Coconut Neera—A Vital Health Beverage from Coconut Palms: Harvesting, Processing and Quality Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Tapping and Harvesting
2.2. Preparation and Usage of the Anti-Fermentation Solution (AFS)
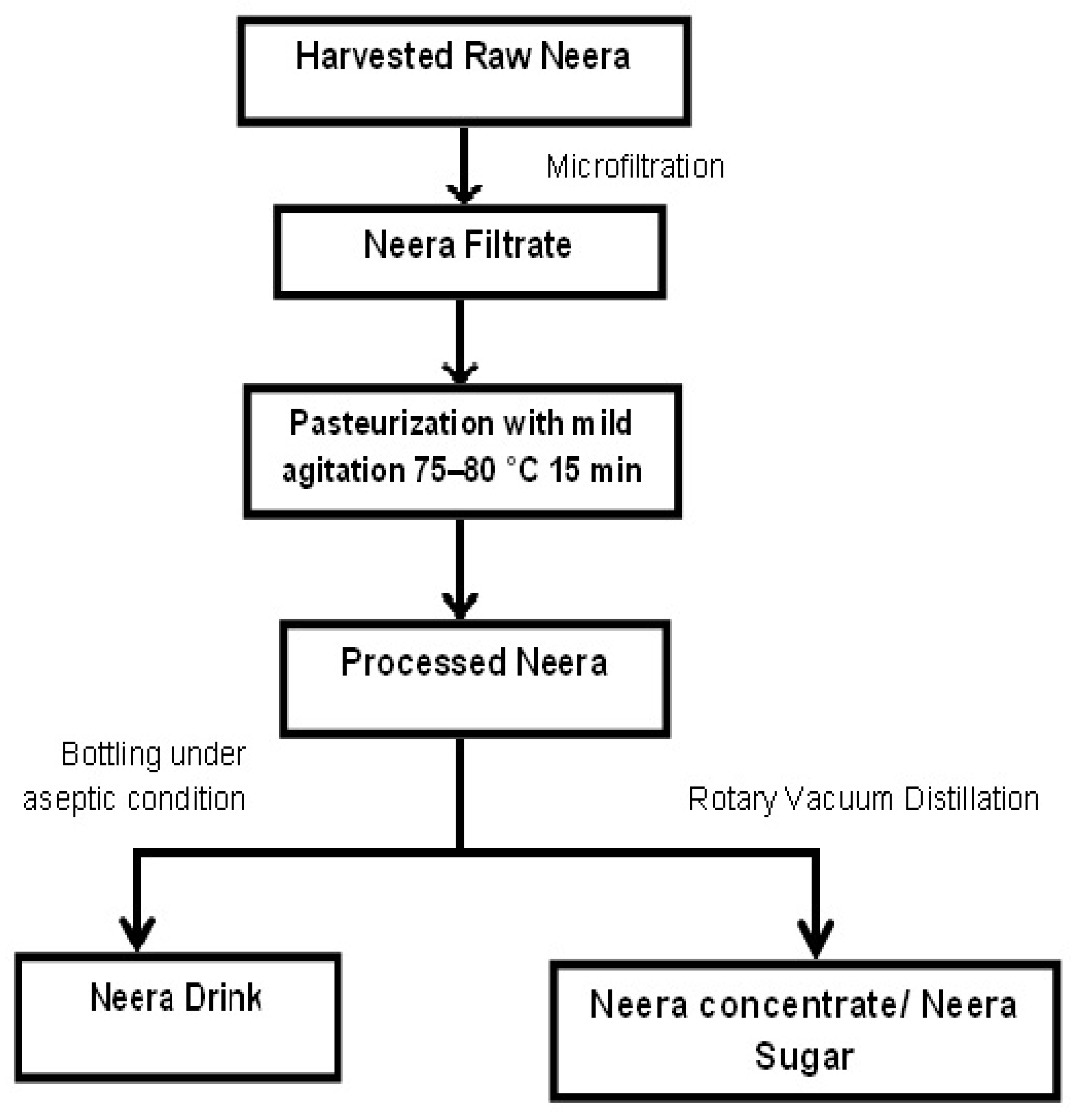
2.3. Processing and Storage of Harvested Neera
- (i)
- Microfiltration: The raw Neera was filtered through micro filters with a pore size of 100 microns under aseptic conditions.
