SARS-CoV-2 Persistence: Data Summary up to Q2 2020
Abstract
1. Introduction
1.1. Background
- Common human coronaviruses: HCoV-OC43 and HCoV-HKU1 (Betacoronavirus) and HCoV-229E and HCoV-NL63 (Alphacoronavirus); they cause common colds and severe lower respiratory infections
- other human Coronaviruses (Betacoronavirus): SARS-CoV, MERS-CoV and 2019-nCoV (now called SARS-CoV-2).
- Handles and public transport handholds;
- Buttons;
- Mobile phones and electronics;
1.2. Summary and Aim
- The study aims to evaluate new coronavirus persistence on different:
- ○
- Surfaces;
- ○
- Temperatures;
- ○
- Humidity conditions;
- ○
- pH.
2. Data Description
2.1. Data Features
- Virus: type of investigated virus;
- Authors and Year: authors, reference (according to journal guidelines), and year of publication;
- Investigated Material: investigated material surfaced by the study;
- Time: persistence time of virus;
- Note on Results: additional notes on results.
2.1.1. Coronavirus Features
- Alphacoronavirus (α-CoV);
- Betacoronavirus (β-CoV);
- Gammacoronavirus (γ-CoV);
- Deltacoronavirus (δ-CoV).
- (1)
- Human Coronavirus 229E (HCoV-229E);
- (2)
- Human Coronavirus OC43 (HCoV-OC43);
- (3)
- Human Coronavirus NL63 (HCoV-NL63);
- (4)
- Human Coronavirus HKU1 (HCoV-HFU1);
- (5)
- Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV)
- (6)
- Middle Eastern Coronavirus Respiratory Syndrome (MERS-CoV)
- (7)
- Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), responsible for the COVID-19 disease.
- (1)
- Human coronavirus 229E (HCoV-229E) is a species of coronavirus that infects humans and bats. The species belongs to the genus Alphacoronavirus and is the only one of the subgenus Duvinacovirus. It is a single-stranded, positive-sense enveloped RNA virus that enters its host cell by binding to the APN (Membrane alanyl aminopeptidase also known as alanyl aminopeptidase (AAP) or aminopeptidase N) receptor. HCoV-229E is associated with a range of respiratory symptoms, ranging from the common cold to high morbidity outcomes such as pneumonia and bronchiolitis.
- (2)
- The human coronavirus OC43 (HCoV-OC43) is a viral strain of the Betacoronavirus 1 virus, which infects humans and livestock, of the genus Betacoronavirus of the subgenus Embecovirus, as it possesses haemagglutinin esterase (HE) among the surface proteins. It is a single-stranded, positive-sense enveloped RNA virus that enters its host cell by binding to the n-acetyl-9-O-acetylneuraminic acid receptor. Together with the human coronavirus 229E, it is one of the viruses responsible for the common cold
- (3)
- Human coronavirus NL63 (HCoV-NL63) is a species of coronavirus identified in late 2004 in a seven-month-old baby with bronchiolitis in the Netherlands. The virus is a positive-sense monofilament RNA virus that enters its host cell via the ACE2 receptor. The virus originated from infected palm owls and bats. Infection with the virus has been confirmed worldwide, associated diseases include mild to moderate upper respiratory tract infections, severe lower respiratory tract infection, and bronchiolitis, also responsible for gastrointestinal manifestations. Recent data suggest an association of HCoV-NL63 infection with Kawasaki disease, a systemic vasculitis in childhood that can result in coronary artery aneurysms [36]. In the developed world, Kawasaki disease is the most common cause of acquired heart disease in children.
- (4)
- Human coronavirus HKU1 (HCoV-HKU1) is a species of virus originating from infected mice, of the genus Betacoronavirus, subgenus Embecovirus, as it has the hemagglutinin esterase (HE) gene. In humans, the infection causes upper respiratory disease with symptoms of the common cold, but it can progress to pneumonia and bronchiolitis.
- (5)
- The severe acute respiratory syndrome coronavirus, abbreviated to SARS-CoV (Severe Acute Respiratory Syndrome—Coronavirus), is a viral strain at the origin of the 2003 SARS epidemic. SARS-CoV is one of the positive polarity single-chain RNA viruses (group IV of the Baltimore classification), of the SARS-related coronavirus species, belonging to the genus of Betacoronavirus.
- (6)
- MERS (Middle East Respiratory Syndrome) or coronavirus Middle East respiratory syndrome is a disease caused by the coronavirus MERS-CoV. The virus causing the pathology is a coronavirus similar to the virus causing SARS, but the disease it causes, although similar to SARS, seems to cause greater mortality, in fact, its lethality rate is approximately 34%, while for SARS it is 10%.
