Identification of High-Risk Areas for the Spread of Highly Pathogenic Avian Influenza in Central Luzon, Philippines
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Data Sources
2.3. Model Formulation
3. Assumptions
4. Results
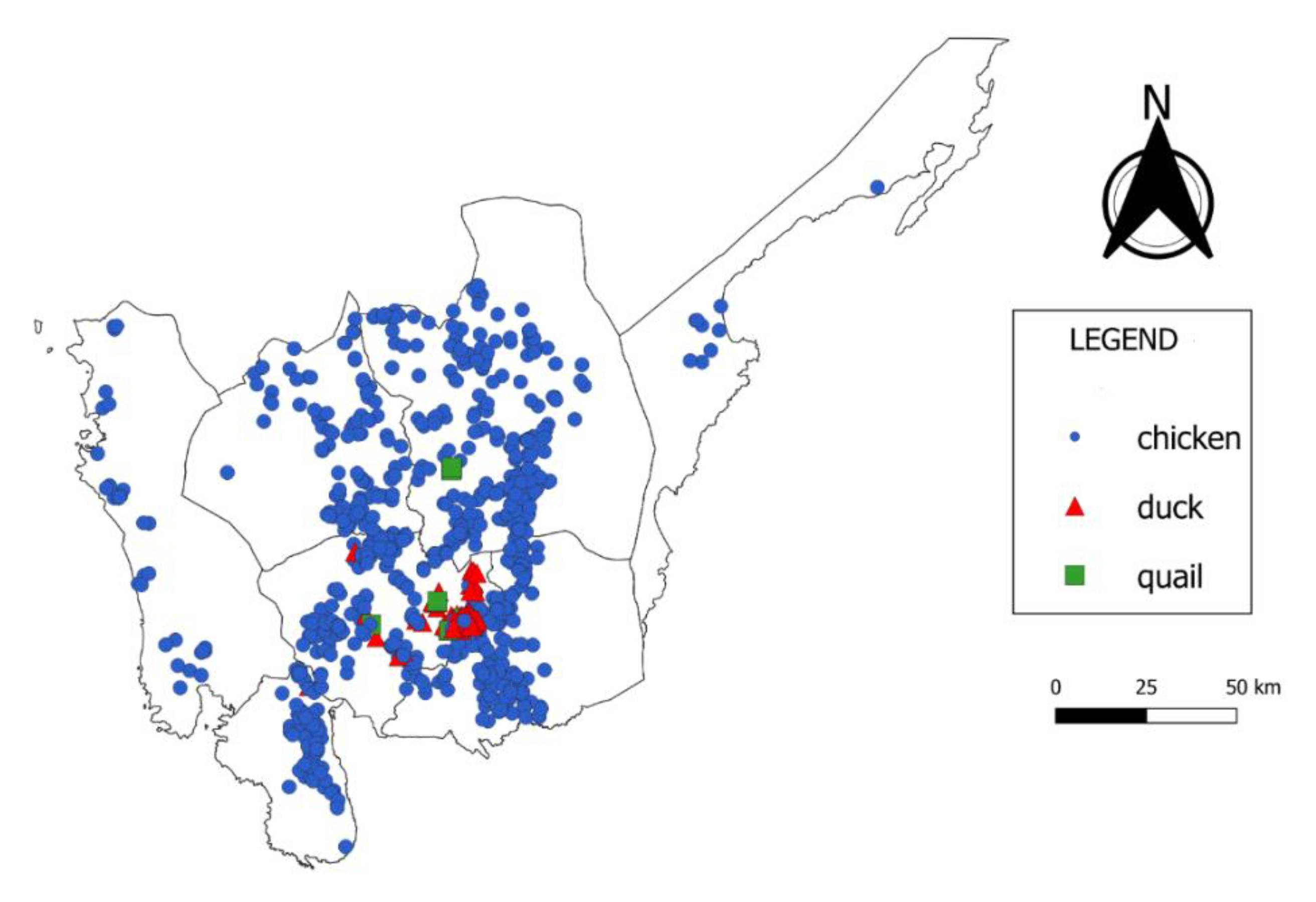
4.1. Farm Location
4.2. Farm Practices
4.3. Transmission Parameters
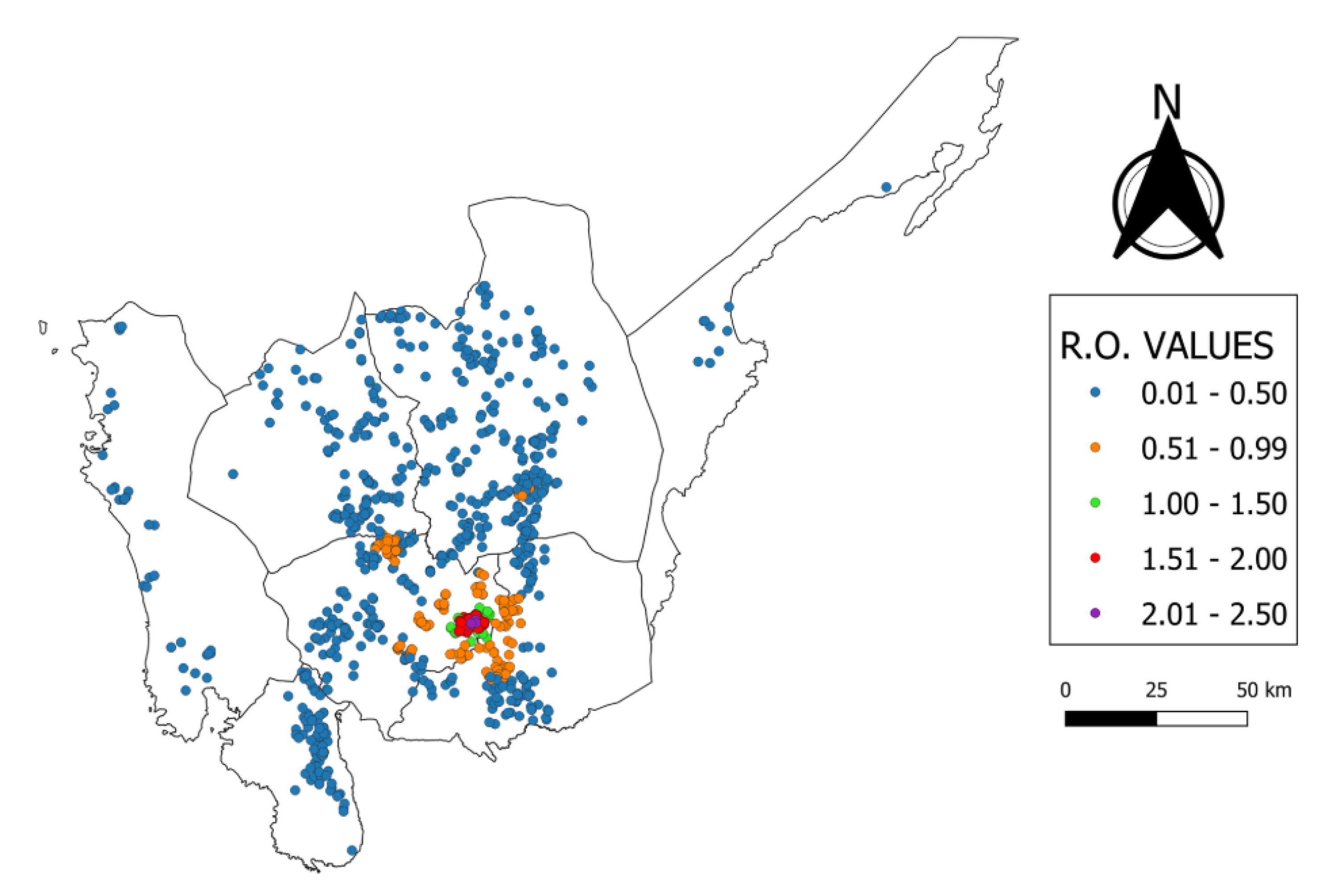
4.4. Farm-Level Basic Reproductive Ratio Values
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- Lee, H.; Lao, A. Transmission dynamics and control strategies assessment of avian influenza A (H5N6) in the Philippines. Infect. Dis. Model. 2018, 3, 35–59. [Google Scholar]
- Loth, L.; Gilbert, M.; Osmani, M.G.; Kalam, A.M.; Xiao, X. Risk factors and clusters of highly pathogenic avian influenza H5N1 outbreaks in Bangladesh. Prev. Vet. Med. 2010, 96, 104–113. [Google Scholar] [CrossRef]
- Loth, L.; Gilbert, M.; Wu, J.; Czarnecki, C.; Hidayat, M.; Xiao, X. Identifying risk factors of highly pathogenic avian influenza (H5N1 subtype) in Indonesia. Prev. Vet. Med. 2011, 102, 50–58. [Google Scholar] [CrossRef]
- Pfeiffer, D.U.; Minh, P.Q.; Martin, V.; Epprecht, M.; Otte, M.J. An analysis of the spatial and temporal patterns of highly pathogenic avian influenza occurrence in Vietnam using national surveillance data. Vet. J. 2007, 174, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, M.S.; Dissanayake, R.; Negi, A.B.; Oberoi, M.; Castellan, D.; Thrusfield, M.; Linard, C.; Gilbert, M. Spatio-temporal epidemiology of highly pathogenic avian influenza (subtype H5N1) in poultry in eastern India. Spat. Spatiotemporal. Epidemiol. 