Use of Computed Tomography and Magnetic Resonance Angiograms Combined with a 3D Surgical Guide in an Elderly Cat with an Occipital Lobe Meningioma
Abstract
Simple Summary
Abstract
1. Introduction
2. Case Presentation
2.1. Case
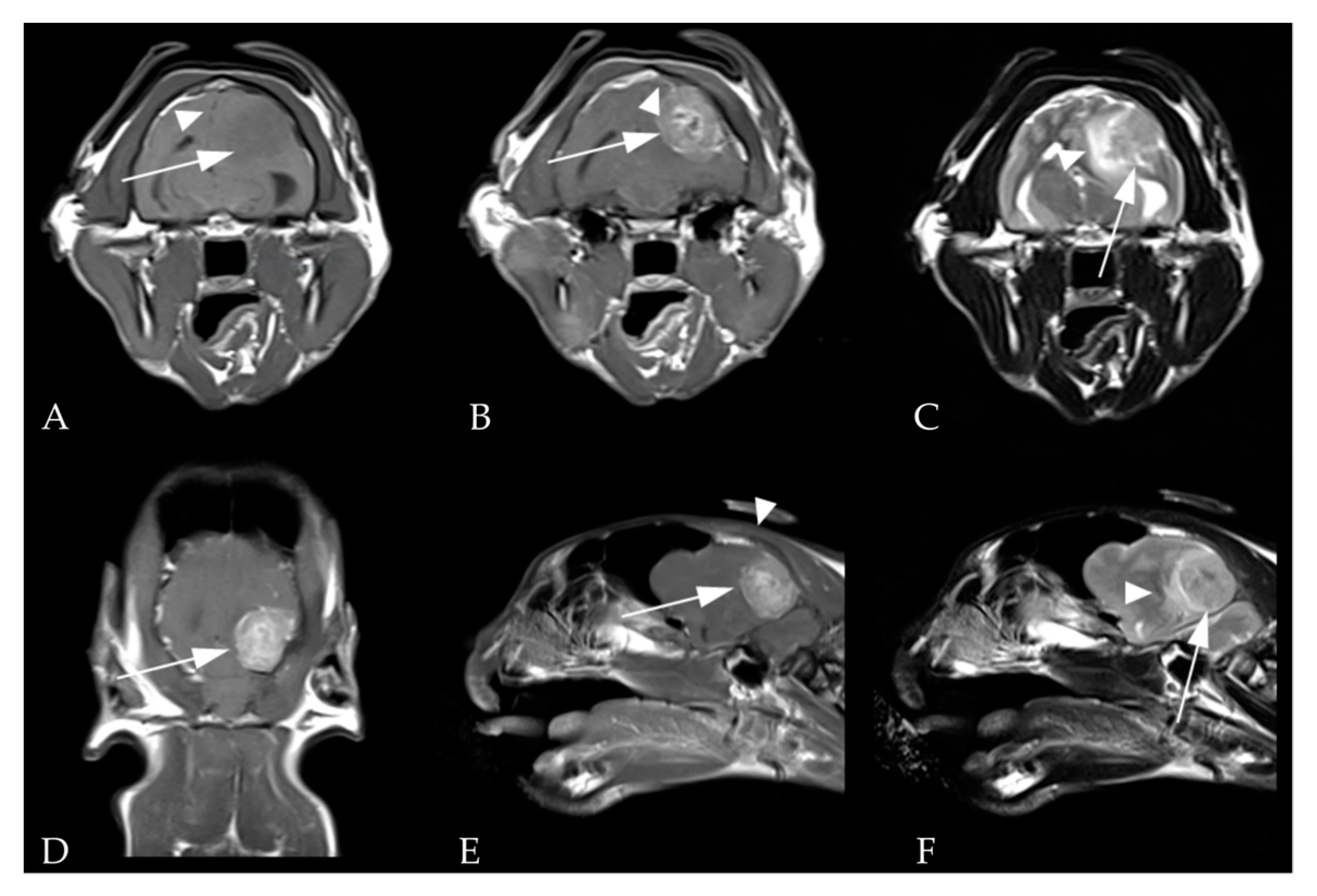
2.2. Images
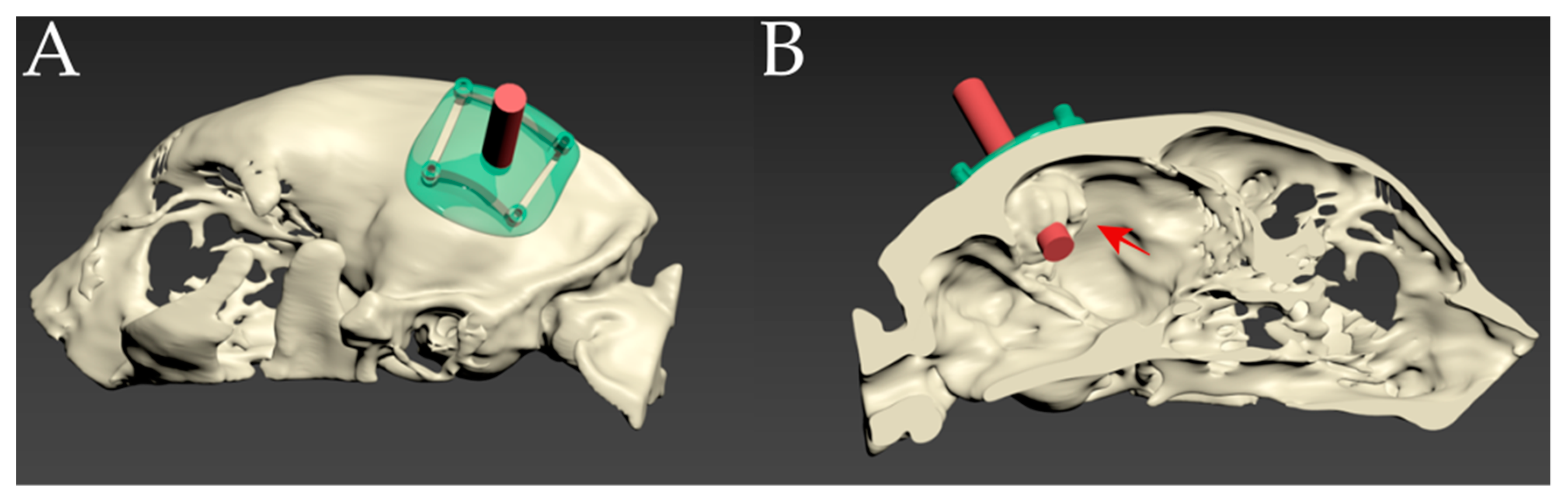
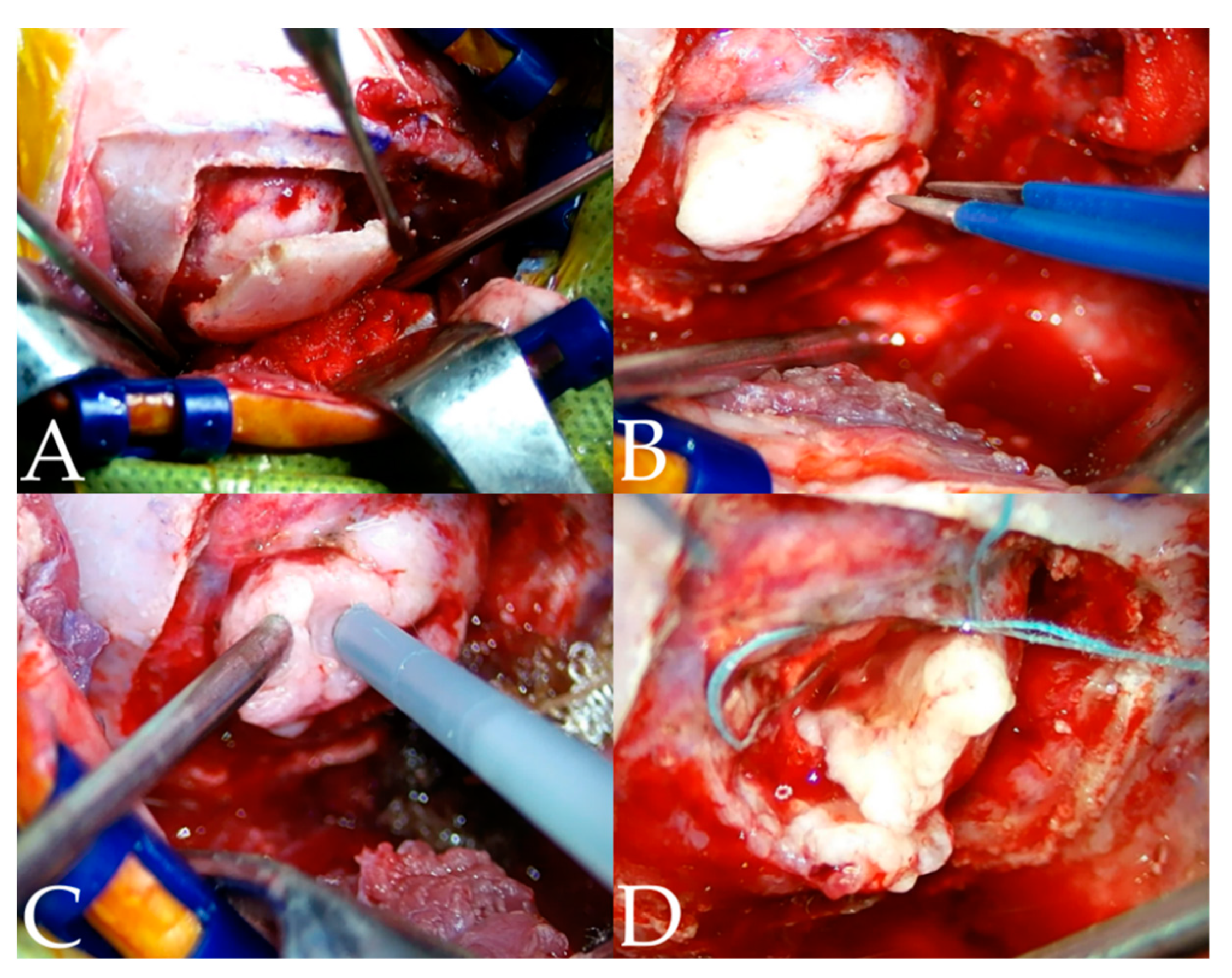
2.3. Surgical Techniques
2.4. Postoperative Care, Outcomes, and Pathological Examination
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Motta, L.; Mandara, M.T.; Skerritt, G.C. Canine and feline intracranial meningiomas: An updated review. Vet. J. 2012, 192, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Troxel, M.T.; Vite, C.H.; Van Winkle, T.J.; Newton, A.L.; Tiches, D.; Dayrell-Hart, B.; Kapatkin, A.S.; Shofer, F.S.; Steinberg, S.A. Feline intracranial neoplasia: Retrospective review of 160 cases (1985–2001). J. Vet. Intern. Med. 2003, 17, 850–859. [Google Scholar] [PubMed]
- Gorden, L.; Thacher, C.; Matthisen, D.; Joseph, R. Results of craniotomy for the treatment of cerebral meningioma in 42 cases. Vet. Surg. 1994, 23, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Adamo, P.F.; Forrest, L.; Dubielzig, R. Canine and feline meningiomas: Diagnosis, treatment, and prognosis. Compendium 2004, 26, 951–966. [Google Scholar]
- Rao, D.; Le, R.T.; Fiester, P.; Patel, J.; Rahmathulla, G. An Illustrative Review of Common Modern Craniotomies. J. Clin. Imaging Sci. 2020, 10, 81. [Google Scholar] [CrossRef]
- Forterre, F.; Fritsch, G.; Kaiser, S.; Matiasek, K.; Brunnberg, L. Surgical approach for tentorial meningiomas in cats: A review of six cases. J. Feline Med. Surg. 2006, 8, 227–233. [Google Scholar] [CrossRef]
- Clarke, D.F.; Tindall, K.; Lee, M.; Patel, B. Bilateral occipital dysplasia, seizure identification, and ablation: A novel surgical technique. Epileptic Disord. 2014, 16, 238–243. [Google Scholar] [CrossRef]
- Corniola, M.V.; Bouthour, W.; Vargas, M.-I.; Meling, T.R. Visual field restoration after Simpson grade I resection of symptomatic occipital lobe meningioma: Illustrative case and review of the literature. Acta Neurochir. 2021, 163, 67–71. [Google Scholar] [CrossRef]