- (7)
- The severe acute respiratory syndrome coronavirus 2, abbreviated to SARS-CoV-2 (acronym from severe acute respiratory syndrome coronavirus 2) [1,2], previously named new coronavirus of 2019 (2019-nCoV [3], or also 2019 nCoV-ARD) [4], is a viral strain of the SARS-related coronavirus/SARS-CoV species, belonging to the genus Betacoronavirus. Already described in the previous chapter.
2.1.2. Coronaviruses Testing Methods
2.2. Data Synthesis
2.2.1. Coronavirus on Different Surfaces
2.2.2. Coronaviruses in Different Temperatures and Humidity
- Resistance of the microorganism;
- Relative humidity of the air;
- Air temperature and sunlight;
- Aerosol composition.
2.2.3. Coronaviruses and Chemo-Physical Treatments
- Alcohols: both ethanol and propanol can interact with natural fibers causing swelling, but also their use on synthetic fibers, normally resistant to alcohol, could cause irreversible damage to colored garments, giving rise to discoloration phenomena or dissolution. In addition, the use of alcohol-based products, especially if used in a sprayed form, represents an additional risk factor related to flammability.
- Sodium hypochlorite and hydrogen peroxide: they are not recommended because they could damage colored garments causing the release of color and the formation of stains.
- Ozone: although it is capable of acting quickly on viruses, and although it has special wardrobes or boxes or other containers on the market to carry out the treatment, its use should be carefully evaluated since its oxidizing power could alter the colors of the garments and the exposure times would be a critical factor to control.
- Other chemical substances: those capable of lowering/raising the pH to denature the proteins of the viruses are strongly discouraged. In fact, natural fibers, but also some synthetic fibers, could be subject to degradation or swelling phenomena below pH 3 and above pH 10–11; moreover, the resistance, appearance, and ecotoxicological characteristics of the garment could be irreparably compromised (the major eco-toxicological specifications provide for a pH for the fabrics between 4.0 and 7.5).
- Dry steam, in principle, is not a problem since it is already used in fabric finishing operations. The transfer of steam, as a means of contrasting the virus in a commercial context, could be practicable by the same sales staff with portable vaporizers even if the time necessary for the heat to be really effective for the complexity of the article is not standardizable, i.e., the creases, seams, turn-ups, etc., which may require a longer steaming time. It should be emphasized that any use of vaporizers should be carried out in separate rooms, to be ventilated abundantly after applying the steam in order to avoid the transfer of any contaminants from the treated fabrics to the operator by means of aerosols.
