Doxorubicin as a Potential Treatment Option in Canine Mammary Tumors
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Cell Culture
2.2. MTT Cell Viability Assay
2.3. Apoptosis Assay through Fluorescence Microscopy
2.4. Confluency Assay
2.5. Cell Cycle Assay using Celigo
2.6. Colony Formation Assay
2.7. Scratch Assay
2.8. Cell Invasion Assay
2.9. Gene Expression Evaluation Using PCR Array
2.10. Statistical Analysis
3. Results
3.1. Doxorubicin Inhibits Cell Viability in Both In Vitro Canine Mammary Cancer Models
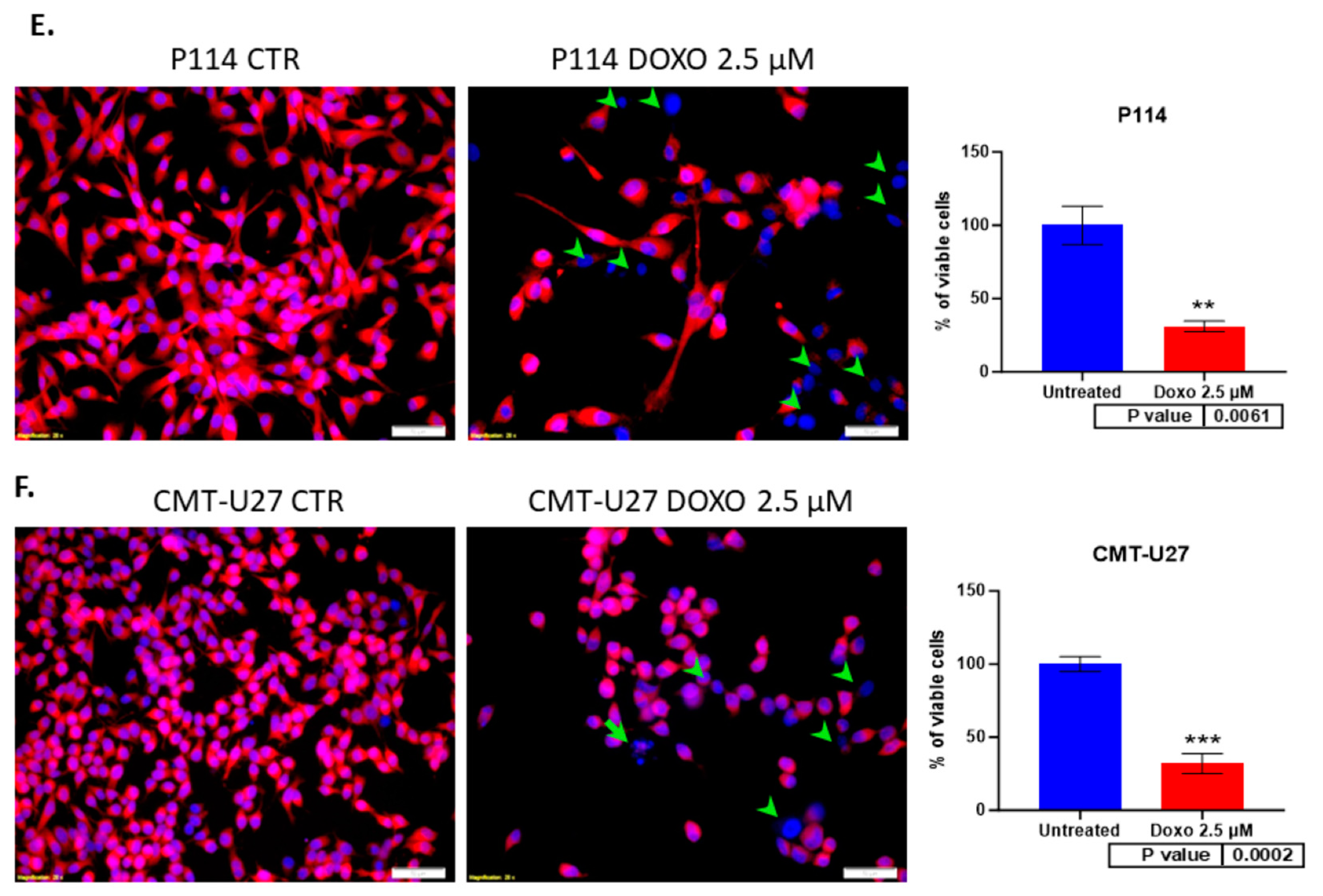
3.2. Doxorubicin Treatment Decreases Cell Viability and Induces Apoptosis in Cell Lines
3.3. Reduced Cell Confluency in the Treated versus Untreated Cells
3.4. Cell Cycle Arrest Induced by Doxorubicin
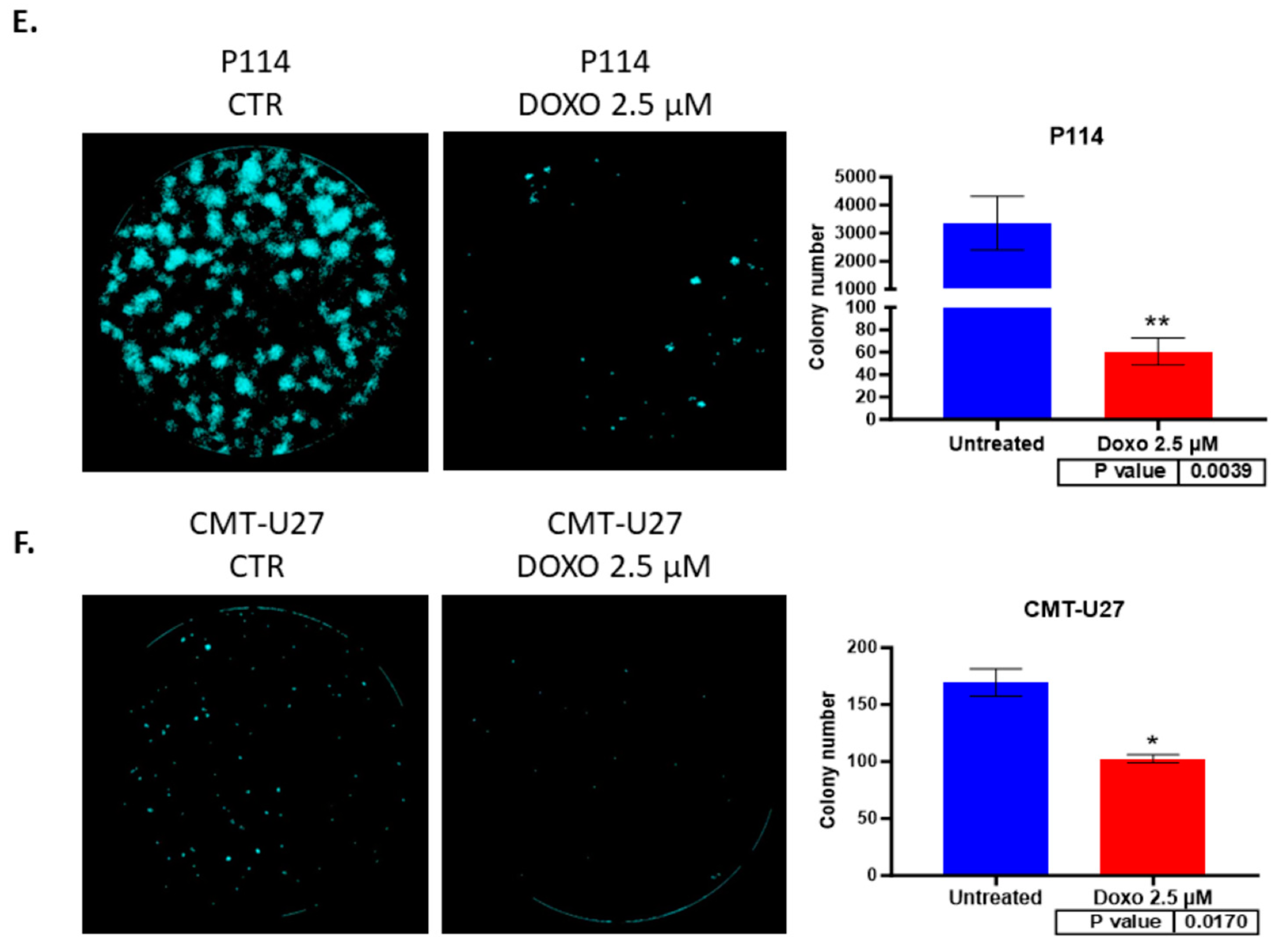
3.5. Doxorubicin Reduces Colony Formation in Both Canine Mammary Cancer Cell Lines
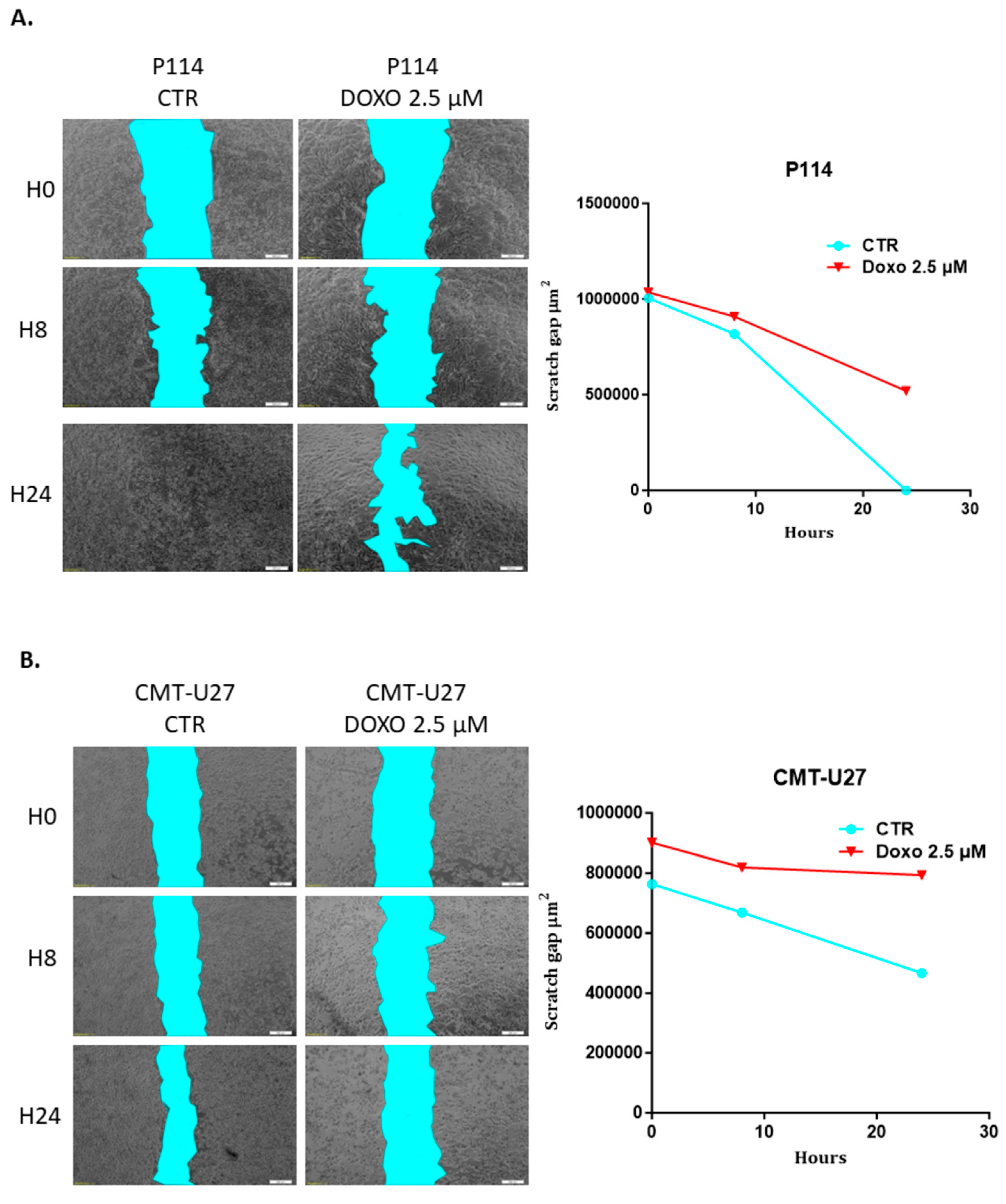
3.6. Doxorubicin Impairs Cell Migration in Both Canine In Vitro Mammary Cancer Models
3.7. Doxorubicin Treatment Suppresses Cell Invasion
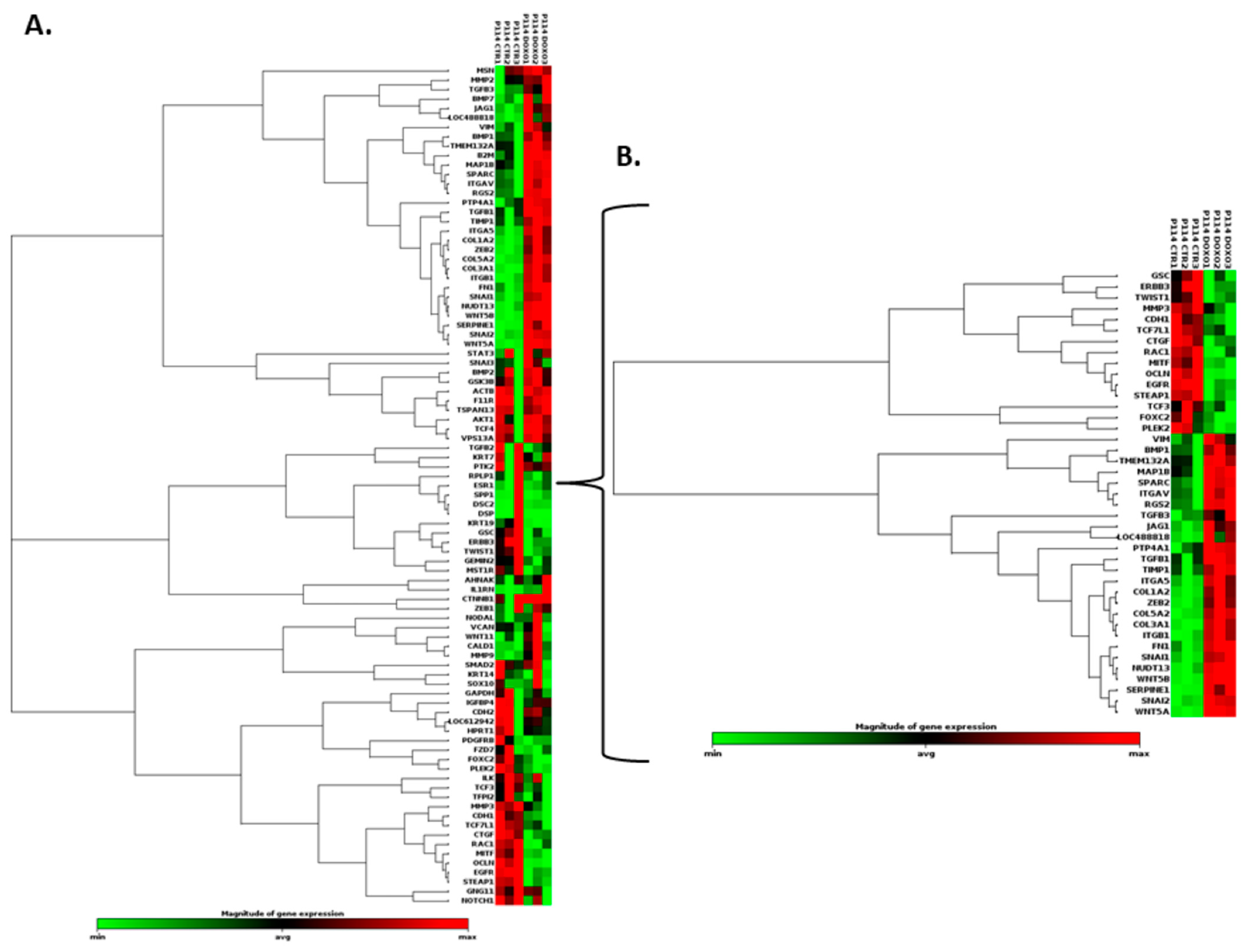
3.8. Doxorubicin Treatment Induces EMT Gene Expression Level Alteration
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Salas, Y.; Marquez, A.; Diaz, D.; Romero, L. Epidemiological Study of Mammary Tumors in Female Dogs Diagnosed during the Period 2002-2012: A Growing Animal Health Problem. PLoS ONE 2015, 10, e0127381. [Google Scholar] [CrossRef]
- Goldschmidt, M.H.; Peña, L.; Zappulli, V. Tumors of the Mammary Gland. In Tumors in Domestic Animals; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; pp. 723–765. [Google Scholar] [CrossRef]
- Burrai, G.P.; Gabrieli, A.; Moccia, V.; Zappulli, V.; Porcellato, I.; Brachelente, C.; Pirino, S.; Polinas, M.; Antuofermo, E. A Statistical Analysis of Risk Factors and Biological Behavior in Canine Mammary Tumors: A Multicenter Study. Anim. Open Access J. 2020, 10, 1687. [Google Scholar] [CrossRef]
- Sorenmo, K. Canine mammary gland tumors. Vet. Clin. N. Am. Small Anim. Pract. 2003, 33, 573–596. [Google Scholar] [CrossRef] [PubMed]
- Raduly, L.; Cojocneanu, R.; Sarpataki, O.; Chira, S.; Atanasov, A.; Braicu, C.; Berindan-Neagoe, I.; Marcus, I. Canis lupus familiaris as relevant animal model for breast cancer—A comparative oncology review. Anim. Sci. Pap. Rep. 2018, 36, 119–148. [Google Scholar]