- Wu, A.; Garcia, M.A.; Magill, S.T.; Chen, W.; Vasudevan, H.N.; Perry, A.; Theodosopoulos, P.V.; McDermott, M.W.; Braunstein, S.E.; Raleigh, D.R. Presenting symptoms and prognostic factors for symptomatic outcomes following resection of meningioma. World Neurosurg. 2018, 111, e149–e159. [Google Scholar] [CrossRef]
- Wu, W.-C.; Smith, T.S.; Henderson, W.G.; Eaton, C.B.; Poses, R.M.; Uttley, G.; Mor, V.; Sharma, S.C.; Vezeridis, M.; Khuri, S.F. Operative blood loss, blood transfusion, and 30-day mortality in older patients after major noncardiac surgery. Ann. Surg. 2010, 252, 11–17. [Google Scholar] [CrossRef]
- Rajagopalan, V.; Chouhan, R.S.; Pandia, M.P.; Lamsal, R.; Rath, G.P. Effect of intraoperative blood loss on perioperative complications and neurological outcome in adult patients undergoing elective brain tumor surgery. J. Neurosci. Rural Pract. 2019, 10, 631–640. [Google Scholar] [CrossRef]
- Gerlach, R.; Raabe, A.; Scharrer, I.; Meixensberger, J.; Seifert, V. Post-operative hematoma after surgery for intracranial meningiomas: Causes, avoidable risk factors and clinical outcome. Neurol. Res. 2004, 26, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; Cha, S.; Higashida, R.; Cullen, S.; Halbach, V.; Dowd, C.; McDermott, M.; Saloner, D. Assessment of vasculature of meningiomas and the effects of embolization with intra-arterial MR perfusion imaging: A feasibility study. Am. J. Neuroradiol. 2007, 28, 1771–1777. [Google Scholar] [CrossRef] [PubMed]
- Garosi, L.S. Cerebrovascular disease in dogs and cats. Vet. Clin. Small Anim. Pract. 2010, 40, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Sherman, J.H.; Hoes, K.; Marcus, J.; Komotar, R.J.; Brennan, C.W.; Gutin, P.H. Neurosurgery for brain tumors: Update on recent technical advances. Curr. Neurol. Neurosci. Rep. 2011, 11, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Jandial, R. Core Techniques in Operative Neurosurgery E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Mert, A.; Buehler, K.; Sutherland, G.R.; Tomanek, B.; Widhalm, G.; Kasprian, G.; Knosp, E.; Wolfsberger, S. Brain tumor surgery with 3-dimensional surface navigation. Oper. Neurosurg. 2012, 71, ons286–ons295. [Google Scholar] [CrossRef]
