Application of Microwaves as an Advanced Technique for the Development of Sherry Vinegar Macerated with Pineapple
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Maceration with Pineapple
2.3. Spectrophotometric Analysis
2.4. Analysis of Volatile Compounds
2.5. Sensory Analysis
2.6. Statistical Study
3. Results and Discussion
3.1. Optimization of the Product Development by Traditional Maceration
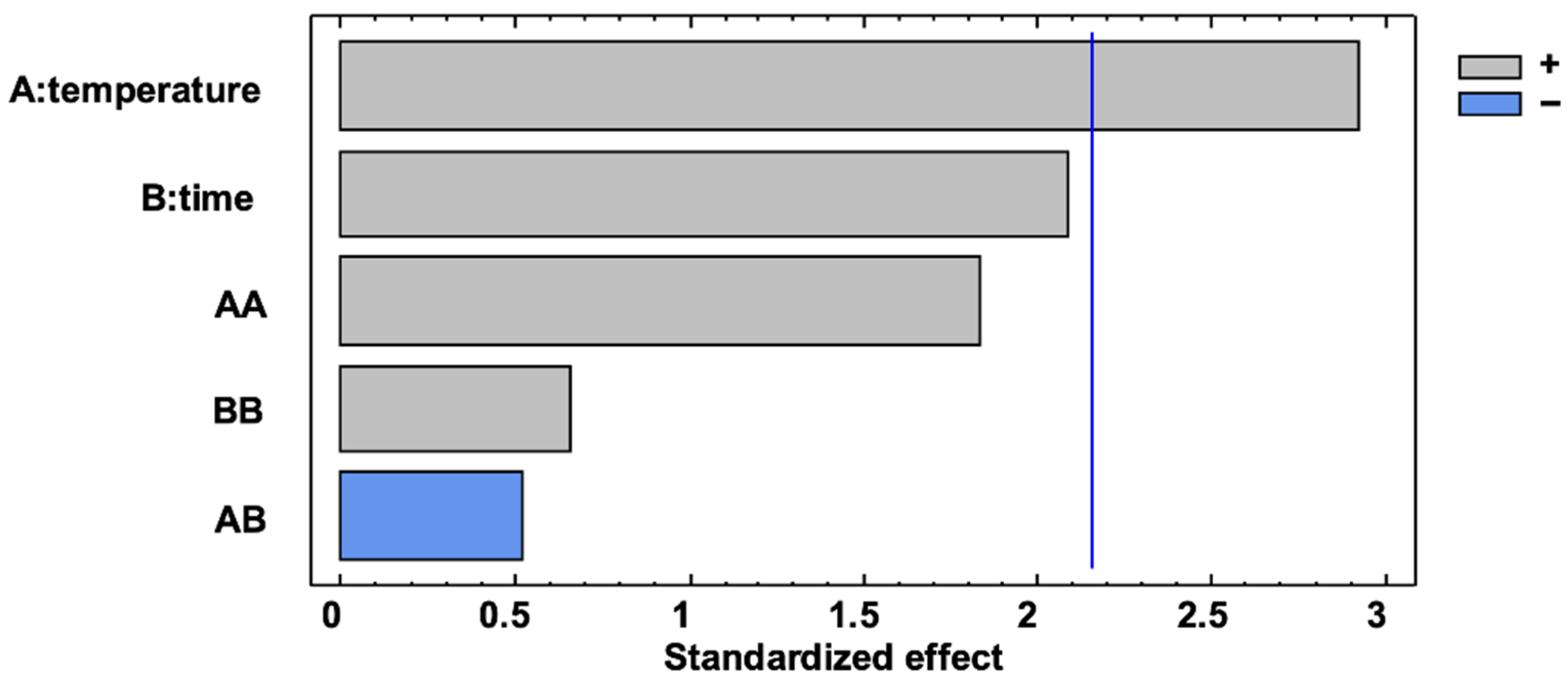
3.2. Optimization of Microwave Maceration
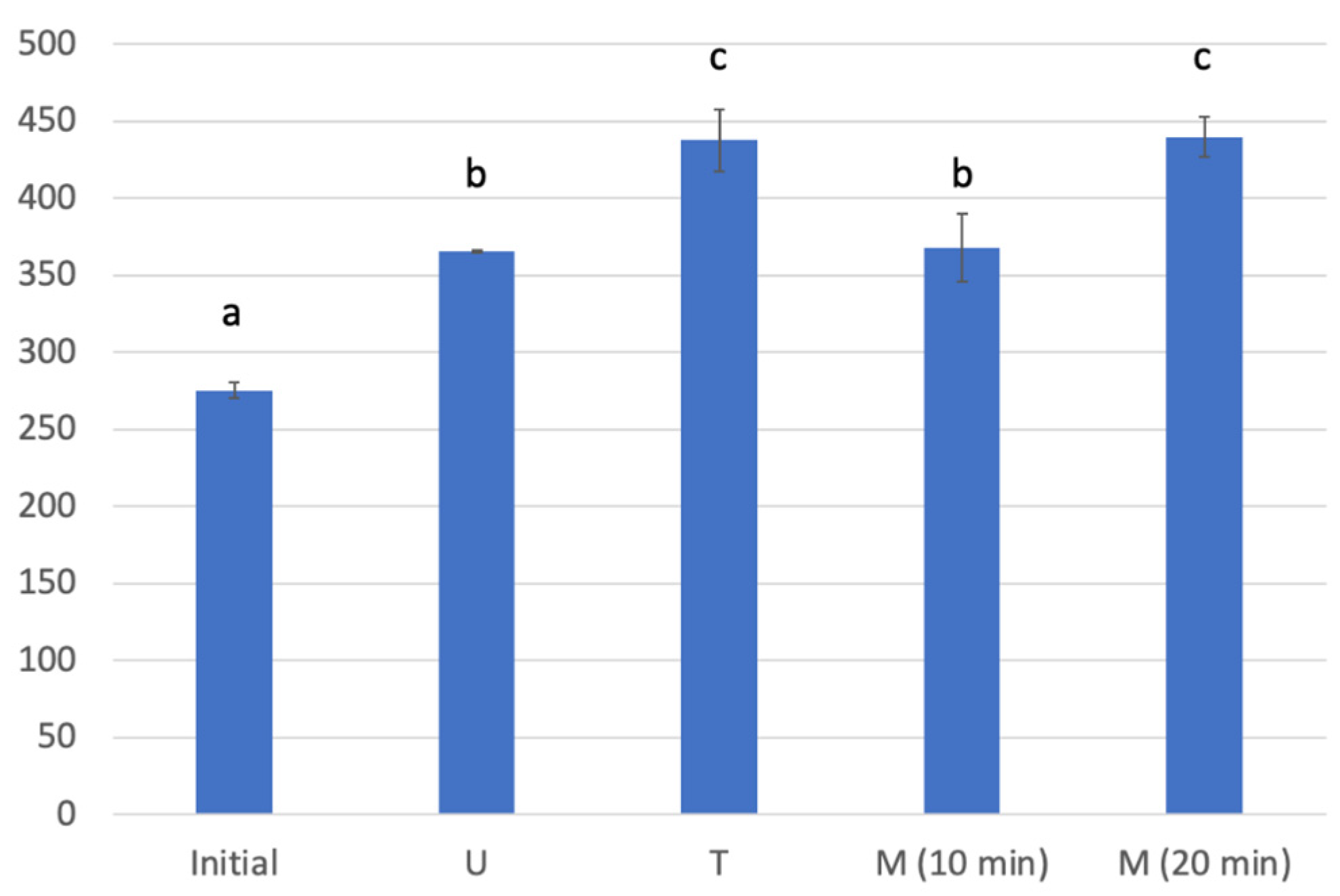
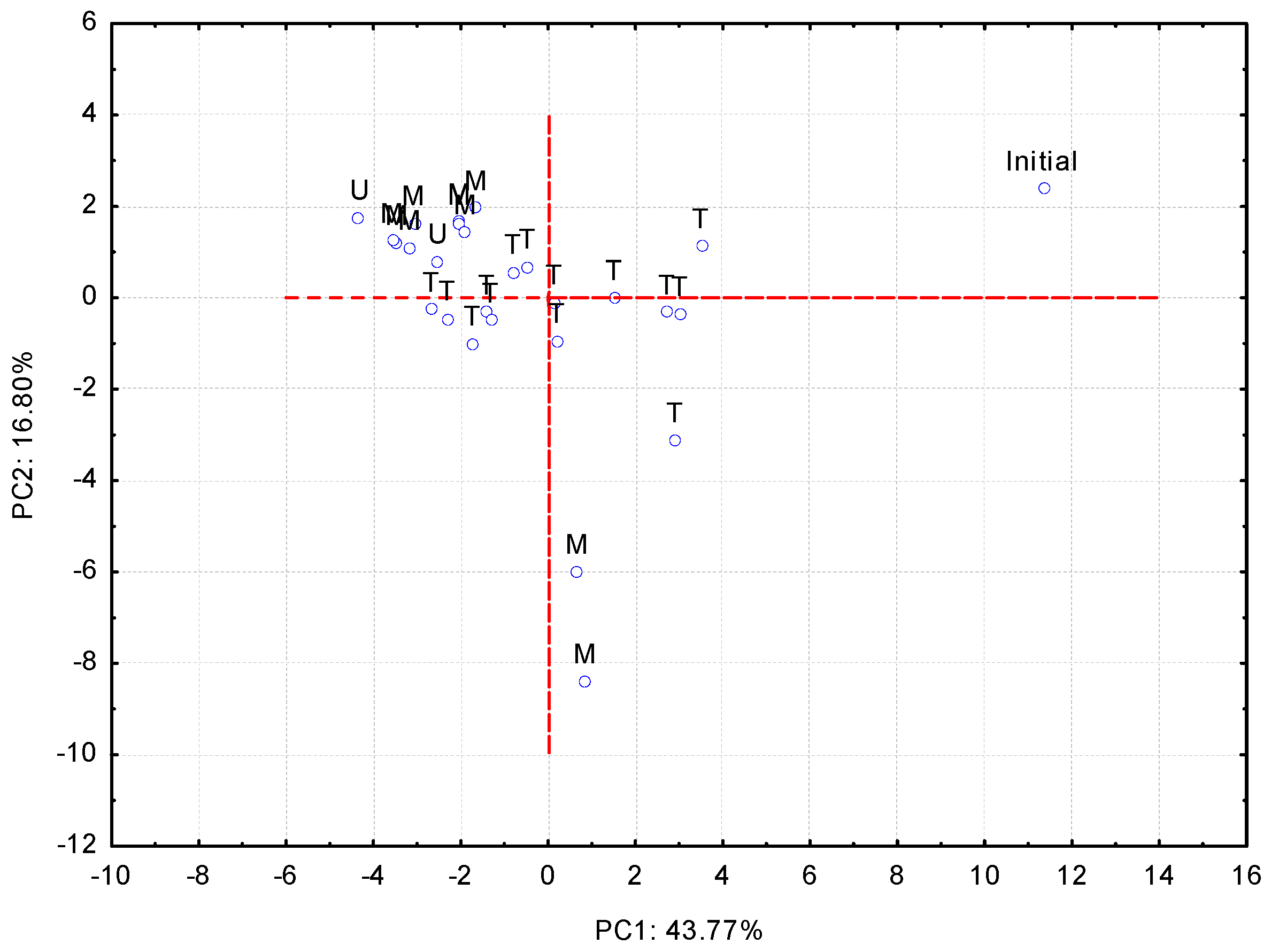
3.3. Comparison of the Extraction Methods
3.4. Analysis of Volatile Compounds
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cejudo-Bastante, M.J.; Dodero, M.C.R.; Guerrero, E.D.; Mejías, R.C.; Marín, R.N.; Barroso, C.G. Development and optimisation by means of sensory analysis of new beverages based on different fruit juices and sherry wine vinegar. J. Sci. Food Agric. 2012, 93, 741–748. [Google Scholar] [CrossRef]
- Bastante, M.J.C.; Guerrero, E.D.; Mejías, R.C.; Marín, R.N.; Dodero, M.C.R.; Barroso, C.G. Study of the Polyphenolic Composition and Antioxidant Activity of New Sherry Vinegar-Derived Products by Maceration with Fruits. J. Agric. Food Chem. 2010, 58, 11814–11820. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.-Y.; Sun, G.-M.; Liu, Y.-G.; Lv, L.-L.; Yang, W.-X.; Zhao, W.-F.; Wei, C.-B. Aroma Volatile Compounds from Two Fresh Pineapple Varieties in China. Int. J. Mol. Sci. 2012, 13, 7383–7392. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.M.; Hashim, N.; Aziz, S.A.; Lasekan, O. Pineapple (Ananas comosus): A comprehensive review of nutritional values, volatile compounds, health benefits, and potential food products. Food Res. Int. 2020, 137, 109675. [Google Scholar] [CrossRef]
- Marrufo-Curtido, A.; Cejudo-Bastante, M.J.; Rodríguez-Dodero, M.C.; Natera-Marín, R.; Castro-Mejías, R.; García-Barroso, C.; Durán-Guerrero, E. Novel vinegar-derived product enriched with dietary fiber: Effect on polyphenolic profile, volatile composition and sensory analysis. J. Food Sci. Technol. 2015, 52, 7608–7624. [Google Scholar] [CrossRef] [PubMed]
