Penetration of Enrofloxacin in Aqueous Humour of Avian Eyes
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Dosing and Sample Collection Protocols
2.3. Determination of Fluoroquinolones
2.4. Determination of Enrofloxacin/Ciprofloxacin Ratios
2.5. Data Analysis
2.6. Minimal Inhibitory Concentrations (MICs) of Enrofloxacin
3. Results
3.1. Tolerability of Enrofloxacin
3.2. Pharmacokinetics of Fluoroquinolones in Serum and Aqueous Humour
3.3. Metabolization of Enrofloxacin to Ciprofloxacin
3.4. Fluoroquinolone Susceptibility to Relevant Bacteria
4. Discussion
4.1. Study Design
4.2. Fluoroquinolone Analysis
4.3. Pharmacokinetics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Korbel, R. Ocular manifestations of systemic diseases in birds. Part 2. Tierarztl. Prax. 1992, 20, 483–491. [Google Scholar] [PubMed]
- Bayón, A.; Almela, R.; Talavera, J. Avian ophthalmology. Eur. J. Companion. Anim. Pract. 2007, 17, 253–266. [Google Scholar]
- Willis, A.M.; Wilkie, D.A. Avian ophthalmology, part 2: Review of ophthalmic diseases. J. Avian Med. Surg. 1999, 13, 245–251. [Google Scholar]
- Nakamura, K.; Abe, F. Ocular lesions in chickens inoculated with Escherichia coli. Can. J. Vet. Res. 1987, 51, 528–530. [Google Scholar]
- Korbel, R. Ocular manifestations of systemic diseases in birds. Part 1. Tierärztl. Prax. 1992, 20, 385–394. [Google Scholar] [PubMed]
- Cunha-Vaz, J.G.; Shakib, M.; Ashton, N. Studies on the permeability of the blood-retinal barrier. I. On the existence, development, and site of a blood-retinal barrier. Br. J. Ophthalmol. 1966, 50, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.; Pennefather, P.M.; Kaye, S.B.; Hart, C.A. Fluoroquinolones. Drugs 2001, 61, 747–761. [Google Scholar] [CrossRef]
- Scope, A. Spezielle Antibiose. In Kompendium der Ziervogelkrankheiten—Papageien, Tauben, Sperlingsvögel; Kaleta, E.F., Krautwald-Junghanns, M.E., Eds.; Schlütersche Verlagsgesellschaft mbH & Co. KG: Hannover, Germany, 2011; pp. 219–223. [Google Scholar]
- Flammer, K.; Aucoin, D.P.; Whitt, D.A. Intramuscular and oral disposition of enrofloxacin in African grey parrots following single and multiple doses. J. Vet. Pharmacol. Ther. 1991, 14, 359–366. [Google Scholar]
- Abd el-Aziz, M.I.; Aziz, M.A.; Soliman, F.A.; Afify, N.A. Pharmcokinetic evaluation of enrofloxacin in chickens. Br. Poult. Sci. 1997, 38, 164–168. [Google Scholar]
- Bailey, T.A.; Sheen, R.S.; Silvanose, C.; Samour, J.H.; Garner, A.; Harron, D.W.G. Pharmacokinetics of enrofloxacin after intravenous, intramuscular and oral administration in houbara bustard (Chlamydotis undulata macqueenii). J. Vet. Pharmacol. Ther. 1998, 21, 288–297. [Google Scholar] [CrossRef]
