Effects of 3 Different Commercial Vaccines Formulations against BVDV and BHV-1 on the Inflammatory Response of Holstein Heifers
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
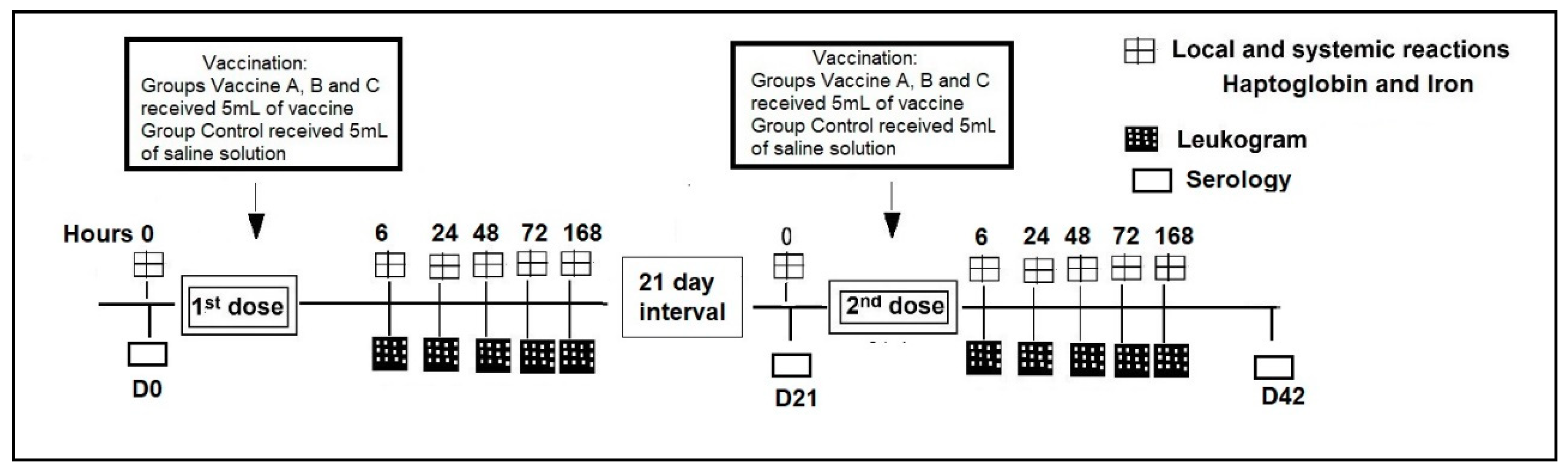
2.2. Vaccination and Management
2.3. Local and Systemic Reaction
2.4. Blood Samples
2.5. White Blood Count (WBC)
2.6. Iron Concentration
2.7. Haptoglobin
2.8. Serum Virus Neutralization (SVN)
2.9. Statistical Analysis
3. Results
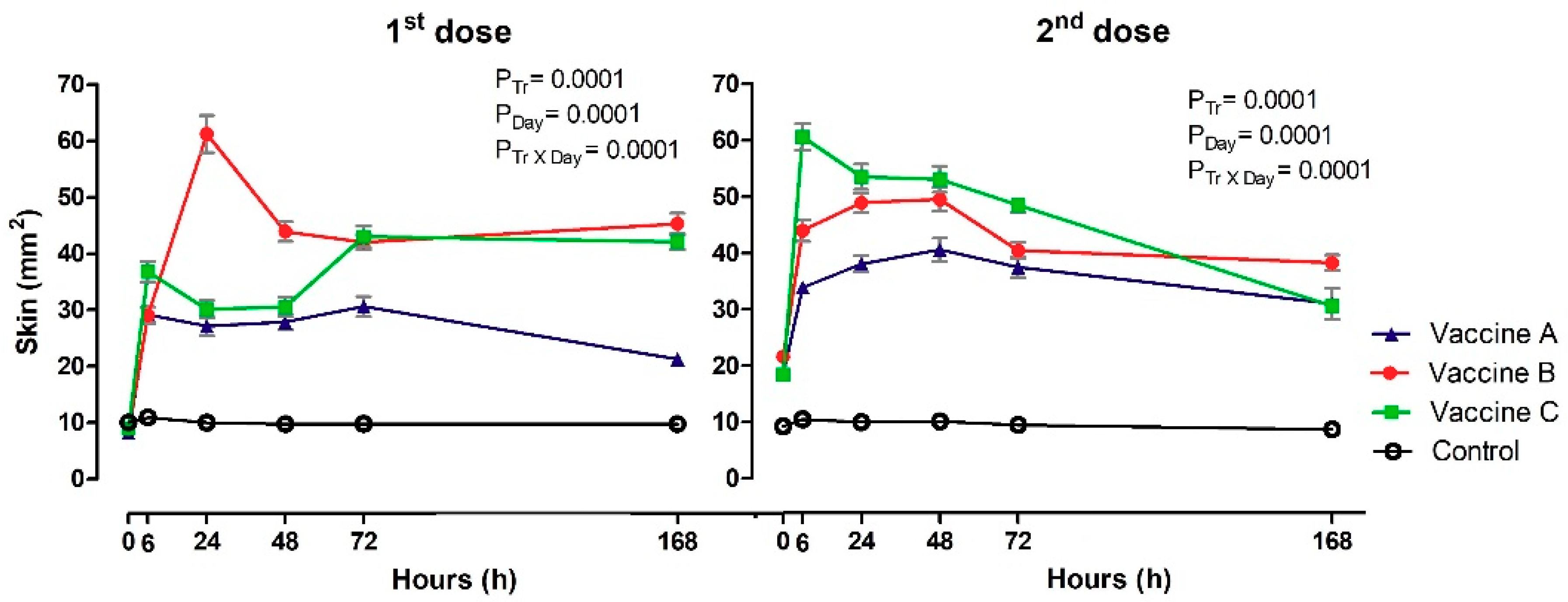
3.1. Local Vaccine Reactions
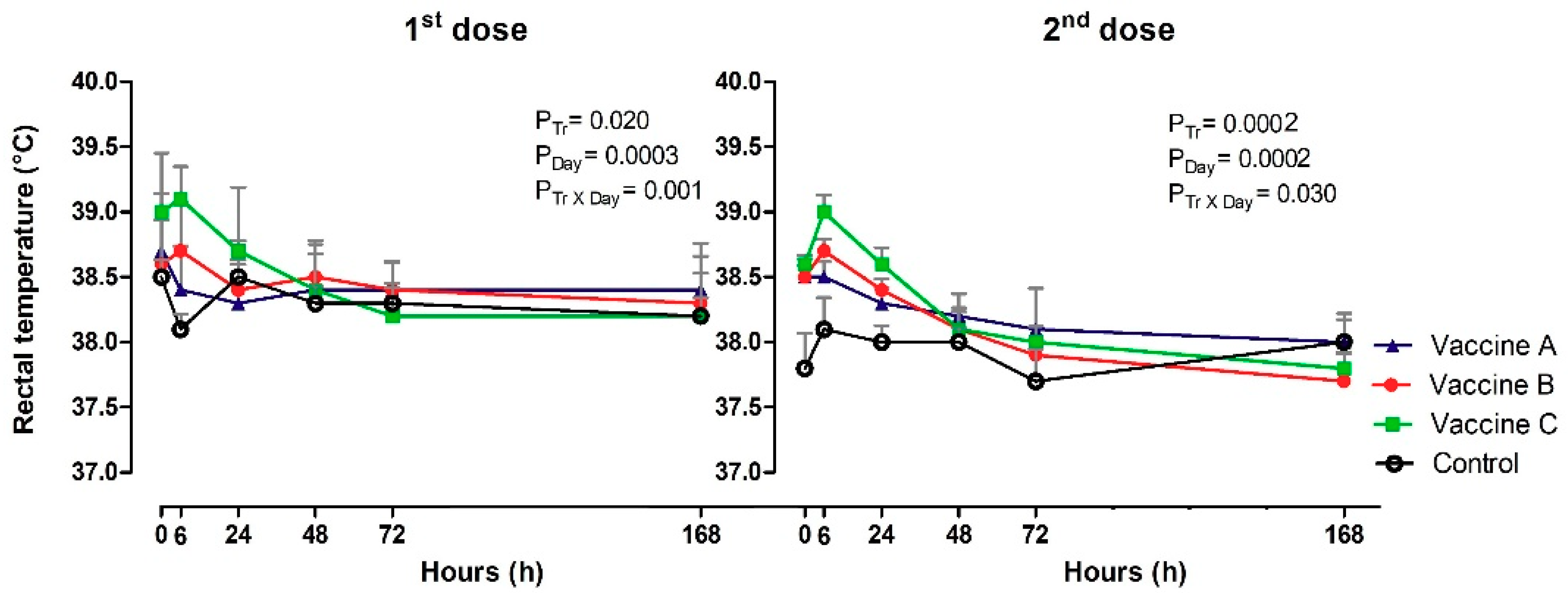
3.2. Systemic Vaccine Reactions
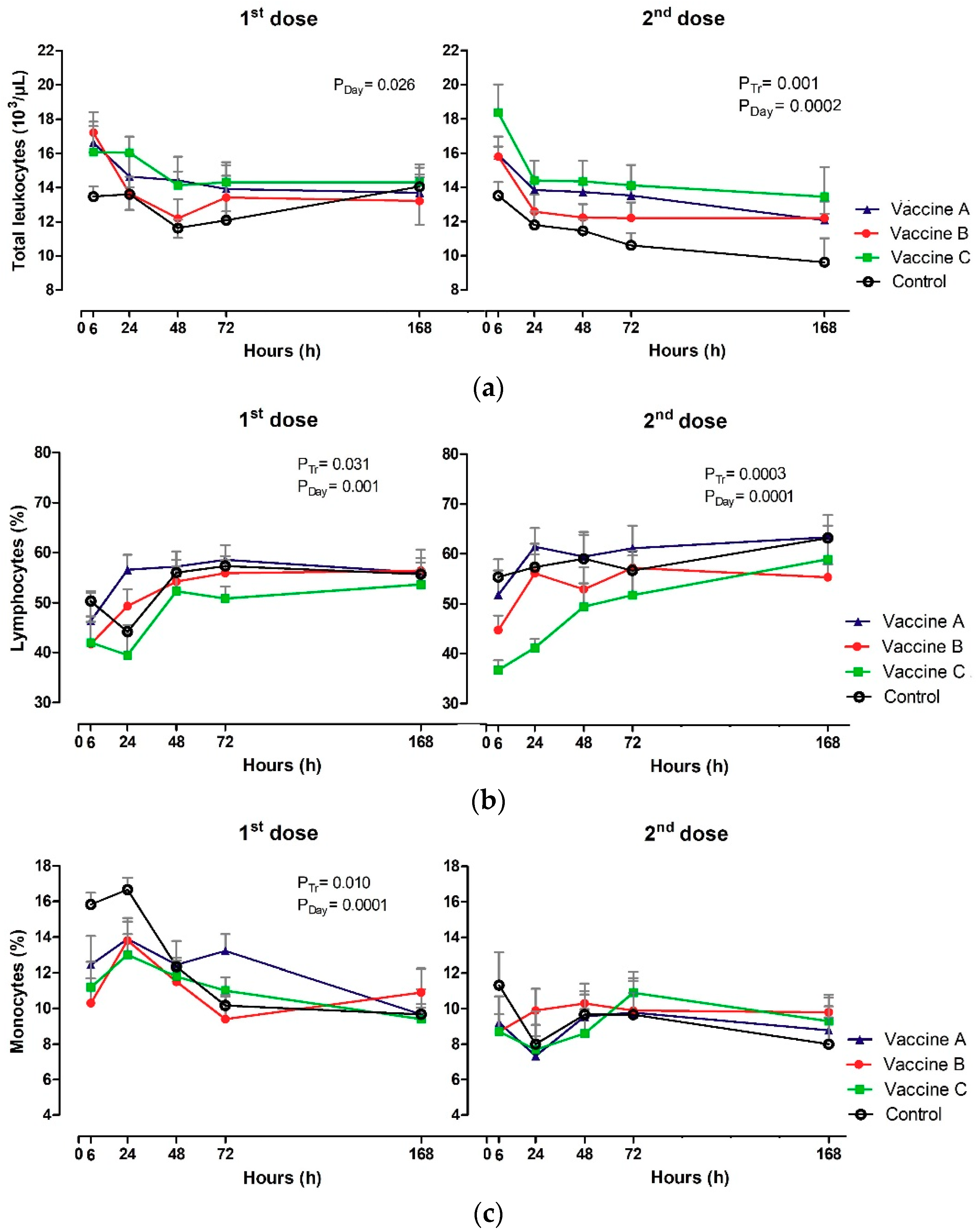
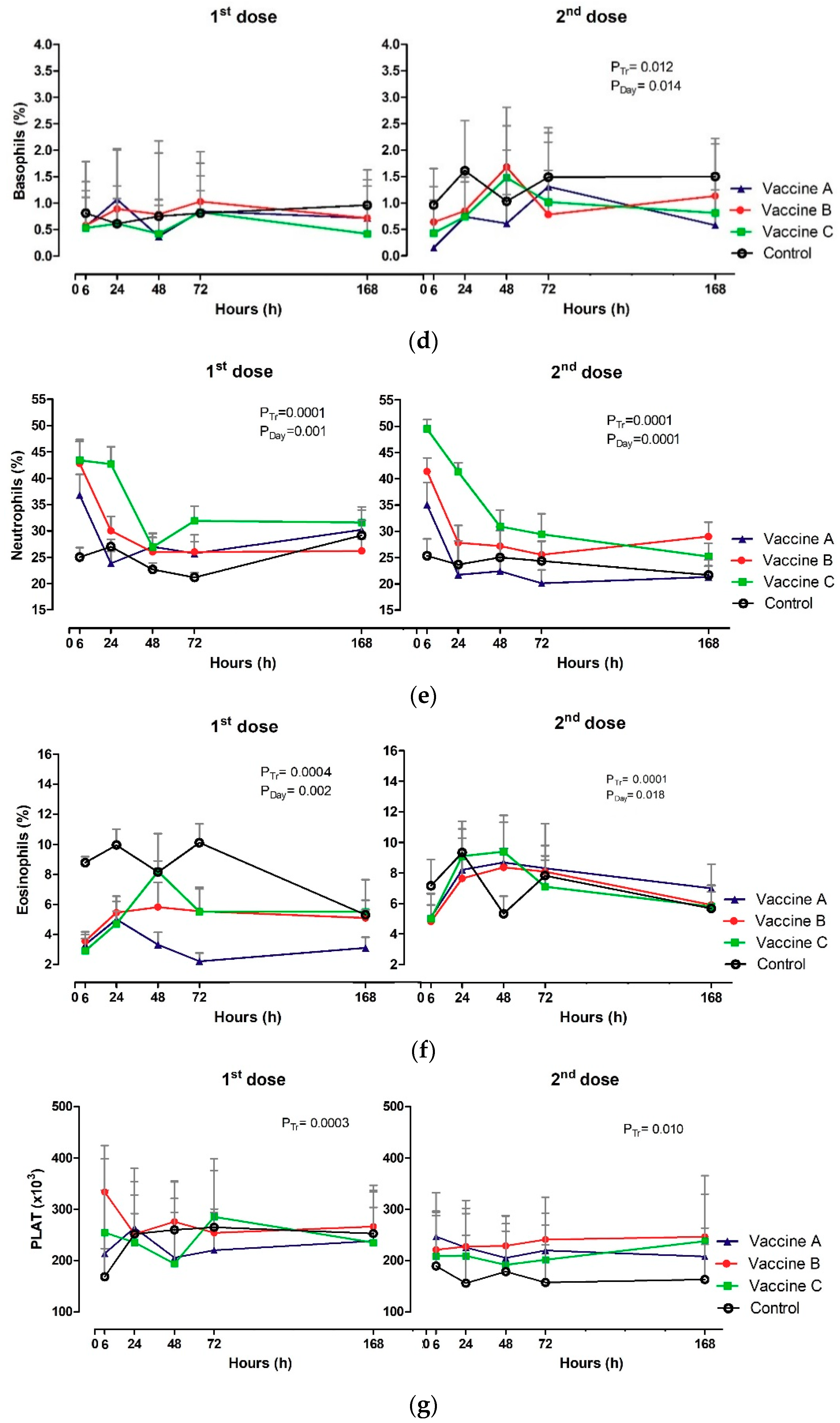
3.3. White Blood Count (WBC)
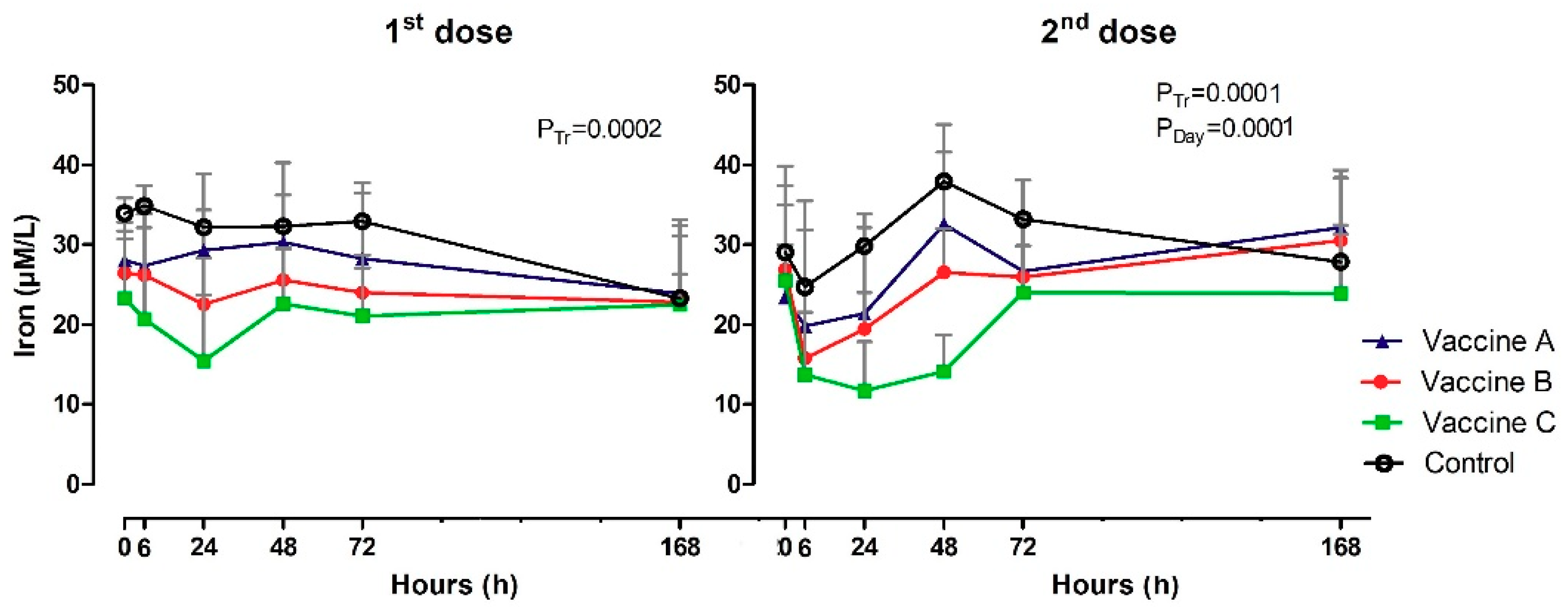
3.4. Serum Iron Levels
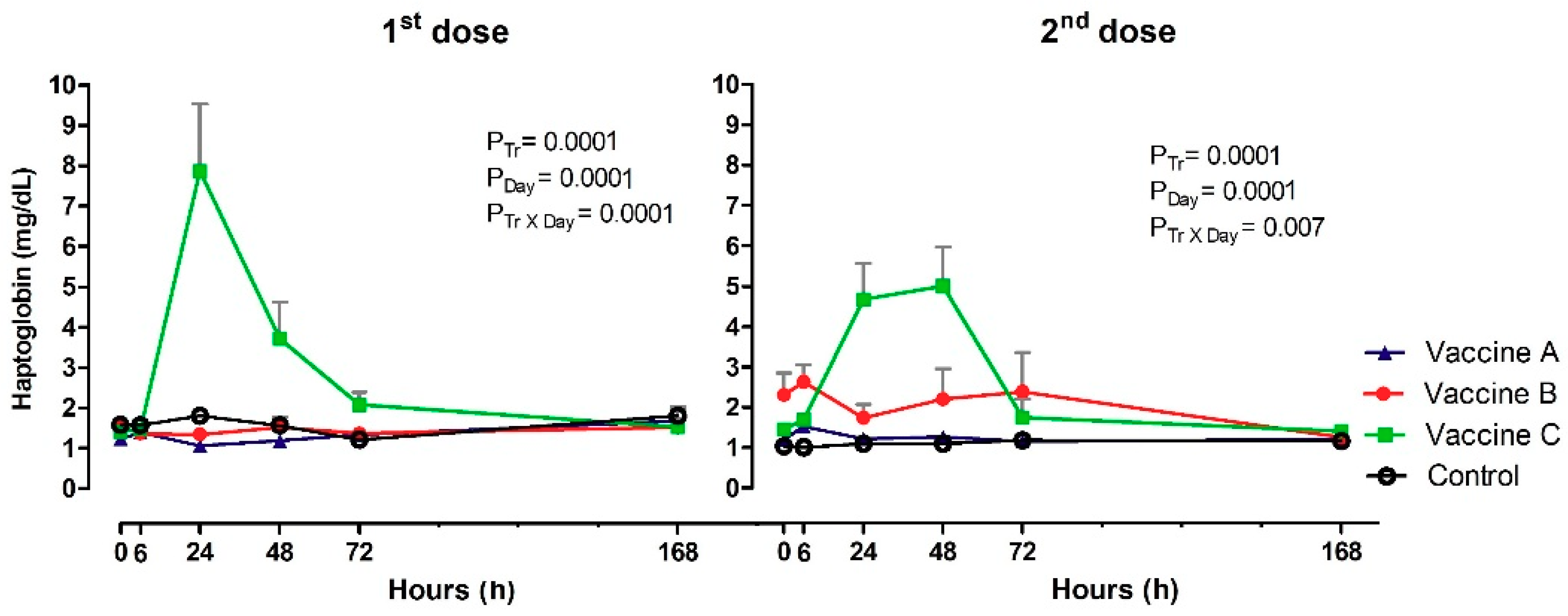
3.5. Haptoglobin Inflammation
3.6. BVDV and BHV-1 Antibody Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix
| Variable | First Dose | Second Dose | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | X2 | Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | X2 | ||
| Pain | 0 h 1 | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - |
| 6 h | 7 (78%) | 7 (70%) | 7 (70%) | 0 (0%) | 0.012 | 8 (89%) | 6 (60%) | 9 (90%) | 0 (0%) | 0.001 | |
| 24 h | 5 (56%) | 7 (70%) | 6 (60%) | 0 (0%) | 0.044 | 6 (67%) | 5 (50%) | 8 (80%) | 0 (0%) | 0.015 | |
| 48 h | 5 (56%) | 5 (50%) | 3 (30%) | 0 (0%) | 0.123 | 7 (78%) | 7 (70%) | 9 (90%) | 0 (0%) | 0.002 | |
| 72 h | 5 (56%) | 8 (80%) | 3 (30%) | 0 (0%) | 0.013 | 3 (33%) | 4 (40%) | 5 (50%) | 0 (0%) | 0.224 | |
| 168 h | 4 (44%) | 7 (70%) | 3 (30%) | 0 (0%) | 0.041 | 1 (11%) | 5 (50%) | 1 (10%) | 0 (0%) | 0.042 | |
| Cochran’s | 0.004 | 0.000 | 0.002 | - | 0.001 | 0.001 | 0.000 | - | |||
| Heat 6 | 0 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - |
| 6 h | 4 (44%) | 4 (40%) | 8 (80%) | 0 (0%) | 0.019 | 4 (44%) | 5 (50%) | 7 (70%) | 0 (0%) | 0.057 | |
| 24 h | 2 (22%) | 7 (70%) | 9 (90%) | 0 (0%) | 0.001 | 6 (67%) | 5 (50%) | 5 (50%) | 0 (0%) | 0.079 | |
| 48 h | 6 (67%) | 7 (70%) | 5 (50%) | 0 (0%) | 0.035 | 2 (22%) | 4 (40%) | 6 (60%) | 0 (0%) | 0.079 | |
| 72 h | 3 (33%) | 8 (80%) | 6 (60%) | 0 (0%) | 0.012 | 2 (22%) | 4 (40%) | 3 (30%) | 0 (0%) | 0.348 | |
| 168 h | 0 (0%) | 2 (20%) | 2 (20%) | 0 (0%) | 0.336 | 1 (11%) | 3 (30%) | 0 0%) | 0 (0%) | 0.148 | |
| Cochran’s | 0.006 | 0.001 | 0.000 | - | 0.004 | 0.025 | 0.000 | - | |||
| Variables | First Dose | Second Dose | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | X2 | Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | X2 | ||
| Cough | 0 h 1 | 0 (0%) | 0 (0%) | 1 (10%) | 0 (0%) | - | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - |
| 6 h | 0 (0%) | 0 (0%) | 1 (10%) | 0 (0%) | 0.616 | 1 (11%) | 2 (20%) | 1 (10%) | 0 (0%) | 0.677 | |
| 24 h | 0 (0%) | 1 (10%) | 2 (20%) | 0 (0%) | 0.377 | 1 (11%) | 2 (20%) | 3 (30%) | 0 (0%) | 0.441 | |
| 48 h | 2 (22%) | 0 (0%) | 2 (20%) | 0 (0%) | 0.133 | 1 (11%) | 4 (40%) | 1 (10%) | 1 (17%) | 0.307 | |
| 72 h | 0 (0%) | 3 (30%) | 1 (10%) | 1 (17%) | 0.117 | 1 (11%) | 4 (40%) | 1 (10%) | 0 (0%) | 0.199 | |
| 168 h | 1 (11%) | 2 (20%) | 1 (10%) | 1 (17%) | 0.915 | 1 (11%) | 2 (20%) | 3 (30%) | 0 (0%) | 0.774 | |
