Changes in Faecal and Plasma Amino Acid Profile in Dogs with Food-Responsive Enteropathy as Indicators of Gut Homeostasis Disruption: A Pilot Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Sample Collection
2.2. Concentration of Free Amino Acids in Plasma Samples
2.3. Concentration of Amino Acids in Faecal Samples by Acid Hydrolysis
2.4. Statistical Analysis
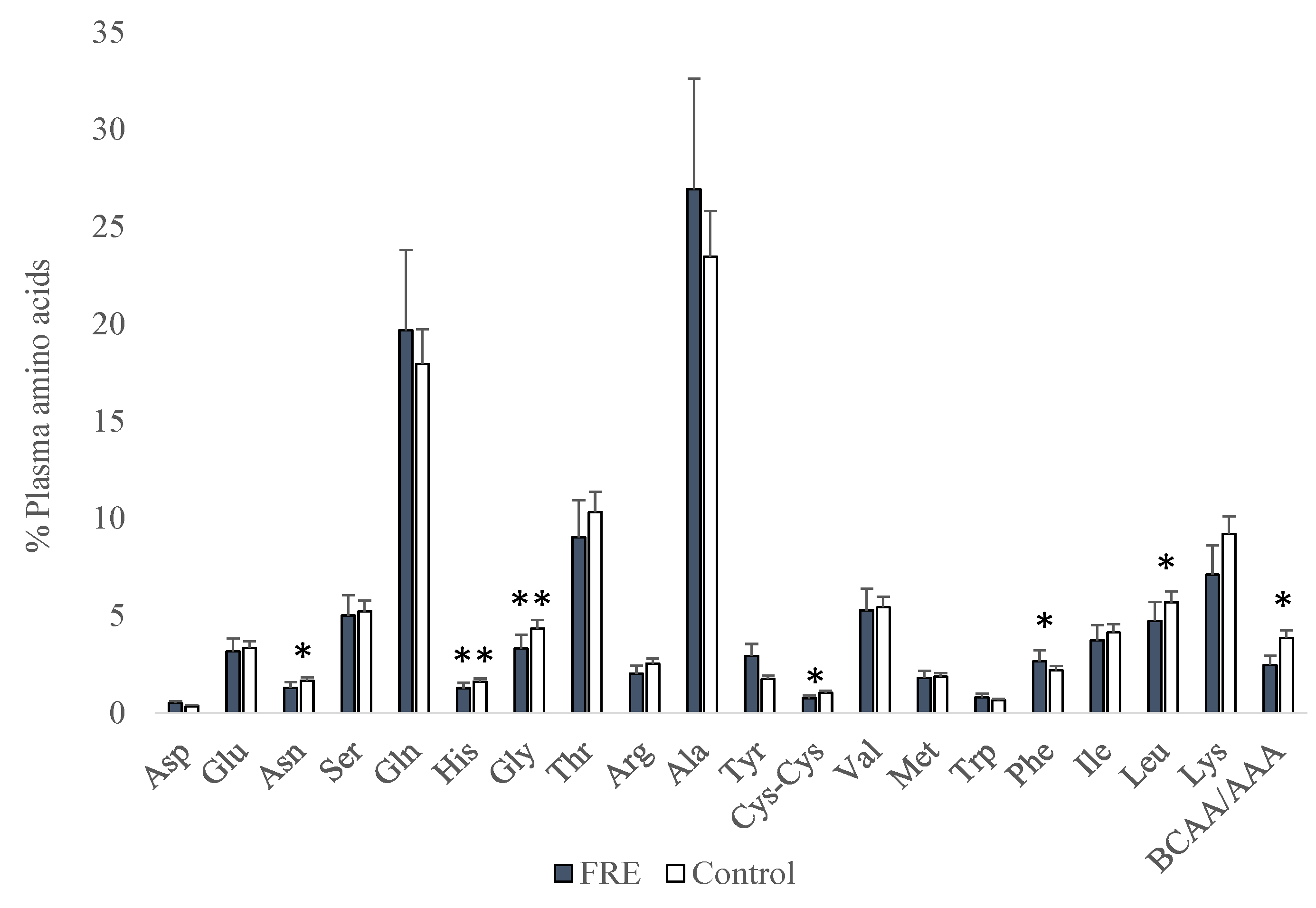
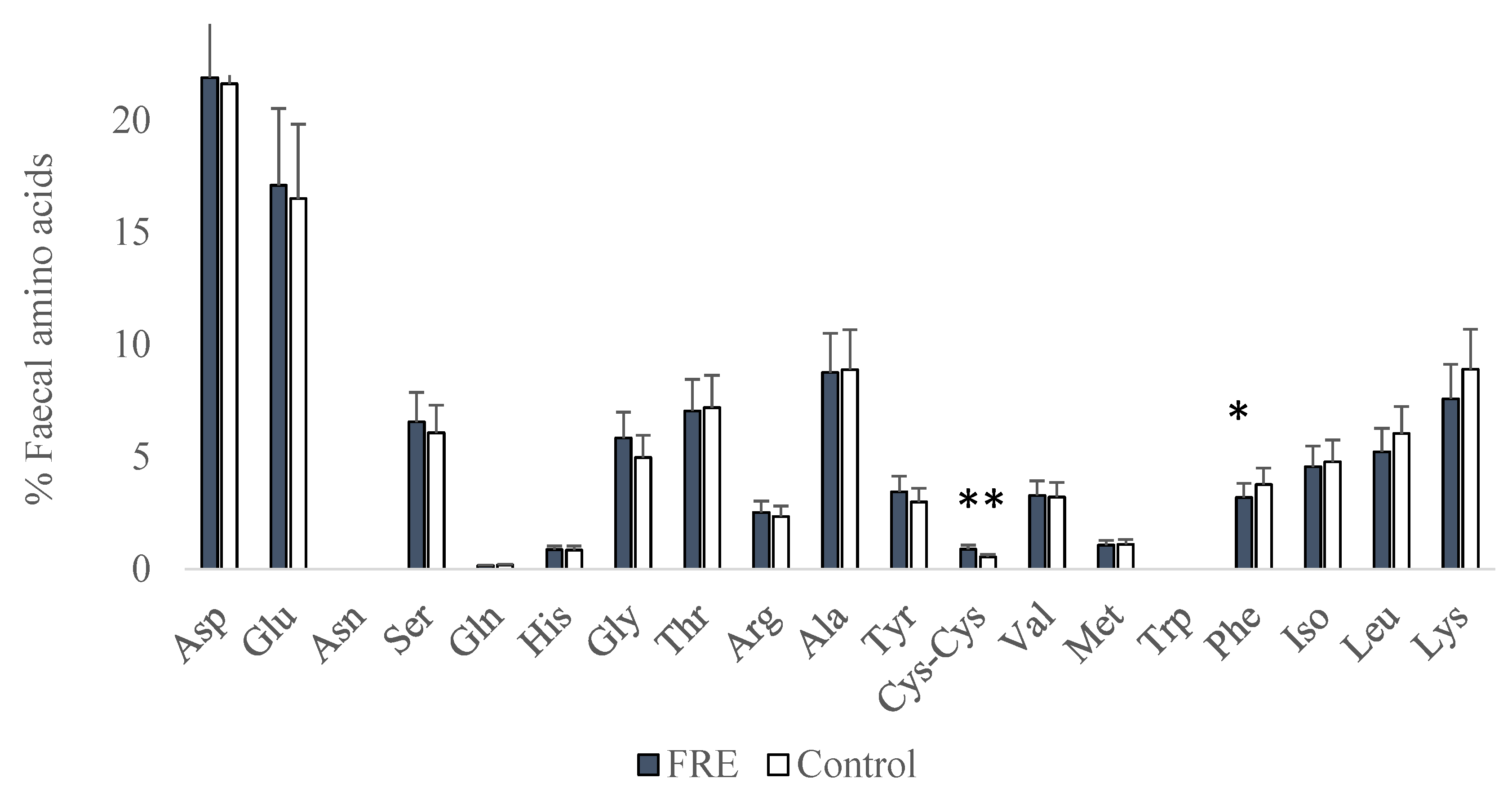
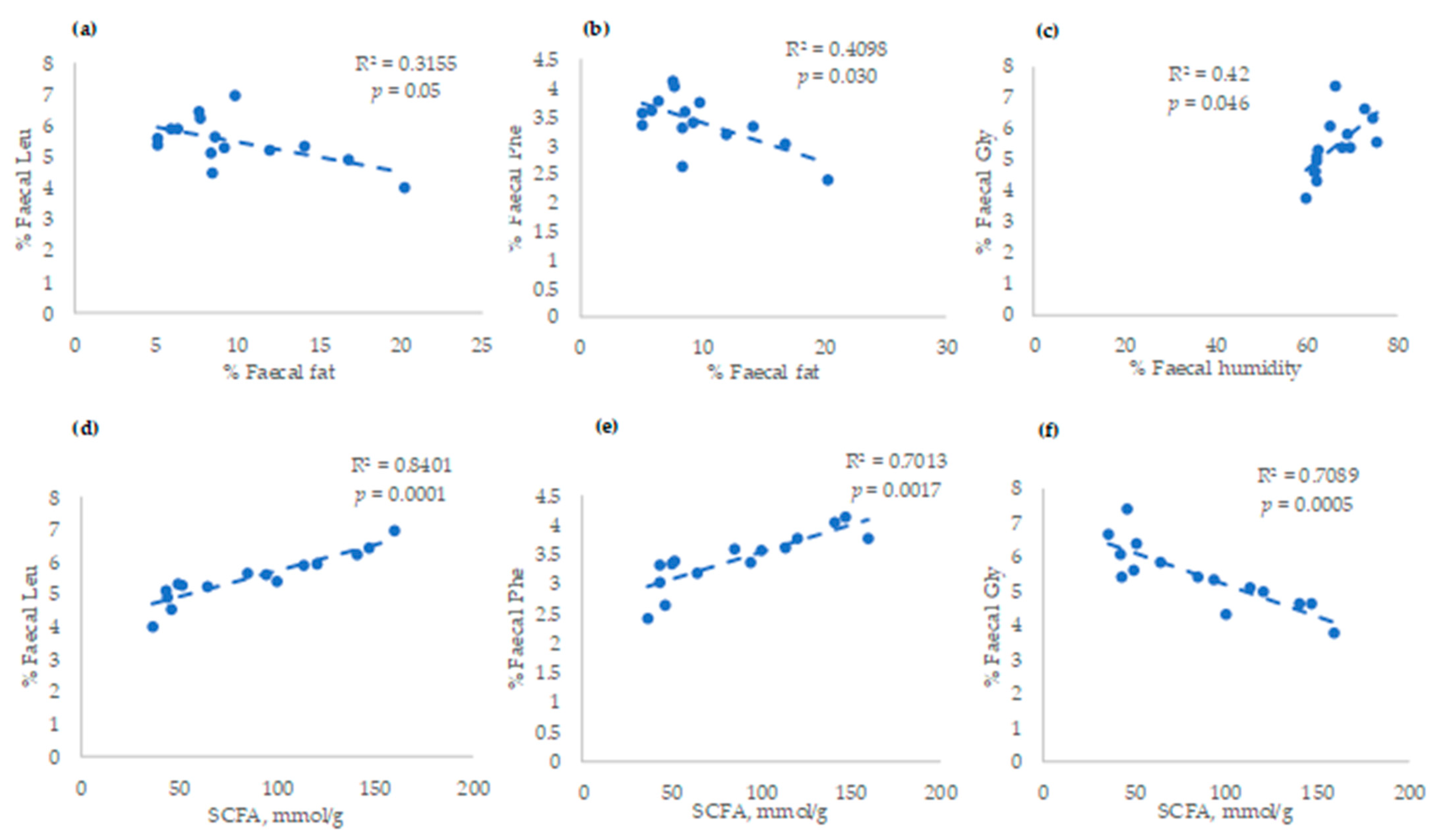
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kamei, Y.; Hatazawa, Y.; Uchitomi, R.; Yoshimura, R.; Miura, S. Regulation of skeletal muscle function by amino acids. Nutrients 2020, 12, 261. [Google Scholar] [CrossRef] [PubMed]
- Tolbert, M.K.; Murphy, M.; Gaylord, L.; Witzel-Rollins, A. Dietary management of chronic enteropathy in dogs. J. Small Anim. Pract. 2022, 63, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R.; Bains, K. Protein, lysine and vitamin D: Critical role in muscle and bone health. Crit. Rev. Food Sci. Nutr. 2022, 62, 2548–2559. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Wu, C.; Li, P.; Li, N.; Zhang, D.; Zhu, Q.; Ren, W.; Peng, Y. Functions and signaling pathways of amino acids in intestinal inflammation. BioMed Res. Int. 2018, 2018, 9171905. [Google Scholar] [CrossRef] [PubMed]
- Bauset, C.; Gisbert-Ferrándiz, L.; Cosín-Roger, J. Metabolomics as a promising resource identifying potential biomarkers for inflammatory bowel disease. J. Clin. Med. 2021, 10, 622. [Google Scholar] [CrossRef] [PubMed]
- Hisamatsu, T.; Okamoto, S.; Hashimoto, M.; Muramatsu, T.; Andou, A.; Uo, M.; Kitazume, M.T.; Matsuoka, K.; Yajima, T.; Inoue, N.; et al. Novel, objective, multivariate biomarkers composed of plasma amino acid profiles for the diagnosis and assessment of inflammatory bowel disease. PLoS ONE 2012, 7, e31131. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Hu, C.A. Therapeutic potential of amino acids in inflammatory bowel disease. Nutrients 2017, 9, 920. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, J.; Dandrieux, J.R.S. From bench top to clinics. Vet. Clin. North Am. Small Anim. Pract. 2021, 51, 137–153. [Google Scholar] [CrossRef]