- Cejudo-Bastante, M.J.; Durán, E.; Castro, R.; Rodríguez-Dodero, M.C.; Natera, R.; García-Barroso, C. Study of the volatile composition and sensory characteristics of new Sherry vinegar-derived products by maceration with fruits. LWT 2013, 50, 469–479. [Google Scholar] [CrossRef]
- Bautista-Ortín, A.B.; Jiménez-Martínez, M.D.; Jurado, R.; Iniesta, J.A.; Terrades, S.; Andrés, A.; Gómez-Plaza, E. Application of high-power ultrasounds during red wine vinification. Int. J. Food Sci. Technol. 2017, 52, 1314–1323. [Google Scholar] [CrossRef]
- Tao, Y.; Zhang, Z.; Sun, D.-W. Experimental and modeling studies of ultrasound-assisted release of phenolics from oak chips into model wine. Ultrason. Sonochem. 2014, 21, 1839–1848. [Google Scholar] [CrossRef]
- Carew, A.L.; Gill, W.; Close, D.C.; Dambergs, R.G. Microwave Maceration with Early Pressing Improves Phenolics and Fermentation Kinetics in Pinot noir. Am. J. Enol. Vitic. 2014, 65, 401–406. [Google Scholar] [CrossRef]
- Casassa, L.F.; Sari, S.E.; Bolcato, E.A.; Fanzone, M.L. Microwave-Assisted Extraction Applied to Merlot Grapes with Contrasting Maturity Levels: Effects on Phenolic Chemistry and Wine Color. Fermentation 2019, 5, 15. [Google Scholar] [CrossRef]
- Delgado-González, M.; Sánchez-Guillén, M.; García-Moreno, M.; Rodríguez-Dodero, M.; García-Barroso, C.; Sánchez, D.G. Study of a laboratory-scaled new method for the accelerated continuous ageing of wine spirits by applying ultrasound energy. Ultrason. Sonochem. 2017, 36, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, I.; Pereira, R.; Clímaco, M.; Belchior, A.; De Sousa, R.B. Improved method for extraction of aroma compounds in aged brandies and aqueous alcoholic wood extracts using ultrasound. Anal. Chim. Acta 2004, 513, 125–134. [Google Scholar] [CrossRef]
- Jiménez-Sánchez, M.; Durán-Guerrero, E.; Rodríguez-Dodero, M.C.; Barroso, C.G.; Castro, R. Use of ultrasound at a pilot scale to accelerate the ageing of sherry vinegar. Ultrason. Sonochem. 2020, 69, 105244. [Google Scholar] [CrossRef] [PubMed]