| Cochran’s | 0.180 | 0.014 | 0.940 | 0.416 | 0.742 | 0.193 | 0.139 | 0.549 | |||
| Nasal discharge | 0 h | 3 33%) | 5 (50%) | 6 (60%) | 1 (17%) | 0.331 | 4 (44%) | 5 (50%) | 6 (60%) | 0 (0%) | 0.116 |
| 6 h | 4 (44%) | 7 (70%) | 6 (60%) | 1 (17%) | 0.124 | 7 (78%) | 5 (50%) | 9 (90%) | 3 (50%) | 0.169 | |
| 24 h | 5 (55%) | 6 (60%) | 6 (60%) | 2 (33%) | 0.722 | 5 (56%) | 8 (80%) | 7 (70%) | 3 (50%) | 0.561 | |
| 48 h | 5 (55%) | 6 (60%) | 7 (70%) | 2 (33%) | 0.551 | 5 (56%) | 9 (90%) | 10 (100%) | 5 (83%) | 0.067 | |
| 72 h | 3 (33%) | 5 (50%) | 6 (60%) | 1 (17%) | 0.331 | 6 (67%) | 10 (100%) | 10 (100%) | 4 (67%) | 0.051 | |
| 168 h | 6 (66%) | 6 (60%) | 7 (70%) | 3 (50%) | 0.865 | 5 (56%) | 7 (70%) | 9 (90%) | 2 (33%) | 0.116 | |
| Cochran’s | 0.539 | 0.920 | 0.974 | 0.709 | 0.335 | 0.009 | 0.052 | 0.044 | |||
| Ocular discharge | 0 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 0 (0%) | 4 (40%) | 1 (10%) | 1 (17%) | 0.117 |
| 6 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 2 (22%) | 4 (40%) | 2 (20%) | 1 (17%) | 0.742 | |
| 24 h | 3 (33%) | 2 (20%) | 0 (0%) | 1 (17%) | 0.116 | 2 (22%) | 5 (50%) | 5 (50%) | 2 (33%) | 0.546 | |
| 48 h | 2 (22%) | 2 (20%) | 3 (30%) | 1 (17%) | 0.926 | 4 (44%) | 8 (80%) | 8 (80%) | 3 (50%) | 0.233 | |
| 72 h | 2 (22%) | 0 (0%) | 2 (20%) | 1 (17%) | 0.334 | 4 (44%) | 6 (60%) | 6 (60%) | 2 (33%) | 0.246 | |
| 168 h | 1 (11%) | 6 (60%) | 4 (40%) | 2 (33%) | 0.178 | 3 (33%) | 4 (40%) | 3 (30%) | 1 (17%) | 0.808 | |
| Cochran’s | 0.079 | 0.002 | 0.018 | 0.543 | 0.218 | 0.155 | 0.014 | 0.649 | |||
| Rectal temperature | 0 h | 4 (44%) | 1 (10%) | 7 (70%) | 0 (0%) | 0.462 | 3 (33%) | 1 (10%) | 3 (30%) | 0 (0%) | 0.152 |
| 6 h | 2 (22%) | 5 (50%) | 4 (40%) | 0 (0%) | 0.151 | 1 (11%) | 4 (40%) | 6 (60%) | 0 (0%) | 0.462 | |
| 24 h | 0 (0%) | 0 (0%) | 1 (10%) | 1 (17%) | 0.151 | 0 (0%) | 0 (0%) | 4 (40%) | 0 (0%) | 0.152 | |
| 48 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | |
| 72 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | |
| 168 h | 1 (11%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | |
| Cochran’s | 0.569 | 0.344 | 0.278 | 0.444 | 0.101 | 0.189 | 0.152 | - | |||
| Fecal consistency | 0 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - |
| 6 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | 1 (11%) | 0 (0%) | 1 (10%) | 0 (0%) | 0.677 | |
| 24 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0.462 | 0 (0%) | 1 (10%) | 0 (0%) | 0 (0%) | 0.416 | |
| 48 h | 3 (33%) | 0 (0%) | 0 (0%) | 2 (33%) | 0.190 | 1 (11%) | 0 (0%) | 0 (0%) | 1 (17%) | 0.661 | |
| 72 h | 2 (22%) | 1 (10%) | 0 (0%) | 1 (17%) | 0.211 | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | |
| 168 h | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0.396 | 2 (22%) | 0 (0%) | 0 (0%) | 0 (0%) | 0.876 | |
| Cochran’s | 0.093 | 0.594 | - | 0.221 | 0.127 | 0.156 | 0.113 | 0.100 | |||
| Sum of health score | 0 h | 2 (22%) | 0 (0%) | 1 (10%) | 0 (0%) | 0.300 | 1 (11%) | 3 (30%) | 1 (10%) | 0 (0%) | 0.356 |
| 6 h | 1 (11%) | 1 (10%) | 2 (20%) | 0 (0%) | 0.677 | 1 (11%) | 3 (30%) | 5 (50%) | 0 (0%) | 0.990 | |
| 24 h | 1 (11%) | 1 (10%) | 3 (30%) | 0 (0%) | 0.356 | 1 (11%) | 3 (30%) | 4 (40%) | 0 (0%) | 0.218 | |