- Sorenmo, K.U.; Rasotto, R.; Zappulli, V.; Goldschmidt, M.H. Development, anatomy, histology, lymphatic drainage, clinical features, and cell differentiation markers of canine mammary gland neoplasms. Vet. Pathol. 2011, 48, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Valdivia, G.; Alonso-Diez, A.; Perez-Alenza, D.; Pena, L. From Conventional to Precision Therapy in Canine Mammary Cancer: A Comprehensive Review. Front. Vet. Sci. 2021, 8, 623800. [Google Scholar] [CrossRef] [PubMed]
- Lavalle, G.E.; De Campos, C.B.; Bertagnolli, A.C.; Cassali, G.D. Canine malignant mammary gland neoplasms with advanced clinical staging treated with carboplatin and cyclooxygenase inhibitors. In Vivo 2012, 26, 375–379. [Google Scholar]
- Kim, J.-H.; Im, K.-S.; Kim, N.-H.; Chon, S.-K.; Doster, A.R.; Sur, J.-H. Inflammatory mammary carcinoma with metastasis to the brain and distant organs in a spayed Shih Tzu dog. J. Vet. Diagn. Investig. 2011, 23, 1079–1082. [Google Scholar] [CrossRef]
- Simon, D.; Schoenrock, D.; Baumgartner, W.; Nolte, I. Postoperative adjuvant treatment of invasive malignant mammary gland tumors in dogs with doxorubicin and docetaxel. J. Vet. Intern. Med. 2006, 20, 1184–1190. [Google Scholar] [CrossRef]
- Ogilvie, G.K.; Obradovich, J.E.; Elmslie, R.E.; Vail, D.M.; Moore, A.S.; Straw, R.C.; Dickinson, K.; Cooper, M.F.; Withrow, S.J. Efficacy of mitoxantrone against various neoplasms in dogs. J. Am. Vet. Med. Assoc. 1991, 198, 1618–1621. [Google Scholar]
- Poirier, V.J.; Hershey, A.E.; Burgess, K.E.; Phillips, B.; Turek, M.M.; Forrest, L.J.; Beaver, L.; Vail, D.M. Efficacy and toxicity of paclitaxel (Taxol) for the treatment of canine malignant tumors. J. Vet. Intern. Med. 2004, 18, 219–222. [Google Scholar] [CrossRef]
- von Euler, H.; Rivera, P.; Nyman, H.; Haggstrom, J.; Borga, O. A dose-finding study with a novel water-soluble formulation of paclitaxel for the treatment of malignant high-grade solid tumours in dogs. Vet. Comp. Oncol. 2013, 11, 243–255. [Google Scholar] [CrossRef]
- Raduly, L.; Gulei, D.; Jurj, A.; Moldovan, C.; Balint, E.; Marcus, I.; Sarpataki, O.; Korban, S.; Berindan-Neagoe, I. Comparative effects of BCL-2 inhibition in canine and human breast cancer in vitro models-a review. Anim. Sci. Pap. Rep. 2022, 40, 5–22. [Google Scholar]
- Mentoor, I.; Engelbrecht, A.M.; van de Vyver, M.; van Jaarsveld, P.J.; Nell, T. The paracrine effects of adipocytes on lipid metabolism in doxorubicin-treated triple negative breast cancer cells. Adipocyte 2021, 10, 505–523. [Google Scholar] [CrossRef]
