Multidrug-Resistant Staphylococcus aureus Strains Thrive in Dairy and Beef Production, Processing, and Supply Lines in Five Geographical Areas in Ethiopia
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Dairy Farms and Abattoirs: Sampling, Sample Types, and Sample Handling
2.3. Bacteriological Isolation and Identification
2.4. Antimicrobial Susceptibility Testing
Multiple Antimicrobial Resistance Index (MARI) for Ranking Isolates and Ecologies
2.5. Data Analysis
3. Results
3.1. Prevalence of S. aureus
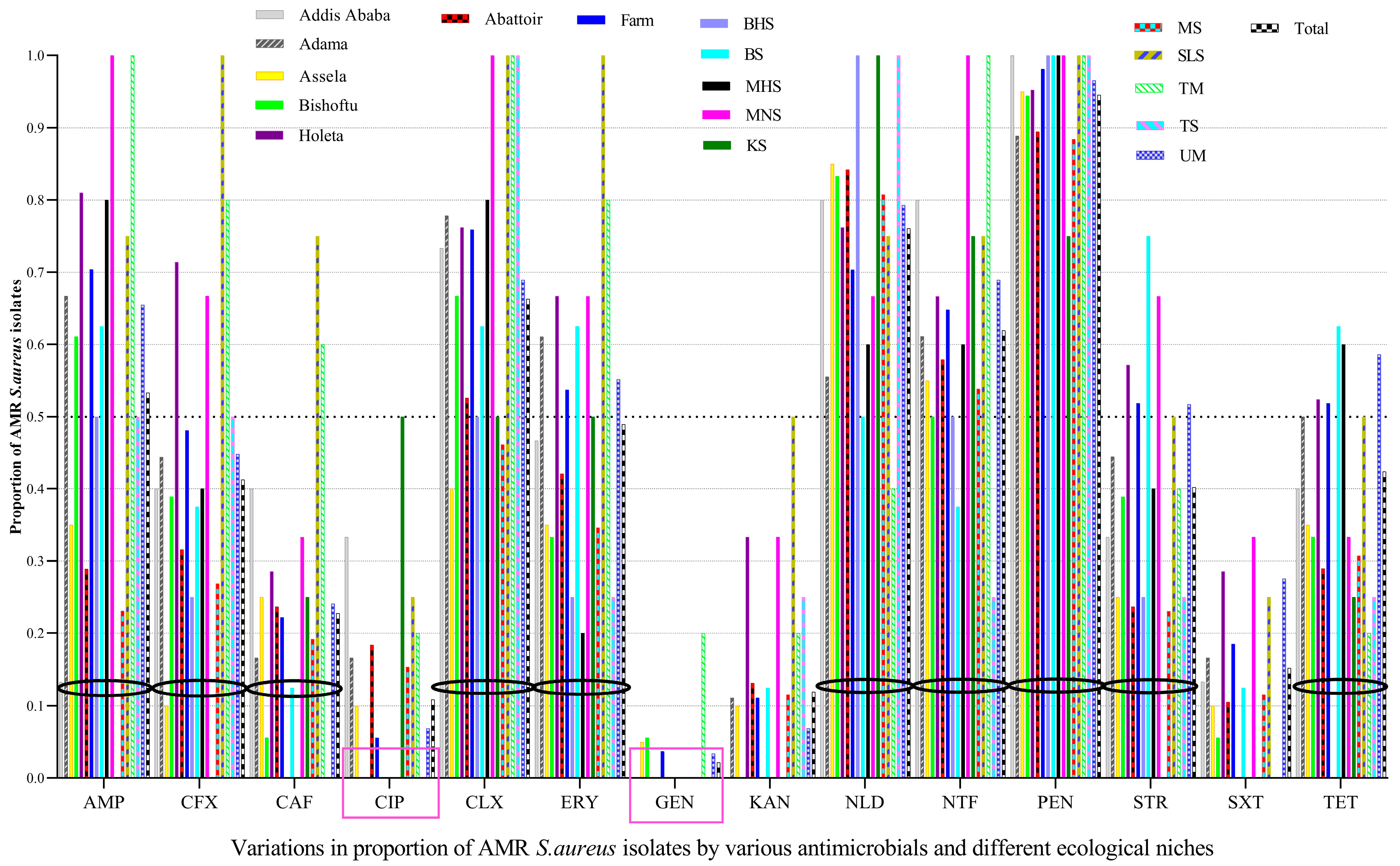
3.2. Prevalence of Antimicrobial Resistance
3.3. MRSA vs. MSSA Isolate Detection by Cefoxitin (FOX) Disc
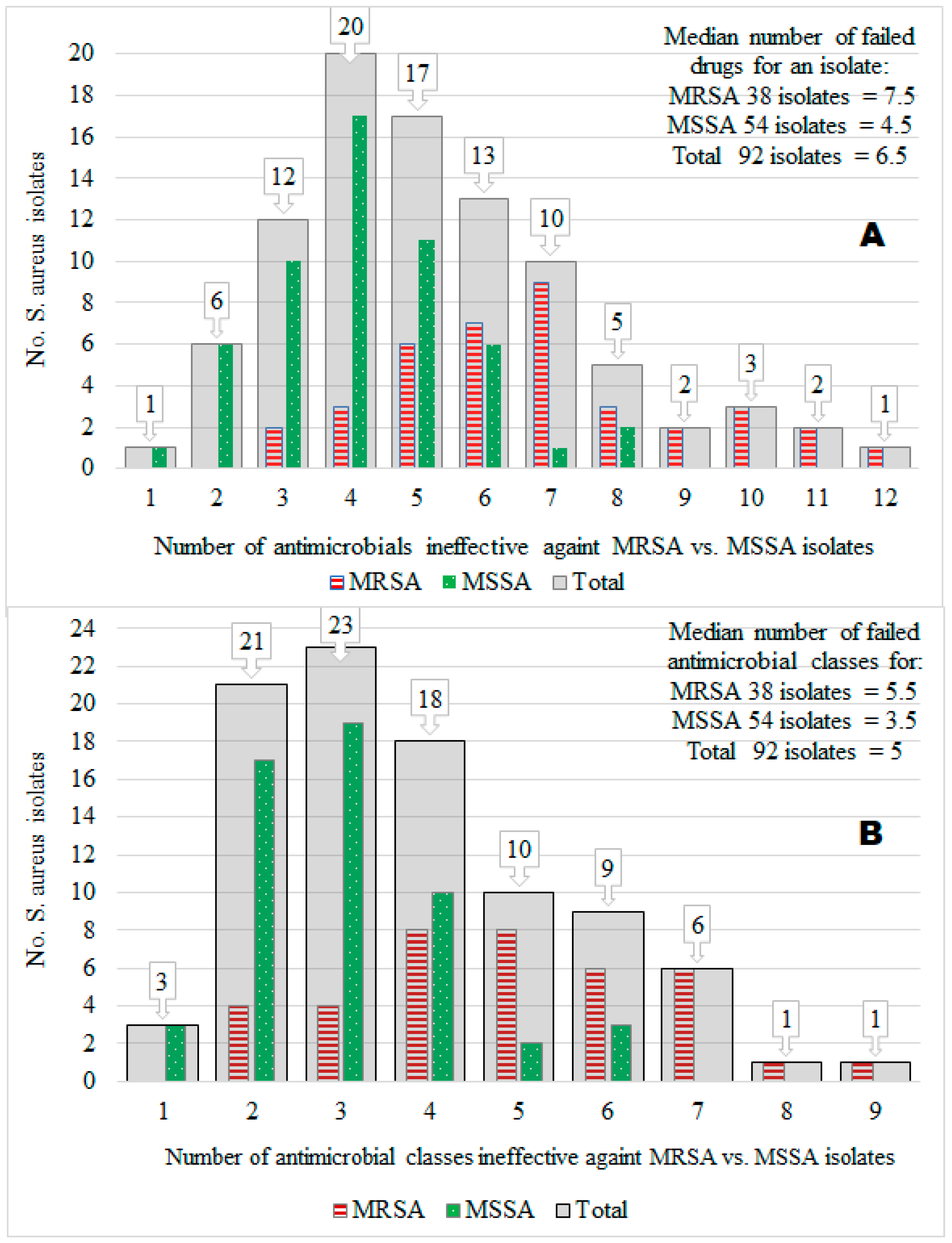
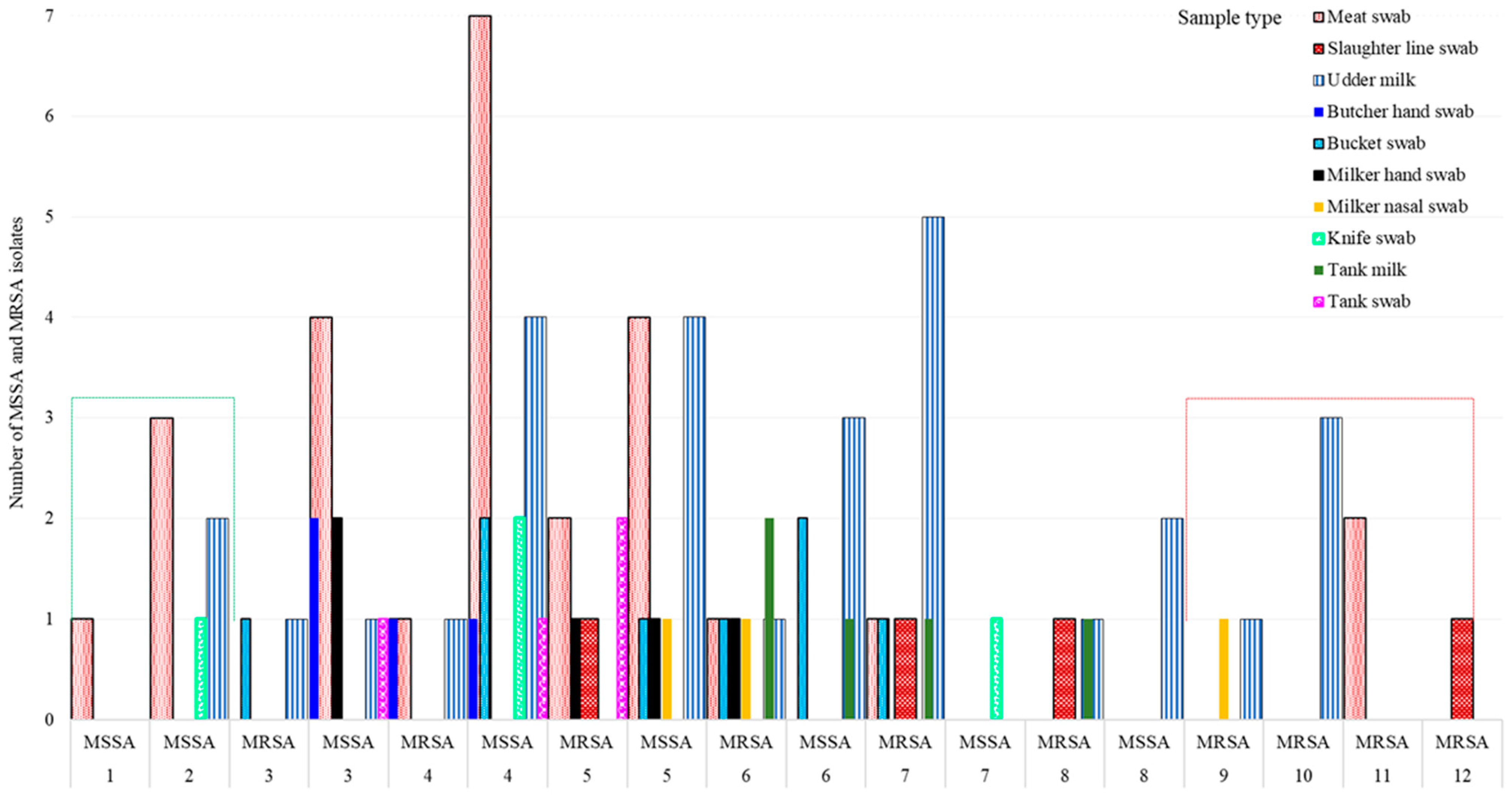
3.4. Multidrug Resistance (MDR)
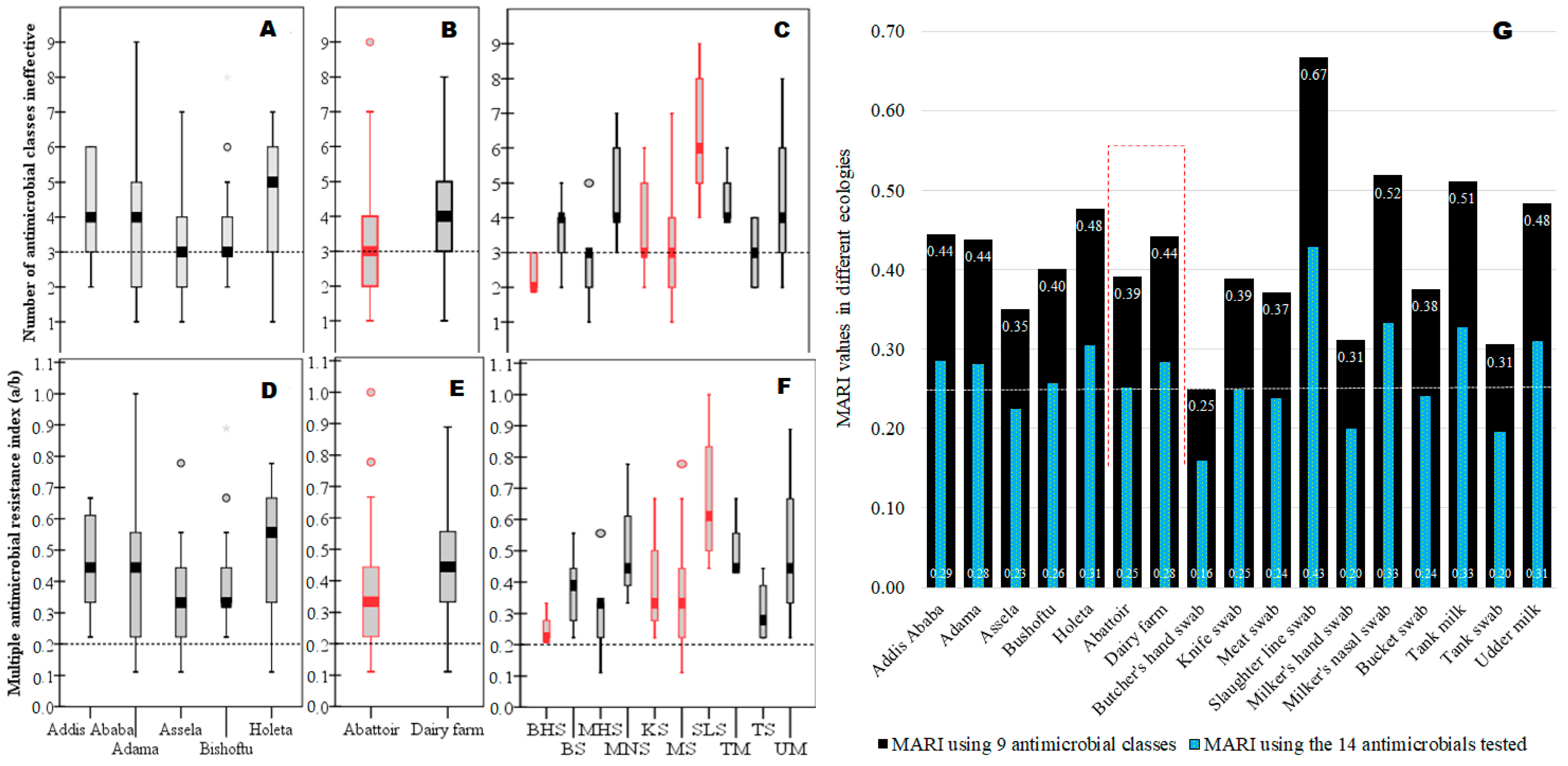
3.5. MARI for “High-Risk” Source Determination
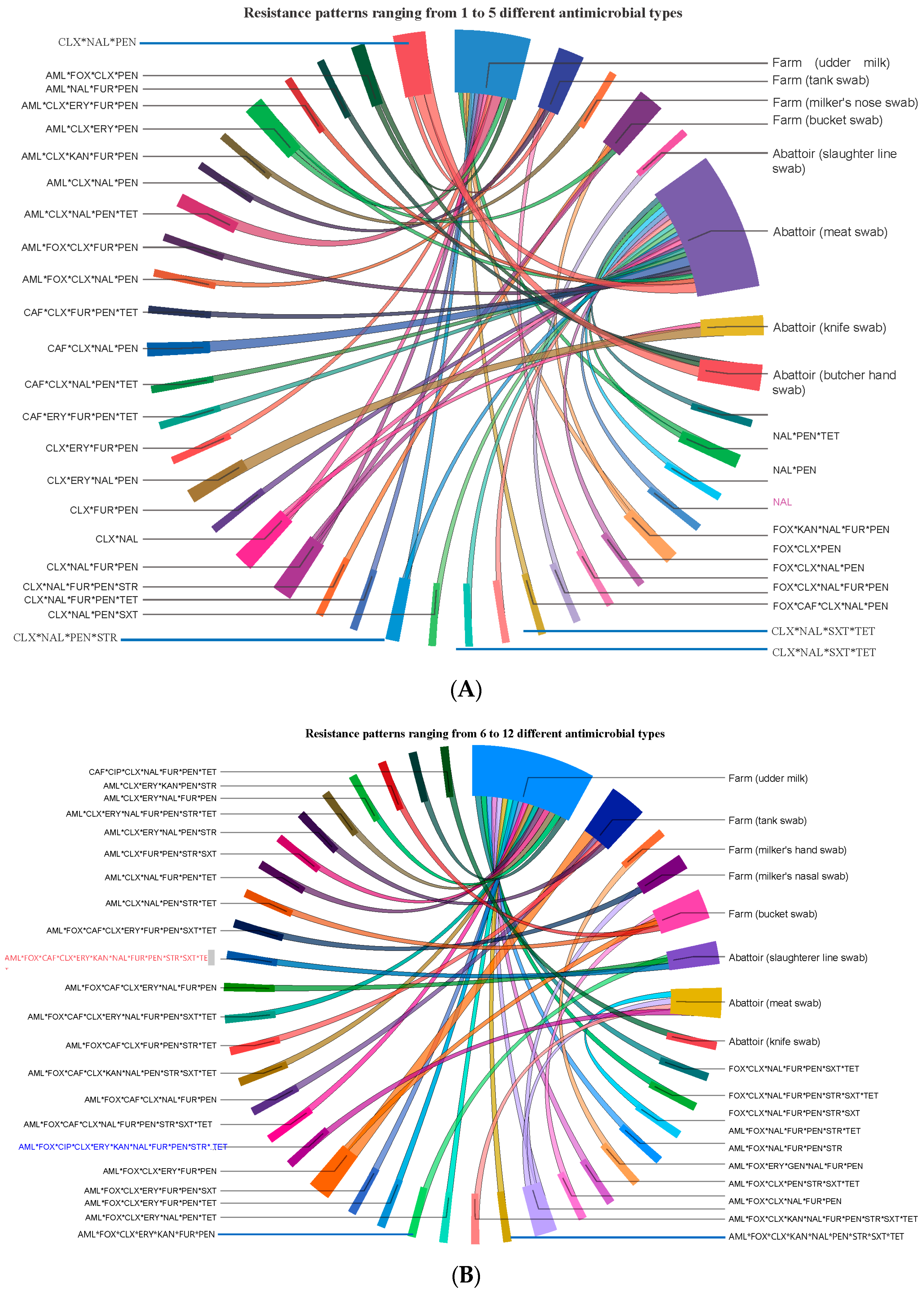
3.6. Antimicrobial Resistance Pattern
4. Discussion
4.1. Antimicrobial Resistance
4.2. MDR and AMR Patterns in Different Sample Sources
4.3. Methicillin Resistance
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fluit, A.C. Livestock-associated Staphylococcus aureus. Clin. Microbiol. Infect. 2012, 18, 735–744. [Google Scholar] [CrossRef]
- Ge, B.; Mukherjee, S.; Hsu, C.-H.; Davis, J.A.; Tran, T.T.T.; Yang, Q.; Abbott, J.W.; Ayers, S.L.; Young, S.R.; Crarey, E.T.; et al. MRSA and multidrug-resistant Staphylococcus aureus in U.S. retail meats, 2010–2011. Food Microbiol. 2017, 62, 289–297. [Google Scholar] [CrossRef]
- Al-Ashmawy, M.A.; Sallam, K.I.; Abd-Elghany, S.M.; Elhadidy, M.; Tamura, T. Prevalence, Molecular Characterization, and Antimicrobial Susceptibility of Methicillin-Resistant Staphylococcus aureus Isolated from Milk and Dairy Products. Foodborne Pathog. Dis. 2016, 13, 156–162. [Google Scholar] [CrossRef]
- Seo, K.S.; Bohach, G.A. Staphylococcal food poisoning. In Pathogens and Toxins in Foods; American Society of Microbiology: Washington, DC, USA, 2010; pp. 119–130. [Google Scholar]
- Klein, E.Y.; Van Boeckel, T.P.; Martinez, E.M.; Pant, S.; Gandra, S.; Levin, S.A.; Goossens, H.; Laxminarayan, R. Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc. Natl. Acad. Sci. USA 2018, 115, E3463. [Google Scholar] [CrossRef]
- Ahmad, M.; Khan, A.U. Global economic impact of antibiotic resistance: A review. J. Glob. Antimicrob. Resist. 2019, 19, 313–316. [Google Scholar] [CrossRef]
- Shallcross, L.J.; Howard, S.J.; Fowler, T.; Davies, S.C. Tackling the threat of antimicrobial resistance: From policy to sustainable action. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140082. [Google Scholar] [CrossRef]
- Anderson, R.M. The pandemic of antibiotic resistance. Nat. Med. 1999, 5, 147–149. [Google Scholar] [CrossRef]