- Harrenstien, L.A.; Tell, L.A.; Vulliet, R.; Needham, M.; Brandt, C.M.; Brondos, A.; Stedman, B.; Kass, P.H. Disposition of Enrofloxacin in Red-tailed Hawks (Buteo jamaicensis) and Great Horned Owls (Bubo virginianus) after a Single Oral, Intramuscular, or Intravenous Dose. J. Avian Med. Surg. 2000, 14, 228–236. [Google Scholar] [CrossRef]
- Kaspar, H.; Steinacker, U.; Römer, A.; Karaalp, A.; Ballhausen, B.; Kluge, M.; Gowik, P. Bericht zur Resistenzmonitoringstudie 2018-Resistenzsituation bei Klinisch Wichtigen Tierpathogenen Bakterien; Bundesamt für Verbraucherschutz und Lebensmittelsicherheit: Berlin, Germany, 2020; p. 98. [Google Scholar]
- Popp, M.; Gerhards, H.; Wollanke, B. Enrofloxacinkonzentrationen im Glaskörper und im Serum an equiner rezidivierender Uveitis (ERU) erkrankter Pferde nach wiederholter intravenöser Verabreichung. Pferdeheilkunde 2013, 29, 574–580. [Google Scholar] [CrossRef]
- Krastev, Z.; Haritova, A.; Lashev, L.; Hubenov, H. Enrofloxacin disposition in aqueous humour after subcutaneous administration in dogs. Bulg. J. Vet. Med. 2011, 14, 221–230. [Google Scholar]
- Cunha-Vaz, J.; Bernardes, R.; Lobo, C. Blood-retinal barrier. Eur. J. Ophthalmol. 2011, 21 (Suppl. S6), 3–9. [Google Scholar] [CrossRef] [PubMed]
- Reese, S.; Hegner, K.; Korbel, R. Zur Anatomie des Ziliarkörpers und der Kammerwinkelregion von Greifvogelaugen als Grundlage für die Gonioskopie. In Proceedings of the XI. Tagung der DVG-Fachgruppe “Geflügelkrankheiten”, München, Germany, 5–6 March 1998; pp. 231–238. [Google Scholar]
- Reese, S.; Korbel, R.; Liebich, H.G. The eye (organum visus). In Avian Anatomy—Textbook and Colour Atlas; König, H.E., Korbel, R., Liebich, H.G., Eds.; 5m Publishing: Sheffield, UK, 2016; pp. 224–231. [Google Scholar]
- Salomon, F.V.; Krautwald-Junghanns, M.E. Anatomie der Vögel. In Anatomie für Die Tiermedizin, 2nd ed.; Salomon, F.V., Geyer, H., Gille, U., Verlag, E., Eds.; Enke Verlag: Stuttgard, Germany, 2008; pp. 754–814. [Google Scholar]
- Greenacre, C.B.; Luna, L.G.; Morishita, T.Y. Backyard Poultry and Waterfowl. In Exotic Animal Formulary, 5th ed.; Carpenter, J.W., Marion, C.J., Eds.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2018; p. 411. [Google Scholar]
- Acaroz, U.; Dietrich, R.; Knauer, M.; Märtlbauer, E. Development of a generic enzyme-immunoassay for the detection of fluoro (quinolone)-residues in foodstuffs based on a highly sensitive monoclonal antibody. Food Anal. Methods 2020, 13, 780–792. [Google Scholar] [CrossRef]
- Kang, J.; Hossain, M.A.; Park, H.-c.; Kim, Y.; Lee, K.-j.; Park, S.-W. Pharmacokinetic and pharmacodynamic integration of enrofloxacin against Salmonella Enteritidis after administering to broiler chicken by per-oral and intravenous routes. J. Vet. Sci. 2019, 20, e15. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hao, H.; Cheng, G.; Wang, X.; Ahmed, S.; Shabbir, M.A.B.; Liu, Z.; Dai, M.; Yuan, Z. The effects of different enrofloxacin dosages on clinical efficacy and resistance development in chickens experimentally infected with Salmonella Typhimurium. Sci. Rep. 2017, 7, 11676. [Google Scholar] [CrossRef]