- Walsh, E.M.; Smith, K.L.; Stearns, V. Management of hormone receptor-positive, HER2-negative early breast cancer. Semin. Oncol. 2020, 47, 187–200. [Google Scholar] [CrossRef]
- Ciocan-Cartita, C.A.; Jurj, A.; Zanoaga, O.; Cojocneanu, R.; Pop, L.A.; Moldovan, A.; Moldovan, C.; Zimta, A.A.; Raduly, L.; Pop-Bica, C.; et al. Correction to: New insights in gene expression alteration as effect of doxorubicin drug resistance in triple negative breast cancer cells. J. Exp. Clin. Cancer Res. 2020, 39, 287. [Google Scholar] [CrossRef] [PubMed]
- Levi, M.; Salaroli, R.; Parenti, F.; De Maria, R.; Zannoni, A.; Bernardini, C.; Gola, C.; Brocco, A.; Marangio, A.; Benazzi, C.; et al. Doxorubicin treatment modulates chemoresistance and affects the cell cycle in two canine mammary tumour cell lines. BMC Vet. Res. 2021, 17, 30. [Google Scholar] [CrossRef] [PubMed]
- Yun, U.-J.; Lee, J.-H.; Shim, J.; Yoon, K.; Goh, S.-H.; Yi, E.H.; Ye, S.-K.; Lee, J.-S.; Lee, H.; Park, J.; et al. Anti-cancer effect of doxorubicin is mediated by downregulation of HMG-Co A reductase via inhibition of EGFR/Src pathway. Lab. Investig. 2019, 99, 1157–1172. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, V.; Varunkumar, K.; Ravikumar, V.; Rajaram, R. Target delivery of doxorubicin tethered with PVP stabilized gold nanoparticles for effective treatment of lung cancer. Sci. Rep. 2018, 8, 3815. [Google Scholar] [CrossRef]
- Ciocan-Cartita, C.A.; Jurj, A.; Raduly, L.; Cojocneanu, R.; Moldovan, A.; Pileczki, V.; Pop, L.A.; Budisan, L.; Braicu, C.; Korban, S.S.; et al. New perspectives in triple-negative breast cancer therapy based on treatments with TGFbeta1 siRNA and doxorubicin. Mol. Cell Biochem. 2020, 475, 285–299. [Google Scholar] [CrossRef]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, N.; Gheldof, A.; Tatari, M.; Christofori, G. EMT as the ultimate survival mechanism of cancer cells. Semin. Cancer Biol. 2012, 22, 194–207. [Google Scholar] [CrossRef]
- Brabletz, T. To differentiate or not--routes towards metastasis. Nat. Rev. Cancer 2012, 12, 425–436. [Google Scholar] [CrossRef]
- Raposo-Ferreira, T.M.M.; Brisson, B.K.; Durham, A.C.; Laufer-Amorim, R.; Kristiansen, V.; Pure, E.; Volk, S.W.; Sorenmo, K. Characteristics of the Epithelial-Mesenchymal Transition in Primary and Paired Metastatic Canine Mammary Carcinomas. Vet. Pathol. 2018, 55, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Hong, W.; Wei, X. The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. J. Hematol. Oncol. 2022, 15, 129. [Google Scholar] [CrossRef]