- Bruna-Maynou, F.J.; Castro, R.; Rodríguez-Dodero, M.C.; Barroso, C.G.; Durán-Guerrero, E. Flavored Sherry vinegar with citric notes: Characterization and effect of ultrasound in the maceration of orange peels. Food Res. Int. 2020, 133, 109165. [Google Scholar] [CrossRef] [PubMed]
- Chanivet, M.; Durán-Guerrero, E.; Rodríguez-Dodero, M.D.C.; Barroso, C.G.; Castro, R. Application of accelerating energies to the maceration of sherry vinegar with citrus fruits. J. Sci. Food Agric. 2021, 101, 2235–2246. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, M.; Rodríguez, M.C.; Sanchez, M.; Guillén, D.A.; Barroso, C.G. Development of an accelerated aging method for Brandy. LWT 2014, 59, 108–114. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Duran-Guerrero, E.; Marín, R.N.; Mejías, R.C.; Barroso, C.G. Optimisation of stir bar sorptive extraction applied to the determination of volatile compounds in vinegars. J. Chromatogr. A 2006, 1104, 47–53. [Google Scholar] [CrossRef]
- Duran-Guerrero, E.; Marín, R.N.; Mejías, R.C.; Barroso, C.G. Stir bar sorptive extraction of volatile compounds in vinegar: Validation study and comparison with solid phase microextraction. J. Chromatogr. A 2007, 1167, 18–26. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Sensory Analysis. Guidelines for the Use of Quantitative Response Scales; ISO: Geneva, Switzerland, 2003; p. 4121. [Google Scholar]
- International Organization for Standardization (ISO). Sensory Analysis. Methodology. Ranking; ISO: Geneva, Switzerland, 2006; p. 8587. [Google Scholar]
- International Organization for Standardization (ISO). Sensory Analysis. General Guidance for the Design of Test Rooms; ISO: Geneva, Switzerland, 2007; p. 8589. [Google Scholar]
- International Organization for Standardization (ISO). Sensory Analysis. Apparatus. Wine-Tasting Glass; ISO: Geneva, Switzerland, 1977; p. 3591. [Google Scholar]