2014, 11, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Boender, G.J.; Hagenaars, T.J.; Bouma, A.; Nodelijk, G.; Elbers, A.R.W.; De Jong, M.C.M.; Van Boven, M. Risk maps for the spread of highly pathogenic avian influenza in poultry. PLoS Comput. Biol. 2007, 3, e71. [Google Scholar] [CrossRef] [PubMed]
- Boender, G.J.; Elbers, A.R.W.; De Jong, M.C.M. Spread of avian influenza in The Netherlands: Identifying areas at high risk. Vet. Ital. 2007, 43, 605–609. [Google Scholar] [PubMed]
- Hayama, Y.; Yamamoto, T.; Kobayashi, S.; Muroga, N.; Tsutsui, T. Evaluation of the transmission risk of foot-and-mouth disease in Japan. Virology 2015, 9, 1167–1170. [Google Scholar] [CrossRef] [PubMed][Green Version]
- McCullagh, M. Detecting hotspots in time and space. Int. Symp. Exhib. Geoinf. 2006, 349, 1–18. [Google Scholar]
- Keeling, M.J.; Woolhouse, E.J.; Shaw, D.J.; Matthews, L.; Chase-Topping, M.; Haydon, D.T.; Cornell, S.J.; Kappey, J.; Wilesmith, J.; Grenfell, B.T. Dynamics of the 2001 UK foot and mouth epidemic: Stochastic dispersal in a heterogeneous landscape. Science 2001, 294, 813–817. [Google Scholar] [CrossRef]
- Boender, G.J.; Van Roermund, H.J.; De Jong, M.C.M.; Hagenaars, T.J. Transmission risks and control of foot-and-mouth disease in The Netherlands: Spatial patterns. Epidemics 2010, 2, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Tildesley, M.J.; Keeling, M.J. Is R 0 a good predictor of final epidemic size: Foot-and-mouth disease in the UK. J. Theor. Biol. 2009, 258, 623–629. [Google Scholar] [CrossRef] [PubMed]