- Higueras, C.; Rey, A.I.; Escudero, R.; Díaz-Regañón, D.; Rodríguez-Franco, F.; García-Sancho, M.; Agulla, B.; Sainz, A. Short-chain and total fatty acid profile of faeces or plasma as predictors of food-responsive enteropathy in dogs: A preliminary study. Animals 2021, 12, 89. [Google Scholar] [CrossRef]
- Kathrani, A.; Allenspach, K.; Fascetti, A.J.; Larsen, J.A.; Hall, E.J. Alterations in serum amino acid concentrations in dogs with protein-losing enteropathy. J. Vet. Intern. Med. 2018, 32, 1026–1032. [Google Scholar] [CrossRef]
- Tamura, Y.; Ohta, H.; Kagawa, Y.; Osuga, T.; Morishita, K.; Sasaki, N.; Takiguchi, M. Plasma amino acid profiles in dogs with inflammatory bowel disease. J. Vet. Intern. Med. 2019, 33, 1602–1607. [Google Scholar] [CrossRef] [PubMed]
- Benvenuti, E.; Pierini, A.; Gori, E.; Bartoli, F.; Erba, P.; Ruggiero, P.; Marchetti, V. Serum amino acid profile in 51 dogs with immunosuppressant-responsive enteropathy (IRE): A pilot study on clinical aspects and outcomes. BMC Vet. Res. 2020, 16, 117. [Google Scholar] [CrossRef] [PubMed]
- Walker, H.K.; Boag, A.M.; Ottka, C.; Lohi, H.; Handel, I.; Gow, A.G.; Mellanby, R. Serum metabolomic profiles in dogs with chronic enteropathy. J. Vet. Intern Med. 2022, 36, 1752–1759. [Google Scholar] [CrossRef] [PubMed]
- Allenspach, K.; Culverwell, C.; Chan, D. Long-term outcome in dogs with chronic enteropathies: 203 cases. Vet. Rec. 2016, 178, 368. [Google Scholar] [CrossRef] [PubMed]
- Jergens, A.E.; Schreiner, C.A.; Frank, D.E.; Niyo, Y.; Ahrens, F.E.; Eckersall, P.D.; Benson, T.; Evans, R. A scoring index for disease activity in canine inflammatory bowel disease. J. Vet. Intern. Med. 2003, 17, 291–297. [Google Scholar] [CrossRef]