- Sommer, S.; Cohen, S.D. Comparison of Different Extraction Methods to Predict Anthocyanin Concentration and Color Characteristics of Red Wines. Fermentation 2018, 4, 39. [Google Scholar] [CrossRef]
- Wojdyło, A.; Samoticha, J.; Chmielewska, J. Effect of different pre-treatment maceration techniques on the content of phenolic compounds and color of Dornfelder wines elaborated in cold climate. Food Chem. 2021, 339, 127888. [Google Scholar] [CrossRef] [PubMed]
- Teai, T.; Claude-Lafontaine, A.; Schippa, C.; Cozzolino, F. Volatile Compounds in Fresh Pulp of Pineapple (Ananas comosus [L.] Merr.) from French Polynesia. J. Essent. Oil Res. 2001, 13, 314–318. [Google Scholar] [CrossRef]
- Tokitomo, Y.; Steinhaus, M.; Büttner, A.; Schieberle, P. Odor-Active Constituents in Fresh Pineapple (Ananas comosus [L.] Merr.) by Quantitative and Sensory Evaluation. Biosci. Biotechnol. Biochem. 2005, 69, 1323–1330. [Google Scholar] [CrossRef]
- Takeoka, G.R.; Buttery, R.G.; Teranishi, R.; Flath, R.A.; Guentert, M. Identification of additional pineapple volatiles. J. Agric. Food Chem. 1991, 39, 1848–1851. [Google Scholar] [CrossRef]
- Lukić, K.; Brnčić, M.; Ćurko, N.; Tomašević, M.; Tušek, A.J.; Ganić, K.K. Quality characteristics of white wine: The short- and long-term impact of high power ultrasound processing. Ultrason. Sonochem. 2020, 68, 105194. [Google Scholar] [CrossRef] [PubMed]
- Pino, J.A.; Queris, O. Analysis of volatile compounds of pineapple wine using solid-phase microextraction techniques. Food Chem. 2010, 122, 1241–1246. [Google Scholar] [CrossRef]
| No. | Pineapple Presentation | Concentration Ratio (g/L) | Stirring |
|---|---|---|---|
| 1 | Fresh | 200 | Shaking |
| 2 | Fresh | 200 | 300 rpm |
| 3 | Fresh | 400 | Shaking |
| 4 | Fresh | 400 | 300 rpm |
| 5 | Dehydrated | 200 | Shaking |
| 6 | Freeze-dried | 26.5 | Shaking |
| ANOVA for TPI | ANOVA for FCI | ||||||
| Source | F-Ratio | p-Value | Source | F-Ratio | p-Value | ||
| A: temperature | 8.58 | 0.0117 | A: temperature | 22.53 | 0.0413 | ||
