Application and Comparative Evaluation of Fluorescent Antibody, Immunohistochemistry and Reverse Transcription Polymerase Chain Reaction Tests for the Detection of Rabies Virus Antigen or Nucleic Acid in Brain Samples of Animals Suspected of Rabies in India
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
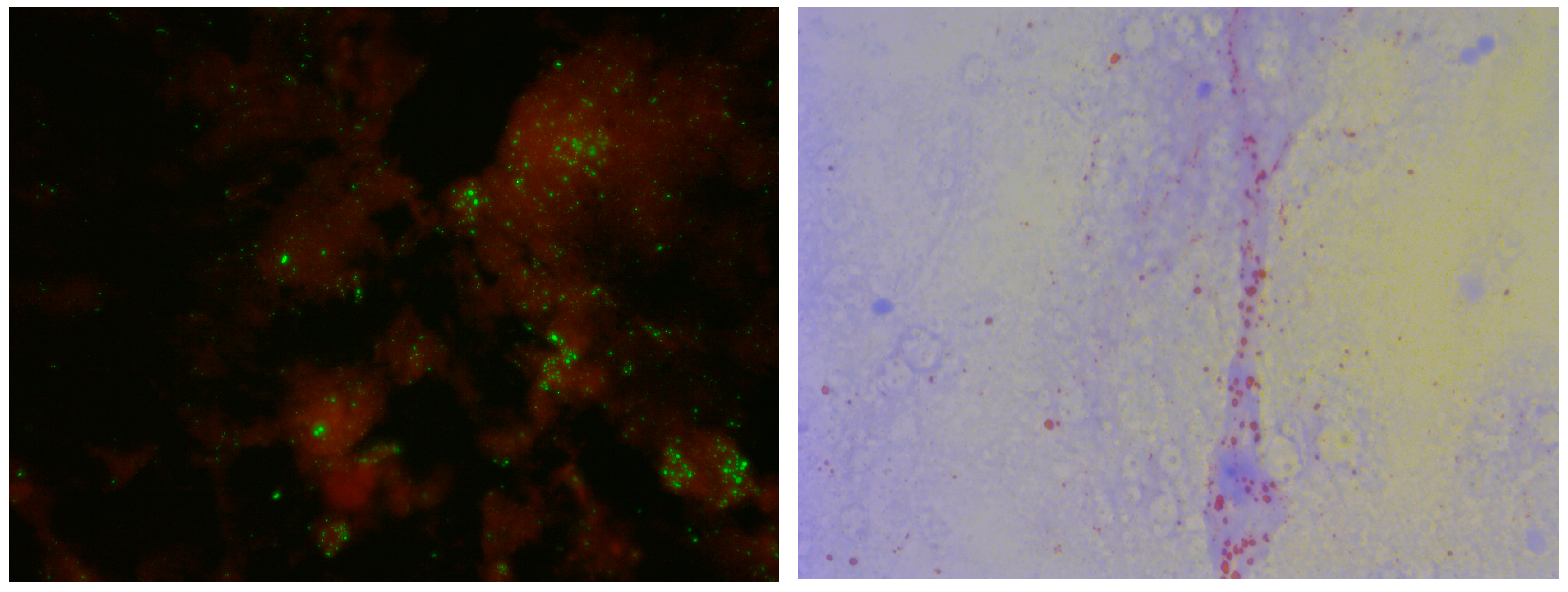
2.2. Direct Fluorescent Antibody (DFA) Test
2.3. Direct Rapid Immunohistochemistry Test (dRIT)
2.4. Cells and Viruses
2.5. Isolation of RNA from Brain Samples
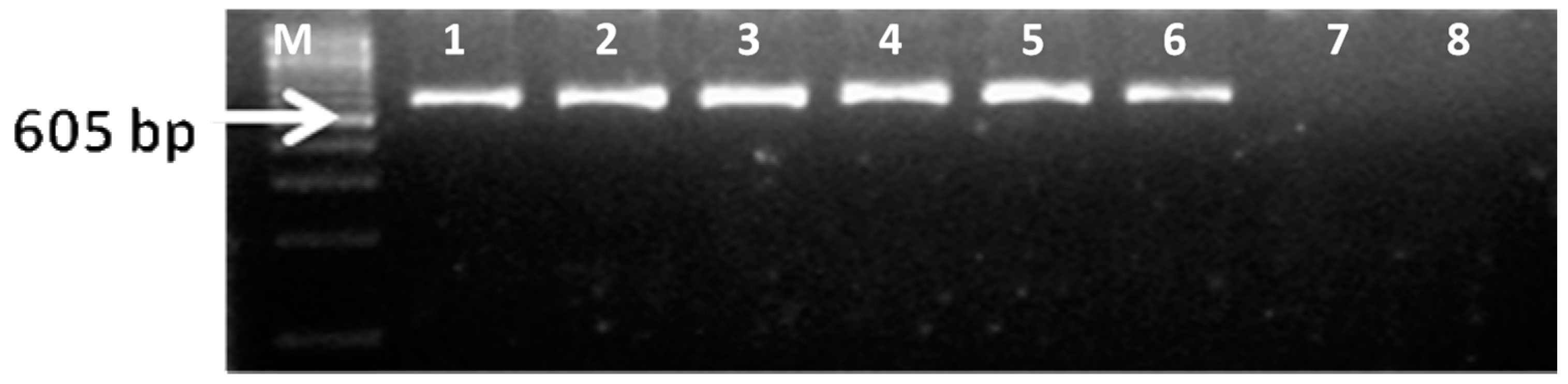
2.6. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anonymous. Take a Bite of Rabies. Available online: https://www.cdc.gov/features/rabies/index.html (accessed on 13 December 2017).
- Blancou, J. The control of rabies in Eurasia: Overview, history and background. Dev. Biol. 2008, 131, 3–15. [Google Scholar]
- Singer, A.; Kauhala, K.; Holmala, K.; Smith, G.C. Rabies in northeastern Europe—The threat from invasive raccoon dogs. J. Wildl. Dis. 2009, 45, 1121–1137. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.S. New aspects of rabies with emphasis on epidemiology, diagnosis, and prevention of the disease in the United States. Clin. Microbiol. Rev. 1996, 9, 166–176. [Google Scholar] [PubMed]
- Dacheux, L.; Wacharapluesadee, S.; Hemachudha, T.; Meslin, F.X.; Buchy, P.; Reynes, J.M.; Bourhy, H. More accurate insight into the incidence of human rabies in developing countries through validated laboratory techniques. PLoS NTD 2010, 4, e765. [Google Scholar] [CrossRef] [PubMed]
- Duong, V.; Tarantola, A.; Ong, S.; Mey, C.; Choeung, R.; Ly, S.; Bourhy, H.; Dussart, P.; Buchy, P. Laboratory diagnostics in dog-mediated rabies: An overview of performance and a proposed strategy for various settings. Int. J. Infect. Dis. 2016, 46, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Wacharapluesadee, S.; Hemachudha, T. Ante- and post-mortem diagnosis of rabies using nucleic acid amplification tests. Expert Rev. Mol. Diagn. 2010, 10, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Woldehiwet, Z. Clinical laboratory advances in the detection of rabies virus. Clin. Chim. Acta 2005, 351, 49–63. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO expert consultation on rabies. Second Report. World Health Organ. Tech. Rep. Ser. 2013, 931, 1–139. [Google Scholar]