- Rey, A.I.; de-Cara, A.; Calvo, L.; Puig, P.; Hechavarría, T. Changes in plasma fatty acids, free amino acids, antioxidant defense, and physiological stress by oleuropein supplementation in pigs prior to slaughter. Antioxidants 2020, 9, 56. [Google Scholar] [CrossRef]
- Jones, B.N.; Pääbo, S.; Stein, S. Amino acid analysis and enzymatic sequence determination of peptides by an improved o-phthaldialdehyde precolumn labeling procedure. J. Liq. Chromatogr. 1981, 4, 565–586. [Google Scholar] [CrossRef]
- Kim, C.; Kovacs-Nolan, J.; Yang, C.; Archbold, T.; Fan, M.; Mine, Y. L-Tryptophan exhibits therapeutic function in a porcine model of dextran sodium sulfate (DSS)-induced colitis. J. Nutr. Biochem. 2010, 21, 468–475. [Google Scholar] [CrossRef]
- Farré, R.; Fiorani, M.; Abdu Rahiman, S.; Matteoli, G. Intestinal permeability, inflammation and the role of nutrients. Nutrients 2020, 12, 1185. [Google Scholar] [CrossRef]
- Hisamatsu, T.; Ono, N.; Imaizumi, A.; Mori, M.; Suzuki, H.; Uo, M.; Hashimoto, M.; Naganuma, M.; Matsuoka, K.; Mizuno, S.; et al. Decreased plasma histidine level predicts risk of relapse in patients with ulcerative colitis in remission. PLoS ONE 2015, 10, e0140716. [Google Scholar] [CrossRef]
- Kelly, B.; Pearce, E.L. Amino assets: How amino acids support immunity. Cell Metab. 2020, 32, 154–175. [Google Scholar] [CrossRef]
- Giaretta, P.R.; Rech, R.R.; Guard, B.C.; Blake, A.B.; Blick, A.K.; Steiner, J.M.; Lidbury, J.; Cook, A.; Hanifeh, M.; Spillmann, T.; et al. Comparison of intestinal expression of the apical sodium-dependent bile acid transporter between dogs with and without chronic inflammatory enteropathy. J. Vet. Intern. Med. 2018, 32, 1918–1926. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.S.; Lin, J.Y.; Chen, W.S.; Liu, M.H.; Cheng, C.W.; Cheng, M.L.; Wang, C. Phenylalanine- and leucine-defined metabolic types identify high mortality risk in patients with severe infection. Int. J. Infect. Dis. 2019, 85, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Mori, E.; Suzuki, H.; Hasebe, M.; Kobayashi, K. Alterations in levels of plasma phenylalanine and its catabolism in the liver of stressed rats. Metabolism 1992, 41, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.E. An overview of phenylalanine and tyrosine kinetics in humans. J. Nutr. 2007, 137, 1549S–1555S. [Google Scholar] [CrossRef]
- Dam, G.; Sørensen, M.; Buhl, M.; Sandahl, T.D.; Møller, N.; Ott, P.; Vilstrup, H. Muscle metabolism and whole blood amino acid profile in patients with liver disease. Scand. J. Clin. Lab. Investig. 2015, 75, 674–680. [Google Scholar]
- Nie, C.; He, T.; Zhang, W.; Zhang, G.; Ma, X. Branched Chain Amino Acids: Beyond nutrition metabolism. Int. J. Mol. Sci. 2018, 19, 954. [Google Scholar] [CrossRef]
- Holeček, M. Relation between glutamine, branched-chain amino acids, and protein metabolism. Nutrition 2002, 18, 130–133. [Google Scholar] [CrossRef]
- Eghtesad, S.; Poustchi, H.; Malekzadeh, R. Malnutrition in liver cirrhosis: The influence of protein and sodium. Middle East J. Dig. Dis. 2013, 5, 65–75. [Google Scholar]
- Mikkola, T.M.; Salonen, M.K.; Kajantie, E.; Kautiainen, H.; Eriksson, J.G. Associations of fat and lean body mass with circulating amino acids in older men and women. J. Gerontol. Ser. A 2020, 75, 885–891. [Google Scholar] [CrossRef]
- Lischka, J.; Schanzer, A.; Baumgartner, M.; de Gier, C.; Greber-Platzer, S.; Zeyda, M. Tryptophan metabolism is associated with BMI and adipose tissue mass and linked to metabolic disease in pediatric obesity. Nutrients 2022, 14, 286. [Google Scholar] [CrossRef] [PubMed]
- Short, K.R.; Chadwick, J.Q.; Teague, A.M.; Tullier, M.A.; Wolbert, L.; Coleman, C.; Copeland, K. Effect of obesity and exercise training on plasma amino acids and amino metabolites in american indian adolescents. J. Clin. Endocrinol. Metab. 2019, 104, 3249–3261. [Google Scholar] [CrossRef] [PubMed]