| B: time | 4.35 | 0.0572 | B: time | 20.47 | 0.0555 | ||
| AA | 3.39 | 0.0886 | AA | 0.11 | 0.7691 | ||
| AB | 0.27 | 0.6111 | AB | 0.02 | 0.9120 | ||
| BB | 0.43 | 0.5231 | BB | 0.32 | 0.6263 | ||
| Blocks | 0.56 | 0.4688 | Blocks | 0.07 | 0.8163 | ||
| Lack of fit | 2.58 | 0.1176 | Lack of fit | 1.64 | 0.4396 | ||
| R2 | 56.9172% | R2 | 62.8244% | ||||
| Adjusted R2 | 37.0329% | Adjusted R2 | 45.6664% | ||||
| PRESS | 10.6002 | PRESS | 23261.6 | ||||
| Predicted R2 | 0.0% | Predicted R2 | 10.2544% | ||||
| Standard error of est. | 0.590299 | Standard error of est. | 27.2252 | ||||
| Mean absolute error | 0.419217 | Mean absolute error | 18.3628 | ||||
| Durbin-Watson statistic | 1.86518 (p = 0.3105) | Durbin-Watson statistic | 2.11939 (p = 0.5191) | ||||
| Residual autocorrelation | −0.0148876 | Residual autocorrelation | −0.154277 | ||||
| Coefficients for TPI | Estimate | Coefficients for FCI | Estimate | ||||
| Constant | 12.419 | Constant | 233.15 | ||||
| A: temperature | 0.101465 | A: temperature | 0.44043 | ||||
| B: time | 0.0452361 | B: time | 12.3087 | ||||
| AA | 0.00159721 | AA | 0.0108245 | ||||
| AB | −0.00207143 | AB | −0.0184524 | ||||
| BB | 0.010459 | BB | −0.337534 | ||||
| No. | Extraction Time (s) | TPI (Mean ± SD) | FCI (Mean ± SD) |
|---|---|---|---|
| 1 | 10 | 11.57 ± 0.09 a | 367.77 ± 21.70 a |
| 2 | 12.5 | 13.03 ± 0.04 b | 403.20 ± 12.12 ab |
| 3 | 15 | 13.45 ± 0.07 c | 397.89 ± 8.35 a |
| 4 | 20 | 14.22 ± 0.09 d | 439.58 ± 13.04 b |
| Criterion | Ranking |
|---|---|
| Pineapple aroma | Initial < U < M (20 min) < M (10 min) < T |
| Overall quality | U < Initial < M (20 min) < T < M (10 min) |
| Volatile Compound | Initial | Microwaves | Ultrasound | Traditional | F | p-Value |
|---|---|---|---|---|---|---|
| Ethyl acetate | 9.166 b | 5.445 ab | 3.958 a | 6.373 ab | 6.68 | 0.0488 * |
| Isobutyl acetate | 1.706 b | 1.166 ab | 0.709 a | 1.344 ab | 8.84 | 0.0307 * |
| Ethyl 2-methylbutyrate | ND a | 0.337 b | 0.022 a | 0.052 a | 52.69 | 0.0011 * |
| Ethyl isovalerate | 0.509 b | 0.335 ab | 0.185 a | 0.395 ab | 10.29 | 0.0236 * |