- Dean, D.J.; Abelseth, M.K.; Atanasiu, P. The Fluorescent Antibody Test, 4th ed.; World Health Organization: Geneva, Switzerland, 1996. [Google Scholar]
- Durr, S.; Naissengar, S.; Mindekem, R.; Diguimbye, C.; Niezgoda, M.; Kuzmin, I.; Rupprecht, C.E.; Zinsstag, J. Rabies diagnosis for developing countries. PLoS NTD 2008, 2, e206. [Google Scholar] [CrossRef] [PubMed]
- Lembo, T.; Niezgoda, M.; Velasco-Villa, A.; Cleaveland, S.; Ernest, E.; Rupprecht, C.E. Evaluation of a direct, rapid immunohistochemical test for rabies diagnosis. Emerg. Infect. Dis. 2006, 12, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, K.N.; Isloor, S.; Veeresh, B.H.; Rathnamma, D.; Yathiraj, S.; Satyanarayana, M.L.; D’Souza, P.; Neelufer, M.S.; Sharada, R.; Rahman, S.A. Application of N-protein monoclonal antibody based direct fluorescent antibody assay (DFAT) and direct rapid immunohistochemistry test (dRIT) for detection of rabies virus in brain samples of animals in India. Commonwealth Vet. J. 2014, 30, 11–16. [Google Scholar]
- David, D.; Yakobson, B.; Rotenberg, D.; Dveres, N.; Davidson, E.; Stram, Y. Rabies virus detection by RT-PCR in decomposed naturally infected brains. Vet. Microbiol. 2002, 87, 111–118. [Google Scholar] [CrossRef]
- Picard-Meyer, E.; Bruyere, V.; Barrat, J.; Tissot, E.; Barrat, M.J.; Cliquet, F. Development of a hemi-nested RT-PCR method for the specific determination of European Bat Lyssavirus 1. Comparison with other rabies diagnostic methods. Vaccine 2004, 22, 1921–1929. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Protocol for Postmortem Diagnosis of Rabies in Animals by Direct Fluorescent Antibody Testing; Centres for Disease Control and Prevention: Atlanta, GA, USA. Available online: https://www.cdc.gov/rabies/pdf/rabiesdfaspv2.pdf (accessed on 12 January 2018).
