Detection and Pulsed-Field Gel Electrophoresis Typing of Listeria monocytogenes Isolates from Milk Vending Machines in Croatia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Milk Samples
2.2. Microbiological Analyses
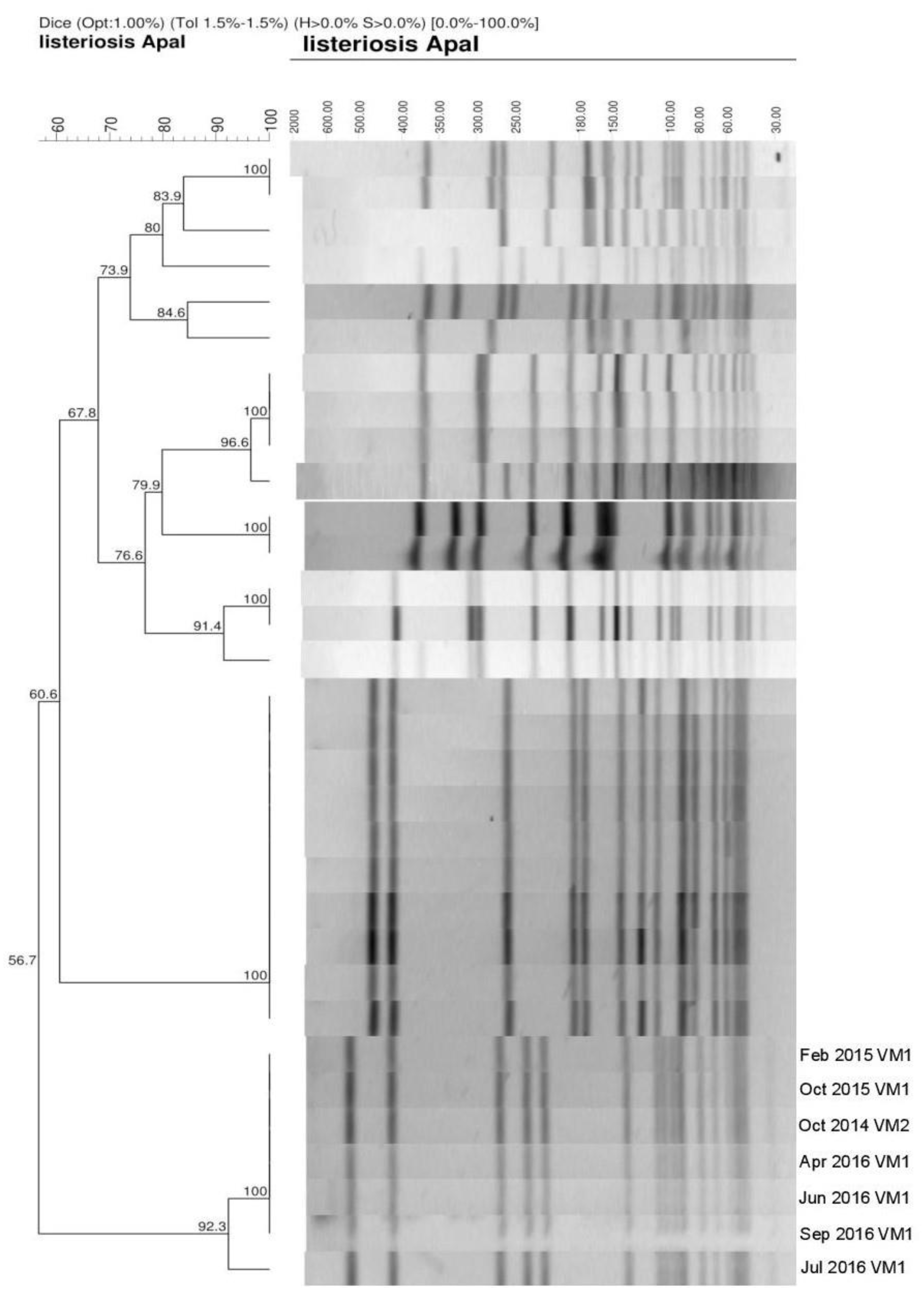
2.3. Pulsed-Field Gel Electrophoresis Typing of Listeria monocytogenes
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tremonte, P.; Tipaldi, L.; Succi, M.; Pannella, G.; Falasca, L.; Capilongo, V.; Coppola, R.; Sorrentino, E. Raw milk from vending machines: Effects of boiling, microwave treatment, and refrigeration on microbiological quality. J. Dairy Sci. 2014, 97, 3314–3320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Czyżak-Runowska, G.; Wójtowski, J.; Niewiadomska, A.; Markiewicz-Keszycka, M. Quality of fresh and stored mare’s milk. Mljekarstvo 2018, 68, 108–115. [Google Scholar] [CrossRef]
- Giacometti, F.; Serraino, A.; Finazzi, G.; Daminelli, P.; Losio, M.N.; Tamba, M.; Garigliani, A.; Mattioli, R.; Riu, R.; Zanoni, R.G. Field handling conditions of raw milk sold in vending machines: Experimental evaluation of the behaviour of Listeria monocytogenes, Escherichia coli O157:H7, Salmonella Typhimurium and Campylobacter jejuni. Ital. J. Anim. Sci. 2012, 11, 132–136. [Google Scholar] [CrossRef]
- Croatian Food Agency. Scientific Opinion on Public Health Risk Related to Raw Milk Consumption. Available online: https://www.hah.hr/wp-content/uploads/2016/12/znanstveno-misljenje-o-javno-zdravstvenom-riziku-vezanom-za-konzumaciju-sirovog-mlijeka.pdf (accessed on 4 November 2018).
- EFSA: Panel on Biological Hazards. Scientific Opinion on the public health risks related to the consumption of raw drinking milk. EFSA J. 2015, 13, 3940. [Google Scholar] [CrossRef]