| 48 h | 4 (44%) | 1 (10%) | 2 (20%) | 1 (17%) | 0.116 | 1 (11%) | 6 (60%) | 6 (60%) | 2 (33%) | 0.024 | |
| 72 h | 2 (22%) | 3 (30%) | 1 (10%) | 1 (17%) | 0.725 | 3 (33%) | 5 (50%) | 1 (10%) | 0 (0%) | 0.081 | |
| 168 h | 1 (11%) | 3 (30%) | 4 (40%) | 1 (17%) | 0.489 | 2 (22%) | 3 (30%) | 3 (30%) | 0 (0%) | 0.501 | |
| Cochran’s | 0.472 | 0.119 | 0.472 | 0.629 | 0.416 | 0.501 | 0.044 | 0.044 | |||
| Variables | First Dose | Second Dose | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | ||
| WBC 6(103/µL) | 6 h 1 | 16.63 | 17.22 a | 16.07 | 13.47 | 15.89 A,b,a | 15.80 A,B,a | 18.36 A,a | 13.50 B,a |
| 24 h | 14.64 | 13.66 b | 16.04 | 13.60 | 13.84 a | 12.58 b | 14.40 b | 11.80 a,b | |
| 48 h | 14.43 | 12.19 b | 14.13 | 11.63 | 13.73 a | 12.23 b | 14.36 b | 11.45 a,b | |
| 72 h | 13.92 | 13.41 b | 14.30 | 12.08 | 13.51 a | 12.21 b | 14.11 b | 10.60 a,b | |
| 168 h | 13.70 | 13.21 b | 14.30 | 14.05 | 12.07 A,B,b | 12.20 A,B,b | 13.45 A,b | 9.62 B,b | |
| LYMPH 7(103/µL) | 6 h | 7.76 | 7.01 | 6.58 | 6.98 | 8.69 | 6.99 | 6.73 | 7.42 |
| 24 h | 8.43 | 6.91 | 6.33 | 6.13 | 8.65 | 7.01 | 5.96 | 6.65 | |
| 48 h | 8.25 | 6.59 | 7.39 | 6.48 | 8.40 | 6.51 | 7.08 | 6.71 | |
| 72 h | 8.09 | 7.37 | 7.25 | 6.82 | 8.45 | 6.96 | 7.22 | 5.98 | |
| 168 h | 8.11 | 7.57 | 7.67 | 7.45 | 7.78 | 6.71 | 8.19 | 5.93 | |
| MONO 8(103/µL) | 6 h | 2.11 a | 1.80 | 1.72 a,b | 2.06 a | 1.40 | 1.32 | 1.52 | 1.58 |
| 24 h | 2.06 a | 1.81 | 1.99 a | 2.22 a | 1.00 | 1.21 | 1.14 | 0.97 | |
| 48 h | 1.86 a,b | 1.44 | 1.71 a,b | 1.43 b | 1.31 | 1.23 | 1.25 | 1.09 | |
| 72 h | 1.85 a,b | 1.35 | 1.59 a | 1.23 b | 1.35 | 1.24 | 1.51 | 1.05 | |
| 168 h | 1.31 b | 1.38 | 1.24 b | 1.31 b | 1.02 | 1.17 | 1.33 | 0.76 | |
| BAS 9(103/µL) | 6 h | 0.68 A,B | 0.68 A,B | 0.41 B | 1.19 A | 0.73 | 0.90 | 0.60 | 1.11 |
| 24 h | 0.86 | 0.94 | 0.84 | 1.36 | 1.01 | 1.10 | 1.28 | 1.27 | |
| 48 h | 0.53 | 0.74 | 0.98 | 1.02 | 1.19 | 1.25 | 1.29 | 0.70 | |
| 72 h | 0.29 | 0.80 | 0.65 | 0.99 | 1.00 | 1.08 | 1.0 | 0.89 | |
| 168 h | 0.46 | 0.70 | 0.63 | 0.69 | 0.87 | 0.68 | 0.89 | 0.65 | |
| NEUTR 10(103/µL) | 6 h | 5.80 A,B,a | 7.38 A,a | 7.28 A,a,b | 3.26 B,a,b | 5.17 B,C,a | 6.54 B,a | 9.10 A,a | 3.43 C,a,b |
| 24 h | 3.21 B,b | 3.80 B,b | 6.92 A,b | 3.60 B,a,b | 2.92 B,b | 3.50 B,b | 5.83 A,b,c | 2.84 B,a,b | |
| 48 h | 3.70 A,b | 3.16 A,b | 3.87 A,c | 2.63 A,b | 2.91 A,b | 3.20 A,b | 4.34 A,c | 2.94 A,a,b | |
| 72 h | 3.54 A,B,b | 3.47 A,B,b | 4.68 A,c | 2.54 B,b | 2.63 B,b | 3.16 A,B,b | 4.17 A,c | 2.62 B,b | |
| 168 h | 3.68 A,b | 3.39 A,b | 4.66 A,c | 4.60 A,b | 2.49 A,B,b | 3.70 A,B,b | 3.92 A,c | 2.16 B,b | |
| EOS 11 (103/µL) | 6 h | 0.54 B,a,b,c | 0.60 B | 0.42 B,b | 1.08 A,a,b | 0.75 a,b,c | 0.78 | 0.96 a,b | 0.96 a,b |
| 24 h | 0.72 B,a,b,c | 0.78 B | 0.74 B,a,b | 1.40 A,a | 1.04 a | 0.98 | 1.39 a | 1.12 a,b | |
| 48 h | 0.45 b,c | 0.65 | 1.10 a,b | 1.00 a,b | 1.08 a | 0.99 | 1.44 a | 0.60 b | |
| 72 h | 0.29 B,c | 0.73 A,B | 0.70 B,a,b | 1.40 A,a | 0.98 a,b | 0.99 | 1.06 a,b | 0.78 a,b | |
| 168 h | 0.40 b,c | 0.59 | 0.68 a,b | 0.57 b | 0.78 a,b,c | 0.68 | 0.81 a,b | 0.60 b | |
| PLAT 12(×103/µL) | 6 h | 214.33 A,B | 333.30 A | 254.20 A,B | 168.17 B | 246.56 | 221.00 | 208.80 | 188.00 |