- Muhie, O.A. Antibiotic Use and Resistance Pattern in Ethiopia: Systematic Review and Meta-Analysis. Int. J. Microbiol. 2019, 2019, 2489063. [Google Scholar] [CrossRef]
- Yadesa, T.M.; Gudina, E.K.; Angamo, M.T. Antimicrobial use-related problems and predictors among hospitalized medical in-patients in Southwest Ethiopia: Prospective observational study. PLoS ONE 2015, 10, e0138385. [Google Scholar] [CrossRef]
- Erku, D.A.; Mekuria, A.B.; Belachew, S.A. Inappropriate use of antibiotics among communities of Gondar town, Ethiopia: A threat to the development of antimicrobial resistance. Antimicrob. Resist. Infect. Control. 2017, 6, 112. [Google Scholar] [CrossRef]
- Beyene, T.; Assefa, S.; Ayana, D.; Beyene, T.J.; Tadesse, F.; Woldemichael, D.N. Assessment of rational veterinary drugs use in livestock at Adama district veterinary clinic, central Ethiopia. J. Vet. Sci. Technol. 2016, 7, 319–326. [Google Scholar] [CrossRef]
- Beyene, T.; Endalamaw, D.; Tolossa, Y.; Feyisa, A. Evaluation of rational use of veterinary drugs especially antimicrobials and anthelmintics in Bishoftu, Central Ethiopia. BMC Res. Notes 2015, 8, 482. [Google Scholar] [CrossRef]
- Suleman, S.; Woliyi, A.; Woldemichael, K.; Tushune, K.; Duchateau, L.; Degroote, A.; Vancauwenberghe, R.; Bracke, N.; De Spiegeleer, B. Pharmaceutical regulatory framework in Ethiopia: A critical evaluation of its legal basis and implementation. Ethiop. J. Health Sci. 2016, 26, 259–276. [Google Scholar] [CrossRef]
- Moges, F.; Endris, M.; Mulu, A.; Tessema, B.; Belyhun, Y.; Shiferaw, Y.; Huruy, K.; Unakal, C.; Kassu, A. The growing challenges of antibacterial drug resistance in Ethiopia. J. Glob. Antimicrob. Resist. 2014, 2, 148–154. [Google Scholar] [CrossRef]
- Abera, B.; Kibret, M.; Mulu, W. Knowledge and beliefs on antimicrobial resistance among physicians and nurses in hospitals in Amhara Region, Ethiopia. BMC Pharmacol. Toxicol. 2014, 15, 26. [Google Scholar] [CrossRef]
- Gebretekle, G.B.; Serbessa, M.K. Exploration of over the counter sales of antibiotics in community pharmacies of Addis Ababa, Ethiopia: Pharmacy professionals’ perspective. Antimicrob. Resist. Infect. Control. 2016, 5, 2. [Google Scholar] [CrossRef]
- Deyno, S.; Fekadu, S.; Astatkie, A. Resistance of Staphylococcus aureus to antimicrobial agents in Ethiopia: A meta-analysis. Antimicrob. Resist. Infect. Control. 2017, 6, 85. [Google Scholar] [CrossRef]
- Eshetie, S.; Tarekegn, F.; Moges, F.; Amsalu, A.; Birhan, W.; Huruy, K. Methicillin resistant Staphylococcus aureus in Ethiopia: A meta-analysis. BMC Infect. Dis. 2016, 16, 689. [Google Scholar] [CrossRef]
- Scott, T.M.; Rose, J.B.; Jenkins, T.M.; Farrah, S.R.; Lukasik, J. Microbial Source Tracking: Current Methodology and Future Directions. Appl. Environ. Microbiol. 2002, 68, 5796. [Google Scholar] [CrossRef]