- Dai, Z.; Yang, R.; Hang, F.; Zhuang, J.; Lin, Q.; Wang, Z.; Lao, Y. Neurosurgical craniotomy localization using interactive 3D lesion mapping for image-guided neurosurgery. IEEE Access 2019, 7, 10606–10616. [Google Scholar] [CrossRef]
- Dickinson, P.J. Advances in diagnostic and treatment modalities for intracranial tumors. J. Vet. Intern. Med. 2014, 28, 1165–1185. [Google Scholar] [CrossRef]
- Baek, S.E.; Lee, S.B.; Kim, Y.W.; Lee, J.H. Usefulness of three-dimensional CT image in meningioma using contrast method. J. Korean Soc. Radiol. 2008, 2, 17–21. [Google Scholar]
- Abe, T.; Matsumoto, K.; Hanakawa, K.; Homma, H.; Kawamura, N.; Ikeda, H.; Horichi, Y.; Hayashi, T. Role of 3D-TOF magnetic resonance angiography for intracranial meningioma. J. Clin. Neurosci. 1998, 5, 387–389. [Google Scholar] [CrossRef]
- Engelhard, H.H. Progress in the diagnosis and treatment of patients with meningiomas: Part I: Diagnostic imaging, preoperative embolization. Surg. Neurol. 2001, 55, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Okahara, M.; Kiyosue, H.; Yamashita, M.; Nagatomi, H.; Hata, H.; Saginoya, T.; Sagara, Y.; Mori, H. Diagnostic accuracy of magnetic resonance angiography for cerebral aneurysms in correlation with 3D–digital subtraction angiographic images: A study of 133 aneurysms. Stroke 2002, 33, 1803–1808. [Google Scholar] [CrossRef] [PubMed]
- Uetani, H.; Akter, M.; Hirai, T.; Shigematsu, Y.; Kitajima, M.; Kai, Y.; Yano, S.; Nakamura, H.; Makino, K.; Azuma, M. Can 3T MR angiography replace DSA for the identification of arteries feeding intracranial meningiomas? Am. J. Neuroradiol. 2013, 34, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Uricchio, M.; Gupta, S.; Jakowenko, N.; Levito, M.; Vu, N.; Doucette, J.; Liew, A.; Papatheodorou, S.; Khawaja, A.M.; Aglio, L.S.; et al. Computed tomography angiography versus digital subtraction angiography for postclipping aneurysm obliteration detection: A meta-analysis. Stroke 2019, 50, 381–388. [Google Scholar] [CrossRef]