- (ii)
- Pasteurization: The filtered raw Neera was stored in a sterilized vessel for pasteurization for 10–15 min at a temperature range of 75–85 °C. A mild agitation was inevitable during pasteurization. The temperature was kept constant at 80 °C during pasteurization by remote control.
- (iii)
- Bottling: The pasteurized Neera was bottled by an automatic bottling machine with a volume (200 mL) in polypropylene (PP) bottles and 300 mL in glass bottles and capped and sealed automatically. The entire steps were done in a closed system for avoiding further contamination during processing.
- (iv)
- Storage: Shelf life was checked periodically by analyzing the pH, brix value and nutritional components.
2.4. Quality Analysis of Neera
2.5. Estimation of Alcohol Content
2.6. Estimation of Vital Nutrients
2.7. Oral Toxicity Test and Glycemic Index of Processed Neera
2.8. Production of Value-Added Products
- (i)
- Neera Concentrate: Neera concentrate—commercially called Neera squash—was developed by concentrating the Neera drink from a brix value 14 to 45–50 using rotary vacuum distillation under controlled temperatures.
- (ii)
- Neera Sugar: The process of making Neera sugar was similar to that of the Neera concentrate. At the level of getting the Neera concentrate of a brix value 45–50, it was again subjected to further vacuum distillation process until the brix value came to the level of 55–60 with a texture of a semisolid form. This semisolid Neera thick concentrate was transferred to a wide metal pan and kept in an incubator at 70 °C for overnight. After proper dehydration, the solidified Neera as white a Neera sugar was powdered in a blender and bottled.
2.9. Statistical Analysis
3. Results
3.1. Tapping and Harvesting
3.2. Anti-Fermentation Solution: Mode of Action of AFS
3.3. Processing and Bottling of Neera
3.4. Microbial Load of Raw and Processed Neera
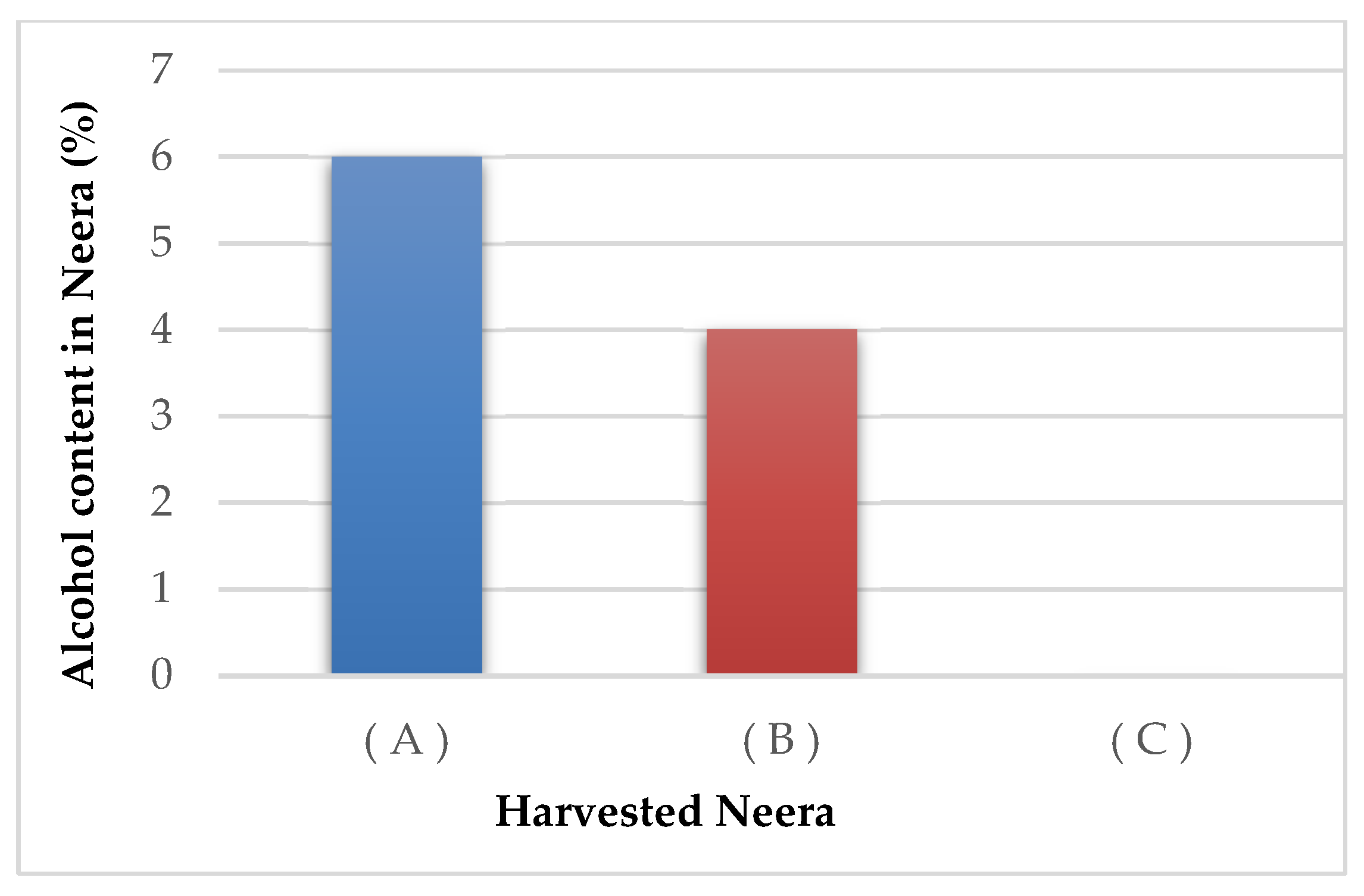
3.5. Alcohol Content
3.6. Nutritional Composition of Processed Neera
3.7. Acute Oral Toxicity Data of the Neera Drink
3.8. Glycemic Index of Neera