- LeMenach, A.; Legrand, J.; Grais, R.F.; Viboud, C.; Valleron, A.-J.; Flahault, A. Modeling spatial and temporal transmission of foot-and-mouth disease in France: Identification of high-risk areas. Vet. Res. 2005, 36, 699–712. [Google Scholar] [CrossRef] [PubMed]
- Hartemink, N.A.; Purse, B.V.; Meiswinkel, R.; Brown, H.E.; De Koeijer, A.; Elbers, A.R.W.; Boender, G.J.; Rogers, D.J.; Heesterbeek, J.A.P. Mapping the basic reproduction number (R0) for vector-borne diseases: A case study on bluetongue virus. Epidemics 2009, 1, 153–161. [Google Scholar] [CrossRef]
- Ster, I.C.; Ferguson, N.M. Transmission parameters of the 2001 foot and mouth epidemic in Great Britain. PLoS ONE 2007, 2, e502. [Google Scholar]
- Boender, G.J.; Meester, R.; Gies, E.; De Jong, M.C.M. The local threshold for geographical spread of infectious diseases between farms. Prev. Vet. Med. 2007, 82, 90–101. [Google Scholar] [CrossRef]
- Diekmann, O.; Heesterbeek, J.A.P.; Metz, J.A.J. On the definition and the computation of the basic reproduction ratio R o in models for infectious diseases in heterogeneous populations. J. Math. Biol. 1990, 28, 365–382. [Google Scholar] [CrossRef]
- Bersales, L.G.S. Duck situation report January–December 2018. Philipp. Stat. Auth. 2019, 1–37, ISSN 2546-0625. [Google Scholar]