- Makanae, Y.; Fujita, S. Role of exercise and nutrition in the prevention of sarcopenia. J. Nutr. Sci. Vitaminol. 2015, 61, S125–S127. [Google Scholar] [CrossRef] [PubMed]
- Columbus, D.A.; Fiorotto, M.L.; Davis, T.A. Leucine is a major regulator of muscle protein synthesis in neonates. Amino Acids 2015, 47, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Escobar, J.; Frank, J.W.; Suryawan, A.; Nguyen, H.v.; Kimball, S.R.; Jefferson, L.S.; Davis, T. Physiological rise in plasma leucine stimulates muscle protein synthesis in neonatal pigs by enhancing translation initiation factor activation. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E914–E921. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.; Schmitt-Kopplin, P. The role of fecal sulfur metabolome in inflammatory bowel diseases. Int. J. Med. Microbiol. 2021, 311, 151513. [Google Scholar] [CrossRef]
- Ovalles, J.F. Estado del arte de la hidrólisis de proteínas y sinopsis sobre la determinación del perfil de aminoácidos. Una educación universitaria de calidad. In Sello Editorial Publicaciones del Vicerrectorado Académico—ULA Mérida; Rosenzweig Levy, P., Celis, M.T., Eds.; Universidad de Los Andes: Mérida, Venezuela, 2015; pp. 879–914. [Google Scholar]
- Balasubramanian, K.; Kumar, S.; Singh, R.R.; Sharma, U.; Ahuja, V.; Makharia, G.K.; Jagannathan, N. Metabolism of the colonic mucosa in patients with inflammatory bowel diseases: An in vitro proton magnetic resonance spectroscopy study. Magn. Reson. Imaging 2009, 27, 79–86. [Google Scholar] [CrossRef]
- Bjerrum, J.T.; Wang, Y.; Hao, F.; Coskun, M.; Ludwig, C.; Günther, U.; Nielsen, O. Metabonomics of human fecal extracts characterize ulcerative colitis, Crohn’s disease and healthy individuals. Metabolomics 2015, 11, 122–133. [Google Scholar] [CrossRef]