- Niezgoda, M.; Rupprecht, C.E. Standard Operating Procedure for the Direct Rapid Immuohistochemistry Test for the Detection of Rabies Virus Antigen; Centers for Disease Control and Prevention, US Department of Health and Human Services: Atlanta, GA, USA, 2006.
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar]
- Chandrashekara, N.; Rathnamma, D.; Veeresh, B.H.; Yathiraj, S.; Satyanarayana, M.L.; Veeregowda, B.M. Application of Reverse Transcription Polymerase Chain Reaction (RT-PCR) for Diagnosis of Rabies in Animals and Its Comparative Evaluation with Direct Fluorescence Antibody Test (DFAT) and Direct Immunoihistochemistry Test (dRIT). In Proceedings of the Association for the Prevenstion of and Control of Rabies in India Conference, Kolkata, India, 6–8 July 2012; p. 22. [Google Scholar]
- Heaton, P.R.; Johnstone, P.; McElhinney, L.M.; Cowley, R.; O’Sullivan, E.; Whitby, J.E. Heminested PCR assay for detection of six genotypes of rabies and rabies-related viruses. J. Clim. Microbiol. 1997, 35, 2762–2766. [Google Scholar]
- Sudarshan, M.K. Assessing the burden of rabies in India: WHO sponsored national multi-centric rabies survey, 2003. Indian J. Community Med. 2005, 30, 100–101. [Google Scholar] [CrossRef]
- Knobel, D.L.; Cleaveland, S.; Coleman, P.G.; Fevre, E.M.; Meltzer, M.I.; Miranda, M.E.G.; Shaw, A.; Zinsstag, J.; Meslin, F.-X. Re-evaluating the burden or rabies in African and Asia. Bull. World Health Organ. 2005, 83, 360–368. [Google Scholar] [PubMed]
- Menezes, R. Rabies in India. CMAJ J. 2008, 178, 564–566. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, R.; Ichhpujani, R.L.; Madhusudana, S.N.; Hemachuda, T. Rabies in South and Southeast Asia. In Program and Abstracts of the WHO Expert Consultation on Rabies; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Mansfield, K.L.; Banyard, A.C.; Fooks, A.R.; Franka, R.; Isloor, S.; Rahman, A. Supporting rabies control in India. Vet. Rec. 2016, 179, 296–297. [Google Scholar] [CrossRef] [PubMed]
- World Organisation for Animal Health. Rabies. In OIE Manual of Diagnostic Tests and Vaccines; World Organization for Animal Health OIE: Paris, France, 2013. [Google Scholar]
- Clavijo, A.; Freire de Carvalho, M.H.; Orciari, L.A.; Velasco-Villa, A.; Ellison, J.A.; Greenberg, L.; Yager, P.A.; Green, D.B.; Vigilato, M.A.; Cosivi, O.; et al. An inter-laboratory proficiency testing exercise for rabies diagnosis in Latin America and the Caribbean. PLoS NTD 2017, 11, e0005427. [Google Scholar] [CrossRef] [PubMed]
- Barrat, J.; Barrat, M.J.; Muller, T.; Cliquet, F. European interlaboratory F.A.T. comparison test 2001. Rabies Bull. Eur. 2003, 27, 7–12. [Google Scholar]
- Robardet, E.; Andrieu, S.; Rasmussen, T.B.; Dobrostana, M.; Horton, D.L.; Hostnik, P.; Jaceviciene, I.; Juhasz, T.; Muller, T.; Mutinelli, F.; et al. Comparative assay of fluorescent antibody test results among twelve European national reference laboratories using various anti-rabies conjugates. J. Virol. Methods 2013, 191, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Robardet, E.; Picard-Meyer, E.; Andrieu, S.; Servat, A.; Cliquet, F. International interlaboratory trials on rabies diagnosis: An overview of results and variation in reference diagnosis techniques (fluorescent antibody test, rabies tissue culture infection test, mouse inoculation test) and molecular biology techniques. J. Virol. Methods 2011, 177, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, J.A.; Franke, M.A.; Davis, A.D. Rabies direct fluorescent antibody test does not inactivate rabies or eastern equine encephalitis viruses. J. Virol. Methods 2016, 234, 52–53. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Selden, D.; Fooks, A.R.; Banyard, A. Inactivation of rabies virus. J. Virol. Methods 2017, 243, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Hamir, A.N.; Moser, G.; Wampler, T.; Hattel, A.; Dietzschold, B.; Rupprecht, C.E. Use of a single anti-nucleocapsid monoclonal antibody to detect rabies antigen in formalin-fixed, paraffin-embedded tissues. Vet. Rec. 1996, 138, 114–115. [Google Scholar] [CrossRef] [PubMed]
- Stein, L.T.; Rech, R.R.; Harrison, L.; Brown, C.C. Immunohistochemical study of rabies virus within the central nervous system of domestic and wildlife species. Vet. Pathol. 2010, 47, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Madhusudana, S.N.; Subha, S.; Thankappan, U.; Ashwin, Y.B. Evaluation of a direct rapid immunohistochemical test (dRIT) for rapid diagnosis of rabies in animals and humans. Virol. Sin. 2012, 27, 299–302. [Google Scholar] [CrossRef] [PubMed]
- Faizee, N.; Hailat, N.Q.; Ababneh, M.M.; Hananeh, W.M.; Muhaidat, A. Pathological, immunological and molecular diagnosis of rabies in clinically suspected animals of different species using four detection techniques in Jordan. Transbound. Emerg. Dis. 2012, 59, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Hamir, A.N.; Moser, G. Immunoperoxidase test for rabies: Utility as a diagnostic test. J. Vet. Diagn. Investig. 1994, 6, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.; Zhang, F.; Zhang, S.; Hu, R. Restudy on effect of various physical and chemical factors on infectivity of a rabies virus isolate. Chin. J. Biol. 2011, 24, 45–47. [Google Scholar]
- Coetzer, A.; Sabeta, C.T.; Markotter, W.; Rupprecht, C.E.; Nel, L.H. Comparison of biotinylated monoclonal and polyclonal antibodies in an evaluation of a direct rapid immunohistochemical test for the routine diagnosis of rabies in southern Africa. PLoS NTD 2014, 8, e3189. [Google Scholar] [CrossRef] [PubMed]
- Rahmadane, I.; Certoma, A.F.; Peck, G.R.; Fitria, Y.; Payne, J.; Colling, A.; Shiell, B.J.; Beddome, G.; Wilson, S.; Yu, M.; et al. Development and validation of an immunoperoxidase antigen detection test for improved diagnosis of rabies in Indonesia. PLoS NTD 2017, 11, e0006079. [Google Scholar] [CrossRef] [PubMed]
- Aravindhbabu, R.P.; Manoharan, S.; Ramadass, P.; Chandran, N.D. Evaluation of RT-PCR assay for routine laboratory diagnosis of rabies in post-mortem brain samples from different species of animals. Indian J. Virol. 2012, 23, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Whitby, J.E.; Heaton, P.R.; Whitby, H.E.; O’Sullivan, E.; Johnstone, P. Rapid detection of rabies and rabies-related viruses by rt-pcr and enzyme-linked immunosorbent assay. J. Virol. Methods 1997, 69, 63–72. [Google Scholar] [CrossRef]
- Johnson, N.; Un, H.; Vos, A.; Aylan, O.; Fooks, A.R. Wildlife rabies in western turkey: The spread of rabies through the western provinces of turkey. Epidemiol. Infect. 2006, 134, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Beltran, F.J.; Dohmen, F.G.; Del Pietro, H.; Cisterna, D.M. Diagnosis and molecular typing of rabies virus in samples stored in inadequate conditions. J. Infect. Dev. Ctries. 2014, 8, 1016–1021. [Google Scholar] [CrossRef] [PubMed]