- Platt, S.R.; Radaelli, S.T.; McDonnell, J.J. The prognostic value of the modified Glasgow Coma Scale in head trauma in dogs. J. Vet. Intern. Med. 2001, 15, 581–584. [Google Scholar] [CrossRef]
- Song, K.; Lee, H.; Jeong, J.; Roh, Y. Multiple Meningioma Resection by Bilateral Extended Rostrotentorial Craniotomy with a 3D-Print Guide in a Cat. Vet. Sci. 2022, 9, 512. [Google Scholar] [CrossRef]
- Otani, N.; Wada, K.; Toyooka, T.; Mori, K. Occipital interhemispheric approach for surgical removal of the middle third falx meningioma: Two case reports. Asian J. Neurosurg. 2018, 13, 789–791. [Google Scholar] [CrossRef]
- Velnar, T.; Pregelj, R.; Limbaeck-Stokin, C. Brain meningioma invading and destructing the skull bone: Replacement of the missing bone. Radiol. Oncol. 2011, 45, 304–309. [Google Scholar] [CrossRef]
- Avecillas-Chasin, J.M.; Saceda-Gutierrez, J.; Alonso-Lera, P.; Garcia-Pumarino, R.; Issa, S.; López, E.; Barcia, J.A. Scalp metastases of recurrent meningiomas: Aggressive behavior or surgical seeding? Wolrd Neurosurg. 2015, 84, 121–131. [Google Scholar] [CrossRef]
- Meuten, D.J. Tumors in Domestic Animals; John Wiley & Sons: Hoboken, NJ, USA, 2020. [Google Scholar]
- Adachi, K.; Kawase, T.; Yoshida, K.; Yazaki, T.; Onozuka, S. ABC Surgical Risk Scale for skull base meningioma: A new scoring system for predicting the extent of tumor removal and neurological outcome. J. Neurosurg. 2009, 111, 1053–1061. [Google Scholar] [CrossRef]
- Du, W.; Zhong, D.; Lü, D.; Li, J.; Huang, H.; Yang, J.; Wu, Y.; Xia, H.; Tang, W.; Sun, X. Dynamic retraction microneurosurgery for the treatment of medial tentorial meningiomas. Zhonghua Yi Xue Za Zhi 2018, 98, 1317–1321. [Google Scholar] [PubMed]
- Ekşi, M.Ş.; Canbolat, Ç.; Akbaş, A.; Özmen, B.B.; Akpınar, E.; Usseli, M.İ.; Güngör, A.; Güdük, M.; Hacıhanefioğlu, M.; Danyeli, A.E. Elderly patients with intracranial meningioma: Surgical considerations in 228 patients with a comprehensive analysis of the literature. World Neurosurg. 2019, 132, e350–e365. [Google Scholar] [CrossRef] [PubMed]