- Dawiskiba, T. Serum and urine metabolomic fingerprinting in diagnostics of inflammatory bowel diseases. World J. Gastroenterol. 2014, 20, 163. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, H.; Chen, T.; Shi, L.; Wang, D.; Tang, D. Regulatory role of short-chain fatty acids in inflammatory bowel disease. Cell Commun. Signal. 2022, 20, 64. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Gibson, G.R.; Beatty, E.; Cummings, J.H. Estimation of short-chain fatty acid production from protein by human intestinal bacteria based on branched-chain fatty acid measurements. FEMS Microbiol. Ecol. 1992, 10, 81–88. [Google Scholar] [CrossRef]
- Portune, K.J.; Beaumont, M.; Davila, A.M.; Tomé, D.; Blachier, F.; Sanz, Y. Gut microbiota role in dietary protein metabolism and health-related outcomes: The two sides of the coin. Trends Food Sci. Technol. 2016, 57, 213–232. [Google Scholar] [CrossRef]
| FRE | Range | Control | Range | |
|---|---|---|---|---|
| Average ± SD | Average ± SD | |||
| Age (years) | 6.2 ± 3.6 | (3–13) | 4.5 ± 2.1 | (3–6) |
| Male | 4 (2 entire, 2 castrated) | 3 (3 entire) | ||
| Female | 5 (1 entire, 4 spayed) | 3 (3 spayed) | ||
| Body weight | 11.4 ± 9.4 | (2.3–31) | 15.5 ± 0.7 | (15–16) |
| Body condition score (scale 9) | 4.2 ± 1.4 | (2–6) | 4.5 ± 0.40 | (4–5) |
| CIBDAI index | 7 ± 1.8 | (5–10) | 0 | (0–0) |
| % | Fat | α-Tocopherol | BCS | CIBDAI |
|---|---|---|---|---|
| Aspartic acid | −0.39 | 0.27 | 0.35 | 0.19 |
| Glutamic acid | −0.20 | 0.48 | 0.28 | −0.35 |
| Asparagine | 0.04 | 0.56 b | −0.39 | −0.32 |
| Serine | 0.41 | −0.07 | −0.14 | −0.07 |
| Glutamine | −0.30 | 0.26 | 0.36 | 0.15 |
| Histidine | 0.23 | 0.15 | −0.30 | −0.38 |
| Glycine | 0.20 | 0.51 b | 0.20 | −0.47 |
| Threonine | 0.35 | −0.02 | −0.22 | −0.12 |
| Arginine | −0.08 | 0.65 a | −0.02 | −0.42 |
| Alanine | 0.15 | −0.49 b | −0.22 | 0.20 |
| Tyrosine | −0.25 | 0.08 | −0.29 | 0.41 |