| 24 h | 263.67 | 251.60 | 235.20 | 251.33 | 225.33 | 227.30 | 209.10 | 155.67 | |
| 48 h | 205.67 | 275.60 | 193.70 | 259.67 | 204.89 | 228.60 | 191.50 | 178.33 | |
| 72 h | 220.00 | 253.70 | 285.00 | 264.67 | 219.67 A,B | 241.00 A | 201.60 A,B | 157.17 B | |
| 168 h | 238.11 | 266.00 | 235.00 | 252.50 | 207.89 | 246.00 | 237.70 | 163.00 | |
| Iron (µM/L) | 0 h | 28 A | 26 A,B | 23 B | 32 | 23 A,b | 27 A,a,b | 25 A,a | 29 A,a,b |
| 6 h | 27 A,B | 26 B | 21 B | 35 | 20 A,B,b | 16 B,c | 14 B,b | 25 A,b | |
| 24 h | 29 A,B | 23 B | 15 C | 32 | 21 A,B,b | 19 B,b,c | 12 C,b | 30 A,a,b | |
| 48 h | 30 A,B | 26 A,B | 23 B | 32 | 33 A,b,a | 27 B,a,b | 14 C,b | 38 A,a | |
| 72 h | 28 A,B | 24 B,C | 21 C | 33 | 27 A,B,a,b | 26 A,B,b | 24 B,a | 33 A,a,b | |
| Variables | First Dose | Second Dose | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | Vaccine A 2 | Vaccine B 3 | Vaccine C 4 | Control 5 | ||
| LYMPH 6(%) | 6 h1 | 46.33 a | 41.70 a | 42.00 a,b | 50.33 a,b,c | 51.78 A,b,a | 44.70 B,C,a | 36.70 C,a | 55.33 A |
| 24 h | 56.56 A,a,b | 49.30 B,a,b | 39.50 C,a | 44.17 B,C,a | 61.44 A,a,b | 56.10 A,b,c | 41.20 B,a,b | 57.33 A | |
| 48 h | 57.22 a,b | 54.20 b | 52.30 c | 56.00 b,c | 59.44 a,b | 52.90 a,b,c | 49.40 b,c | 59.00 | |
| 72 h | 58.56 b | 55.90 b | 50.80 b,c | 57.33 b,c | 61.11 a,b | 57.20 c | 51.70 c | 56.67 | |
| 168 h | 56.00 a,b | 56.40 b | 53.60 c | 55.67 a,b | 63.33 b | 55.30 c | 58.90 c | 63.17 | |
| MONO 7(%) | 6 h | 12.44 A,B,a,b | 10.30 A,a | 11.20 A,a,b | 15.83 C,a,b | 9.22 | 8.70 | 8.70 | 11.33 |
| 24 h | 13.89 A,B,C,a | 13.80 A,B,C,b | 13.00 A,B,C,a | 16.67 C,b | 7.33 | 9.90 | 7.70 | 8.00 | |
| 48 h | 12.44 a,b | 11.50 a,b | 11.80 a,b | 12.33 a,c,d | 9.56 | 10.30 | 8.60 | 9.67 | |
| 72 h | 13.22 A,a,b | 9.40 B,a | 11.00 A,B,a,b | 10.17 A,B,c,d | 9.78 | 9.90 | 10.90 | 9.67 | |
| 168 h | 9.67 b | 10.90 a,b | 9.40 b | 9.67 d | 8.78 | 9.80 | 9.30 | 8.00 | |
| BAS 8(%) | 6 h | 0.56 a | 0.50 | 0.40 | 0.67 | 0.45 A,a | 0.50 A,B,a | 0.30 A,B,a | 0.83 B,ª |
| 24 h | 1.11 b | 0.90 | 0.50 | 0.61 | 0.67 A,a,b | 0.70 A,a | 0.60 A,a | 1.67 B,ª | |
| 48 h | 0.37 a | 0.80 | 0.30 | 0.83 | 0.44 A,a | 1.80 B,b | 1.50 B,b | 1.00 A,B,a | |
| 72 h | 0.67 a | 0.90 | 0.70 | 0.67 | 1.33 b | 0.70 a | 1.00 a,b | 1.50ª | |
| 168 h | 0.67 a | 0.60 | 0.30 | 0.96 | 0.44 A,a | 1.10 A,B,a,b | 0.70 A,B,a | 1.50 B,a | |
| NEUTR 9(%) | 6 h | 36.80 A,a | 42.81 A,a | 43.40 A,a | 25.00 B | 35.00 B,a,b | 41.36 A,B,a | 49.50 A,a | 25.33 C |
| 24 h | 23.80 B,c,d,e | 30.00 B,b | 42.70 A,a | 27.00 B | 21.70 B,c,d,e | 27.81 B,b | 41.30 A,a | 23.67 B | |
| 48 h | 27.00 a,b,c,d | 26.00 b | 26.90 b | 22.67 | 22.40 c,d,e | 27.18 b | 30.90 b | 25.00 A | |
| 72 h | 25.70 A,B,b,c | 26.00 A,B,b | 31.90 A,b | 21.17 B | 20.10 e | 25.54 b | 29.40 b | 24.33 | |
| 168 h | 30.20 a,b,c | 26.18 b | 31.60 b | 29.16 | 21.30 B,d,e | 29.00 A,b | 25.20 A,B,b | 21.66 B | |
| EOS 10(%) | 6 h | 3.30 B,b,c | 3.54 B,c | 2.90 B,b | 8.78 A,a,b,c | 5.10 B,a,b,c | 4.81 B,b,c | 5.00 B,a,b | 7.16 A,a,b,c |
| 24 h | 5.00 B,a,b,c | 5.45 B,a,b,c | 4.70 B,a,b | 9.96 A,a,b | 8.20 a,b | 7.64 a,b | 9.10 a | 9.33 a,b,c | |
| 48 h | 3.30 b,c | 5.82 a,b,c | 8.20 a | 8.16 a,b,c | 8.70 A,a | 8.36 A,a | 9.40 A,a | 5.33 B,A,c | |