- Bersales, L.G.S. Chicken situation report January–December 2018. Philipp. Stat. Auth. 2019, 1–45, ISSN 2546-0625. [Google Scholar]
- Truscott, J.; Garske, T.; Chis-ster, I.; Guitian, J.; Pfeiffer, D.; Snow, L.; Wilesmith, J.; Ferguson, N.M.; Ghani, A.C. Control of a highly pathogenic H5N1 avian influenza outbreak in the GB poultry flock. Proc. R. Soc. B Biol. Sci. 2007, 1623, 2287–2295. [Google Scholar] [CrossRef]
- Bonney, P.J.; Malladi, S.; Boender, G.J.; Weaver, J.T.; Ssematimba, A.; Halvorson, D.A.; Cardona, C.J. Spatial transmission of H5N2 highly pathogenic avian influenza between Minnesota poultry premises during the 2015 outbreak. PLoS ONE 2018, 13, e0204262. [Google Scholar] [CrossRef] [PubMed]
- Dorigatti, I.; Mulatti, P.; Rosà, R.; Pugliese, A.; Busani, L. Modelling the spatial spread of H7N1 avian in fl uenza virus among poultry farms in Italy. Epidemics 2010, 2, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Hayama, Y.; Yamamoto, T.; Kobayashi, S.; Muroga, N.; Tsutsui, T. Mathematical model of the 2010 foot-and-mouth disease epidemic in Japan and evaluation of control measures. Prev. Vet. Med. 2013, 112, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Keeling, M.J. Models of foot-and-mouth disease. Proc. R. Soc. B Biol. Sci. 2005, 272, 1195–1202. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008; ISBN 3-900051-07-0. [Google Scholar]
- QGIS Development Team QGIS Geographic Information System. Open Source Geospatial Foundation Project 2019. Available online: http://www.qgis.org (accessed on 5 January 2020).