- Zhang, X.; Guo, M.; Xie, D.; Chen, Y.; Zhang, C.; Cao, Y.; Wu, Y. Antibiotic resistance of Mycoplasma Synoviae strains isolated in China from 2016 to 2019. BMC Vet. Res. 2022, 18, 8. [Google Scholar] [CrossRef]
- de Jong, A.; Youala, M.; Klein, U.; El Garch, F.; Simjee, S.; Moyaert, H.; Rose, M.; Gautier-Bouchardon, A.V.; Catania, S.; Ganapathy, K.; et al. Minimal inhibitory concentration of seven antimicrobials to Mycoplasma gallisepticum and Mycoplasma synoviae isolates from six European countries. Avian Pathol. 2021, 50, 161–173. [Google Scholar] [CrossRef]
- Morrow, C.J.; Kreizinger, Z.; Achari, R.R.; Bekő, K.; Yvon, C.; Gyuranecz, M. Antimicrobial susceptibility of pathogenic mycoplasmas in chickens in Asia. Vet. Microbiol. 2020, 250, 108840. [Google Scholar] [CrossRef]
- Catania, S.; Bottinelli, M.; Fincato, A.; Gastaldelli, M.; Barberio, A.; Gobbo, F.; Vicenzoni, G. Evaluation of Minimum Inhibitory Concentrations for 154 Mycoplasma synoviae isolates from Italy collected during 2012–2017. PloS ONE 2019, 14, e0224903. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Jiang, L.; Lan, W.; Jiang, Y.; Wang, Z. In vivo pharmacokinetic/Pharmacodynamic modeling of Enrofloxacin against Escherichia coli in broiler chickens. BMC Vet. Res. 2018, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- Chuppava, B.; Keller, B.; El-Wahab, A.A.; Meißner, J.; Kietzmann, M.; Visscher, C. Resistance of Escherichia coli in turkeys after therapeutic or environmental exposition with enrofloxacin depending on flooring. Int. J. Environ. Res. Public Health 2018, 15, 1993. [Google Scholar] [CrossRef] [PubMed]
- Wisely, C.E.; Sayed, J.A.; Tamez, H.; Zelinka, C.; Abdel-Rahman, M.H.; Fischer, A.J.; Cebulla, C.M. The chick eye in vision research: An excellent model for the study of ocular disease. Prog. Retin. Eye Res. 2017, 61, 72–97. [Google Scholar] [CrossRef] [PubMed]
- Holtzapple, C.K.; Buckley, S.A.; Stanker, L.H. Determination of fluoroquinolones in serum using an on-line clean-up column coupled to high-performance immunoaffinity–reversed-phase liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 2001, 754, 1–9. [Google Scholar] [CrossRef]
- Suhren, G.; Hammer, P.; Heeschen, W. Detection of residues of quinolones in milk. In Proceedings of the Euroresidue III: Conference on Residues of Veterinary Drugs in Food, RIVM, Bilthoven, The Netherlands, 6–8 May 1996; pp. 917–921. [Google Scholar]
- Slana, M.; Pahor, V.; Cvitkovic Maricic, L.; Sollner-Dolenc, M. Excretion pattern of enrofloxacin after oral treatment of chicken broilers. J. Vet. Pharmacol. Ther. 2014, 37, 611–614. [Google Scholar] [CrossRef]