- Silbergeld, E.K.; Graham, J.; Price, L.B. Industrial food animal production, antimicrobial resistance, and human health. Annu. Rev. Public Health 2008, 29, 151–169. [Google Scholar] [CrossRef]
- Van Boeckel, T.P.; Brower, C.; Gilbert, M.; Grenfell, B.T.; Levin, S.A.; Robinson, T.P.; Teillant, A.; Laxminarayan, R. Global trends in antimicrobial use in food animals. Proc. Natl. Acad. Sci. USA 2015, 112, 5649. [Google Scholar] [CrossRef]
- Liu, Y.; Han, C.; Chen, Z.; Guo, D.; Ye, X. Relationship between livestock exposure and methicillin-resistant Staphylococcus aureus carriage in humans: A systematic review and dose–response meta-analysis. Int. J. Antimicrob. Agents 2020, 55, 105810. [Google Scholar] [CrossRef]
- Smith, T.C. Livestock-associated Staphylococcus aureus: The United States experience. PLoS Path 2015, 11, e1004564. [Google Scholar] [CrossRef]
- Alemu, Y.F.; Jemberu, W.T.; Mekuriaw, Z.; Abdi, R.D. Incidence and Predictors of Calf Morbidity and Mortality from Birth to 6-Months of Age in Dairy Farms of Northwestern Ethiopia. Front. Vet. Sci. 2022, 9, 859401. [Google Scholar] [CrossRef]
- Headey, D.; Nguyen, P.; Kim, S.; Rawat, R.; Ruel, M.; Menon, P. Is Exposure to Animal Feces Harmful to Child Nutrition and Health Outcomes? A Multicountry Observational Analysis. Am. J. Trop. Med. Hyg. 2017, 96, 961–969. [Google Scholar] [CrossRef]
- Seleshe, S.; Jo, C.; Lee, M. Meat consumption culture in Ethiopia. Korean J. Food. Sci. Anim. Resour. 2014, 34, 7–13. [Google Scholar] [CrossRef]
- Gutema, F.D.; Agga, G.E.; Abdi, R.D.; Jufare, A.; Duchateau, L.; De Zutter, L.; Gabriël, S. Assessment of Hygienic Practices in Beef Cattle Slaughterhouses and Retail Shops in Bishoftu, Ethiopia: Implications for Public Health. Int. J. Environ. Res. Public Health 2021, 18, 2729. [Google Scholar] [CrossRef]
- Nyokabi, N.S.; Phelan, L.; Gemechu, G.; Berg, S.; Lindahl, J.F.; Mihret, A.; Wood, J.L.N.; Moore, H.L. From farm to table: Exploring food handling and hygiene practices of meat and milk value chain actors in Ethiopia. BMC Public Health 2023, 23, 899. [Google Scholar] [CrossRef]
- Zeaki, N.; Johler, S.; Skandamis, P.N.; Schelin, J. The Role of Regulatory Mechanisms and Environmental Parameters in Staphylococcal Food Poisoning and Resulting Challenges to Risk Assessment. Front. Microbiol. 2019, 10, 1307. [Google Scholar] [CrossRef]
- Tiwari, A. Themes in urban infrastructure research in Ethiopian cities. In Urban Infrastructure Research: A Review of Ethiopian Cities; Tiwari, A., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 7–34. [Google Scholar]
- Harrigan, W.F. Laboratory Methods in Food Microbiology, 3rd ed.; Academic Press: San Diego, CA, USA, 1998; p. 532. [Google Scholar]
