Primary Prostatic Carcinoma with Metastasis to Epaxial Muscles and Myocardium in a Dog
Simple Summary
Abstract
1. Introduction
2. Case Presentation
2.1. Clinical Examination
2.2. Laboratory Findings
2.3. Diagnostic Imaging
2.4. Treatment and Outcome
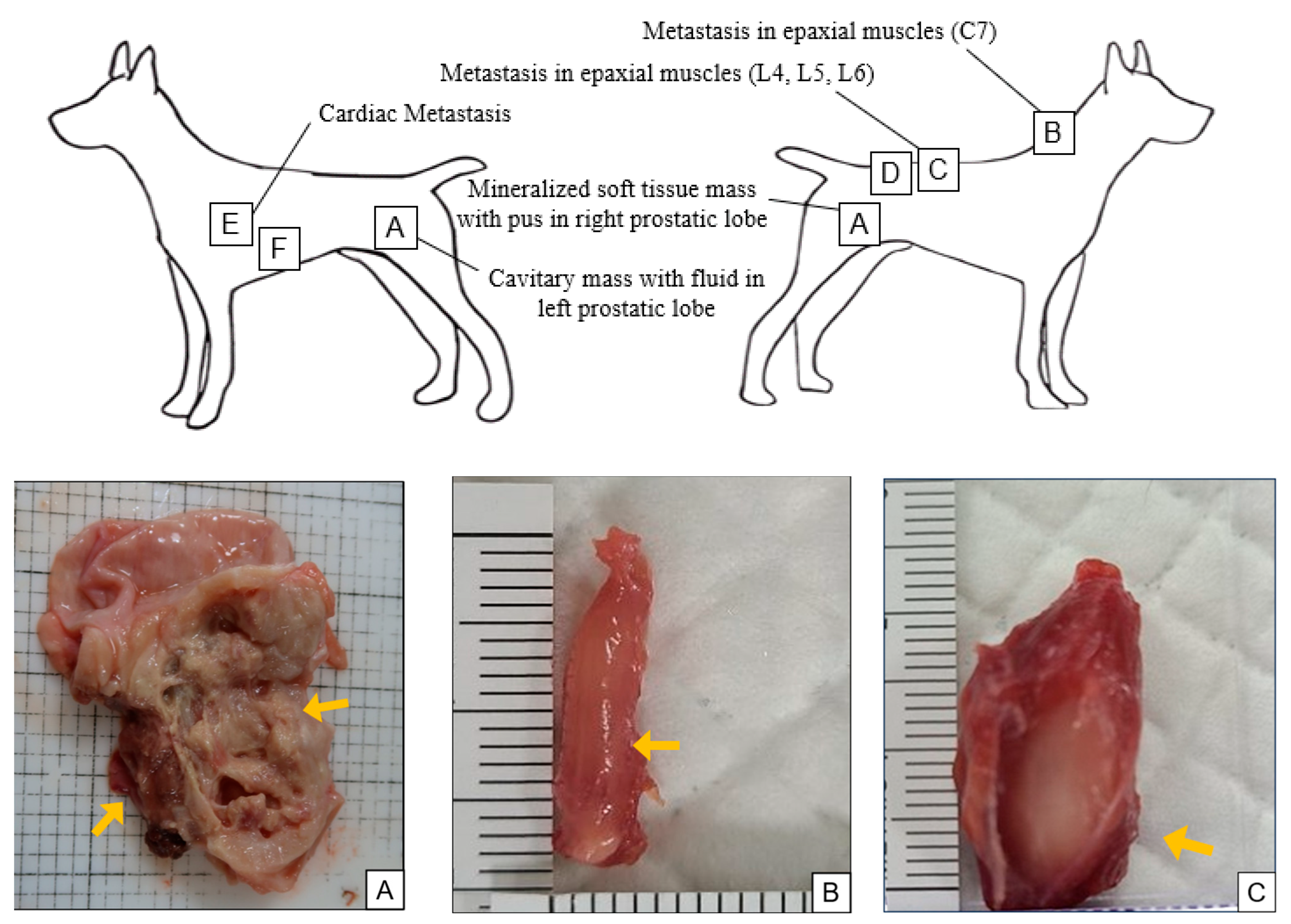
2.5. Necropsy Findings
2.6. Histopathological and Immunohistochemical Findings
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BUN | Blood urea nitrogen |
| CRP | C-reactive protein |
| CT | Computed tomography |
| H&E | Hematoxylin and eosin |
| IHC | Immunohistochemistry |
| MMVD | Myxomatous mitral valve disease |
| PCV | Packed cell volume |
| RI | Reference interval |
| SDMA | Symmetric dimethylarginine |
| TEG | Thromboelastography |
| R | Reaction time (TEG parameter) |
| K | Clot formation time (TEG parameter) |
| α angle | Clot acceleration angle (TEG parameter) |
| MA | Maximum amplitude (TEG parameter) |
| G | Clot strength (TEG parameter) |
| LY30 | Lysis at 30 min (TEG parameter) |
| EPL | Estimated percent lysis (TEG parameter) |
References
- Cornell, K.K.; Bostwick, D.G.; Cooley, D.M.; Hall, G.; Harvey, H.J.; Hendrick, M.J.; Waters, D.J. Clinical and pathologic aspects of spontaneous canine prostate carcinoma: A retrospective analysis of 76 cases. Prostate 2000, 45, 173–183. [Google Scholar] [CrossRef]
- Bryan, J.N.; Keeler, M.R.; Henry, C.J.; Bryan, M.E.; Hahn, A.W.; Caldwell, C.W. A population study of neutering status as a risk factor for canine prostate cancer. Prostate 2007, 67, 1174–1181. [Google Scholar] [CrossRef]
- Palmieri, C.; Fonseca-Alves, C.E.; Laufer-Amorim, R. A review on canine and feline prostate pathology. Front. Vet. Sci. 2022, 9, 881232. [Google Scholar] [CrossRef]
- Bell, F.W.; Klausner, J.S.; Hayden, D.W.; Feeney, D.A.; Johnston, S.D.; Ewing, C.M. Clinical and pathologic features of prostatic adenocarcinoma in the dog. Vet. Pathol. 2013, 50, 778–785. [Google Scholar] [CrossRef]
- Leroy, B.E.; Northrup, N. Prostate cancer in dogs: Comparative and clinical aspects. Vet. J. 2009, 180, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.X.; Lei, L.; Zhu, Y.C.; Du, K.Q.; Li, X.F.; Chen, H.F.; Wang, W.X.; Xu, C.W. A prostate cancer patient with isolated lung metastases: A case report. Transl. Cancer Res. 2020, 9, 2064–2068. [Google Scholar] [CrossRef] [PubMed]
- Reinstatler, L.; Dupuis, J.; Dillon, J.L.; Black, C.C.; Phillips, J.D.; Hyams, E.S. Lung malignancy in prostate cancer: A report of both metastatic and primary lung lesions. Urol. Case Rep. 2017, 16, 119–122. [Google Scholar] [CrossRef]