- Pirlog, R.; Chiroi, P.; Rusu, I.; Jurj, A.M.; Budisan, L.; Pop-Bica, C.; Braicu, C.; Crisan, D.; Sabourin, J.C.; Berindan-Neagoe, I. Cellular and Molecular Profiling of Tumor Microenvironment and Early-Stage Lung Cancer. Int. J. Mol. Sci. 2022, 23, 5346. [Google Scholar] [CrossRef] [PubMed]
- Groza, I.M.; Braicu, C.; Jurj, A.; Zanoaga, O.; Lajos, R.; Chiroi, P.; Cojocneanu, R.; Paun, D.; Irimie, A.; Korban, S.S.; et al. Cancer-Associated Stemness and Epithelial-to-Mesenchymal Transition Signatures Related to Breast Invasive Carcinoma Prognostic. Cancers 2020, 12, 3053. [Google Scholar] [CrossRef]
- Mirzaei, S.; Abadi, A.J.; Gholami, M.H.; Hashemi, F.; Zabolian, A.; Hushmandi, K.; Zarrabi, A.; Entezari, M.; Aref, A.R.; Khan, H.; et al. The involvement of epithelial-to-mesenchymal transition in doxorubicin resistance: Possible molecular targets. Eur. J. Pharmacol. 2021, 908, 174344. [Google Scholar] [CrossRef]
- Hellmen, E. Characterization of four in vitro established canine mammary carcinoma and one atypical benign mixed tumor cell lines. Vitr. Cell. Dev. Biol. J. Tissue Cult. Assoc. 1992, 28A, 309–319. [Google Scholar] [CrossRef]
- Rao, N.A.; van Wolferen, M.E.; Gracanin, A.; Bhatti, S.F.; Krol, M.; Holstege, F.C.; Mol, J.A. Gene expression profiles of progestin-induced canine mammary hyperplasia and spontaneous mammary tumors. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2009, 60 (Suppl. S1), 73–84. [Google Scholar]
- Krol, M.; Pawlowski, K.M.; Skierski, J.; Rao, N.A.; Hellmen, E.; Mol, J.A.; Motyl, T. Transcriptomic profile of two canine mammary cancer cell lines with different proliferative and anti-apoptotic potential. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2009, 60 (Suppl. S1), 95–106. [Google Scholar]
- Hong, D.; Fritz, A.J.; Zaidi, S.K.; van Wijnen, A.J.; Nickerson, J.A.; Imbalzano, A.N.; Lian, J.B.; Stein, J.L.; Stein, G.S. Epithelial-to-mesenchymal transition and cancer stem cells contribute to breast cancer heterogeneity. J. Cell Physiol. 2018, 233, 9136–9144. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef] [PubMed]