- Bautista-Trujillo, G.U.; Solorio-Rivera, J.L.; Rentería-Solórzano, I.; Carranza-Germán, S.I.; Bustos-Martínez, J.A.; Arteaga-Garibay, R.I.; Baizabal-Aguirre, V.M.; Cajero-Juárez, M.; Bravo-Patiño, A.; Valdez-Alarcón, J.J. Performance of culture media for the isolation and identification of Staphylococcus aureus from bovine mastitis. J. Med. Microbiol. 2013, 62, 369–376. [Google Scholar] [CrossRef]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 32nd ed.; CLSI supplement M100; CLSI: Wayne, PE, USA, 2022; Volume 42. [Google Scholar]
- Swenson, J.M.; Tenover, F.C. Results of Disk Diffusion Testing with Cefoxitin Correlate with Presence of mecA in Staphylococcus spp. J. Clin. Microbiol. 2005, 43, 3818. [Google Scholar] [CrossRef]
- Broekema, N.M.; Van, T.T.; Monson, T.A.; Marshall, S.A.; Warshauer, D.M. Comparison of cefoxitin and oxacillin disk diffusion methods for detection of mecA-mediated resistance in Staphylococcus aureus in a large-scale study. J. Clin. Microbiol. 2009, 47, 217. [Google Scholar] [CrossRef]
- Magiorakos, A.P.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, C.G.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Liljequist, B.; et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef]
- Krumperman, P.H. Multiple antibiotic resistance indexing of Escherichia coli to identify high-risk sources of fecal contamination of foods. Appl. Environ. Microbiol. 1983, 46, 165. [Google Scholar] [CrossRef]
- Woh, P.Y.; Yeung, M.P.S.; Goggins, W.B., III. Multiple antibiotic resistance index (MARI) of human-isolated Salmonella species: A practical bacterial antibiotic surveillance tool. J. Antimicrob. Chemother. 2023, 78, 1295–1299. [Google Scholar] [CrossRef]
- Chitanand, M.P.; Kadam, T.A.; Gyananath, G.; Totewad, N.D.; Balhal, D.K. Multiple antibiotic resistance indexing of coliforms to identify high risk contamination sites in aquatic environment. Indian J. Microbiol. 2010, 50, 216–220. [Google Scholar] [CrossRef]
- Davis, R.; Brown, P.D. Multiple antibiotic resistance index, fitness and virulence potential in respiratory Pseudomonas aeruginosa from Jamaica. J. Med. Microbiol. 2016, 65, 261–271. [Google Scholar] [CrossRef]
- Chen, C.; Wu, Y.; Xia, R. A painless way to customize Circos plot: From data preparation to visualization using TBtools. iMeta 2022, 1, e35. [Google Scholar] [CrossRef]
- Graveland, H.; Duim, B.; van Duijkeren, E.; Heederik, D.; Wagenaar, J.A. Livestock-associated methicillin-resistant Staphylococcus aureus in animals and humans. Int. J. Med. Microbiol. 2011, 301, 630–634. [Google Scholar] [CrossRef]
- Grundmann, H.; Aires-de-Sousa, M.; Boyce, J.; Tiemersma, E. Emergence and resurgence of meticillin-resistant Staphylococcus aureus as a public-health threat. Lancet 2006, 368, 874–885. [Google Scholar] [CrossRef]