- Soe, A.M.; Bordia, S.; Xiao, P.Q.; Lopez-Morra, H.; Tejada, J.; Atluri, S.; Krishnaiah, M. A rare presentation of metastasis of prostate adenocarcinoma to the stomach and rectum. J. Gastric Cancer 2014, 14, 271–274. [Google Scholar] [CrossRef][Green Version]
- Moshref, L.; Abidullah, M.; Czaykowski, P.; Chowdhury, A.; Wightman, R.; Hebbard, P. Prostate cancer metastasis to stomach: A case report and review of literature. Curr. Oncol. 2023, 30, 3901–3914. [Google Scholar] [CrossRef]
- Muñoz López, A.Y.; Domínguez Castillo, R.E.; Rodenas Gil, E.A.; Navarro Ruesga, I.; Morales Montor, J.G.; Pacheco Gahbler, C. Primary neuroendocrine prostate cancer with adrenal gland metastasis. Urol. Case Rep. 2021, 40, 101896. [Google Scholar] [CrossRef]
- Pouessel, D.; Gallet, B.; Bibeau, F.; Avancès, C.; Iborra, F.; Sénesse, P.; Culine, S. Liver metastases in prostate carcinoma: Clinical characteristics and outcome. BJU Int. 2007, 99, 807–811. [Google Scholar] [CrossRef]
- McCutcheon, I.E.; Eng, D.Y.; Logothetis, C.J. Brain metastasis from prostate carcinoma: Antemortem recognition and outcome after treatment. Cancer 1999, 86, 2301–2311. [Google Scholar] [CrossRef]
- Hallemeier, C.L.; Kohli, M.; Chandan, V.S.; Miller, R.C.; Choo, R. Multiple urinary bladder masses from metastatic prostate adenocarcinoma. Rare Tumors 2010, 2, e65. [Google Scholar] [CrossRef] [PubMed]
- Thudi, N.K.; Shu, S.T.; Martin, C.K.; Lanigan, L.G.; Nadella, M.V.; Van Bokhoven, A.; Werbeck, J.L.; Simmons, J.K.; Murahari, S.; Kisseberth, W.C.; et al. Development of a brain metastatic canine prostate cancer cell line. Prostate 2011, 71, 1251–1263. [Google Scholar] [CrossRef] [PubMed]
- Klusa, D.; Lohaus, F.; Furesi, G.; Rauner, M.; Benešová, M.; Krause, M.; Kurth, I.; Peitzsch, C. Metastatic spread in prostate cancer patients influencing radiotherapy response. Front. Oncol. 2021, 10, 627379. [Google Scholar] [CrossRef]
- Karaman, S.; Detmar, M. Mechanisms of lymphatic metastasis. J. Clin. Investig. 2014, 124, 922–928. [Google Scholar] [CrossRef]
- Jayarangaiah, A.; Kemp, A.K.; Theetha Kariyanna, P. Bone metastasis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025; Available online: https://www.ncbi.nlm.nih.gov/books/NBK507911 (accessed on 8 September 2024).
- Datta, K.; Muders, M.; Zhang, H.; Tindall, D.J. Mechanism of lymph node metastasis in prostate cancer. Future Oncol. 2010, 6, 823–836. [Google Scholar] [CrossRef]