- Swayne, D.E.; Beck, J.R. Experimental study to determine if low-pathogenicity and high-pathogenicity avian influenza viruses can be present in chicken breast and thigh meat following intranasal virus inoculation. Avian Dis. 2005, 49, 81–85. [Google Scholar] [CrossRef]
- Tian, G.; Zhang, S.; Li, Y.; Bu, Z.; Liu, P.; Zhou, J.; Li, C.; Shi, J.; Yu, K.; Chen, H. Protective efficacy in chickens, geese and ducks of an H5N1-inactivated vaccine developed by reverse genetics. Virology 2005, 341, 153–162. [Google Scholar] [CrossRef]
- Van Der Goot, J.A.; Koch, G.; De Jong, M.C.M.; Van Boven, M. Quantification of the effect of vaccination on transmission of avian influenza (H7N7) in chickens. Proc. Natl. Acad. Sci. USA 2005, 102, 18141–18146. [Google Scholar] [CrossRef]
- Hénaux, V.; Samuel, M.D.; Bunck, C.M. Model-based evaluation of highly and low pathogenic avian influenza dynamics in wild birds. PLoS ONE 2010, 5, e10997. [Google Scholar] [CrossRef]
- Spekreijse, D.; Bouma, A.; Koch, G.; Stegeman, J.A. Airborne transmission of a highly pathogenic avian influenza virus strain H5N1 between groups of chickens quantified in an experimental setting. Vet. Microbiol. 2011, 152, 88–95. [Google Scholar] [CrossRef]
- Van der Goot, J.A.; De Jong, M.C.M.; Koch, G.; Boven, V.M. Comparison of the transmission characteristics of low and high pathogenicity avian influenza A virus (H5N2). Epidemiol. Infect. 2003, 131, 1003–1013. [Google Scholar] [CrossRef]
- Dorea, F.C.; Vieira, A.R.; Hofacre, C.; Waldrip, D.; Cole, D.J. Stochastic model of the potential spread of highly pathogenic avian influenza from an infected commercial broiler operation in Georgia. Avian Dis. 2010, 5, e172–e173. [Google Scholar] [CrossRef]
- Elbers, A.A.R.W.; Fabri, T.H.F.; De Vries, T.S.; De Wit, J.J.; Pijpers, A.; Koch, G. The highly pathogenic Avian influenza A (H7N7) virus epidemic in The Netherlands in 2003—Lessons learned from the first five outbreaks. Avian Dis. 2004, 48, 691–705. [Google Scholar] [CrossRef] [PubMed]
- Savill, N.J.; Rose, S.G.S.; Woolhouse, M.E.J.; Interface, J.R.S.; Savill, N.J.; Rose, S.G.S.; Woolhouse, M.E.J. Detection of mortality clusters associated with highly pathogenic avian influenza in poultry: A theoretical analysis. J. R. Soc. Interf. 2008, 5, 1409–1419. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jiao, P.; Cui, J.; Song, Y.; Song, H.; Zhao, Z.; Wu, S.; Qu, N.; Wang, N.; Ouyang, G.; Liao, M. New reassortant H5N6 highly pathogenic avian influenza viruses in Southern China, 2014. Front. Microbiol. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Lee, E.K.; Lee, Y.N.; Kye, S.J.; Lewis, N.S.; Brown, I.H.; Sagong, M.; Heo, G.B.; Kang, Y.M.; Cho, H.K.; Kang, H.M.; et al. Characterization of a novel reassortant H5N6 highly pathogenic avian influenza virus clade 2.3.4.4 in Korea, 2017 correspondence. Emerg. Microbes Infect. 2018, 7, 1–8. [Google Scholar]
- Moore, D.A.; Carpenter, T.E. Spatial analytical methods and GIS: Use in health research and epidemiology. Epidemiol. Rev. 1999, 21, 143–161. [Google Scholar] [CrossRef]
- Ahmed, S.S.U.; Ersbøll, A.K.; Biswas, P.K.; Christensen, J.P. The space-time clustering of highly pathogenic avian influenza (HPAI) H5N1 outbreaks in Bangladesh. Epidemiol. Infect. 2010, 138, 843–852. [Google Scholar] [CrossRef]
- Tiensin, T.; Ahmed, S.S.U.; Rojanasthien, S.; Songserm, T.; Ratanakorn, P.; Chaichoun, K.; Kalpravidh, W.; Wongkasemjit, S.; Patchimasiri, T.; Chanachai, K.; et al. Ecologic risk factor investigation of clusters of Avian influenza A (H5N1) virus infection in Thailand. J. Infect. Dis. 2009, 199, 1735–1743. [Google Scholar] [CrossRef]
- Gilbert, M.; Chaitaweesub, P.; Parakamawongsa, T.; Premashthira, S.; Tiensin, T.; Kalpravidh, W.; Wagner, H.; Slingenbergh, J. Free-grazing ducks and highly pathogenic Avian Influenza, Thailand. Emerg. Infect. Dis. J. 2006, 12, 227–234. [Google Scholar] [CrossRef]
- Wibawa, H.; Bingham, J.; Nuradji, H.; Lowther, S.; Payne, J.; Harper, J.; Junaidi, A.; Middleton, D.; Meers, J. Experimentally infected domestic ducks show efficient transmission of Indonesian H5N1 highly pathogenic Avian influenza virus, but lack persistent viral shedding. PLoS ONE 2014, 9, e83417. [Google Scholar] [CrossRef]
- Tiensin, T.; Nielen, M.; Vernooij, H.; Songserm, T.; Kalpravidh, W.; Chotiprasatintara, S.; Chaisingh, A.; Wongkasemjit, S.; Chanachai, K.; Thanapongtham, W.; et al. Transmission of the highly pathogenic Avian influenza virus H5N1 within flocks during the 2004 epidemic in Thailand. J. Infect. Dis. 2007, 196, 1679–1684. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.C.; Uyeki, T.M.; Jadhao, S.; Maines, T.; Shaw, M.; Matsuoka, Y.; Smith, C.; Rowe, T.; Lu, X.; Hall, H.; et al. Isolation and characterization of Avian influenza viruses, including highly pathogenic H5N1, from poultry in live bird markets in Hanoi, Vietnam, in 2001. J. Virol. 2005, 79, 4201–4212. [Google Scholar] [CrossRef] [PubMed]