- Johnson, A.P. Methicillin-resistant Staphylococcus aureus: The European landscape. J. Antimicrob. Chemother. 2011, 66, iv43–iv48. [Google Scholar] [CrossRef]
- Daka, D.; Gsilassie, S.; Yihdego, D. Antibiotic-resistance Staphylococcus aureus isolated from cow’s milk in the Hawassa area, South Ethiopia. Ann. Clin. Microbiol. 2012, 11, 26. [Google Scholar] [CrossRef]
- Haftu, R.; Taddele, H.; Gugsa, G.; Kalayou, S. Prevalence, bacterial causes, and antimicrobial susceptibility profile of mastitis isolates from cows in large-scale dairy farms of Northern Ethiopia. Trop. Anim. Health Prod. 2012, 44, 1765–1771. [Google Scholar] [CrossRef]
- Guven, K.; Mutlu, M.B.; Gulbandilar, A.; Cakir, P. Occurrence and characterization of Staphylococcus aureus isolated from meat and dairy products consumed in Italy. J. Food Saf. 2010, 30, 196–212. [Google Scholar] [CrossRef]
- Saini, V.; McClure, J.T.; Léger, D.; Dufour, S.; Sheldon, A.G.; Scholl, D.T.; Barkema, H.W. Antimicrobial use on Canadian dairy farms. J. Dairy Sci. 2012, 95, 1209–1221. [Google Scholar] [CrossRef]
- Saini, V.; McClure, J.T.; Scholl, D.T.; DeVries, T.J.; Barkema, H.W. Herd-level association between antimicrobial use and antimicrobial resistance in bovine mastitis Staphylococcus aureus isolates on Canadian dairy farms. J. Dairy Sci. 2012, 95, 1921–1929. [Google Scholar] [CrossRef]
- Auguet, O.T.; Betley, J.R.; Stabler, R.A.; Patel, A.; Ioannou, A.; Marbach, H.; Hearn, P.; Aryee, A.; Goldenberg, S.D.; Otter, J.A.; et al. Evidence for community transmission of community-associated but not health-care-associated methicillin-resistant Staphylococcus aureus strains linked to social and material deprivation: Spatial analysis of cross-sectional aata. PLoS Med. 2016, 13, e1001944. [Google Scholar] [CrossRef]
- Pol, M.; Ruegg, P.L. Relationship between antimicrobial drug usage and antimicrobial susceptibility of gram-positive mastitis pathogens. J. Dairy Sci. 2007, 90, 262–273. [Google Scholar] [CrossRef]
- Jørgensen, H.J.; Mørk, T.; Høgåsen, H.R.; Rørvik, L.M. Enterotoxigenic Staphylococcus aureus in bulk milk in Norway. J. Appl. Microbiol. 2005, 99, 158–166. [Google Scholar] [CrossRef]
- Jamali, H.; Paydar, M.; Radmehr, B.; Ismail, S.; Dadrasnia, A. Prevalence and antimicrobial resistance of Staphylococcus aureus isolated from raw milk and dairy products. Food Control 2015, 54, 383–388. [Google Scholar] [CrossRef]
- Haileselassie, M.; Taddele, H.; Adhana, K.; Kalayou, S. Food safety knowledge and practices of abattoir and butchery shops and the microbial profile of meat in Mekelle City, Ethiopia. Asian Pac. J. Trop. Biomed. 2013, 3, 407–412. [Google Scholar] [CrossRef]