3.9. Value-Added Products
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chinnamma, M.; Bhasker, S.; Madhav, H.; Devasia, R.M.; Shashidharan, A.; Pillai, B.C.; Thevannoor, P. Production of Coconut methyl ester (CME) and glycerol from Coconut (Cocos nucifera) oil and the functional feasibility of CME as Biofuel in diesel engine. Fuel 2015, 140, 4–9. [Google Scholar] [CrossRef]
- Ghosh, D.K.; Bandyopadhyay, A.; Das, S.; Hebbar, K.B.; Biswas, B. Coconut sap (neera)-untapped opportunity of spinoff gains in West Bengal, India. Int. J. Curr. Microbiol. App. Sci. 2018, 7, 1883–1897. [Google Scholar]
- Siddharameswara, G.M. Coconut neera production and processing in Karnataka. Indian Coconut J. 2013, 31–33. [Google Scholar]
- Atputharajah, J.D.; Widanapathirana, S.; Samarajeewa, U. Microbiology and biochemistry of natural fermentation of coconut palm sap. Food Microbiol. 1986, 3, 273–280. [Google Scholar] [CrossRef]
- Abdulmumeen, H.A.; Risikat, A.N.; Sururah, A.R. Food: Its preservatives, additives and applications. Int. J. Chem. Biochem. Sci. 2012, 1, 36–47. [Google Scholar]
- Indian Standard. Method for Yeast and Mould Count of Foodstuffs and Animal Feeds; Bureau of Indian Standard: Manak Bhawan, India, 1999; Volume 1, pp. 1–3.
- Indian Standard. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Micro-Organisms-Colony-Count Technique at 30 °C; Bureau of Indian Standard: Manak Bhawan, India, 2012; Volume 2, pp. 1–8.
- Sumbhate, S.; Nayak, S.; Goupale, D.; Tiwari, A.; Jadon, R.S. Colorimetric method for the estimation of ethanol in alcoholic-drinks. J. Anal. Tech. 2012, 1, 1–6. [Google Scholar]
- Indian Standard. Method for Determination of Protein in Foods and Feeds; Bureau of Indian Standard: Manak Bhawan, India, 1973; pp. 3–10.
- Indian Standard. Specification for Edible Groundnut Flour (Expeller Pressed); Bureau of Indian Standard: Manak Bhawan, India, 1975; Volume 1, pp. 13–14.
- Indian Standard. Milk-Cereal Based Complementary Foods—Specification; Bureau of Indian Standard: Manak Bhawan, India, 2007; Volume 4, pp. 1–6.