| Cystine | −0.13 | 0.60 b | 0.28 | −0.40 |
| Valine | −0.13 | −0.03 | 0.28 | −0.25 |
| Methionine | 0.10 | 0.18 | −0.39 | 0.08 |
| Tryptophan | −0.58 b | 0.06 | 0.36 | 0.32 |
| Phenylalanine | −0.50 b | 0.03 | 0.14 | 0.53 b |
| Isoleucine | −0.08 | 0.32 | 0.12 | −0.37 |
| Leucine | 0.15 | 0.38 | 0.48 b | −0.69 a |
| Lysine | 0.04 | 0.36 | 0.43 | −0.59 b |
| BCAA 1 | 0.00 | 0.26 | 0.33 | −0.49 b |
| AAA 2 | −0.32 | 0.08 | −0.24 | 0.47 |
| BCAA/AAA | 0.40 | 0.03 | 0.21 | −0.67 a |
| % Faeces | % Faecal Fat | % Faecal Humidity | BCS | CIBDAI |
|---|---|---|---|---|
| Aspartic acid | 0.37 | 0.07 | −0.18 | −0.12 |
| Glutamic acid | 0.25 | −0.05 | −0.02 | 0.22 |
| Serine | 0.45 | 0.48 | −0.21 | 0.52 |
| Histidine | −0.13 | −0.38 | 0.33 | −0.09 |
| Glycine | 0.26 | 0.59 b | −0.39 | 0.40 |
| Threonine | 0.41 | 0.29 | −0.03 | 0.15 |
| Arginine | 0.29 | 0.16 | 0.40 | 0.23 |
| Alanine | −0.24 | 0.27 | 0.13 | 0.15 |
| Tyrosine | −0.39 | 0.32 | −0.04 | 0.41 |
| Cystine | 0.35 | 0.35 | 0.22 | 0.48 |
| Valine | −0.43 | −0.21 | 0.47 | 0.13 |
| Methionine | −0.10 | −0.34 | 0.31 | −0.1 |
| Phenylalanine | −0.62 b | −0.53 | 0.28 | −0.45 |
| Isoleucine | −0.57 | −0.47 | 0.44 | −0.19 |
| Leucine | −0.57 b | −0.66 b | 0.44 | −0.46 |
| Lysine | −0.65 b | −0.43 | −0.02 | −0.39 |
| % Faeces | ∑SCFAs | C2 | C3 | IC4 | C4 | IC5 | C5 |
|---|---|---|---|---|---|---|---|
| Aspartic acid | −0.42 | −0.12 | −0.11 | −0.14 | −0.54 | 0.02 | −0.24 |
| Glutamic acid | 0.04 | −0.55 | −0.24 | 0.04 | 0.37 | 0.29 | 0.39 |
| Serine | −0.49 | −0.58 | −0.53 | 0.10 | −0.03 | −0.47 | −0.10 |
| Histidine | 0.25 | −0.33 | 0.05 | 0.64 b | 0.39 | 0.13 | 0.24 |
| Glycine | −0.85 a | −0.10 | −0.48 | −0.22 | −0.78 a | −0.59 b | −0.61 b |
| Threonine | 0.01 | −0.24 | 0.07 | −0.19 | 0.25 | 0.14 | 0.01 |
| Arginine | 0.27 | 0.12 | −0.04 | −0.36 | 0.42 | −0.24 | 0.32 |
| Alanine | 0.14 | 0.53 | 0.19 | −0.16 | 0.05 | −0.23 | −0.26 |