- Lyu, J.H.; Park, D.W.; Huang, B.; Kang, S.H.; Lee, S.J.; Lee, C.; Bae, Y.S.; Lee, J.G.; Baek, S.H. RGS2 suppresses breast cancer cell growth via a MCPIP1-dependent pathway. J. Cell Biochem. 2015, 116, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Ye, Q.; Cao, Y.; Tan, J.; Wang, F.; Jiang, J.; Cao, Y. Downregulation of regulator of G protein signaling 2 expression in breast invasive carcinoma of no special type: Clinicopathological associations and prognostic relevance. Oncol. Lett. 2018, 15, 213–220. [Google Scholar] [CrossRef]
- Tassi, E.; Al-Attar, A.; Aigner, A.; Swift, M.R.; McDonnell, K.; Karavanov, A.; Wellstein, A. Enhancement of fibroblast growth factor (FGF) activity by an FGF-binding protein. J. Biol. Chem. 2001, 276, 40247–40253. [Google Scholar] [CrossRef]
- Zhang, Y.; Di, X.; Chen, G.; Liu, J.; Zhang, B.; Feng, L.; Cheng, S.; Wang, Y. An immune-related signature that to improve prognosis prediction of breast cancer. Am. J. Cancer Res. 2021, 11, 1267–1285. [Google Scholar]
- Mani, S.A.; Yang, J.; Brooks, M.; Schwaninger, G.; Zhou, A.; Miura, N.; Kutok, J.L.; Hartwell, K.; Richardson, A.L.; Weinberg, R.A. Mesenchyme Forkhead 1 (FOXC2) plays a key role in metastasis and is associated with aggressive basal-like breast cancers. Proc. Natl. Acad. Sci. USA 2007, 104, 10069–10074. [Google Scholar] [CrossRef]
- Hollier, B.G.; Tinnirello, A.A.; Werden, S.J.; Evans, K.W.; Taube, J.H.; Sarkar, T.R.; Sphyris, N.; Shariati, M.; Kumar, S.V.; Battula, V.L.; et al. FOXC2 expression links epithelial-mesenchymal transition and stem cell properties in breast cancer. Cancer Res. 2013, 73, 1981–1992. [Google Scholar] [CrossRef]
- Werden, S.J.; Sphyris, N.; Sarkar, T.R.; Paranjape, A.N.; LaBaff, A.M.; Taube, J.H.; Hollier, B.G.; Ramirez-Pena, E.Q.; Soundararajan, R.; den Hollander, P.; et al. Phosphorylation of serine 367 of FOXC2 by p38 regulates ZEB1 and breast cancer metastasis, without impacting primary tumor growth. Oncogene 2016, 35, 5977–5988. [Google Scholar] [CrossRef]
- Suhaimi, S.A.; Chan, S.C.; Rosli, R. Matrix Metallopeptidase 3 Polymorphisms: Emerging genetic Markers in Human Breast Cancer Metastasis. J. Breast Cancer 2020, 23, 1–9. [Google Scholar] [CrossRef]
- Argote Camacho, A.X.; Gonzalez Ramirez, A.R.; Perez Alonso, A.J.; Rejon Garcia, J.D.; Olivares Urbano, M.A.; Torne Poyatos, P.; Rios Arrabal, S.; Nunez, M.I. Metalloproteinases 1 and 3 as Potential Biomarkers in Breast Cancer Development. Int. J. Mol. Sci. 2021, 22, 9012. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.; Kumar, B.V.; Singh, S.; Verma, R. Development of recombinant matrix metalloproteinase-3 based sandwich ELISA for sero-diagnosis of canine mammary carcinomas. J. Immunoass. Immunochem. 2017, 38, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Grunewald, T.G.P.; Ranft, A.; Esposito, I.; da Silva-Buttkus, P.; Aichler, M.; Baumhoer, D.; Schaefer, K.L.; Ottaviano, L.; Poremba, C.; Jundt, G.; et al. High STEAP1 expression is associated with improved outcome of Ewing’s sarcoma patients. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2012, 23, 2185–2190. [Google Scholar] [CrossRef] [PubMed]
- Gomes, I.M.; Arinto, P.; Lopes, C.; Santos, C.R.; Maia, C.J. STEAP1 is overexpressed in prostate cancer and prostatic intraepithelial neoplasia lesions, and it is positively associated with Gleason score. Urol. Oncol. 2014, 32, 53.e23–53.e29. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Chen, S.L.; Sung, W.W.; Lai, H.W.; Hsieh, M.J.; Yen, H.H.; Su, T.C.; Chiou, Y.H.; Chen, C.Y.; Lin, C.Y.; et al. The Prognostic Role of STEAP1 Expression Determined via Immunohistochemistry Staining in Predicting Prognosis of Primary Colorectal Cancer: A Survival Analysis. Int. J. Mol. Sci. 2016, 17, 592. [Google Scholar] [CrossRef]
- Grunewald, T.G.; Diebold, I.; Esposito, I.; Plehm, S.; Hauer, K.; Thiel, U.; da Silva-Buttkus, P.; Neff, F.; Unland, R.; Muller-Tidow, C.; et al. STEAP1 is associated with the invasive and oxidative stress phenotype of Ewing tumors. Mol. Cancer Res. 2012, 10, 52–65. [Google Scholar] [CrossRef]
- Xie, J.; Yang, Y.; Sun, J.; Jiao, Z.; Zhang, H.; Chen, J. STEAP1 Inhibits Breast Cancer Metastasis and Is Associated with Epithelial-Mesenchymal Transition Procession. Clin. Breast Cancer 2019, 19, e195–e207. [Google Scholar] [CrossRef] [PubMed]



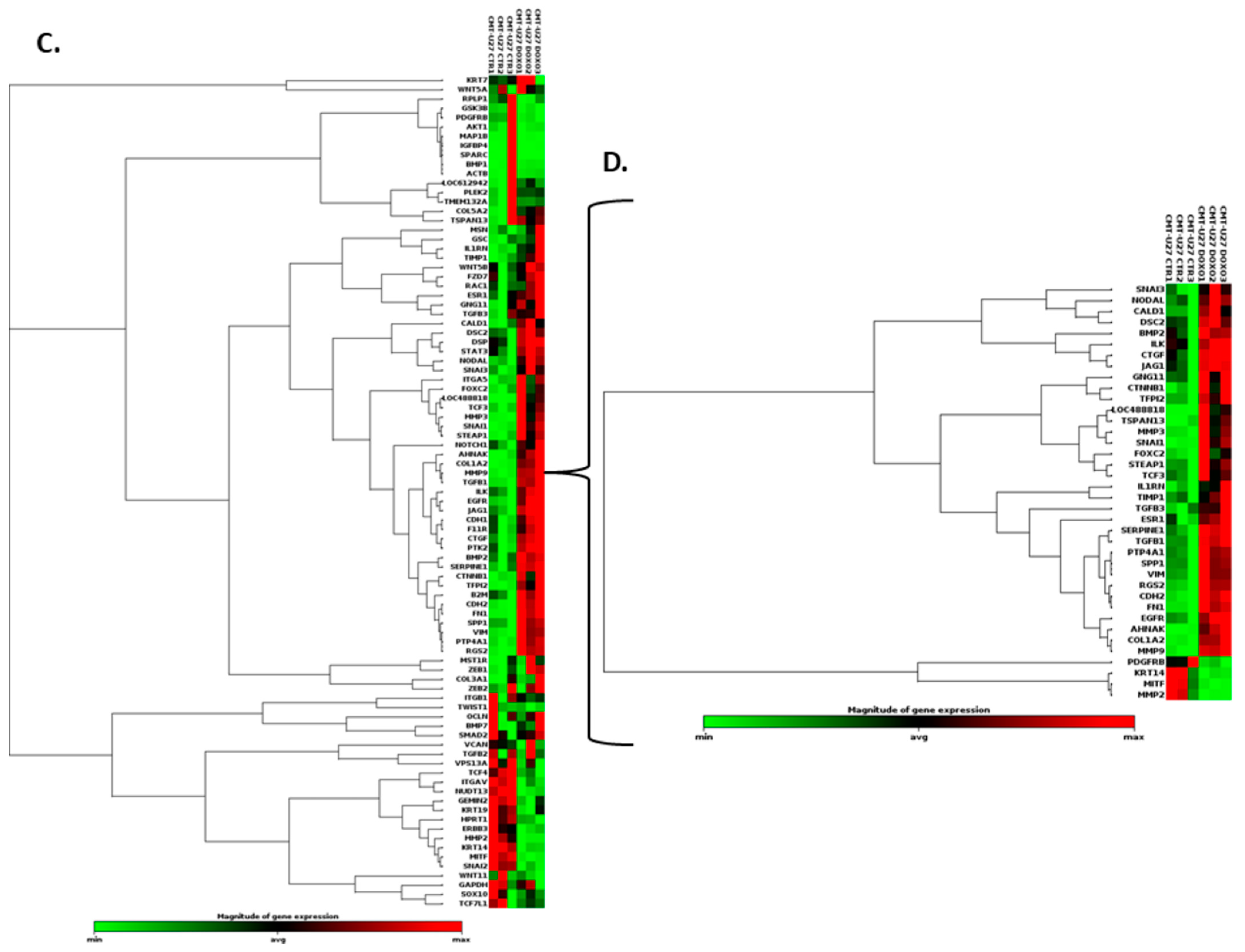
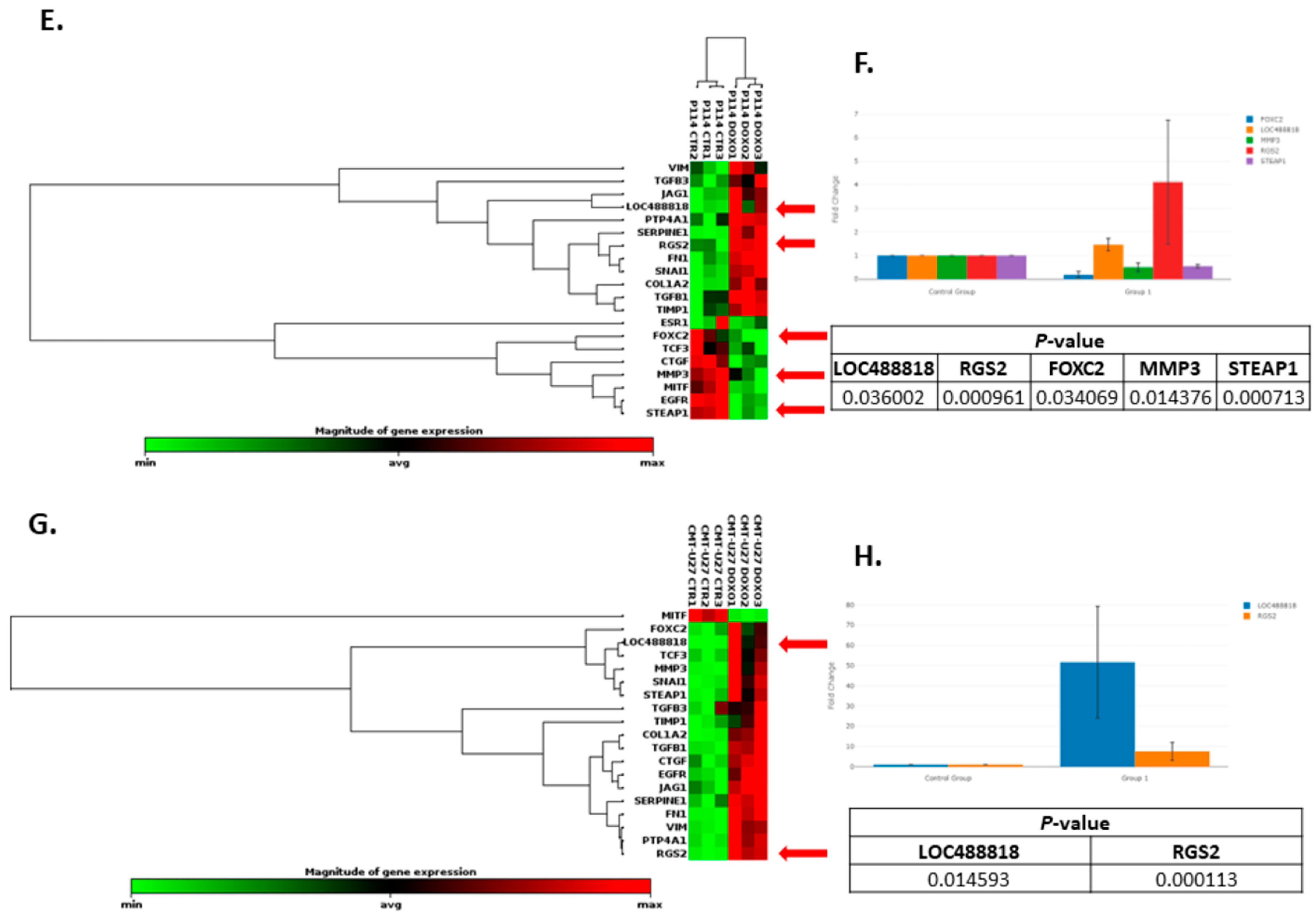
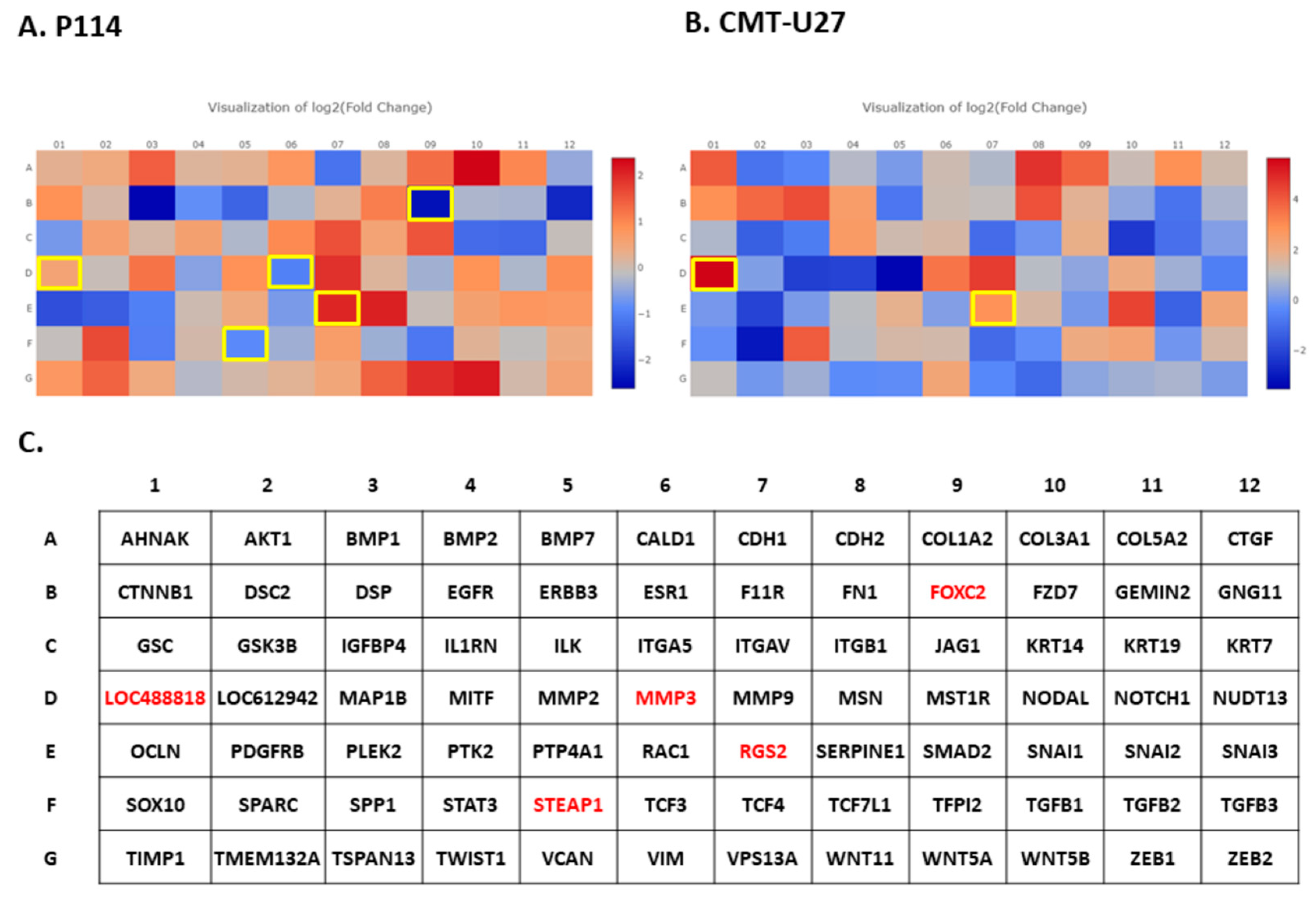
| Cell Line | Mean Number of Cells (%) | |||||
|---|---|---|---|---|---|---|
| G0/G1 | S | G2/M | G0/G1 | S | G2/M | |
| CTR | DOXO | |||||
| P114 | 31.34 | 2.47 | 62.17 | 25.28 | 1.37 | 71.84 |
| CMT-U27 | 76.35 | 10.86 | 9.69 | 63.95 | 17.79 | 10.20 |
| P114 | ||
|---|---|---|
| Genes | Fold Change (Compared to Control Group) | Biological Processes |
| BMP1 | 2.73 | EMT, Cell growth and proliferation |
| CDH1 | 0.44 | EMT |
| COL1A2 | 2.43 | EMT |
| COL3A1 | 5.24 | EMT |
| COL5A2 | 2.00 | EMT |
| CTGF | 0.73 | fJAG |
| EGFR | 0.57 | Cell growth and proliferation |