- Miller, D.S.; Payne, P.R. A ballistic bomb calorimeter. Br. J. Nutr. 1959, 13, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Charrondiere, U.R.; Chevassus-Agnes, S.; Marroni, S.; Burlingame, B. Impact of different macronutrient definitions and energy conversion factors on energy supply estimations. J. Food Comp. Anal. 2004, 17, 339–360. [Google Scholar] [CrossRef]
- Fat (Total, Saturated and Unsaturated) in Foods, hydrolytic extraction gas chromatographic method. In AOAC Official Method 996.06, 18th ed.; AOAC International: Arlington, TX, USA, 2001.
- Kaneda, T.; Nakajima, A.; Fujimoto, K.; Kobayashi, T.; Kiriyama, S.; Ebihara, K.; Innami, T.; Tsuji, K.; Tsuji, E.; Kinumaki, T.; et al. Quantitative analysis of cholesterol in foods by gas-liquid chromatography. J. Nutr. Sci. Vitaminol. 1980, 26, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, T.G.; Goldberg, J.V. The anthrone method for the determination of carbohydrates in foods and in oral rinsing. J. Dent. Res. 1956, 35, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Morris, D.L. Quantitative determination of carbohydrates with dreywood’s anthrone reagent. Science 1948, 107, 254–255. [Google Scholar] [CrossRef] [PubMed]
- Dreywood, R. Qualitative test for carbohydrate material. Ind. Eng. Chem. Anal. Ed. 1946, 18, 499. [Google Scholar] [CrossRef]
- Ellefson, W. HPLC of mono- and disaccharides using refractive index detection. Curr. Prot. Food Anal. Chem. 2002, 6, E1.2.1–E1.2.9. [Google Scholar] [CrossRef]
- Food Safety and Standards Authority of India. Manual of Methods of Analysis of Foods Metals; Fssai, Ministry of Health and Family Welfare: New Delhi, India, 2016; pp. 3–82.
- White, J.A.; Hart, R.T. Derivatization Methods for Liquid Chromatographic Separation of Amino Acid in Food Analysis; Marcel Dekker, Inc.: New York, NY, USA, 1992; pp. 53–74. [Google Scholar]
- Nollet, L.M.L. Handbook of Food Analysis: Physical Characterization and Nutrient Analysis; Marcel Dekker, Inc.: New York, NY, USA, 2004; Volume 1, pp. 83–110. [Google Scholar]
- Eitenmiller, R.R.; Landen, W.O., Jr. Vitamin Analysis for the Health and Food Sciences; CRC Press: Boca Raton, FL, USA, 1999; pp. 3–67, 271–494. [Google Scholar]
- OECD Guideline for the Testing of Chemicals: Acute Oral Toxicity—Up-and-down Procedure. No. 420, Section 4: Health Effects. Available online: https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/oecd/oecd_gl420.pdf (accessed on 17 December 2001).
- Rigo, J.; Ziegler, E.E. (Eds.) Protein and energy requirements in infancy and childhood. In Nestlé Nutr Workshop Ser Pediatr Program; Nestlé Nutrition Institute: Vevey, Switzerland, 2006; Volume 58, pp. 109–119. [Google Scholar]
- Imura, K.; Okada, A. Amino Acid Metabolism in Pediatric Patients—Review. Nutrient 1998, 14, 143–148. [Google Scholar] [CrossRef]





| Organism | Raw Neera without AFS | Raw Neera with AFS |
|---|---|---|
| Total Bacterial Count/mL (CFU/mL) | 102,000 | Less than 10 CFU |
| Total Fungal Count/mL (CFU/mL) | Less than 10 CFU | Less than 10 CFU |
| Components | Quantity/100 mL | S.D. Mean | |
|---|---|---|---|
| Proximate composition | Food Energy | 74.86 Kcal | 0.51 |
| Protein | 0.28 g | 0.004 | |