- UV radiation, in particular of the UV-C spectrum between 207–222 nm, was able to eradicate type A influenza viruses (H1N1 virus) and it can be assumed that a few minutes of application would be sufficient to also inactivate SARS-CoV-2 on clothing and clothing (both sunlight or artificial lamp). Although the use of germicidal lamps has been widespread for decades and they are already used in sanitization processes, the following critical factors for UV radiation’s potential use in the textile field should be carefully considered:
- Poor penetration; it does not penetrate paper, glass, clothing and, if the virus is nested in the fabric, it risks not being reached;
- Minimum distance from UV source to treated material;
- Energy and lamp costs (replacement every 8000 h);
- Dependence on environmental conditions (relative humidity);
- Containment of operator exposure considering mutagenicity for humans (skin, eyes): in this case the 222 nm radiation, the most active on viruses, is the least penetrating in the skin and dangerous for humans but the eyes should still be protected;
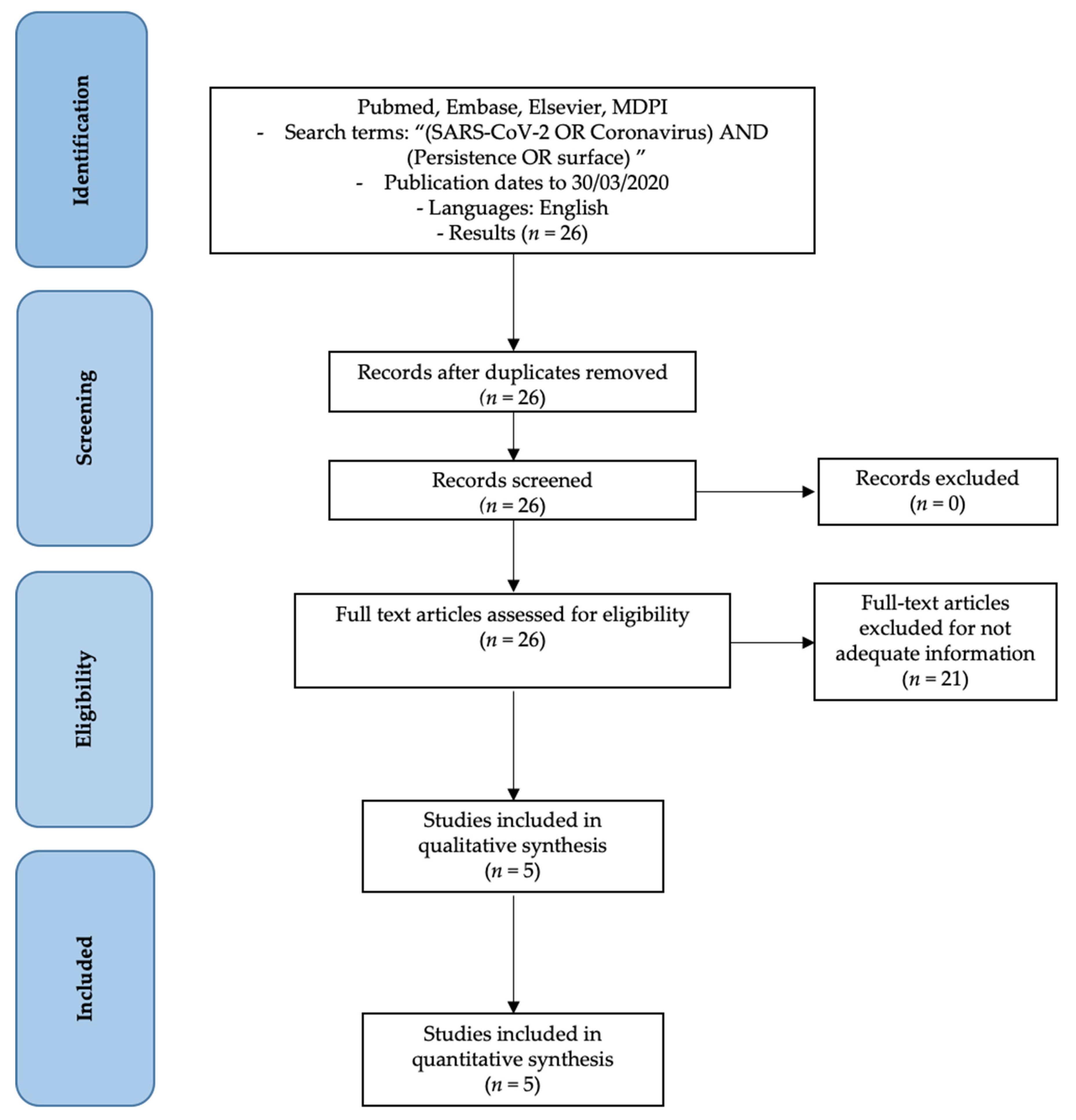
3. Methods
- SARS-CoV-2 features articles;
- SARS-CoV-2 persistence;
- The persistence of other coronaviruses.
- Not enough information regarding the topic;
- No access to the title and abstract.
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control and Prevention. Symptoms of Coronavirus. Available online: https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Fabout%2Fsymptoms.html (accessed on 17 August 2020).
- Expert Reaction to News Reports That the China Coronavirus May Spread before Symptoms Show. Available online: https://www.sciencemediacentre.org/expert-reaction-to-news-reports-that-the-china-coronavirus-may-spread-before-symptoms-show/ (accessed on 17 August 2020).
- Zhang, Y.; Xu, J.; Li, H.; Cao, B. A novel coronavirus (COVID-19) outbreak: A call for action. Chest 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, Y.; Yang, L.; Wang, C. Suspending Classes without Stopping Learning: China’s Education Emergency Management Policy in the COVID-19 Outbreak. J. Risk Financ. Manag. 2020, 13, 55. [Google Scholar] [CrossRef]
- Yu, H.; Sun, X.; Solvang, W.D.; Zhao, X. Reverse Logistics Network Design for Effective Management of Medical Waste in Epidemic Outbreaks: Insights from the Coronavirus Disease 2019 (COVID-19) Outbreak in Wuhan (China). Int. J. Environ. Res. Public Health 2020, 17, 1770. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Shang, W.; Rao, X. Facing the COVID-19 outbreak: What should we know and what could we do? J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Ravalli, S.; Musumeci, G. Coronavirus Outbreak in Italy: Physiological Benefits of Home-Based Exercise During Pandemic. J. Funct. Morphol. Kinesiol. 2020, 5, 31. [Google Scholar] [CrossRef]
- Morawska, L.; Tang, J.W.; Bahnfleth, W.; Bluyssen, P.M.; Boerstra, A.; Buonanno, G.; Cao, J.; Dancer, S.; Floto, A.; Franchimon, F.; et al. How can airborne transmission of COVID-19 indoors be minimised? Environ. Int. 2020, 142, 105832. [Google Scholar] [CrossRef]