- Waters, A.E.; Contente-Cuomo, T.; Buchhagen, J.; Liu, C.M.; Watson, L.; Pearce, K.; Foster, J.T.; Bowers, J.; Driebe, E.M.; Engelthaler, D.M.; et al. Multidrug-Resistant Staphylococcus aureus in US Meat and Poultry. Clin. Infect. Dis. 2011, 52, 1227–1230. [Google Scholar] [CrossRef]
- Pesavento, G.; Ducci, B.; Comodo, N.; Nostro, A.L. Antimicrobial resistance profile of Staphylococcus aureus isolated from raw meat: A research for methicillin resistant Staphylococcus aureus (MRSA). Food Control. 2007, 18, 196–200. [Google Scholar] [CrossRef]
- Capurro, A.; Aspán, A.; Unnerstad, H.E.; Waller, K.P.; Artursson, K. Identification of potential sources of Staphylococcus aureus in herds with mastitis problems. J. Dairy Sci. 2010, 93, 180–191. [Google Scholar] [CrossRef]
- Roberson, J.R.; Fox, L.K.; Hancock, D.D.; Gay, J.M.; Besser, T.E. Sources of Intramammary Infections from Staphylococcus aureus in Dairy Heifers at First Parturition1. J. Dairy Sci. 1998, 81, 687–693. [Google Scholar] [CrossRef]
- Wertheim, H.F.L.; Melles, D.C.; Vos, M.C.; van Leeuwen, W.; van Belkum, A.; Verbrugh, H.A.; Nouwen, J.L. The role of nasal carriage in Staphylococcus aureus infections. Lancet Infect. Dis. 2005, 5, 751–762. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, C.-M.; Lu, L.-M.; Ren, G.-W.N.; Cao, X.-Y.; Shen, J.-Z. Macrolide–lincosamide-resistant phenotypes and genotypes of Staphylococcus aureus isolated from bovine clinical mastitis. Vet. Microbiol. 2008, 130, 118–125. [Google Scholar] [CrossRef]
- Abdi, R.D.; Gillespie, B.E.; Vaughn, J.; Merrill, C.; Headrick, S.I.; Ensermu, D.B.; D’Souza, D.H.; Agga, G.E.; Almeida, R.A.; Oliver, S.P.; et al. Antimicrobial resistance of Staphylococcus aureus isolates from dairy cows and genetic diversity of resistant isolates. Foodborne Pathog. Dis. 2018, 15, 449–458. [Google Scholar] [CrossRef]
- Oliveira, L.; Langoni, H.; Hulland, C.; Ruegg, P.L. Minimum inhibitory concentrations of Staphylococcus aureus recovered from clinical and subclinical cases of bovine mastitis. J. Dairy Sci. 2012, 95, 1913–1920. [Google Scholar] [CrossRef]
- Parhizgari, N.; Khoramrooz, S.S.; Hosseini, S.A.A.M.; Marashifard, M.; Yazdanpanah, M.; Emaneini, M.; Gharibpour, F.; Mirzaii, M.; Darban-Sarokhalil, D.; Moein, M.; et al. High frequency of multidrug-resistant Staphylococcus aureus with SCCmec type III and Spa types t037 and t631 isolated from burn patients in southwest of Iran. APMIS 2015, 124, 221–228. [Google Scholar] [CrossRef]
- Hendriksen, R.S.; Mevius, D.J.; Schroeter, A.; Teale, C.; Meunier, D.; Butaye, P.; Franco, A.; Utinane, A.; Amado, A.; Moreno, M.; et al. Prevalence of antimicrobial resistance among bacterial pathogens isolated from cattle in different European countries: 2002–2004. Acta Vet. Scand. 2008, 50, 28. [Google Scholar] [CrossRef]