| 2-Methyl-1-propanol | ND a | 0.011 ab | 0.034 c | 0.021 bc | 17.17 | 0.0094 * |
| Isoamyl acetate | 13.489 b | 10.227 ab | 5.554 a | 10.935 ab | 9.83 | 0.0256 * |
| 2,6-Dimethyl-4-heptanone | ND | 0.032 | 0.027 | 0.069 | 2.40 | 0.2076 |
| Methyl hexanoate | ND a | 0.153 a | 0.389 a | 1.458 b | 38.58 | 0.0020 * |
| Ethyl hexanoate | 0.023 a | 1.440 b | 0.098 a | 0.394 a | 30.32 | 0.0032 * |
| Acetoin | 0.032 b | 0.013 a | 0.021 ab | 0.021 ab | 9.74 | 0.0260 * |
| 3-Hexen-1-ol acetate | 0.005 | 0.003 | 0.002 | 0.004 | 3.44 | 0.1316 |
| Ethyl heptanoate | ND a | 0.019 c | 0.001 ab | 0.009 b | 30.37 | 0.0032 * |
| 2-Butyl acetate | ND a | 0.016 b | 0.014 b | 0.018 b | 92.52 | 0.0003 * |
| Methyl octanoate | ND a | 0.065 ab | 0.060 ab | 0.365 b | 9.30 | 0.0281 * |
| Furfural | 0.009 b | 0.003 a | 0.003 a | 0.003 a | 53.29 | 0.0011 * |
| Linalool oxide | ND a | 0.002 b | 0.002 b | 0.002 b | 40.60 | 0.0018 * |
| 2,3-Butanediol diacetate | 0.026 | 0.022 | 0.018 | 0.022 | 6.24 | 0.0545 |
| Benzaldehyde | 0.069 b | 0.043 a | 0.048 a | 0.053 a | 22.16 | 0.0059 * |
| Isobutyric acid | 0.096 | 0.035 | 0.046 | 0.053 | 5.00 | 0.0768 |
| Pentanoic acid | ND a | 0.004 ab | 0.006 b | 0.005 ab | 8.44 | 0.0332 * |
| 3-Methylbutanoic acid | 2.024 b | 0.854 a | 0.836 a | 0.897 a | 6.97 | 0.0456 * |
| Benzyl acetate | 0.023 b | 0.015 a | 0.014 a | 0.024 b | 28.72 | 0.0036 * |
| Methylbenzeneacetic acid | 0.003 | 0.003 | 0.003 | 0.005 | 6.11 | 0.0563 |
| Methyl salicilate | 0.018 c | 0.011 b | 0.008 a | 0.011 b | 73.33 | 0.0005 * |
| Ethyl phenyl acetate | 0.322 c | 0.237 ab | 0.179 a | 0.239 b | 31.68 | 0.0030 * |
| 2-Phenetyl acetate | 9.559 c | 7.249 b | 5.373 a | 7.118 b | 47.70 | 0.0013 * |
| Hexanoic acid | 0.143 | 0.125 | 0.076 | 0.100 | 2.33 | 0.2154 |
| Benzenemethanol | 1.070 b | 0.707 a | 0.659 a | 0.785 ab | 12.33 | 0.0172 * |
| p-Ethylguaiacol | 0.093 b | 0.063 a | 0.058 a | 0.064 a | 88.13 | 0.0004 * |
| Octanoic acid | 0.871 b | 0.595 a | 0.416 a | 0.498 a | 20.71 | 0.0067 * |