- Speroni, S.; Cicciù, M.; Maridati, P.; Grossi, G.B.; Maiorana, C. Clinical investigation of mucosal thickness stability after soft tissue grafting around implants: A 3-year retrospective study. Indian J. Dent. Res. 2010, 21, 474–479. [Google Scholar] [CrossRef]
- Isola, G.; Matarese, M.; Ramaglia, L.; Cicciù, M.; Matarese, G. Evaluation of the efficacy of celecoxib and ibuprofen on postoperative pain, swelling, and mouth opening after surgical removal of impacted third molars: A randomized, controlled clinical trial. Int. J. Oral Maxillofac. Surg. 2019, 48, 1348–1354. [Google Scholar] [CrossRef]
- Cicciù, M.; Risitano, G.; Lo Giudice, G.; Bramanti, E. Periodontal health and caries prevalence evaluation in patients affected by Parkinson’s disease. Parkinson’s Disease 2012. [Google Scholar] [CrossRef]
- Xu, B.; Kraemer, M.U.G. Open access epidemiological data from the COVID-19 outbreak. Lancet Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Pan, R.; Wan, X.; Tan, Y.; Xu, L.; Ho, C.S.; Ho, R.C. Immediate Psychological Responses and Associated Factors during the Initial Stage of the 2019 Coronavirus Disease (COVID-19) Epidemic among the General Population in China. Int. J. Environ. Res. Public Health 2020, 17, 1729. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Cheng, Z.; Yue, X.-G.; McAleer, M. Risk Management of COVID-19 by Universities in China. J. Risk Financ. Manag. 2020, 13, 36. [Google Scholar] [CrossRef]
- The, L. COVID-19: Fighting panic with information. Lancet 2020, 395, 537. [Google Scholar] [CrossRef]
- Tetro, J.A. Is COVID-19 Receiving ADE From Other Coronaviruses? Microbes Infect. 2020. [Google Scholar] [CrossRef]
- Sixto-Costoya, A.; Aleixandre-Benavent, R.; Lucas-Domínguez, R.; Vidal-Infer, A. The Emergency Medicine Facing the Challenge of Open Science. Data 2020, 5, 28. [Google Scholar] [CrossRef]
- Lavrova, A.I.; Esmedljaeva, D.S.; Belik, V.; Postnikov, E.B. Matrix Metalloproteinases as Markers of Acute Inflammation Process in the Pulmonary Tuberculosis. Data 2019, 4, 137. [Google Scholar] [CrossRef]
- Nishiura, H. Backcalculating the Incidence of Infection with COVID-19 on the Diamond Princess. J. Clin. Med. 2020, 9, 657. [Google Scholar] [CrossRef]
- Nguyen, T.; Duong Bang, D.; Wolff, A. 2019 Novel Coronavirus Disease (COVID-19): Paving the Road for Rapid Detection and Point-of-Care Diagnostics. Micromachines 2020, 11, 306. [Google Scholar] [CrossRef]
- McAleer, M. Prevention Is Better Than the Cure: Risk Management of COVID-19. J. Risk Financ. Manag. 2020, 13, 46. [Google Scholar] [CrossRef]
- Liu, Z.; Magal, P.; Seydi, O.; Webb, G. Understanding Unreported Cases in the COVID-19 Epidemic Outbreak in Wuhan, China, and the Importance of Major Public Health Interventions. Biology 2020, 9, 50. [Google Scholar] [CrossRef] [PubMed]
- Fiorillo, L.; Cervino, G.; Matarese, M.; D’Amico, C.; Surace, G.; Paduano, V.; Fiorillo, M.T.; Moschella, A.; La Bruna, A.; Romano, G.L.; et al. COVID-19 Surface Persistence: A Recent Data Summary and Its Importance for Medical and Dental Settings. Int. J. Environ. Res. Public Health 2020, 17, 3132. [Google Scholar] [CrossRef] [PubMed]