- Kivelev, J.; Koskela, E.; Setälä, K.; Niemelä, M.; Hernesniemi, J. Long-term visual outcome after microsurgical removal of occipital lobe cavernomas. J. Neurosurg. 2012, 117, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Morishita, T.; Okun, M.S.; Burdick, A.; Jacobson, C.E., IV; Foote, K.D. Cerebral venous infarction: A potentially avoidable complication of deep brain stimulation surgery. Neuromodulation Technol. Neural Interface 2013, 16, 407–413. [Google Scholar] [CrossRef]
- Barness, J.A. Cardiovascular system. In Atlas of Feline Anatomy for Veterinarians, 2nd ed.; Hudson, L., Hamilton, W., Eds.; Teton NewMedia: Jackson, WY, USA, 2010; pp. 94–95. [Google Scholar]
- Gallagher, J.; Berg, J.; Knowles, K.; Williams, L.; Bronson, R. Prognosis after surgical excision of cerebral meningiomas in cats: 17 cases (1986–1992). J. Am. Vet. Med. Assoc. 1993, 203, 1437–1440. [Google Scholar]
- Dhandapani, S.; Sharma, K. Is “en-bloc” excision, an option for select large vascular meningiomas. Surg. Neurol. Int. 2013, 4, 102. [Google Scholar] [CrossRef]
- Ma, B.; Park, T.; Chun, I.; Yun, K. The accuracy of a 3D printing surgical guide determined by CBCT and model analysis. J. Adv. Prosthodont. 2018, 10, 279–285. [Google Scholar] [CrossRef]
- So, J.; Lee, H.; Jeong, J.; Forterre, F.; Roh, Y. Endoscopy-assisted resection of a sphenoid-wing meningioma using a 3D-printed patient-specific pointer in a dog: A case report. Front. Vet. Sci. 2022, 9, 979290. [Google Scholar] [CrossRef]
- Ishihara, H.; Ishihara, S.; Niimi, J.; Neki, H.; Kakehi, Y.; Uemiya, N.; Kohyama, S.; Yamane, F.; Kato, H.; Suzuki, T. The safety and efficacy of preoperative embolization of meningioma with N-butyl cyanoacrylate. Interv. Neuroradiol. 2015, 21, 624–630. [Google Scholar] [CrossRef]
- Nikoubashman, O.; Jablawi, F.; Dekeyzer, S.; Oros-Peusquens, A.; Abbas, Z.; Lindemeyer, J.; Othman, A.; Shah, N.; Wiesmann, M. MRI appearance of intracerebral iodinated contrast agents: Is it possible to distinguish extravasated contrast agent from hemorrhage? Am. J. Neuroradiol. 2016, 37, 1418–1421. [Google Scholar] [CrossRef]