- Aupperle, H.; März, I.; Ellenberger, C.; Buschatz, S.; Reischauer, A.; Schoon, H.A. Primary and secondary heart tumours in dogs and cats. J. Comp. Pathol. 2007, 136, 18–26. [Google Scholar] [CrossRef]
- Grimes, C.N.; Fry, M.M. Nonregenerative Anemia: Mechanisms of Decreased or Ineffective Erythropoiesis. Vet. Pathol. 2014, 52, 298–311. [Google Scholar] [CrossRef]
- Ettinger, S.J.; Feldman, E.C. Textbook of Veterinary Internal Medicine, 8th ed.; Elsevier: St. Louis, MO, USA, 2017; pp. 2200–2300. [Google Scholar]
- Kristensen, A.T.; Wiinberg, B.; Jessen, L.R.; Andreasen, E.; Jensen, A.L. Evaluation of human recombinant tissue factor-activated thromboelastography in 49 dogs with neoplasia. J. Vet. Intern. Med. 2008, 22, 140–147. [Google Scholar] [CrossRef]
- Wojtukiewicz, M.Z.; Sierko, E.; Hempel, D.; Tucker, S.C.; Honn, K.V. Platelets and cancer angiogenesis nexus. Cancer Metastasis Rev. 2017, 36, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Michalak, S.; Woerde, D.; Wilson, S.; Alonso, F.; Hardy, B. Mandibular metastasis of a prostatic carcinoma in a dog. Vet. Med. Sci. 2021, 7, 513. [Google Scholar] [CrossRef] [PubMed]
- Vignoli, M.; Terragni, R.; Rossi, F.; Frühauf, L.; Bacci, B.; Ressel, L.; Capitani, O.; Marconato, L. Whole body computed tomographic characteristics of skeletal and cardiac muscular metastatic neoplasia in dogs and cats. Vet. Radiol. Ultrasound 2013, 54, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Prisca, A.E.; Rajaonarison, L.H.; Adrienne, R.K.; Ahmad, A. Muscle metastasis from prostate cancer. West Afr. J. Radiol. 2016, 24, 192759. [Google Scholar] [CrossRef]
- Koutserimpas, C.; Dargaras, N.; Naoum, S.; Arkoudis, N.A.; Bafaloukos, D.; Kourea, H.; Samonis, G. Soleus muscle single metastasis from hepatocellular carcinoma. Maedica 2022, 17, 714–719. [Google Scholar] [CrossRef]
- Kim, Y.W.; Seo, K.J.; Lee, S.L.; Kwon, K.W.; Hur, J.; An, H.J.; Ko, Y.H.; Kim, J.S.; Won, H.S. Skeletal muscle metastases from breast cancer: Two case reports. J. Breast Cancer 2013, 16, 117–121. [Google Scholar] [CrossRef]
- Seely, S. Possible reasons for the high resistance of muscle to cancer. Med. Hypotheses 1980, 6, 133–137. [Google Scholar] [CrossRef]
- Bar-Yehuda, S.; Barer, F.; Volfsson, L.; Fishman, P. Resistance of muscle to tumor metastases: A role for A3 adenosine receptor agonists. Neoplasia 2001, 3, 125–131. [Google Scholar] [CrossRef]