- Mascitti, M.; Campisi, G. Dental Public Health Landscape: Challenges, Technological Innovation and Opportunities in the 21st Century and COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2020, 17, 3636. [Google Scholar] [CrossRef]
- Cervino, G.; Oteri, G. COVID-19 Pandemic and Telephone Triage before Attending Medical Office: Problem or Opportunity? Medicina 2020, 56, 250. [Google Scholar] [CrossRef] [PubMed]
- Roosa, K.; Lee, Y.; Luo, R.; Kirpich, A.; Rothenberg, R.; Hyman, J.M.; Yan, P.; Chowell, G. Short-term Forecasts of the COVID-19 Epidemic in Guangdong and Zhejiang, China: 13–23 February 2020. J. Clin. Med. 2020, 9, 596. [Google Scholar] [CrossRef] [PubMed]
- Repici, A.; Maselli, R.; Colombo, M.; Gabbiadini, R.; Spadaccini, M.; Anderloni, A.; Carrara, S.; Fugazza, A.; Di Leo, M.; Galtieri, P.A.; et al. Coronavirus (COVID-19) outbreak: What the department of endoscopy should know. Gastrointest. Endosc. 2020. [Google Scholar] [CrossRef]
- Qin, C.; Liu, F.; Yen, T.C.; Lan, X. (18)F-FDG PET/CT findings of COVID-19: A series of four highly suspected cases. Eur. J. Nucl. Med. Mol. Imaging 2020. [Google Scholar] [CrossRef]
- Porcheddu, R.; Serra, C.; Kelvin, D.; Kelvin, N.; Rubino, S. Similarity in Case Fatality Rates (CFR) of COVID-19/SARS-COV-2 in Italy and China. J. Infect. Dev. Ctries 2020, 14, 125–128. [Google Scholar] [CrossRef]
- Peeri, N.C.; Shrestha, N.; Rahman, M.S.; Zaki, R.; Tan, Z.; Bibi, S.; Baghbanzadeh, M.; Aghamohammadi, N.; Zhang, W.; Haque, U. The SARS, MERS and novel coronavirus (COVID-19) epidemics, the newest and biggest global health threats: What lessons have we learned? Int. J. Epidemiol. 2020. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Chin, A.W.H.; Chu, J.T.S.; Perera, M.R.A.; Hui, K.P.Y.; Yen, H.-L.; Chan, M.C.W.; Peiris, M.; Poon, L.L.M. Stability of SARS-CoV-2 in different environmental conditions. Lancet Microbe 2020, 1, e10. [Google Scholar] [CrossRef]
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmann, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020, 104, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Warnes, S.L.; Little, Z.R.; Keevil, C.W. Human Coronavirus 229E Remains Infectious on Common Touch Surface Materials. mBio 2015, 6, e01697. [Google Scholar] [CrossRef] [PubMed]
- Fiorillo, L.; De Stefano, R.; Cervino, G.; Crimi, S.; Bianchi, A.; Campagna, P.; Herford, A.S.; Laino, L.; Cicciù, M. Oral and psychological alterations in haemophiliac patients. Biomedicines 2019, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Kahn, J.S.; McIntosh, K. History and recent advances in coronavirus discovery. Pediatr. Infect. Dis. J. 2005, 24, S223–S227. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Shi, L.; Zhang, W.; He, J.; Liu, C.; Zhao, C.; Kong, S.K.; Loo, J.F.C.; Gu, D.; Hu, L. Prevalence and genetic diversity analysis of human coronaviruses among cross-border children. Virol. J. 2017, 14, 230. [Google Scholar] [CrossRef]
- Paoli, A.; Musumeci, G. Elite Athletes and COVID-19 Lockdown: Future Health Concerns for an Entire Sector. J. Funct. Morphol. Kinesiol. 2020, 5, 30. [Google Scholar] [CrossRef]