- Biswal, M.; Ratho, R.K.; Mishra, B. Usefulness of reverse transcriptase-polymerase chain reaction for detection of rabies in archival samples. Jpn. J. Infect. Dis. 2007, 60, 298–299. [Google Scholar] [PubMed]
- Crepin, P.; Audry, L.; Rotivel, Y.; Gacoin, A.; Caroff, C.; Bourhy, H. Intravitam diagnosis of human rabies by PCR using saliva and cerebrospinal fluid. J. Clin. Microbiol. 1998, 36, 1117–1121. [Google Scholar] [PubMed]
- Lopes, M.C.; Venditti, L.L.; Queiroz, L.H. Comparison between RT-PCR and the mouse inoculation test for detection of rabies virus in samples kept for long periods under different conditions. J. Virol. Methods 2010, 164, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.; Takaoka, N.; Brandao, P.; Carnieli, P., Jr.; Macedo, C.; Castilho, J.; Carrieri, M.L.; Kotait, I. Postmortem confirmation of human rabies source. Emerg. Infect. Dis. 2006, 12, 867–869. [Google Scholar] [CrossRef] [PubMed]
- Rojas Anaya, E.; Loza-Rubio, E.; Banda Ruiz, V.M.; Hernandez Baumgarten, E. Use of reverse transcription-polymerase chain reaction to determine the stability of rabies virus genome in brains kept at room temperature. J. Vet. Diagn. Investig. 2006, 18, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Macedo, C.I.; Carnieli, P., Jr.; Brandao, P.E.; Travassos da Rosa, E.S.; Oliveira Rde, N.; Castilho, J.G.; Medeiros, R.; Machado, R.R.; Oliveira, R.C.; Carrieri, M.L.; et al. Diagnosis of human rabies cases by polymerase chain reaction of neck-skin samples. Braz. J. Infect. Dis. 2006, 10, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.; Fooks, A.; McColl, K. Reexamination of human rabies case with long incubation, Australia. Emerg. Infect. Dis. 2008, 14, 1950–1951. [Google Scholar] [CrossRef] [PubMed]
- McElhinney, L.M.; Marston, D.A.; Brookes, S.M.; Fooks, A.R. Effects of carcase decomposition on rabies virus infectivity and detection. J. Virol. Methods 2014, 207, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.K.; Shin, E.K.; Oh, Y.I.; Lee, K.W.; Lee, C.S.; Kim, S.Y.; Lee, J.A.; Song, J.Y. Comparison of four diagnostic methods for detecting rabies viruses circulating in Korea. J. Vet. Sci. 2012, 13, 43–48. [Google Scholar] [CrossRef] [PubMed]
- McColl, K.A.; Gould, A.R.; Selleck, P.W.; Hooper, P.T.; Westbury, H.A.; Smith, J.S. Polymerase chain reaction and other laboratory techniques in the diagnosis of long incubation rabies in Australia. Aust. Vet. J. 1993, 70, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.; Wimalaratne, O.; Dahal, N.; Khawplod, P.; Nanayakkara, S.; Rinzin, K.; Perera, D.; Karunanayake, D.; Matsumoto, T.; Nishizono, A. Evaluation of a monoclonal antibody-based rapid immunochromatographic test for direct detection of rabies virus in the brain of humans and animals. Am. J. Trop. Med. Hyg. 2012, 86, 736–740. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.; Oh, J.; Lee, C.; Park, B.K.; Park, Y.; Hong, K.; Lee, K.; Cho, B.; Song, D. Evaluation of a rapid immunodiagnostic test kit for rabies virus. J. Virol. Methods 2007, 145, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Lechenne, M.; Naissengar, K.; Lepelletier, A.; Alfaroukh, I.O.; Bourhy, H.; Zinsstag, J.; Dacheux, L. Validation of a rapid rabies diagnostic tool for field surveillance in developing countries. PLoS NTD 2016, 10, e0005010. [Google Scholar] [CrossRef] [PubMed]