| 4-Ethylphenol | 0.151 b | 0.089 a | 0.078 a | 0.090 a | 43.97 | 0.0016 * |
| Decanoic acid | 0.228 a | 0.456 b | 0.109 a | 0.104 a | 36.31 | 0.0023 * |
| Volatile Compound | PC1 | Volatile Compound | PC2 |
|---|---|---|---|
| Methyl salicilate | 0.063 | Ethyl heptanoate | 0.164 |
| Ethyl phenyl acetate | 0.062 | Ethyl hexanoate | 0.159 |
| Benzenemethanol | 0.060 | Ethyl 2-methylbutyrate | 0.148 |
| 2-Phenetyl acetate | 0.058 | Decanoic acid | 0.080 |
| 4-Ethylphenol | 0.057 | 3-Methyl-1-butanol | 0.032 |
| p-Ethylguaiacol | 0.051 | Linalool oxide | 0.028 |
| 3-Methylbutanoic acid | 0.051 | Acetoin | 0.026 |
| 2,3-Butanediol diacetate | 0.049 | Isobutyric acid | 0.025 |
| Octanoic acid | 0.049 | Isoamyl acetate | 0.021 |
| Ethyl isovalerate | 0.048 | ||
| Isobutyl acetate | 0.045 | ||
| 3-Hexen-1-ol acetate | 0.044 | ||
| Isoamyl acetate | 0.043 | ||
| Ethyl acetate | 0.043 | ||
| Furfural | 0.038 | ||
| Isobutyric acid | 0.037 | ||
| Hexanoic acid | 0.037 | ||
| Benzaldehyde | 0.031 | ||
| Benzyl acetate | 0.021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Párraga, S.; Durán-Guerrero, E.; Castro, R. Application of Microwaves as an Advanced Technique for the Development of Sherry Vinegar Macerated with Pineapple. Beverages 2021, 7, 18. https://doi.org/10.3390/beverages7020018
Párraga S, Durán-Guerrero E, Castro R. Application of Microwaves as an Advanced Technique for the Development of Sherry Vinegar Macerated with Pineapple. Beverages. 2021; 7(2):18. https://doi.org/10.3390/beverages7020018
Chicago/Turabian StylePárraga, Samuel, Enrique Durán-Guerrero, and Remedios Castro. 2021. "Application of Microwaves as an Advanced Technique for the Development of Sherry Vinegar Macerated with Pineapple" Beverages 7, no. 2: 18. https://doi.org/10.3390/beverages7020018
APA StylePárraga, S., Durán-Guerrero, E., & Castro, R. (2021). Application of Microwaves as an Advanced Technique for the Development of Sherry Vinegar Macerated with Pineapple. Beverages, 7(2), 18. https://doi.org/10.3390/beverages7020018