- Giudice, A.; Barone, S.; Muraca, D.; Averta, F.; Diodati, F.; Antonelli, A.; Fortunato, L. Can Teledentistry Improve the Monitoring of Patients during the Covid-19 Dissemination? A Descriptive Pilot Study. Int. J. Environ. Res. Public Health 2020, 17, 3399. [Google Scholar] [CrossRef]
- Cavallo, L.; Marcianò, A.; Cicciù, M.; Oteri, G. 3D Printing beyond Dentistry during COVID 19 Epidemic: A Technical Note for Producing Connectors to Breathing Devices. Prosthesis 2020, 2, 46–52. [Google Scholar]
- Fiorillo, L.; Leanza, T. Worldwide 3D Printers against the New Coronavirus. Prosthesis 2020, 2, 9. [Google Scholar] [CrossRef]
- Demongeot, J.; Flet-Berliac, Y.; Seligmann, H. Temperature Decreases Spread Parameters of the New Covid-19 Case Dynamics. Biology 2020, 9, 94. [Google Scholar] [CrossRef] [PubMed]
- Roques, L.; Klein, E.K.; Papaïx, J.; Sar, A.; Soubeyrand, S. Using Early Data to Estimate the Actual Infection Fatality Ratio from COVID-19 in France. Biology 2020, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Kenyon, C. COVID-19 Infection Fatality Rate Associated with Incidence—A Population-Level Analysis of 19 Spanish Autonomous Communities. Biology 2020, 9, 128. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, G.; De Vito, D.; Rengo, S.; Tatullo, M. COVID-19 Outbreak: An Overview on Dentistry. Int. J. Environ. Res. Public Health 2020, 17, 2094. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, L.R.; Consani, R.L.X.; Mesquita, M.F.; Sinhoreti, M.A.C. Effect of chemical and microwave disinfection on the surface microhardness of acrylic resin denture teeth. J. Prosthodont. 2013, 22, 298–303. [Google Scholar] [CrossRef]
- Koscova, J.; Hurnikova, Z.; Pistl, J. Degree of Bacterial Contamination of Mobile Phone and Computer Keyboard Surfaces and Efficacy of Disinfection with Chlorhexidine Digluconate and Triclosan to Its Reduction. Int. J. Environ. Res. Public Health 2018, 15, 2238. [Google Scholar] [CrossRef]
- Kampf, G. Biocidal Agents Used for Disinfection Can Enhance Antibiotic Resistance in Gram-Negative Species. Antibiotics 2018, 7, 110. [Google Scholar] [CrossRef]
- Haseeb, R.; Lau, M.; Sheah, M.; Montagner, F.; Quiram, G.; Palmer, K.; Stefan, M.C.; Rodrigues, D.C. Synthesis and Characterization of New Chlorhexidine-Containing Nanoparticles for Root Canal Disinfection. Materials 2016, 9, 452. [Google Scholar] [CrossRef]
- Tang, B.; Scarabel, F.; Bragazzi, N.L.; McCarthy, Z.; Glazer, M.; Xiao, Y.; Heffernan, J.M.; Asgary, A.; Ogden, N.H.; Wu, J. De-Escalation by Reversing the Escalation with a Stronger Synergistic Package of Contact Tracing, Quarantine, Isolation and Personal Protection: Feasibility of Preventing a COVID-19 Rebound in Ontario, Canada, as a Case Study. Biology 2020, 9, 100. [Google Scholar] [CrossRef]
- Nastro, E.; Musolino, C.; Allegra, A.; Oteri, G.; Cicciù, M.; Alonci, A.; Quartarone, E.; Alati, C.; De Ponte, F.S. Bisphosphonate-associated osteonecrosis of the jaw in patients with multiple myeloma and breast cancer. Acta Haematol. 2007, 117, 181–187. [Google Scholar] [CrossRef]