- Latorre, A.A.; Pradhan, A.K.; Van Kessel, J.A.; Karns, J.S.; Boor, K.J.; Rice, D.H.; Mangione, K.J.; Gröhn, Y.T.; Schukken, Y.H. Quantitative risk assessment of listeriosis due to consumption of raw milk. J. Food Prot. 2011, 74, 1268–1281. [Google Scholar] [CrossRef] [PubMed]
- Hunt, K.; Drummond, N.; Murphy, M.; Butler, F.; Buckley, J.; Jordan, K. A case of bovine raw milk contamination with Listeria monocytogenes. Ir. Vet. J. 2012, 65, 13. [Google Scholar] [CrossRef] [PubMed]
- Owusu-Kwarteng, J.; Wuni, A.; Akabanda, F.; Jespersen, L. Prevalence and Characteristics of Listeria monocytogenes Isolates in Raw Milk, Heated Milk and Nunu, a Spontaneously Fermented Milk Beverage, in Ghana. Beverages 2018, 4, 40. [Google Scholar] [CrossRef]
- Bianchi, D.M.; Barbaro, A.; Gallina, S.; Vitale, N.; Chiavacci, L.; Caramelli, M.; Decastelli, L. Monitoring of foodborne pathogenic bacteria in vending machine raw milk in Piedmont. Food Control 2013, 32, 435–439. [Google Scholar] [CrossRef]
- ISO. ISO 11290-1:2017 Microbiology of the Food Chain—Horizontal Method for the Detection and Enumeration of Listeria Monocytogenes and of Listeria spp.—Part 1: Detection Method; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- ISO. ISO 11290-2:2017 Microbiology of the Food Chain—Horizontal Method for the Detection and Enumeration of Listeria Monocytogenes and of Listeria spp.—Part 2: Enumeration Method; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- Pažin, V.; Jankuloski, D.; Kozačinski, L.; Dobranić, V.; Njari, B.; Cvrtila, Ž.; Lorenzo, J.M.; Zdolec, N. Tracing of Listeria monocytogenes Contamination Routes in Fermented Sausage Production Chain by Pulsed-Field Gel Electrophoresis Typing. Foods 2018, 7, 198. [Google Scholar] [CrossRef] [PubMed]
- PulseNet, PFGE Protocols. Available online: https://www.cdc.gov/pulsenet/pdf/listeria-pfge-protocol508c.pdf (accessed on 13 February 2019).