- Kérouanton, A.; Hennekinne, J.A.; Letertre, C.; Petit, L.; Chesneau, O.; Brisabois, A.; De Buyser, M.L. Characterization of Staphylococcus aureus strains associated with food poisoning outbreaks in France. Int. J. Food Microbiol. 2007, 115, 369–375. [Google Scholar] [CrossRef]
- Okeke, I.N.; Lamikanra, A.; Edelman, R. Socioeconomic and behavioral factors leading to acquired bacterial resistance to antibiotics in developing countries. Emerg. Infect. Dis. 1999, 5, 18–27. [Google Scholar] [CrossRef]
- Guzmán-Blanco, M.; Mejía, C.; Isturiz, R.; Alvarez, C.; Bavestrello, L.; Gotuzzo, E.; Labarca, J.; Luna, C.M.; Rodríguez-Noriega, E.; Salles, M.J.C.; et al. Epidemiology of meticillin-resistant Staphylococcus aureus (MRSA) in Latin America. Int. J. Antimicrob. Agents 2009, 34, 304–308. [Google Scholar] [CrossRef]
| Nine Antimicrobial Classes | 14 Antimicrobial Agents (Potency) | Susceptibility Breakpoint (mm) |
|---|---|---|
| 1. Aminoglycosides | Gentamicin, GEN (10 µg) | ≥15 |
| Kanamycin, KAN (30 µg) | ≥18 | |
| Streptomycin, STR (10 µg) | ≥15 | |
| 2. Cephalosporins (b-lactams) | Cefoxitin, FOX (30 µg) | ≥22 |
| 3. Folate pathway inhibitors | Sulfamethoxazole/Trimethoprim, SXT (25 µg) | ≥16 |
| 4. Macrolides | Erythromycin, ERY (15 µg) | ≥23 |
| 5. Nitrofurans | Nitrofurantoin, FUR (50 µg) | ≥17 |
| 6. Penicillins | Amoxicillin, AML (25 µg) | ≥20 |
| Cloxacillin, CLX (5 µg) | 13 | |
| Penicillin G, PEN (10 units) | ≥29 | |
| 7. Phenicols | Chloramphenicol, CAF (30 µg) | ≥18 |
| 8. Quinolones | Ciprofloxacin, CIP (5 µg) | ≥21 |
| Nalidixic acid, NAL (30 µg) | ≥19 | |
| 9. Tetracyclines | Tetracycline, TET (30 µg) | ≥19 |
| Origin | Sample Source | No. | Positive | Prevalence (%) | χ2 | p-Value |
|---|---|---|---|---|---|---|
| Livestock setting | Dairy farm | 514 | 115 | 22.4 | 20.00 | 0.000 |
| Abattoir | 487 | 57 | 11.7 | |||
| Sample types from the abattoir | Butcher’s hand swab | 37 | 7 | 18.9 | 26.32 | 0.03 |
| Slaughter line swab | 37 | 7 | 18.9 | |||
| Abattoir knife swab | 37 | 5 | 13.5 | |||
| Carcass/meat swab | 361 | 38 | 10.5 | |||
| Butcher nasal swab | 15 | 0 | 0.0 | |||
| Sample types from the dairy farm | Tank milk | 50 | 14 | 28.0 | ||
| Milker nasal swab | 17 | 4 | 23.5 | |||
| Udder milk of cow | 297 | 67 | 22.6 | |||
| Bucket swab | 50 | 10 | 20.0 | |||
| Milker’s hand swab | 50 | 10 | 20.0 | |||
| Tank swab | 50 | 10 | 20.0 | |||
| Total | 1001 | 172 | 17.2 |
| Variables | Mean No. of Antimicrobial Classes Ineffective | Generalized Linear Model for Predicting Infective Drug Classes Using Risk Factors | |||||
|---|---|---|---|---|---|---|---|
| Area | Mean | Std. Error | B | Std. Error | 95% CI Lower | 95% CI Upper | Sig. |
| Addis Ababa | 3.94 | 0.374 | −0.567 | 0.5007 | −1.548 | 0.415 | 0.258 |