- Cervino, G.; Fiorillo, L.; Arzukanyan, A.V.; Spagnuolo, G.; Cicciù, M. Dental restorative digital workflow: Digital smile design from aesthetic to function. Dent. J. 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Boyce, J.M. Modern technologies for improving cleaning and disinfection of environmental surfaces in hospitals. Antimicrob. Resist. Infect. Control. 2016, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.H.; Ge, L.; Li, L. The PRISMA Extension Statement. Ann. Intern. Med. 2015, 163, 566. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhou, X.; Yu, G.; Sun, X. The effects of the PRISMA statement to improve the conduct and reporting of systematic reviews and meta-analyses of nursing interventions for patients with heart failure. Int. J. Nurs. Pract. 2019. [Google Scholar] [CrossRef]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
| Diameter (µm) | Velocity (m/h) |
|---|---|
| 0.1 | 0.003 |
| 1 | 0.11 |
| 2 | 0.43 |
| 3 | 0.97 |
| 5 | 2.7 |
| 10 | 10.8 |
| 20 | 42 |
| Virus | Authors and Year | Investigated Material | Time | Note on Results |
|---|---|---|---|---|
| 2019-nCoV | Van Doremalen et al. 2020 [32] | aerosols | 3 h | Reduction from 103.5 to 102.7 TCID50 per liter of air |
| plastic | 72 h | Reduction from 103.7 to 100.6 TCID50 per millimeter | ||
| stainless steel | 48 h | from 103.7 to 100.6 TCID50 per millimeter | ||
| copper | 4 h | No viable SARS-CoV-2 | ||
| cardboard | 24 h | No viable SARS-CoV-2 | ||
| 2019-nCoV | Chin et al. [33] | Paper | 3 h | From 4.76 TCID50 at 0 min to 2.18 TCID50 at 30 min |
| Wood | 48 h | From 5.66 TCID50 at 0 min to 2.47 TCID50 at 6 h | ||
| Cloth | 48 h | From 4.84 TCID50 at 0 min to 2.25 TCID50 at 6 h | ||
| Glass | 96 h | From 5.83 TCID50 at 0 min to 5.06 TCID50 at 6 h | ||
| Stainless Steel | 7 days | From 5.80 TCID50 at 0 min to 5.24 TCID50 at 6 h | ||
| Plastic | 7 days | From 5.81 TCID50 at 0 min to 4.68 TCID50 at 6 h | ||
| Mask (inner layer) | 7 days | From 5.88 TCID50 at 0 min to 5.01 TCID50 at 6 h | ||
| Mask (outer layer) | / | From 5.78 TCID50 at 0 min to 4.97 TCID50 at 6 h to 2.79 at 7 days | ||
| Other coronaviruses | Van Doremalen et al. 2020 [32] | aerosols | 3 h | reduction from 104.3 to 103.5 TCID50 per liter of air |
| plastic | 72 h | from 103.4 to 100.7 TCID50 per millimeter | ||
| stainless steel | 48 h | from 103.6 to 100.6 TCID50 per millimeter | ||
| copper | 8 h | No viable SARS-CoV-1 | ||
| cardboard | 8 h | No viable SARS-CoV-1 | ||
| Kampf et al. 2020 [34] | paper | 5 min up to 5 days | 105 TCID50 per millimeter | |
| glass | 4–5 days | 104 TCID50 per millimeter | ||
| plastic | 2–9 days | 106 TCID50 per millimeter | ||
| PVC | 5 days | 103 TCID50 per millimeter | ||
| silicon rubber | 5 days | 103 TCID50 per millimeter | ||
| surgical gloves (latex) | 5 days | 103 TCID50 per millimeter | ||
| disposable gowns | 1–2 days | 105 TCID50 per millimeter | ||
| Warnes et al. 2015 [35] | polyfluorotetraethylene (PTFE) | 5 days | 103 TCID50 per millimeter | |