- Markotter, W.; York, D.; Sabeta, C.T.; Shumba, W.; Zulu, G.; Le Roux, K.; Nel, L.H. Evaluation of a rapid immunodiagnostic test kit for detection of African lyssaviruses from brain material. Onderstepoort J. Vet. Res. 2009, 76, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Nishizono, A.; Khawplod, P.; Ahmed, K.; Goto, K.; Shiota, S.; Mifune, K.; Yasui, T.; Takayama, K.; Kobayashi, Y.; Mannen, K.; et al. A simple and rapid immunochromatographic test kit for rabies diagnosis. Microbiol. Immunol. 2008, 52, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Servat, A.; Picard-Meyer, E.; Robardet, E.; Muzniece, Z.; Must, K.; Cliquet, F. Evaluation of a rapid immunochromatographic diagnostic test for the detection of rabies from brain material of European mammals. Biologicals 2012, 40, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Voehl, K.M.; Saturday, G.A. Evaluation of a rapid immunodiagnostic rabies field surveillance test on samples collected from military operations in Africa, Europe, and the Middle East. U.S. Army Med. Dept. J. 2014, 27–32. [Google Scholar]
- Eggerbauer, E.; de Benedictis, P.; Hoffmann, B.; Mettenleiter, T.C.; Schlottau, K.; Ngoepe, E.C.; Sabeta, C.T.; Freuling, C.M.; Muller, T. Evaluation of six commercially available rapid immunochromatographic tests for the diagnosis of rabies in brain material. PLoS NTD 2016, 10, e0004776. [Google Scholar] [CrossRef] [PubMed]
- Boldbaatar, B.; Inoue, S.; Sugiura, N.; Noguchi, A.; Orbina, J.R.; Demetria, C.; Miranda, M.E.; Yamada, A. Rapid detection of rabies virus by reverse transcription loop-mediated isothermal amplification. Jpn. J. Infect. Dis. 2009, 62, 187–191. [Google Scholar] [PubMed]
- Muleya, W.; Namangala, B.; Mweene, A.; Zulu, L.; Fandamu, P.; Banda, D.; Kimura, T.; Sawa, H.; Ishii, A. Molecular epidemiology and a loop-mediated isothermal amplification method for diagnosis of infection with rabies virus in Zambia. Virus Res. 2012, 163, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Saitou, Y.; Kobayashi, Y.; Hirano, S.; Mochizuki, N.; Itou, T.; Ito, F.H.; Sakai, T. A method for simultaneous detection and identification of Brazilian dog- and vampire bat-related rabies virus by reverse transctiption loop-mediated isothermal amplification assay. J. Virol. Methods 2010, 168, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Fooks, A.R.; Johnson, N.; Freuling, C.M.; Wakeley, P.R.; Banyard, A.C.; McElhinney, L.M.; Marston, D.A.; Dastjerdi, A.; Wright, E.; Weiss, R.A.; et al. Emerging technologies for the detection of rabies virus: Challenges and hopes in the 21st century. PLoS NTD 2009, 3, e530. [Google Scholar] [CrossRef] [PubMed]

| State or Union Territory | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Andhra Pradesh * | Gujarat | Karnataka | Kerala | Maharashtra | Manipur | Pondicherry | Punjab | Rajasthan | Tamil Nadu | Uttar Pradesh | Total | |
| Dog | 1 | - | 126 | 45 | 9 | 1 | 1 | 6 | 3 | 17 | 2 | 211 |
| Cattle | - | 3 | 9 | 4 | - | - | - | 4 | - | - | 1 | 21 |
| Buffalo | - | 1 | 1 | - | - | - | - | 6 | - | - | 2 | 10 |
| Cat | - | 1 | 3 | 1 | - | - | - | - | - | - | - | 5 |