- Richter, A.; Scherkl, R. Allgemeine Charakteristika von Antibiotika und Begriffsbestimmungen. In Lehrbuch der Pharmakologie und Toxikologie für die Veterinärmedizin, 4th ed.; Löscher, W., Richter, A., Eds.; Enke Verlag Stuttgard: Stuttgard, Germany, 2016; pp. 404–409. [Google Scholar]
- Donnenfeld, E.D.; Comstock, T.L.; Proksch, J.W. Human aqueous humor concentrations of besifloxacin, moxifloxacin, and gatifloxacin after topical ocular application. J. Cataract Refract. Surg. 2011, 37, 1082–1089. [Google Scholar] [CrossRef]
- Westermeyer, H.D.; Hendrix, D.V.; Ward, D.A.; Cox, S.K. Tear, cornea, and aqueous humor concentrations of ciprofloxacin and moxifloxacin after topical ocular application in ophthalmologically normal horses. Am. J. Vet. Res. 2011, 72, 398–403. [Google Scholar] [CrossRef]
- Popp, M.K. Enrofloxacin im Glaskörper an Equiner Rezidivierender Uveitis Erkrankter Pferde. Ph.D. Thesis, Ludwig-Maximilians-University, Munich, Germany, 2011. [Google Scholar]
- Bleeker, G.; Maas, E. Penetration of penethamate, a penicillin ester, into the tissues of the eye. AMA Arch. Ophthalmol. 1958, 60, 1013–1020. [Google Scholar] [CrossRef]
- Barza, M. Factors affecting the intraocular penetration of antibiotics. The influence of route, inflammation, animal species and tissue pigmentation. Scand. J. Infect. Dis. Suppl. 1978, 14, 151–159. [Google Scholar]
- McCulley, J.P.; Caudle, D.; Aronowicz, J.; Shine, W. Fourth-generation fluoroquinolone penetration into the aqueous humor in humans. Ophthalmology 2006, 113, 955–959. [Google Scholar] [CrossRef] [PubMed]
- Bugyei, K.; Black, W.D.; McEwen, S. Pharmacokinetics of enrofloxacin given by the oral, intravenous and intramuscular routes in broiler chickens. Can. J. Vet. Res. 1999, 63, 193–200. [Google Scholar] [PubMed]
- Hawkins, M.G.; Sanchez-Migallon Guzman, D.; Beaufrère, H.; Lennox, A.M.; Carpenter, J.W. Birds. In Exotic Animal Formulary, 5th ed.; Carpenter, J.W., Marion, C.J., Eds.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2018; pp. 178–179. [Google Scholar]
- Pees, M. Medikamentenverzeichnis. In Leitsymptome bei Papageien und Sittichen; Pees, M., Ed.; Enke Verlag: Stuttgard, Germany, 2011; p. 323. [Google Scholar]
- Doneley, B. Formulary. In Avian Medicine and Surgery in Practice: Companion and Aviary Birds, 2nd ed.; Doneley, B., Ed.; CRC Press, Taylor and Francis Group: New York, NY, USA, 2016; pp. 425–454. [Google Scholar]
- Divers, T.; Irby, N.; Mohammed, H.; Schwark, W. Ocular penetration of intravenously administered enrofloxacin in the horse. Equine Vet. J. 2008, 40, 167–170. [Google Scholar] [CrossRef]
- Çelebi, S.; Ay, S.; Aykan, U.; Bulut, V.; Alagöz, G.; Celiker, U. Penetration of oral and topical ciprofloxacin into human aqueous humor. Acta Ophthalmol. Scand. 1998, 76, 683–685. [Google Scholar] [CrossRef] [PubMed]