- Gazzeri, R.; Galarza, M.; Neroni, M.; Alfieri, A.; Giordano, M. Hemostatic matrix sealant in neurosurgery: A clinical and imaging study. Acta Neurochir. 2011, 153, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Kamamoto, D.; Kanazawa, T.; Ishihara, E.; Yanagisawa, K.; Tomita, H.; Ueda, R.; Jinzaki, M.; Yoshida, K.; Toda, M. Efficacy of a topical gelatin-thrombin hemostatic matrix, FLOSEAL®, in intracranial tumor resection. Surg. Neurol. Int. 2020, 11, 16. [Google Scholar] [CrossRef] [PubMed]
- Shores, A.; Brisson, B.A. Current Techniques in Canine and Feline Neurosurgery; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Miyamoto, K.; Masuda, K.; Inoue, N.; Okuma, M.; Muehleman, C.; An, H.S. Anti-adhesion properties of a thrombin-based hemostatic gelatin in a canine laminectomy model: A biomechanical, biochemical, and histologic study. Spine 2006, 31, E91–E97. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byun, P.; Roh, Y.; Lee, H.; Jeong, J. Use of Computed Tomography and Magnetic Resonance Angiograms Combined with a 3D Surgical Guide in an Elderly Cat with an Occipital Lobe Meningioma. Vet. Sci. 2023, 10, 264. https://doi.org/10.3390/vetsci10040264
Byun P, Roh Y, Lee H, Jeong J. Use of Computed Tomography and Magnetic Resonance Angiograms Combined with a 3D Surgical Guide in an Elderly Cat with an Occipital Lobe Meningioma. Veterinary Sciences. 2023; 10(4):264. https://doi.org/10.3390/vetsci10040264
Chicago/Turabian StyleByun, Pillmoo, Yoonho Roh, Haebeom Lee, and Jaemin Jeong. 2023. "Use of Computed Tomography and Magnetic Resonance Angiograms Combined with a 3D Surgical Guide in an Elderly Cat with an Occipital Lobe Meningioma" Veterinary Sciences 10, no. 4: 264. https://doi.org/10.3390/vetsci10040264
APA StyleByun, P., Roh, Y., Lee, H., & Jeong, J. (2023). Use of Computed Tomography and Magnetic Resonance Angiograms Combined with a 3D Surgical Guide in an Elderly Cat with an Occipital Lobe Meningioma. Veterinary Sciences, 10(4), 264. https://doi.org/10.3390/vetsci10040264