| ERBB3 | 0.38 | Cell growth and proliferation |
| FN1 | 2.16 | EMT |
| FOXC2 | 0.18 | EMT, Differentiation and development, Cell growth and proliferation, fJAG, Transcription factors |
| GSC | 0.63 | EMT |
| ITGA5 | 1.96 | EMT |
| ITGAV | 3.04 | EMT |
| ITGB1 | 1.45 | fJAG |
| JAG1 | 2.93 | Differentiation and development, Cell growth and proliferation, Cell migration and motility |
| LOC488818 | 1.46 | EMT, Cell growth and proliferation |
| MAP1B | 2.33 | Cytoskeleton regulation |
| MITF | 0.68 | EMT, Differentiation and development, Transcription factors |
| MMP3 | 0.50 | EMT, fJAG |
| NUDT13 | 1.91 | EMT |
| OCLN | 0.32 | EMT |
| PLEK2 | 0.50 | Cytoskeleton regulation |
| PTP4A1 | 1.35 | Differentiation and development |
| RAC1 | 0.64 | Cell morphogenesis, Cell migration and motility, Cytoskeleton regulation, fJAG |
| RGS2 | 4.12 | EMT |
| SERPINE1 | 4.26 | EMT, fJAG |
| SNAI1 | 1.52 | EMT, Differentiation and development, Cell morphogenesis |
| SNAI2 | 1.75 | EMT, Transcription factors |
| SPARC | 3.18 | EMT |
| STEAP1 | 0.55 | EMT |
| TCF3 | 0.77 | Transcription factors |
| TCF7L1 | 0.77 | Transcription factors |
| TGFB1 | 1.17 | Cell morphogenesis, Cell migration and motility, fJAG |
| TGFB3 | 1.32 | Differentiation and development, Cell growth and proliferation, Cell morphogenesis |
| TIMP1 | 1.69 | EMT, Cell growth and proliferation, fJAG |
| TMEM132A | 2.66 | EMT |
| TWIST1 | 0.87 | EMT, Differentiation and development, Cell morphogenesis, Transcription factors |
| VIM | 1.18 | EMT, Cell migration and motility, Cytoskeleton regulation |
| WNT5A | 3.87 | EMT, Differentiation and development, Cell morphogenesis, Signal transduction |
| WNT5B | 4.46 | EMT, Differentiation and development, Signal transduction |
| ZEB2 | 1.46 | Transcription factors |
| CMT-U27 | ||
|---|---|---|
| Genes | Fold Change (Compared to Control Group) | Biological Processes |
| AHNAK | 16.47 | EMT |
| BMP2 | 1.93 | Differentiation and development |
| CALD1 | 2.51 | EMT, Cell migration and motility |
| CDH2 | 28.15 | EMT, fJAG |
| COL1A2 | 14.38 | EMT |
| CTGF | 2.61 | fJAG |
| CTNNB1 | 7.76 | Differentiation and development, Cell morphogenesis, Cell growth and proliferation, fJAG, Transcription factors |
| DSC2 | 13.75 | fJAG |
| EGFR | 5.92 | Cell growth and proliferation |
| ESR1 | 2.46 | Transcription factors |
| FN1 | 18.71 | EMT, Cell migration and motility, fJAG |