- Kump, D.S. Mechanisms underlying the rarity of skeletal muscle cancers. Int. J. Mol. Sci. 2024, 25, 6480. [Google Scholar] [CrossRef]
- Jacob, S. Abdomen. In Human Anatomy; Jacob, S., Ed.; Churchill Livingstone: Edinburgh, UK, 2008; pp. 71–123. [Google Scholar] [CrossRef]
- Ariete, V.; Barnert, N.; Gómez, M.; Mieres, M.; Pérez, B.; Gutierrez, J.C. Morphometrical Study of the Lumbar Segment of the Internal Vertebral Venous Plexus in Dogs: A Contrast CT-Based Study. Animals 2021, 11, 1502. [Google Scholar] [CrossRef]
- Lucà, F.; Parrini, I.; Canale, M.L.; Rao, C.M.; Nucara, M.; Pelaggi, G.; Murrone, A.; Oliva, S.; Bisceglia, I.; Sergi, A.; et al. Cardiac Metastasis: Epidemiology, Pathophysiology, and Clinical Management. Life 2025, 15, 291. [Google Scholar] [CrossRef]
- Basuoni, A.; Burney, I.; Al-Hashami, Z.; Ahmed, M.; Kamel, A.H.M.; de Oliveira Manoel, A.L.; Al Bulushi, Y.; Dawelbeit, W. When cancer reaches the heart: A case series on rare myocardial and endocardial metastases from squamous cell carcinoma. Arch. Clin. Cases 2025, 12, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Urlings, P.; Gramer, I.; Jones, C.; Dirrig, H. Computed tomographic features of canine prostatic carcinoma. Vet. Radiol. Ultrasound 2024, 65, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Doo, S.W.; Kim, W.B.; Kim, B.K.; Yang, W.J.; Yoon, J.H.; Song, Y.S.; Choi, I.H. Skeletal muscle metastases from urothelial cell carcinoma. Korean J. Urol. 2012, 53, 63–66. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yamac, A.H.; Insanic, D.; Bockmeyer, C. Cardiac metastasis from a urothelial cell carcinoma: A commented case report. Cardiovasc. Pathol. 2014, 23, 178–180. [Google Scholar] [CrossRef]
- Gorospe, L.; García-Santana, E.; Jane-Soler, P.; Gómez-Martínez, M.D.V.; Gómez-Dos-Santos, V.; García-Gómez-Muriel, I.; García-Latorre, R. Cardiac and skeletal muscle metastases from squamous cell carcinoma of the bladder: Positron emission tomography-computed tomography findings. Urology 2017, 100, e5–e6. [Google Scholar] [CrossRef]
- Charney, V.A.; Miller, M.A.; Heng, H.G.; Weng, H.Y.; Knapp, D.W. Skeletal metastasis of canine urothelial carcinoma: Pathologic and computed tomographic features. Vet. Pathol. 2017, 54, 380–386. [Google Scholar] [CrossRef]
- Polton, G.; Borrego, J.F.; Clemente-Vicario, F.; Clifford, C.A.; Jagielski, D.; Kessler, M.; Kobayashi, T.; Lanore, D.; Queiroga, F.L.; Rowe, A.T.; et al. Melanoma of the dog and cat: Consensus and guidelines. Front. Vet. Sci. 2024, 11, 1359426. [Google Scholar] [CrossRef]
