. 2015, 205, 33–37. [Google Scholar] [CrossRef]
- Kaka, U.; Rahman, N.-A.; Abubakar, A.A.; Goh, Y.M.; Fakurazi, S.; Omar, M.A.; Chen, H.C. Pre-Emptive multimodal analgesia with tramadol and ketamine–lidocaine infusion for suppression of central sensitization in a dog model of ovariohysterectomy. J. Pain Res. 2018, 11, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Michelsen, J.; Heller, J.; Wills, F.; Noble, G. Effect of surgeon experience on postoperative plasma cortisol and c-reactive protein concentrations after ovariohysterectomy in the dog: A randomised trial. Aust. Vet. J. 2012, 90, 474–478. [Google Scholar] [CrossRef]
- Kraus, B.L.H. Spotlight on the perioperative use of maropitant citrate. Vet. Med. Res. Rep. 2017, 8, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Scallan, E.M.; Simon, B.T. The effects of acupuncture point pericardium 6 on hydromorphone-induced nausea and vomiting in healthy dogs. Vet. Anaesth. Analg. 2016, 43, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Narjes, H.; Türck, D.; Busch, U.; Heinzel, G.; Nehmiz, G. Pharmacokinetics and tolerability of meloxicam after i.m. administration. Br. J. Clin. Pharmacol. 1996, 41, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Busch, U.; Schmid, J.; Heinzel, G.; Schmaus, H.; Baierl, J.; Huber, C.; Roth, W. Pharmacokinetics of meloxicam in animals and the relevance to humans. Drug Metab. Dispos. 1998, 26, 576–584. [Google Scholar] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomacheuski, R.M.; Taffarel, M.O.; Cardoso, G.S.; Derussi, A.A.P.; Ferrante, M.; Volpato, R.; Luna, S.P.L. Postoperative Analgesic Effects of Laserpuncture and Meloxicam in Bitches Submitted to Ovariohysterectomy. Vet. Sci. 2020, 7, 94. https://doi.org/10.3390/vetsci7030094
Tomacheuski RM, Taffarel MO, Cardoso GS, Derussi AAP, Ferrante M, Volpato R, Luna SPL. Postoperative Analgesic Effects of Laserpuncture and Meloxicam in Bitches Submitted to Ovariohysterectomy. Veterinary Sciences. 2020; 7(3):94. https://doi.org/10.3390/vetsci7030094
Chicago/Turabian StyleTomacheuski, Rubia M., Marilda O. Taffarel, Guilherme S. Cardoso, Ana A. P. Derussi, Marcos Ferrante, Rodrigo Volpato, and Stelio P. L. Luna. 2020. "Postoperative Analgesic Effects of Laserpuncture and Meloxicam in Bitches Submitted to Ovariohysterectomy" Veterinary Sciences 7, no. 3: 94. https://doi.org/10.3390/vetsci7030094
APA StyleTomacheuski, R. M., Taffarel, M. O., Cardoso, G. S., Derussi, A. A. P., Ferrante, M., Volpato, R., & Luna, S. P. L. (2020). Postoperative Analgesic Effects of Laserpuncture and Meloxicam in Bitches Submitted to Ovariohysterectomy. Veterinary Sciences, 7(3), 94. https://doi.org/10.3390/vetsci7030094