| Adama | 4.02 | 0.494 | −0.483 | 0.5785 | −1.617 | 0.651 | 0.404 |
| Assela | 3.47 | 0.367 | −1.04 | 0.4761 | −1.972 | −0.106 | 0.029 |
| Bishoftu | 3.75 | 0.418 | −0.750 | 0.4676 | −1.666 | 0.167 | 0.109 |
| Holeta | 4.50 | 0.396 | Ref a | − | − | − | − |
| Sample source | |||||||
| Abattoir | 4.71 | 0.727 | 1.542 | 1.047 | −0.511 | 3.595 | 0.141 |
| Farm | 3.17 | 0.383 | Ref a | − | − | − | − |
| Sample type | |||||||
| Butcher hand swab | 2.25 | 0.702 | −3.71 | 1.09 | −5.844 | −1.57 | 0.001 |
| Bucket swab | 3.38 | 0.625 | −1.00 | 0.449 | −1.883 | −0.125 | 0.025 |
| Milker hand swab | 2.80 | 0.724 | −1.51 | 0.627 | −2.740 | −0.282 | 0.016 |
| Milker nasal swab | 4.67 | 1.157 | 0.658 | 1.101 | −1.500 | 2.817 | 0.550 |
| Abattoir knife swab | 3.50 | 0.858 | −2.10 | 1.195 | −4.443 | 0.240 | 0.079 |
| Meat swab | 3.35 | 0.613 | −2.47 | 1.052 | −4.532 | −0.409 | 0.019 |
| Slaughter line swab | 6.00 | 0.803 | 1.66 | 0.857 | −0.049 | 3.359 | 0.057 |
| Tank milk | 4.60 | 0.598 | 0.252 | 0.4583 | −0.646 | 1.151 | 0.582 |
| Tank swab | 2.75 | 0.758 | −1.41 | 0.6414 | −2.662 | −0.148 | 0.028 |
| Udder milk(cow) | 4.34 | 0.520 | Ref a | − | − | − | − |
| Total | 3.94 | 0.251 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gizaw, F.; Kekeba, T.; Teshome, F.; Kebede, M.; Abreham, T.; Berhe, H.H.; Ayana, D.; Edao, B.M.; Waktole, H.; Tufa, T.B.; et al. Multidrug-Resistant Staphylococcus aureus Strains Thrive in Dairy and Beef Production, Processing, and Supply Lines in Five Geographical Areas in Ethiopia. Vet. Sci. 2023, 10, 663. https://doi.org/10.3390/vetsci10120663
Gizaw F, Kekeba T, Teshome F, Kebede M, Abreham T, Berhe HH, Ayana D, Edao BM, Waktole H, Tufa TB, et al. Multidrug-Resistant Staphylococcus aureus Strains Thrive in Dairy and Beef Production, Processing, and Supply Lines in Five Geographical Areas in Ethiopia. Veterinary Sciences. 2023; 10(12):663. https://doi.org/10.3390/vetsci10120663
Chicago/Turabian StyleGizaw, Fikru, Tolera Kekeba, Fikadu Teshome, Matewos Kebede, Tekeste Abreham, Halefom Hishe Berhe, Dinka Ayana, Bedaso Mammo Edao, Hika Waktole, Takele Beyene Tufa, and et al. 2023. "Multidrug-Resistant Staphylococcus aureus Strains Thrive in Dairy and Beef Production, Processing, and Supply Lines in Five Geographical Areas in Ethiopia" Veterinary Sciences 10, no. 12: 663. https://doi.org/10.3390/vetsci10120663
APA StyleGizaw, F., Kekeba, T., Teshome, F., Kebede, M., Abreham, T., Berhe, H. H., Ayana, D., Edao, B. M., Waktole, H., Tufa, T. B., Abunna, F., Beyi, A. F., & Abdi, R. D. (2023). Multidrug-Resistant Staphylococcus aureus Strains Thrive in Dairy and Beef Production, Processing, and Supply Lines in Five Geographical Areas in Ethiopia. Veterinary Sciences, 10(12), 663. https://doi.org/10.3390/vetsci10120663