- Maggs, D.J. Chapter 3: Ocular Pharmacology and Therapeutics. In Slatters Fundamentals of Veterinary Ophthalmology, 5th ed.; Maggs, D.J., Miller, P.E., Ofri, R., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2013; pp. 27–59. [Google Scholar]
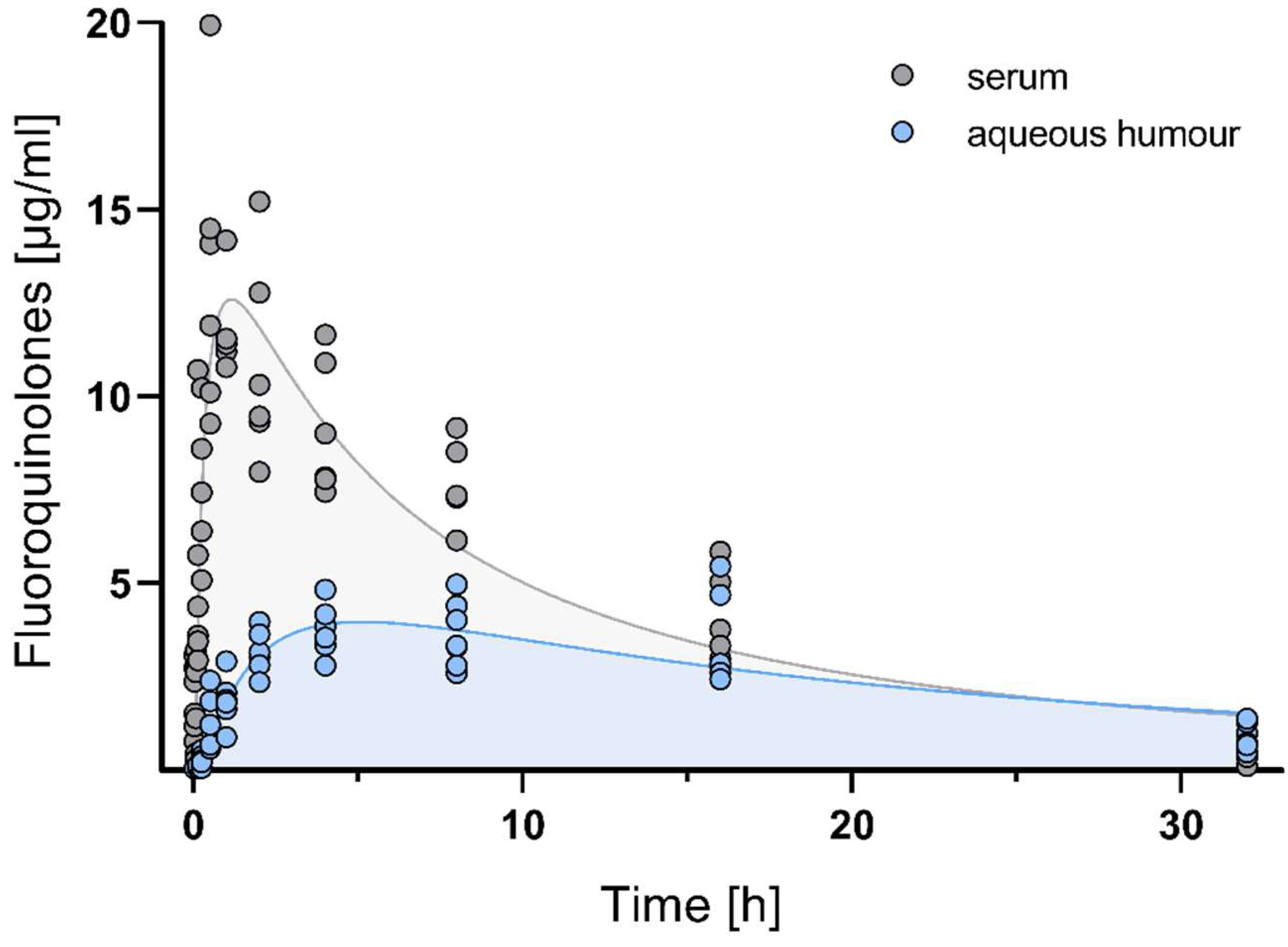
| Serum | Aqueous Humour | |||
|---|---|---|---|---|
| Parameter | Mean ± SD | Range | Mean ± SD | Range |
| Cmax (µg/mL) | 10.67 ± 0.5 | 10.2–11.3 | 4.52 ± 1.2 | 3.2–6.6 |
| tmax (h) | 2.82 ± 0.1 | 2.7–2.9 | 7.54 ± 1.0 | 6.2–8.7 |
| AUC0-inf (µg·h/mL) | 155.40 ± 39.1 | 117.58–204.75 | 99.57 ± 26.8 | 71.88–137.83 |
| MRT (h) | 9.52 ± 1.2 | 8.18–11.28 | 16.03 ± 1.9 * | 12.84–17.89 |
| Vd (L/kg) | 1.11 ± 0.2 | 0.88–1.46 | 3.16 ± 0.91 * | 1.97–4.2 |
| Cl (L/h/kg) | 0.17 ± 0.04 | 0.18–0.21 | 0.27 ± 0.06 * | 0.18–0.35 |
| t1/2 ß (h) | 4.36 ± 0.38 | 4.07–5.06 | 8.79 ± 0.93 * | 7.54–10.37 |
| Serum | Aqueous Humour | |||||
|---|---|---|---|---|---|---|
| Time Point | ENR (µg/mL) | CPR (µg/mL) | Proportion of CPR (%) | ENR (µg/mL) | CPR (µg/mL) | Proportion of CPR (%) |
| 1 h | 8.74 | 0.17 | 1.9 | 1.3 | 0.02 | 1.4 |
| 4 h | 6.53 | 0.72 | 9.9 | 1.46 | 0.05 | 3.3 |
| 8 h | 4.93 | 0.71 | 12.6 | 1.48 | 0.08 | 5.4 |
| Bacteria | MIC | Avian Host | Geographic Area | References |
|---|---|---|---|---|
| Salmonella enteritidis | 0.0625–1 µg/mL | Broiler | South Korea | [22] |
| Salmonella typhimurium | 0.12–16 µg/mL | Chicken | China | [23] |
| Mycoplasmopsis (Mycoplasma) synoviae | 4–32 µg/mL | Chicken | China | [24] |
| 0.031–32 µg/mL | Poultry | Europe | [25] | |
| 0.625–>10 µg/mL | Chicken | Asia | [26] | |
| 1.0–>16 µg/mL | Poultry | Italy | [27] | |
| Mycoplasma gallisepticum | 0.031–16 µg/mL | Poultry | Europe | [25] |
| ≤0.039–5 µg/mL | Chicken | Asia | [26] | |
| Escherichia coli | 8–256 µg/mL | Chicken | Asia | [23] |
| 0.5 µg/mL | Chicken | Asia | [28] | |
| 0.016–>16 µg/mL | Poultry | Germany | [29] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuchs, K.; Rinder, M.; Dietrich, R.; Banspach, L.; Ammer, H.; Korbel, R. Penetration of Enrofloxacin in Aqueous Humour of Avian Eyes. Vet. Sci. 2023, 10, 5. https://doi.org/10.3390/vetsci10010005
Fuchs K, Rinder M, Dietrich R, Banspach L, Ammer H, Korbel R. Penetration of Enrofloxacin in Aqueous Humour of Avian Eyes. Veterinary Sciences. 2023; 10(1):5. https://doi.org/10.3390/vetsci10010005
Chicago/Turabian StyleFuchs, Katrin, Monika Rinder, Richard Dietrich, Leena Banspach, Hermann Ammer, and Rüdiger Korbel. 2023. "Penetration of Enrofloxacin in Aqueous Humour of Avian Eyes" Veterinary Sciences 10, no. 1: 5. https://doi.org/10.3390/vetsci10010005
APA StyleFuchs, K., Rinder, M., Dietrich, R., Banspach, L., Ammer, H., & Korbel, R. (2023). Penetration of Enrofloxacin in Aqueous Humour of Avian Eyes. Veterinary Sciences, 10(1), 5. https://doi.org/10.3390/vetsci10010005