- Dalzini, E.; Bernini, V.; Bertasi, B.; Daminelli, P.; Losio, M.N.; Varisco, G. Survey of prevalence and seasonal variability of Listeria monocytogenes in raw cow milk from Northern Italy. Food Control 2016, 60, 466–470. [Google Scholar] [CrossRef]
- Godic Torkar, K.; Kirbiš, A.; Vadnjal, S.; Biasizzo, M.; Galicic, A.; Jevšnik, M. The microbiological quality of Slovenian raw milk from vending machines and their hygienic-technical conditions. Br. Food J. 2017, 119, 377–389. [Google Scholar] [CrossRef]
- Gelbícová, T.; Karpísková, R. Occurrence and typing of Listeria monocytogenes isolated from raw cow’s milk collected on farms and from vending machines. Klin. Mikrobiol. A Infekcni Lek. 2012, 18, 38–42. [Google Scholar]
- Tahoun, A.B.M.B.; Abou Elez, R.M.M.; Abdelfatah, E.N.; Elsohaby, I.; El-Gedawy, A.A.; Elmoslemany, A.M. Listeria monocytogenes in raw milk, milking equipment and dairy workers: Molecular characterization and antimicrobial resistance patterns. J. Glob. Antimicrob. Resist. 2017, 10, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Castro, H.; Ruusunen, M.; Lindström, M. Occurrence and growth of Listeria monocytogenes in packaged raw milk. Int. J. Food Microbiol. 2017, 261, 1–10. [Google Scholar] [CrossRef] [PubMed]
| Sample ID | Molecular Detection Assay | HRN ISO 11290:1 | HRN ISO 11290:2 (cfu/mL) |
|---|---|---|---|
| 1 | + | + | 60 |
| 2 | + | + | 50 |
| 3 | + | − | <10 |
| 4 | + | − | <10 |
| 5 | + | + | 90 |
| 6 | + | − | <10 |
| 7 | + | − | <10 |
| 8 | + | − | <10 |
| 9 | + | + | 60 |
| 10 | + | − | <10 |
| 11 | + | + | 70 |
| 12 | + | + | 100 |
| 13 | + | + | 60 |
| 14 | + | + | 50 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zdolec, N.; Jankuloski, D.; Kiš, M.; Hengl, B.; Mikulec, N. Detection and Pulsed-Field Gel Electrophoresis Typing of Listeria monocytogenes Isolates from Milk Vending Machines in Croatia. Beverages 2019, 5, 46. https://doi.org/10.3390/beverages5030046
Zdolec N, Jankuloski D, Kiš M, Hengl B, Mikulec N. Detection and Pulsed-Field Gel Electrophoresis Typing of Listeria monocytogenes Isolates from Milk Vending Machines in Croatia. Beverages. 2019; 5(3):46. https://doi.org/10.3390/beverages5030046
Chicago/Turabian StyleZdolec, Nevijo, Dean Jankuloski, Marta Kiš, Brigita Hengl, and Nataša Mikulec. 2019. "Detection and Pulsed-Field Gel Electrophoresis Typing of Listeria monocytogenes Isolates from Milk Vending Machines in Croatia" Beverages 5, no. 3: 46. https://doi.org/10.3390/beverages5030046
APA StyleZdolec, N., Jankuloski, D., Kiš, M., Hengl, B., & Mikulec, N. (2019). Detection and Pulsed-Field Gel Electrophoresis Typing of Listeria monocytogenes Isolates from Milk Vending Machines in Croatia. Beverages, 5(3), 46. https://doi.org/10.3390/beverages5030046