| Carbohydrates | 18.03 g | 0.03 | |
| Fats | 0.18 g | 0.006 | |
| Lipids | Unsaturated fatty acids (Oleic & Linoleic acids) | 80 mg | 0.10 |
| Cholesterol | NIL | 100 mg | |
| Saturated fatty acids (Lauric and Myristic acids) | NIL | 0.47 |
| Components | Quantity/100 mL | S.D. Mean | |
|---|---|---|---|
| Sugars | Glucose | 1.5 g | 0.02 |
| Fructose | 0.6 g | 0.02 | |
| Sucrose | 13.4 g | 0.41 | |
| Minerals | Sodium | 15.2 mg | 0.05 |
| Potassium | 100 mg | 0.47 | |
| Iron | 0.36 mg | 0.003 | |
| Zinc | 40 µg | 0.11 | |
| Magnesium | 6 mg | 0.01 | |
| Calcium | 1.8 mg | 0.04 | |
| Manganese | 10 µg | 0.04 | |
| Selenium | 10 µg | 0.07 | |
| Copper | 27 µg | 0.06 |
| Components | Quantity/100 mL | S.D. Mean | |
|---|---|---|---|
| Amino acids | Aspartic acid | 4 mg | 0.08 |
| Valine | 2.19 mg | 0.09 | |
| Threonine | 3.95 mg | 0.07 | |
| Alanine | 2.78 mg | 0.12 | |
| Methionine | 12 mg | 0.66 | |
| Leucine | 0.47 mg | 0.008 | |
| Isoleucine | 2.19 mg | 0.02 | |
| Histidine | 99.6 mg | 0.42 | |
| Cysteine | 14 mg | 0.13 | |
| Arginine | 7 mg | 0.14 | |
| Tyrosine | 7.11 mg | 0.18 | |
| Vitamins | Vitamin B2 | 15.4 µg | 0.51 |
| Vitamin B3 | 210 µg | 0.78 | |
| Vitamin B6 | 30 µg | 0.56 | |
| Vitamin C | 65 mg | 0.77 | |
| Vitamin A (As retinol) (One IU Vitamin A is equivalent to 0.3 µg of retinol) | 4.88 IU | 0.15 |
| Groups | Serum Glucose (mmol/L) | |||||||
|---|---|---|---|---|---|---|---|---|
| O min | 15 min | 30 min | 45 min | 60 min | 90 min | 120 min | ||
| Glucose (1 g/Kg rat b.w.) | Mean | 4.00 | 9.91 | 9.43 | 9.19 | 8.72 | 7.50 | 6.28 |
| S.D. | 0.20 | 0.41 | 1.58 | 0.58 | 0.41 | 0.39 | 0.52 | |
| S.E.M. | 0.11 | 0.24 | 0.91 | 0.33 | 0.23 | 0.23 | 0.30 | |
| Neera (1 mL/Kg rat b.w.) | Mean | 4.03 | 4.41 | 4.37 | 4.37 | 4.25 | 4.08 | 4.13 |
| S.D. | 0.29 | 0.36 | 0.30 | 0.26 | 0.27 | 0.35 | 0.24 | |
| S.E.M. | 0.17 | 0.21 | 0.17 | 0.15 | 0.15 | 0.20 | 0.14 | |
| Groups | Increment Area under Curve | |
|---|---|---|
| Glucose (1 g/Kg rat b.w) | Mean | 973.25 |
| S.D. | 46.00 | |
| S.E.M. | 26.57 | |
| Neera (1 mL/kg rat b.w) | Mean | 507.76 |
| S.D. | 35.08 | |
| S.E.M. | 20.26 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chinnamma, M.; Bhasker, S.; Binitha Hari, M.; Sreekumar, D.; Madhav, H. Coconut Neera—A Vital Health Beverage from Coconut Palms: Harvesting, Processing and Quality Analysis. Beverages 2019, 5, 22. https://doi.org/10.3390/beverages5010022
Chinnamma M, Bhasker S, Binitha Hari M, Sreekumar D, Madhav H. Coconut Neera—A Vital Health Beverage from Coconut Palms: Harvesting, Processing and Quality Analysis. Beverages. 2019; 5(1):22. https://doi.org/10.3390/beverages5010022
Chicago/Turabian StyleChinnamma, Mohankumar, Salini Bhasker, Malavika Binitha Hari, Divyaa Sreekumar, and Harish Madhav. 2019. "Coconut Neera—A Vital Health Beverage from Coconut Palms: Harvesting, Processing and Quality Analysis" Beverages 5, no. 1: 22. https://doi.org/10.3390/beverages5010022
APA StyleChinnamma, M., Bhasker, S., Binitha Hari, M., Sreekumar, D., & Madhav, H. (2019). Coconut Neera—A Vital Health Beverage from Coconut Palms: Harvesting, Processing and Quality Analysis. Beverages, 5(1), 22. https://doi.org/10.3390/beverages5010022