- Carloni, A.; Terragni, R.; Morselli-Labate, A.M.; Paninarova, M.; Graham, J.; Valenti, P.; Alberti, M.; Albarello, G.; Millanta, F.; Vignoli, M. Prevalence, distribution, and clinical characteristics of hemangiosarcoma-associated skeletal muscle metastases in 61 dogs: A whole body computed tomographic study. J. Vet. Intern. Med. 2019, 33, 812–819. [Google Scholar] [CrossRef]
- Sorenmo, K.U.; Goldschmidt, M.H.; Shofer, F.S.; Goldkamp, C.; Ferracone, J. Evaluation of cyclooxygenase-1 and cyclooxygenase-2 expression and the effect of cyclooxygenase inhibitors in canine prostatic carcinoma. Vet. Comp. Oncol. 2004, 2, 13–23. [Google Scholar] [CrossRef]
- Gibson, E.A.; Culp, W.T.N. Canine prostate cancer: Current treatments and the role of interventional oncology. Vet. Sci. 2024, 11, 169. [Google Scholar] [CrossRef]




| Results | Unit | Reference Interval | Parameters | |
|---|---|---|---|---|
| HCT | 34.3 | % | 37–55% | ↓ |
| WBC | 14.46 | ×103/µL | 6–17 × 103/µL | - |
| MCV | 60.9 | fL | 60–74 fL | - |
| MCHC | 35.6 | g/dL | 31–36 g/dL | - |
| PLT | 375 | ×103/µL | 200–500 × 103/µL | - |
| BUN | 80 | mg/dL | 8–26 mg/dL | ↑ |
| Creatinine | 2.2 | mg/dL | 0.5–1.8 mg/dL | ↑ |
| SDMA | 48 | µg/dL | 0–14 µg/dL | ↑ |
| CRP | 5 | mg/dL | <1 mg/dL | ↑ |
| D-dimer | 2436.69 | ng/mL | 0–250 ng/mL | ↑ |
| TEG R | 3.9 | min | 5–10 min | ↓ |
| TEG K | 0.8 | min | 1–3 min | ↓ |
| α angle | 79.3 | ° | 53–72° | ↑ |
| MA | 81.3 | mm | 50–70 mm | ↑ |
| LY30 | 0 | % | 0–8% | - |
| EPL | 0 | % | 0–15% | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Han, H.-J.; Kim, D.Y. Primary Prostatic Carcinoma with Metastasis to Epaxial Muscles and Myocardium in a Dog. Vet. Sci. 2025, 12, 1045. https://doi.org/10.3390/vetsci12111045
Kim H, Han H-J, Kim DY. Primary Prostatic Carcinoma with Metastasis to Epaxial Muscles and Myocardium in a Dog. Veterinary Sciences. 2025; 12(11):1045. https://doi.org/10.3390/vetsci12111045
Chicago/Turabian StyleKim, Hyouju, Hyun-Jung Han, and Dae Young Kim. 2025. "Primary Prostatic Carcinoma with Metastasis to Epaxial Muscles and Myocardium in a Dog" Veterinary Sciences 12, no. 11: 1045. https://doi.org/10.3390/vetsci12111045
APA StyleKim, H., Han, H.-J., & Kim, D. Y. (2025). Primary Prostatic Carcinoma with Metastasis to Epaxial Muscles and Myocardium in a Dog. Veterinary Sciences, 12(11), 1045. https://doi.org/10.3390/vetsci12111045