- Sturm-Ramirez, K.M.; Hulse-Post, D.J.; Govorkova, E.A.; Humberd, J.; Seiler, P.; Puthavathana, P.; Buranathai, C.; Nguyen, T.D.; Chaisingh, A.; Long, H.T.; et al. Are ducks contributing to the endemicity of highly pathogenic H5N1 influenza virus in Asia? J. Virol. 2005, 79, 11269–11279. [Google Scholar] [CrossRef] [PubMed]
- Thanawat, T.; Prasit, C.; Thaweesak, S.; Arunee, C.; Wirongrong, H.; Chantanee, B.; Tippawon, P.; Sith, P.; Alongkorn, A.; Marius, G.; et al. Highly pathogenic Avian influenza H5N1, Thailand, 2004. Emerg. Infect. Dis. J. 2005, 11, 1664. [Google Scholar]
- Boonyapisitsopa, S.; Chaiyawong, S.; Nonthabenjawan, N. Sentinel model for influenza A virus monitoring in free-grazing ducks in Thailand. Vet. Microbiol. 2016, 182, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Hulse-Post, D.J.; Sturm-Ramirez, K.M.; Humberd, J.; Seiler, P.; Govorkova, E.A.; Krauss, S.; Scholtissek, C.; Puthavathana, P.; Buranathai, C.; Nguyen, T.D.; et al. Role of domestic ducks in the propagation and biological evolution of highly pathogenic H5N1 influenza viruses in Asia. Proc. Natl. Acad. Sci. USA 2005, 102, 10682–10687. [Google Scholar] [CrossRef]
- Melendres, R.G. The utilization of Candaba swamp from prehistoric to present time: Evidences from archaeology, history and ethnography. Bhatter Coll. J. Multidiscip. Stud. 2014, 4, 81–93. [Google Scholar]
- Biswas, P.K.; Christensen, J.P.; Ahmed, S.S.U.; Barua, H.; Das, A.; Rahman, M.H.; Giasuddin, M.; Hannan, A.S.M.A.; Habib, A.M.; Debnath, N.C. Risk factors for infection with highly pathogenic influenza A virus (H5N1) in commercial chickens in Bangladesh. Vet. Rec. 2009, 164, 743–746. [Google Scholar] [CrossRef]
- Ward, M.P.; Maftei, D.; Apostu, C.; Suru, A. Environmental and anthropogenic risk factors for highly pathogenic avian influenza subtype H5N1 outbreaks in Romania, 2005—2006. Vet. Res. Commun. 2008, 32, 627–634. [Google Scholar] [CrossRef]
- Chunxiang, C.A.O.; Min, X.U.; Chaoyi, C.; Yong, X.U.E.; Shaobo, Z.; Liqun, F.; Wuchun, C.A.O.; Hao, Z.; Mengxu, G.A.O.; Qisheng, H.E.; et al. Risk analysis for the highly pathogenic avian influenza in Mainland China using meta-modeling. Chin. Sci. Bull. 2010, 55, 4168–4178. [Google Scholar]
- Fang, L.; Vlas, S.J.D.; Liang, S.; Looman, C.W.N.; Gong, P.; Xu, B.; Yang, H.; Richardus, J.H.; Cao, W. Environmental factors contributing to the spread of H5N1 Avian influenza in Mainland China. PLoS ONE 2008, 3, e2268. [Google Scholar] [CrossRef] [PubMed]