| Horse | - | - | 3 | - | - | - | - | 1 | - | 1 | - | 5 |
| Pig | - | - | - | - | - | - | - | 1 | - | - | - | 1 |
| Goat | - | - | - | 1 | - | - | - | - | - | - | - | 1 |
| Jackal | - | - | - | - | - | - | - | 1 | - | - | - | 1 |
| Leopard | - | - | 1 | - | - | - | - | - | - | - | - | 1 |
| Wolf | - | - | 1 | - | - | - | - | - | - | - | - | 1 |
| TOTAL | 1 | 5 | 144 | 51 | 9 | 1 | 1 | 19 | 3 | 18 | 5 | 257 |
| Species | Samples Collected | Positive by DFA | Positive by dRIT | Positive by RT-PCR | Unfit for DFA/dRIT | Negative |
|---|---|---|---|---|---|---|
| Dogs | 211 | 136 | 136 | 168 | 32 | 42 |
| Cattle | 20 | 16 | 16 | 16 | 1 | 6 |
| Buffalo | 10 | 10 | 10 | 10 | 0 | 0 |
| Cats | 5 | 0 | 0 | 2 | 2 | 3 |
| Horses | 5 | 2 | 2 | 2 | 0 | 3 |
| Pig | 1 | 1 | 1 | 1 | 0 | 0 |
| Goat | 1 | 0 | 0 | 1 | 1 | 0 |
| Jackal | 1 | 1 | 1 | 1 | 0 | 0 |
| Leopard | 1 | 0 | 0 | 0 | 0 | 1 |
| Wolf | 1 | 1 | 1 | 1 | 0 | 0 |
| TOTAL | 257 | 167 | 167 | 202 | 36 | 55 |
| DFA | dRIT | RT-PCR | ICT | |
|---|---|---|---|---|
| Equipment required | Fluorescent microscope | Light microscope | Thermal cycler | None |
| Fixative used | Acetone | Formalin | Various | None |
| Inactivation of the virus | Incomplete | Yes | Dependent on method | Incomplete |
| Applicability to decomposed tissue | No | No | Yes | Not always |
| Requirement for further standardization and validation | No | No | Yes | Yes |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prabhu, K.N.; Isloor, S.; Veeresh, B.H.; Rathnamma, D.; Sharada, R.; Das, L.J.; Satyanarayana, M.L.; Hegde, N.R.; Rahman, S.A. Application and Comparative Evaluation of Fluorescent Antibody, Immunohistochemistry and Reverse Transcription Polymerase Chain Reaction Tests for the Detection of Rabies Virus Antigen or Nucleic Acid in Brain Samples of Animals Suspected of Rabies in India. Vet. Sci. 2018, 5, 24. https://doi.org/10.3390/vetsci5010024
Prabhu KN, Isloor S, Veeresh BH, Rathnamma D, Sharada R, Das LJ, Satyanarayana ML, Hegde NR, Rahman SA. Application and Comparative Evaluation of Fluorescent Antibody, Immunohistochemistry and Reverse Transcription Polymerase Chain Reaction Tests for the Detection of Rabies Virus Antigen or Nucleic Acid in Brain Samples of Animals Suspected of Rabies in India. Veterinary Sciences. 2018; 5(1):24. https://doi.org/10.3390/vetsci5010024
Chicago/Turabian StylePrabhu, K. Nithin, Shrikrishna Isloor, B. Hanchinal Veeresh, Doddamane Rathnamma, R. Sharada, Lekshmi J. Das, M.L. Satyanarayana, Nagendra R. Hegde, and Sira Abdul Rahman. 2018. "Application and Comparative Evaluation of Fluorescent Antibody, Immunohistochemistry and Reverse Transcription Polymerase Chain Reaction Tests for the Detection of Rabies Virus Antigen or Nucleic Acid in Brain Samples of Animals Suspected of Rabies in India" Veterinary Sciences 5, no. 1: 24. https://doi.org/10.3390/vetsci5010024
APA StylePrabhu, K. N., Isloor, S., Veeresh, B. H., Rathnamma, D., Sharada, R., Das, L. J., Satyanarayana, M. L., Hegde, N. R., & Rahman, S. A. (2018). Application and Comparative Evaluation of Fluorescent Antibody, Immunohistochemistry and Reverse Transcription Polymerase Chain Reaction Tests for the Detection of Rabies Virus Antigen or Nucleic Acid in Brain Samples of Animals Suspected of Rabies in India. Veterinary Sciences, 5(1), 24. https://doi.org/10.3390/vetsci5010024