| Tyrosine | −0.15 | 0.24 | −0.29 | −0.29 | −0.01 | −0.59 b | −0.13 |
| Cystine | −0.61 b | −0.42 | −0.53 | −0.40 | −0.23 | −0.60 b | −0.16 |
| Valine | 0.40 | −0.05 | −0.01 | 0.07 | 0.67 b | −0.24 | 0.38 |
| Methionine | 0.53 | −0.24 | 0.25 | 0.23 | 0.68 b | 0.34 | 0.50 |
| Phenylalanine | 0.80 a | 0.41 | 0.62 b | 0.39 | 0.56 | 0.50 | 0.26 |
| Isoleucine | 0.75 a | 0.22 | 0.31 | 0.06 | 0.77 a | 0.19 | 0.54 |
| Leucine | 0.92 a | 0.24 | 0.56 | 0.22 | 0.80 a | 0.47 | 0.59 b |
| Lysine | 0.51 | 0.48 | 0.42 | 0.54 | 0.12 | 0.30 | 0.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Higueras, C.; Escudero, R.; Rebolé, A.; García-Sancho, M.; Rodríguez-Franco, F.; Sainz, Á.; Rey, A.I. Changes in Faecal and Plasma Amino Acid Profile in Dogs with Food-Responsive Enteropathy as Indicators of Gut Homeostasis Disruption: A Pilot Study. Vet. Sci. 2023, 10, 112. https://doi.org/10.3390/vetsci10020112
Higueras C, Escudero R, Rebolé A, García-Sancho M, Rodríguez-Franco F, Sainz Á, Rey AI. Changes in Faecal and Plasma Amino Acid Profile in Dogs with Food-Responsive Enteropathy as Indicators of Gut Homeostasis Disruption: A Pilot Study. Veterinary Sciences. 2023; 10(2):112. https://doi.org/10.3390/vetsci10020112
Chicago/Turabian StyleHigueras, Cristina, Rosa Escudero, Almudena Rebolé, Mercedes García-Sancho, Fernando Rodríguez-Franco, Ángel Sainz, and Ana I. Rey. 2023. "Changes in Faecal and Plasma Amino Acid Profile in Dogs with Food-Responsive Enteropathy as Indicators of Gut Homeostasis Disruption: A Pilot Study" Veterinary Sciences 10, no. 2: 112. https://doi.org/10.3390/vetsci10020112
APA StyleHigueras, C., Escudero, R., Rebolé, A., García-Sancho, M., Rodríguez-Franco, F., Sainz, Á., & Rey, A. I. (2023). Changes in Faecal and Plasma Amino Acid Profile in Dogs with Food-Responsive Enteropathy as Indicators of Gut Homeostasis Disruption: A Pilot Study. Veterinary Sciences, 10(2), 112. https://doi.org/10.3390/vetsci10020112