| ceramic | 5 days | 103 TCID50 per millimeter | ||
| glass | 5 days | 103 TCID50 per millimeter | ||
| stainless steel | 5 days | 103 TCID50 per millimeter | ||
| polyvinyl chloride (PVC) | 5 days | 103 TCID50 per millimeter | ||
| silicon rubber | 3 days | 103 TCID50 per millimeter | ||
| brasses containing copper | <40 min | 103 TCID50 per millimeter | ||
| copper nickels | 120 min | 103 TCID50 per millimeter | ||
| zinc | 60 min |
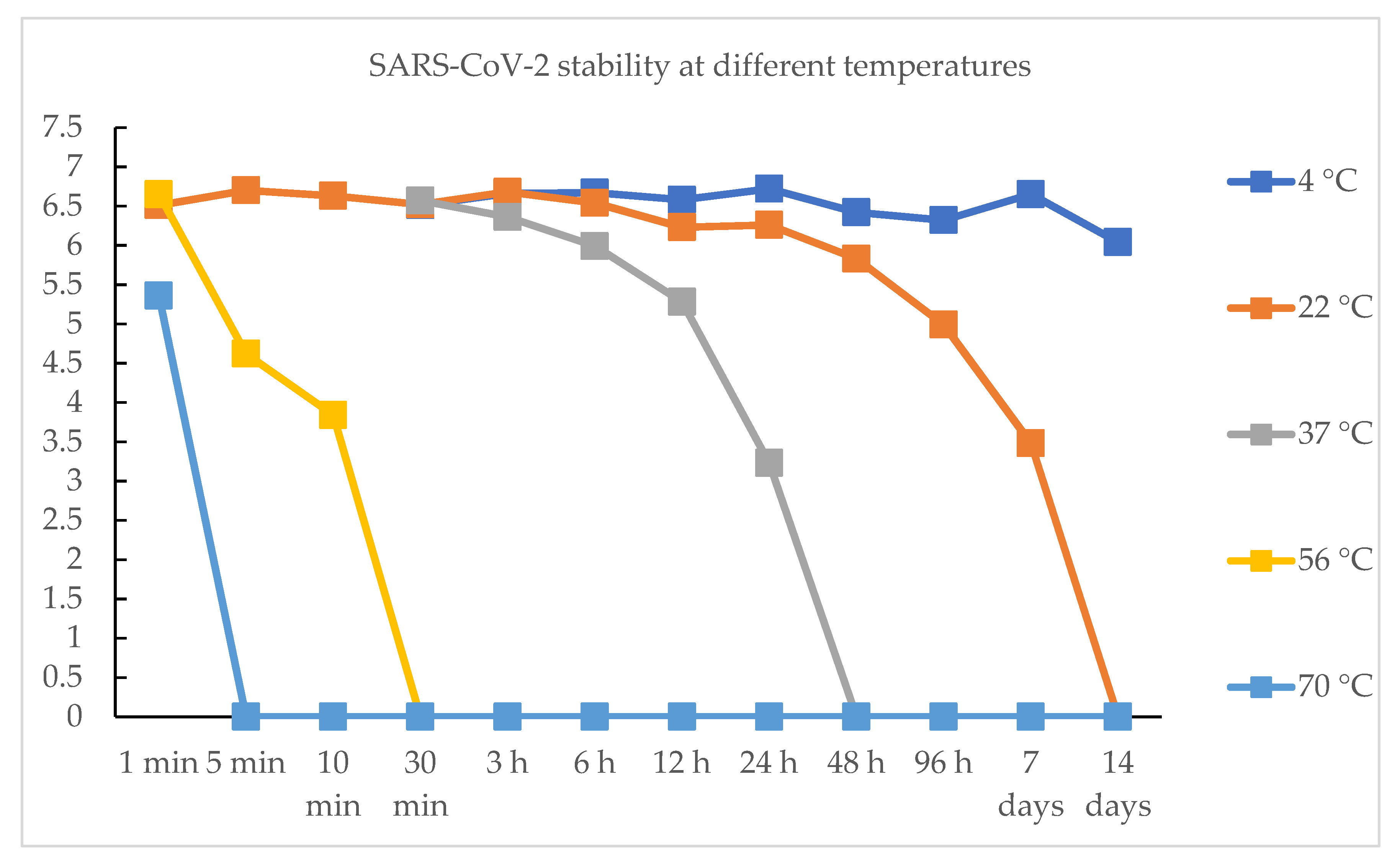
| Time | 4 °C | 22 °C | 37 °C | 56 °C | 70 °C |
|---|---|---|---|---|---|
| 1 min | / | 6.51 TCID50 | / | 6.65 TCID50 | 5.36 TCID50 |
| 5 min | / | 6.7 TCID50 | / | 4.62 TCID50 | / |
| 30 min | 6.51 TCID50 | 6.52 TCID50 | 6.57 TCID50 | / | / |
| 6 h | 6.67 TCID50 | 6.54 TCID50 | 5.99 TCID50 | / | / |
| 12 h | 6.58 TCID50 | 6.23 TCID50 | 5.28 TCID50 | / | / |
| 24 h | 6.72 TCID50 | 6.26 TCID50 | 3.23 TCID50 | / | / |
| 7 days | 6.65 TCID50 | 3.48 TCID50 | / | / | / |
| 14 days | 6.04 TCID50 | / | / | / | / |
| pH | Log TCID50/mL |
|---|---|
| 3 | 5.55 |
| 4 | 5.67 |
| 5 | 5.73 |
| 6 | 5.75 |
| 7 | 5.58 |
| 8 | 5.70 |
| 9 | 5.54 |
| 10 | 5.51 |
| Type of Surface or Material | Detergent/Disinfectant |
|---|---|
| Surfaces in stone, metal, or glass except wood | Neutral detergent and disinfectant virucidal—sodium hypochlorite 0.1% or ethanol (ethyl alcohol) at 70% or other concentration, provided that virucidal is specified |
| Wooden surfaces | Neutral detergent and virucidal disinfectant (against viruses) based on ethanol (70%) or quaternary ammoniums (e.g., benzalkonium chloride; DDAC (Didecyldimethylammonium chloride)) |
| Toilets | Cleaning with detergent and disinfection with disinfectant based on sodium hypochlorite at least 0.1% sodium hypochlorite |
| Textiles (e.g., cotton, linen) | Washing with hot water (70 °C–90 °C) and normal laundry detergent; alternatively: low temperature washing with bleach or other disinfectants for laundry. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cervino, G.; Fiorillo, L.; Surace, G.; Paduano, V.; Fiorillo, M.T.; De Stefano, R.; Laudicella, R.; Baldari, S.; Gaeta, M.; Cicciù, M. SARS-CoV-2 Persistence: Data Summary up to Q2 2020. Data 2020, 5, 81. https://doi.org/10.3390/data5030081
Cervino G, Fiorillo L, Surace G, Paduano V, Fiorillo MT, De Stefano R, Laudicella R, Baldari S, Gaeta M, Cicciù M. SARS-CoV-2 Persistence: Data Summary up to Q2 2020. Data. 2020; 5(3):81. https://doi.org/10.3390/data5030081
Chicago/Turabian StyleCervino, Gabriele, Luca Fiorillo, Giovanni Surace, Valeria Paduano, Maria Teresa Fiorillo, Rosa De Stefano, Riccardo Laudicella, Sergio Baldari, Michele Gaeta, and Marco Cicciù. 2020. "SARS-CoV-2 Persistence: Data Summary up to Q2 2020" Data 5, no. 3: 81. https://doi.org/10.3390/data5030081
APA StyleCervino, G., Fiorillo, L., Surace, G., Paduano, V., Fiorillo, M. T., De Stefano, R., Laudicella, R., Baldari, S., Gaeta, M., & Cicciù, M. (2020). SARS-CoV-2 Persistence: Data Summary up to Q2 2020. Data, 5(3), 81. https://doi.org/10.3390/data5030081