| FOXC2 | 3.41 | EMT, Differentiation and development, Cell growth and proliferation, fJAG, Transcription factors |
| GNG11 | 1.76 | EMT, Signal transduction |
| IL1RN | 6.20 | EMT |
| ILK | 2.57 | Cell growth and proliferation, fJAG |
| JAG1 | 3.76 | Differentiation and development, Cell growth and proliferation, Cell migration and motility |
| KRT14 | 0.21 | Differentiation and development |
| LOC488818 | 51.71 | EMT, Cell growth and proliferation |
| MITF | 0.25 | EMT, Differentiation and development, Transcription Factors |
| MMP2 | 0.09 | EMT, fJAG |
| MMP3 | 11.63 | EMT, fJAG |
| MMP9 | 24.81 | EMT, fJAG |
| NODAL | 4.15 | Differentiation and development, Cell growth and proliferation, Cell migration and motility |
| PDGFRB | 0.25 | Cell growth and proliferation, Cell migration and motility, Signal transduction |
| PTP4A1 | 3.61 | Differentiation and development |
| RGS2 | 7.56 | EMT |
| SERPINE1 | 2.90 | EMT, fJAG |
| SNAI1 | 22.24 | EMT, Differentiation and development, Cell morphogenesis |
| SPP1 | 16.28 | EMT, fJAG |
| STEAP1 | 2.83 | EMT |
| TCF3 | 2.67 | Transcription factors |
| TFPI2 | 4.10 | EMT |
| TGFB1 | 4.92 | Cell morphogenesis, Cell migration and motility, fJAG |
| TGFB3 | 2.79 | Differentiation and development, Cell growth and proliferation, Cell morphogenesis |
| TIMP1 | 2.23 | EMT, Cell growth and proliferation, fJAG |
| TSPAN13 | 1.55 | EMT |
| VIM | 4.84 | EMT, Cell migration and motility, Cytoskeleton regulation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gherman, M.L.; Zanoaga, O.; Budisan, L.; Raduly, L.; Berindan-Neagoe, I. Doxorubicin as a Potential Treatment Option in Canine Mammary Tumors. Vet. Sci. 2023, 10, 654. https://doi.org/10.3390/vetsci10110654
Gherman ML, Zanoaga O, Budisan L, Raduly L, Berindan-Neagoe I. Doxorubicin as a Potential Treatment Option in Canine Mammary Tumors. Veterinary Sciences. 2023; 10(11):654. https://doi.org/10.3390/vetsci10110654
Chicago/Turabian StyleGherman, Madalina Luciana, Oana Zanoaga, Liviuta Budisan, Lajos Raduly, and Ioana Berindan-Neagoe. 2023. "Doxorubicin as a Potential Treatment Option in Canine Mammary Tumors" Veterinary Sciences 10, no. 11: 654. https://doi.org/10.3390/vetsci10110654
APA StyleGherman, M. L., Zanoaga, O., Budisan, L., Raduly, L., & Berindan-Neagoe, I. (2023). Doxorubicin as a Potential Treatment Option in Canine Mammary Tumors. Veterinary Sciences, 10(11), 654. https://doi.org/10.3390/vetsci10110654