| 72 h | 2.20 B,c | 5.55 B,a,b,c | 5.50 B,a,b | 10.12 A,a | 8.30 a | 8.09 a,b | 7.10 a,b | 7.83 a,b,c | |
| 168 h | 3.10 c | 5.09 a,b,c | 5.50 a,b | 5.30 A,c | 7.00 A,a,b,c | 5.91 B,a,b,c | 5.80 B,a,b | 5.67 B,b,c | |
References
- Gunn, G.J.; Stott, A.W.; Humphry, R.W. Modelling and costing BVD outbreaks in beef herds. Vet. J. 2004, 167, 143–149. [Google Scholar] [CrossRef]
- Richter, V.; Lebl, K.; Baumgartner, W.; Obritzhauser, W.; Käsbohrer, A.; Pinior, B. A systematic worldwide review of the direct monetary losses in cattle due to bovine viral diarrhea virus infection. Vet. J. 2017, 220, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Maresca, C.; Scoccia, E.; Dettori, A.; Felici, A.; Guarcini, R.; Petrini, S.; Quaglia, A.; Filipini, G. National surveillance plan for infectious bovine rhinotracheitis (IBR) in autochthonous Italian cattle breeds: Results of first year of activity. Vet. Microbiol. 2018, 219, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Newcomer, B.W.; Chamorro, M.F.; Walz, P.H. Vaccination of cattle against bovine viral diarrhea virus. Vet. Microbiol. 2017, 206, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Scharnböck, B.; Roch, F.F.; Richter, V.; Funke, C.; Firth, C.L.; Obritzhauser, W.; Käsbohrer, A.; Pinior, B. A meta-analysis of bovine viral diarrhea virus (BVDV) prevalence sin the global cattle population. Sci. Rep. 2018, 8, 14420. [Google Scholar] [CrossRef] [PubMed]
- Chase, C.C.L. Dairy Vaccines and Approaches to Best Results. In Proceedings of the 7° International Symposium—Leite Integral in, Curitiba, Brazil, 29 and 30 March 2017. [Google Scholar]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. The roles of TLRs, RLRs and NLRs in pathogen recognition. Int. Immunol. 2009, 21, 317–337. [Google Scholar] [CrossRef]
- Lee, S.R.; Pharr, G.T.; Boyd, B.L.; Pinchuk, L.M. Bovine viral diarrhea viroses modulate toll-like receptors, cytokines and co-stimulatory molecules genes expression in bovine peripheral blood monocytes. Comp. Immunol. Microbiol. Infect. Dis. 2008, 31, 403–418. [Google Scholar] [CrossRef]
- Marin, M.S.; Quintana, S.; Leunda, M.R.; Odeón, A.C.; Pérez, S.E. Toll-like receptor expression in the nervous system of bovine alpha-herpesvirus –infected calves. Res. Vet. Sci. 2014, 97, 422–429. [Google Scholar] [CrossRef]
- Kindt, T.J.; Goldsby, R.A.; Osborne, B.A.; Kuby, J. Kuby Immunology, 7th ed.; WH Freeman: New York, NY, USA, 2013; p. 169. [Google Scholar]
- Chase, C.C.L. Immunology Review/Refresher with Emphasis on Vaccinology. Am. Assoc. Bov. Pract. 2007, 40, 4–9. [Google Scholar]
- Catron, D.M.; Itano, A.A.; Pape, K.A.; Mueller, D.L.; Jenkins, M.K. Visualizing the first 50hr of the primary immune response to a soluble antigen. Immunity. 2004, 21, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Roark, C.L.; Simonian, P.L.; Fontenot, A.P.; Born, W.K.; O’brien, R.L. γδTcells: An important source of IL-17. Curr. Opin. Immunol. 2008, 20, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Chase, C.C.L.; Daniels, C.; Garcia, R. Needle-free injection technology in swine: Progress to ward vaccine efficacy and pork quality. J. Swine Health Prod. 2008, 16, 254–261. [Google Scholar]