- Perez, D.R.; Lim, W.; Seiler, J.P.; Yi, G.; Peiris, M.; Shortridge, K.F.; Webster, R.G. Role of quail in the interspecies transmission of H9 influenza A viruses: Molecular changes on HA that correspond to adaptation from ducks to chickens. J. Virol. 2003, 77, 3148–3156. [Google Scholar] [CrossRef] [PubMed]
- Webster, R.G.; Guan, Y.; Peiris, M.; Walker, D.; Krauss, S.; Zhou, N.N.; Govorkova, E.A.; Ellis, T.M.; Dyrting, K.C.; Sit, T.; et al. Characterization of H5N1 influenza viruses that continue to circulate in geese in Southeastern China. J. Virol. 2002, 76, 118–126. [Google Scholar] [CrossRef] [PubMed]
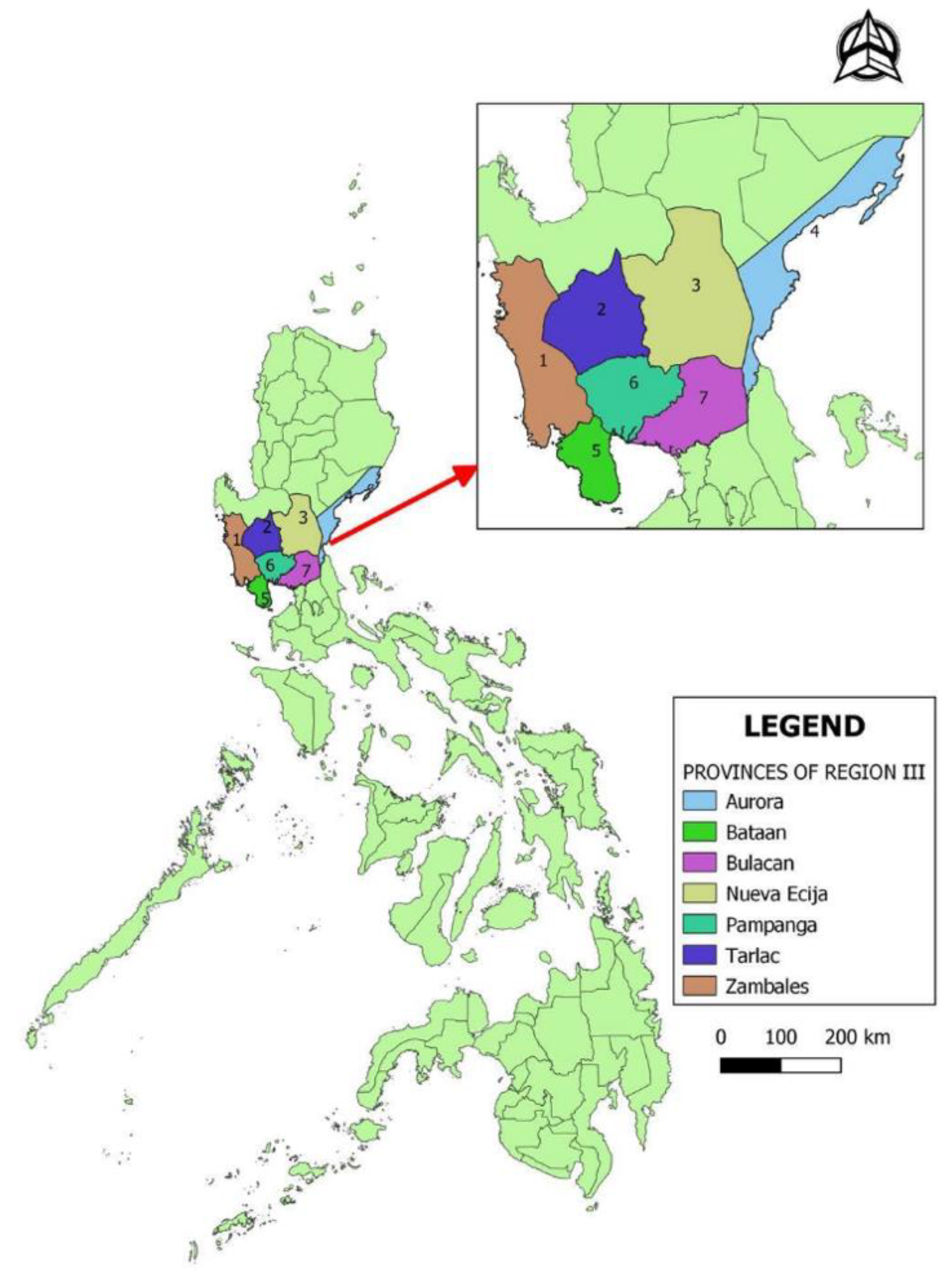
| Province | Poultry Farms | Average Population |
|---|---|---|
| Aurora | 9 | 8222 |
| Bataan | 90 | 54,147 |
| Bulacan | 154 | 92,858 |
| Nueva Ecija | 268 | 57,823 |
| Pampanga | 485 | 27,257 |
| Tarlac | 112 | 87,231 |
| Zambales | 33 | 104,164 |
| R0 | Ducks | Quail | Chickens | Total |
|---|---|---|---|---|
| 0.01–0.50 | 8 | 6 | 705 | 719 |
| 0.51–0.99 | 18 | 1 | 144 | 163 |
| 1.00–1.50 | 8 | 3 | 12 | 23 |
| 1.51–2.00 | 135 | 0 | 15 | 150 |
| 2.01–2.5 | 91 | 1 | 4 | 96 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salvador, R.; Tanquilut, N.; Na Lampang, K.; Chaisowwong, W.; Pfeiffer, D.; Punyapornwithaya, V. Identification of High-Risk Areas for the Spread of Highly Pathogenic Avian Influenza in Central Luzon, Philippines. Vet. Sci. 2020, 7, 107. https://doi.org/10.3390/vetsci7030107
Salvador R, Tanquilut N, Na Lampang K, Chaisowwong W, Pfeiffer D, Punyapornwithaya V. Identification of High-Risk Areas for the Spread of Highly Pathogenic Avian Influenza in Central Luzon, Philippines. Veterinary Sciences. 2020; 7(3):107. https://doi.org/10.3390/vetsci7030107
Chicago/Turabian StyleSalvador, Roderick, Neil Tanquilut, Kannika Na Lampang, Warangkhana Chaisowwong, Dirk Pfeiffer, and Veerasak Punyapornwithaya. 2020. "Identification of High-Risk Areas for the Spread of Highly Pathogenic Avian Influenza in Central Luzon, Philippines" Veterinary Sciences 7, no. 3: 107. https://doi.org/10.3390/vetsci7030107
APA StyleSalvador, R., Tanquilut, N., Na Lampang, K., Chaisowwong, W., Pfeiffer, D., & Punyapornwithaya, V. (2020). Identification of High-Risk Areas for the Spread of Highly Pathogenic Avian Influenza in Central Luzon, Philippines. Veterinary Sciences, 7(3), 107. https://doi.org/10.3390/vetsci7030107