- Janeway, C.A.; Travers, P.; Walport, M.; Shlomchik, M.J. Immunobiology: Innate Immunity, 5th ed.; Garland Science: New York, NY, USA, 2001. [Google Scholar]
- Lippolis, K.D.; Cooke, R.F.; Schubach, K.M.; Marques, R.S.; Bohnert, D.W. Effects of intravenous lipopolysaccharide administration on feed intake, ruminal forage degradability, and liquid parameters and physiological responses in beef cattle. J. Dairy Sci. 2017, 95, 2859–2870. [Google Scholar] [CrossRef]
- Martinod, S. Risk assessment related to veterinary biologicals: Side-effects in target animals. Rev. Sci. Tech. Off. Int. Epiz. 1995, 14, 979–989. [Google Scholar] [CrossRef]
- Levy, J.H.; Roizen, M.F.; Morris, J.M. Anaphylactic and anaphylactoid reactions. A review. Spine 1986, 11, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Ridpath, J.F.; Dominowski, P.; Mannan, R.; Yancey, R., Jr.; Jackson, J.A.; Taylor, L.; Mediratta, S.; Eversole, R.; Mackenzie, C.D.; Neill, J.D. Evaluation of three experimental bovine viral diarrhea virus killed vaccines adjuvanted with combinations of QuilA cholesterol and dimethyl dioctadecyl ammonium (DDA) bromide. Vet. Res. Commun. 2010, 34, 691–702. [Google Scholar] [CrossRef][Green Version]
- Burny, W.; Callegaro, A.; Bechtold, V.; Clement, F.; Delhaye, S.; Fissette, L.; Janssens, M.; Leroux-Roels, G.; Marchant, A.; vanden Berg, R.A.; et al. Different Adjuvants Induce Common Innate Pathways that Are Associated with Enhanced Adaptive Responses againsta Model Antigenin Humans. Front. Immunol. 2017, 8, 943. [Google Scholar] [CrossRef]
- Basqueira, N.S.; Martin, C.C.; Costa, J.F.R.; Okuda, L.H.; Pituco, M.E.; Batista, C.F.; DellaLibera, A.M.M.P.; Gomes, V. Bovine Respiratory Disease (BRD) Complexas a Signal for Bovine Viral Diarrhea Virus (BVDV) Presence in the Herd. Acta Sci. Vet. 2017, 45, 1434. [Google Scholar]
- McGuirk, S.M. Disease management of dairy calves and heifers. Vet. Clin. N. Am. Food Anim. Pract. 2008, 24, 139–153. [Google Scholar] [CrossRef]
- Jones, G.E.; Mould, D.L. Adaptation of the guaiacol (peroxidase) test for haptoglobins to a microtitration plate system. Res. Vet. Sci. 1984, 37, 87–92. [Google Scholar] [CrossRef]
- Bastos, B.L.; Loureiro, D.; Raynal, J.T.; Guedes, M.T.; Vale, V.L.; Moura-Costa, L.F.; Guimarães, J.E.; Azevedo, V.; Portela, R.W.; Meyer, R. Association between haptoglobina and IgM levels and the clinical progression of caseous lymphadenitis in sheep. BMC Vet. Res. 2013, 13, 254. [Google Scholar] [CrossRef]
- OIE-World Organization for Animal Health. Bovine viral diarrhea. In Manual of Diagnostic Tests and Vaccines for Terrestrial Animals; Office international des epizooties: Paris, France, 2015; Chapter 2.4.7; pp. 1–22. [Google Scholar]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Ryman, V.E.; Packiriswamy, N.; Sordillo, L.M. Role of endothelial cells in bovine mammary gland health and disease. Anim. Health Res. Rev. 2015, 16, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Robins, S.L.; Cotran, R.S. Tecido de renovação e reparação: Regeneração, cicatrização e fibrose. In Patologia: Bases Patológicas Das Doenças, 7th ed.; Fausto, N., Kumar, R.V., Abbas, A.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; p. 91. [Google Scholar]
- Leal, P.V.; Pupin, R.C.; Santos, A.C.; Faccin, T.C.; Surdi, E.; Leal, C.R.B.; Brumatti, R.C.; Lemos, R.A.A. Estimativas de perdas econômicas causadas por reação granulomatosa local após uso de vacina oleosa contra febre aftosa em bovinos de Mato Grosso do Sul. Pesq. Vet. Bras. 2014, 34, 738–742. [Google Scholar] [CrossRef]
- Tisoncik, J.R.; Korth, M.J.; Simmons, C.P.; Farrar, J.; Martin, T.R.; Katze, M.G. Intotheeyeofthecytokinestorm. Microbiol. Mol. Biol. Rev. 2012, 76, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Rubenfeld, G.D.; Caldwell, E.; Peabody, E.; Weaver, J.; Martin, D.P.; Neff, M.; Stern, E.J.; Hudson, L.D. Incidence and outcomes of acute lung injury. N. Engl. J. Med. 2005, 353, 1685–1693. [Google Scholar] [CrossRef] [PubMed]
- Isobe, Y.; Kato, T.; Arita, M. Emerging roles of eosinophils and eosinophil-derived lipid mediators in the resolution of inflammation. Front. Immunol. 2012, 3, 270. [Google Scholar] [CrossRef]
- Tornquist, S.J.; Rigas, J. Interpretation of ruminant leukocyte. In Schalm’s Veterinary Hematology, 6nd ed.; Weiss, D.J., Wardrop, K.J., Eds.; Lea and Febiger: Philadelphia, PA, USA, 2010; pp. 307–312. [Google Scholar]
- Ganz, T.; Nemeth, E. Hepcidin and iron homeostasis. Biochim. Biophys. Acta 2012, 1823, 1434–1443. [Google Scholar] [CrossRef]
- Donovan, A.; Lima, C.A.; Pinkus, J.L.; Pinkus, G.S.; Zon, L.I.; Robine, S.; Andrews, N.C. The iron exporter ferroportinis essential for iron homeostasis. Cell Metab. 2005, 1, 191–200. [Google Scholar] [CrossRef]
- Antunes, S.A.; Canziani, M.E.F. Hepcidin: An important iron metabolism regulatorin chronic kidney disease. J. Bras. Nefrol. 2016, 38, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Murata, H.; Shimada, N.; Yoshioka, M. Current research on acute phase proteins in veterinary diagnosis: An overview. Vet. J. 2004, 168, 28–40. [Google Scholar] [CrossRef]
- Petersen, H.; Nielsen, J.P.; Heegaard, P.M. Application of acute phase protein measurements in veterinary clinical chemistry. Vet. Res. 2004, 35, 163–187. [Google Scholar] [CrossRef] [PubMed]
- Stokka, G.L.; Edwards, A.J.; Spire, M.F.; Brandt, R.T., Jr.; Smith, J.E. Inflammatory response to clostridial vaccines in feedlot cattle. J. Am. Vet. Med. Assoc. 1994, 204, 415–419. [Google Scholar] [PubMed]
- Rodrigues, M.C.; Cooke, R.F.; Marques, R.S.; Cappellozza, B.I.; Arispe, S.A.; Keisler, D.H.; Bohnert, D.W. Effects of vaccination against respiratory pathogens on feed intake, metabolic, and inflammatory responses in beef heifers. J. Anim. Sci. 2015, 93, 4443–4452. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.C.L.; Cooke, R.F.; Marques, R.S.; Fernandes, H.J.; Fernandes, C.E.; Stelato, R.; Franco, G.L.; Lemos, R.A.A. Effects of vaccination against foot and-mouth disease virus on reproductive performance of Bos indicus beef cows. J. Anim. Sci. 2016, 94, 401–405. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Zou, Y.; Hu, Z. Advances in aluminum hydroxide-based adjuvant research and its mechanism. Hum. Vaccin Immunother. 2015, 11, 477–488. [Google Scholar] [CrossRef]
- Vartak, A.; Sucheck, S.J. Recent Advances in Subunit Vaccine Carriers. Vaccines 2016, 4, 12. [Google Scholar] [CrossRef]
- Van Oirschot, J.T.; Kaashoek, M.J.; Rijsewijk, F.A. Advances in the development and evaluation of bovine herpesvirus 1 vaccines. Vet. Microbiol. 1996, 53, 43–54. [Google Scholar] [CrossRef]
- Carroll, J.A.; Forsberg, N.E. Influence of stress and nutrition on cattle immunity. Vet. Clin. N. Am. Food Anim. Pract. 2007, 23, 105–149. [Google Scholar] [CrossRef]
- Ortolani, E.L.; Sucupira, M.C.A. Deficiências de macro e micro elementos em gado de corte. In Bovinocultura de Corte, 1st ed.; FEALQ: Piracicaba, Brazil, 2010; Volume 1, pp. 351–437. [Google Scholar]
- Palomares, R.A.; Hurley, D.J.; Bittar, J.H.; Saliki, J.T.; Woolums, A.R.; Moliere, F.; Havenga, L.J.; Norton, N.A.; Clifton, S.J.; Sigmund, A.B.; et al. Effects of injectable trace minerals on humoral and cell-mediated immune responses to Bovine viral diarrhea virus, Bovine herpes virus1 and Bovinere spiratory syncytial virus following administration of a modified-live virus vaccine in dairy calves. Vet. Immunol. Immunopathol. 2016, 178, 88–98. [Google Scholar] [CrossRef] [PubMed]
| Groups | Adjuvant Composed | Strains | Others Antigens |
|---|---|---|---|
| Vaccine A (n = 9) | Aluminum hydroxide (Alhydrogel®) | Bovine viral diarrhea virus (BVDV)-1 (Singer) and BVDV-2 inactivated, strains from INTA 1 and CEVAN 2; bovine herpesvirus 1 (BHV)-1 (Los Angeles) inactivated | Campylobacter fetus; Campylobacter fetus veneralis; Leptospira interrogans pomona; Histophilus somni. |
| Vaccine B (n = 10) | Oil-in-water adjuvant (mineral oil-based) | BVDV-1 and BVDV 2 (inactivated); BHV-1 and BoHV-5 (inactivated) | Leptospirahardjo, L. icterohaemorrhagiae, L. Bratislava, L. Pomona and L. wolfi |
| Vaccine C (n = 10) | Amphigen and Quil A cholesterol and dimethyl dioctadecylammonium (DDA) bromide (QAD) adjuvant ® | BVDV-1 (5960) and BVDV-2 (53,637) inactivated; BHV-1 (RBL106) thermosensitive | Leptospira canicola, L. grippotyphosa, L. hardjo, L. icterohaemorrhagiae and L. pomona |
| Control (n = 6) | - | Saline Solution | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costa Baccili, C.; Martin, C.C.; Decaris, N.; Medici Madureira, K.; Chase, C.; Gomes, V. Effects of 3 Different Commercial Vaccines Formulations against BVDV and BHV-1 on the Inflammatory Response of Holstein Heifers. Vet. Sci. 2019, 6, 69. https://doi.org/10.3390/vetsci6030069
Costa Baccili C, Martin CC, Decaris N, Medici Madureira K, Chase C, Gomes V. Effects of 3 Different Commercial Vaccines Formulations against BVDV and BHV-1 on the Inflammatory Response of Holstein Heifers. Veterinary Sciences. 2019; 6(3):69. https://doi.org/10.3390/vetsci6030069
Chicago/Turabian StyleCosta Baccili, Camila, Camila Cecilia Martin, Nathália Decaris, Karina Medici Madureira, Christopher Chase, and Viviani Gomes. 2019. "Effects of 3 Different Commercial Vaccines Formulations against BVDV and BHV-1 on the Inflammatory Response of Holstein Heifers" Veterinary Sciences 6, no. 3: 69. https://doi.org/10.3390/vetsci6030069
APA StyleCosta Baccili, C., Martin, C. C., Decaris, N., Medici Madureira, K., Chase, C., & Gomes, V. (2019). Effects of 3 Different Commercial Vaccines Formulations against BVDV and BHV-1 on the Inflammatory Response of Holstein Heifers. Veterinary Sciences, 6(3), 69. https://doi.org/10.3390/vetsci6030069







