Simultaneous Voltammetric Determination of Gallic and Protocatechuic Acids in Mango Juice Using a Reduced Graphene Oxide-Based Electrochemical Sensor
Abstract
:1. Introduction
2. Experimental
2.1. Chemicals
2.2. Instruments
2.3. Preparation of the rGO/GCE
2.4. Sample Preparation
3. Results and Discussion
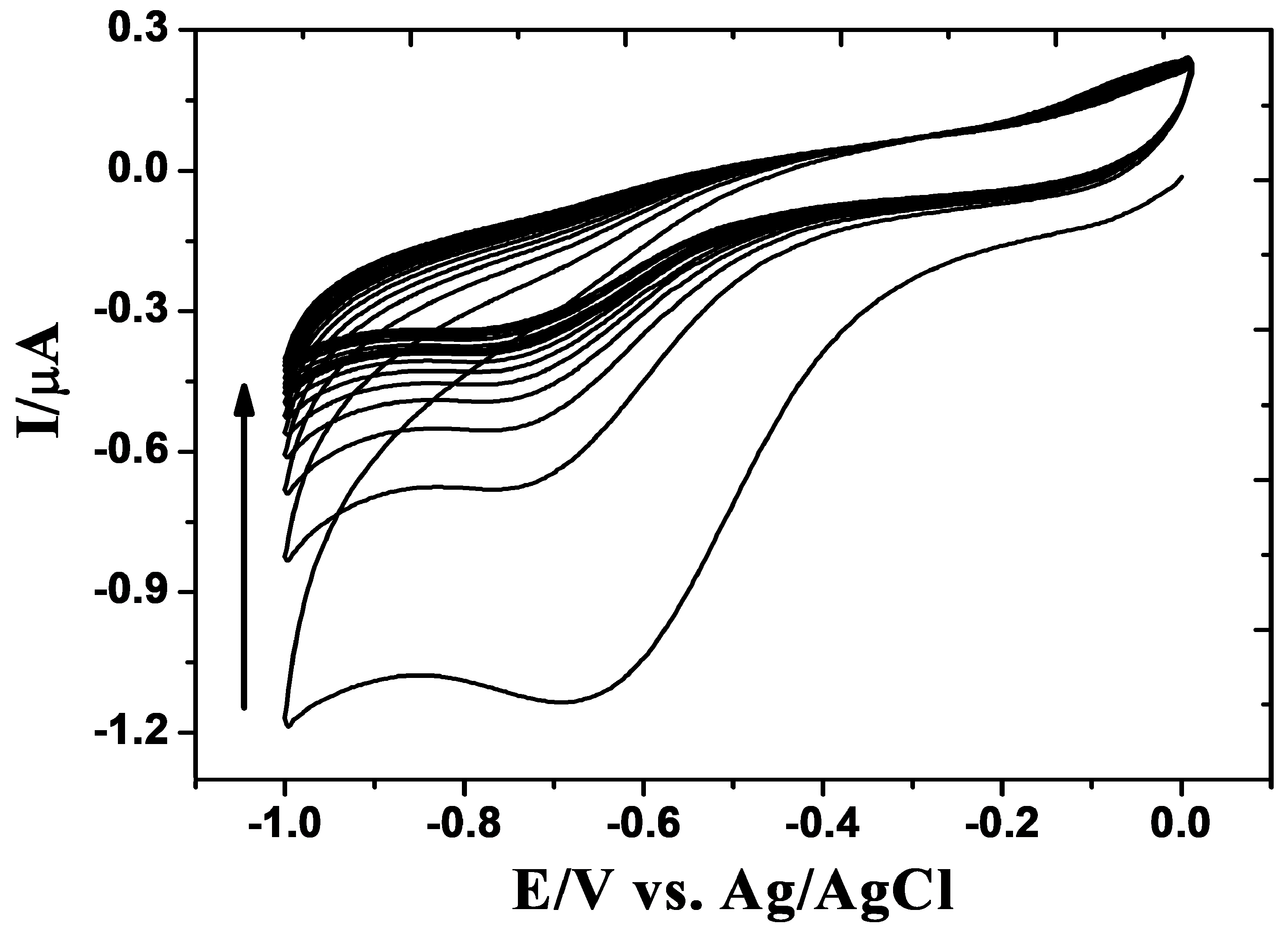
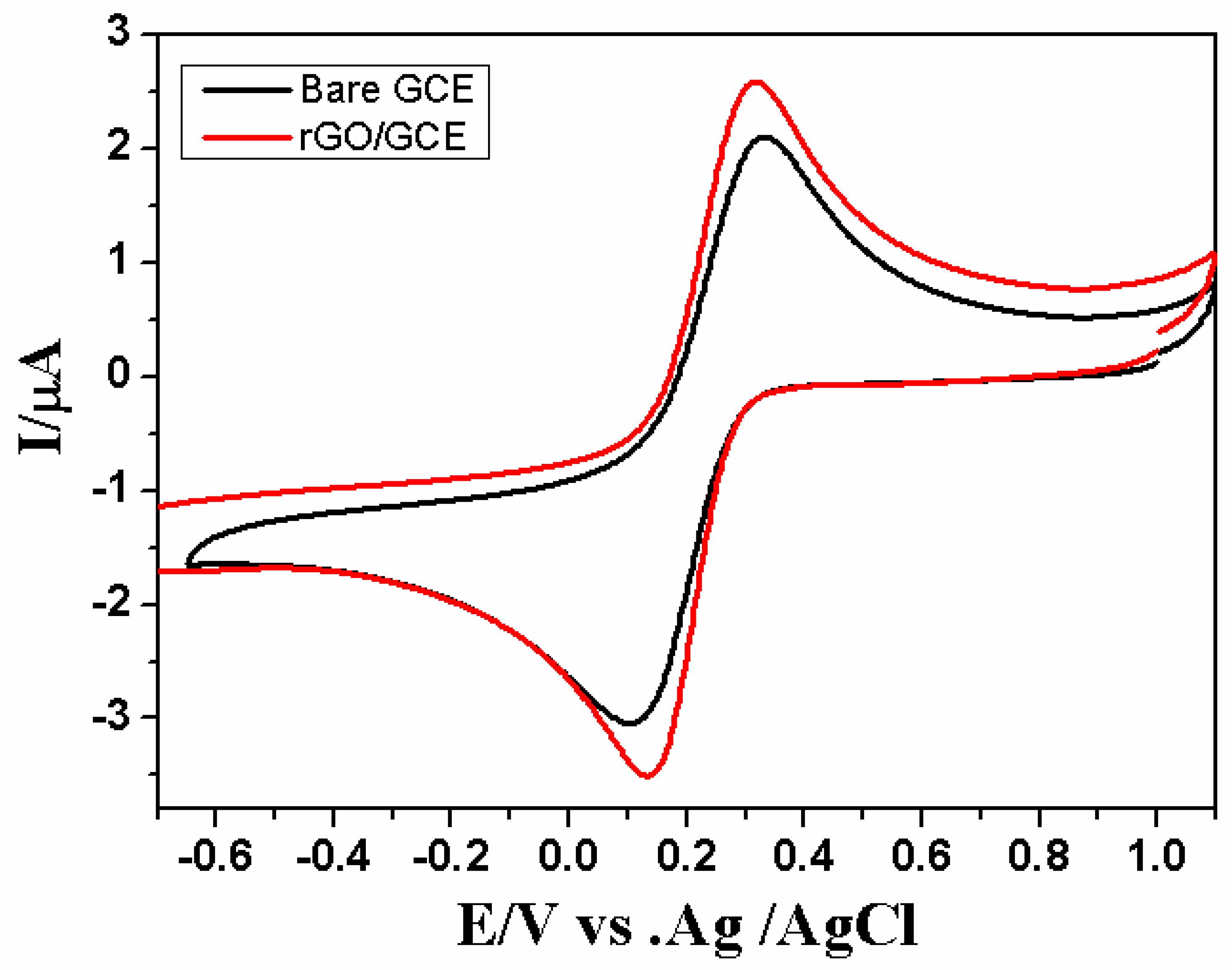
3.1. Electrochemical Synthesis and Characterization of the rGO/GCE
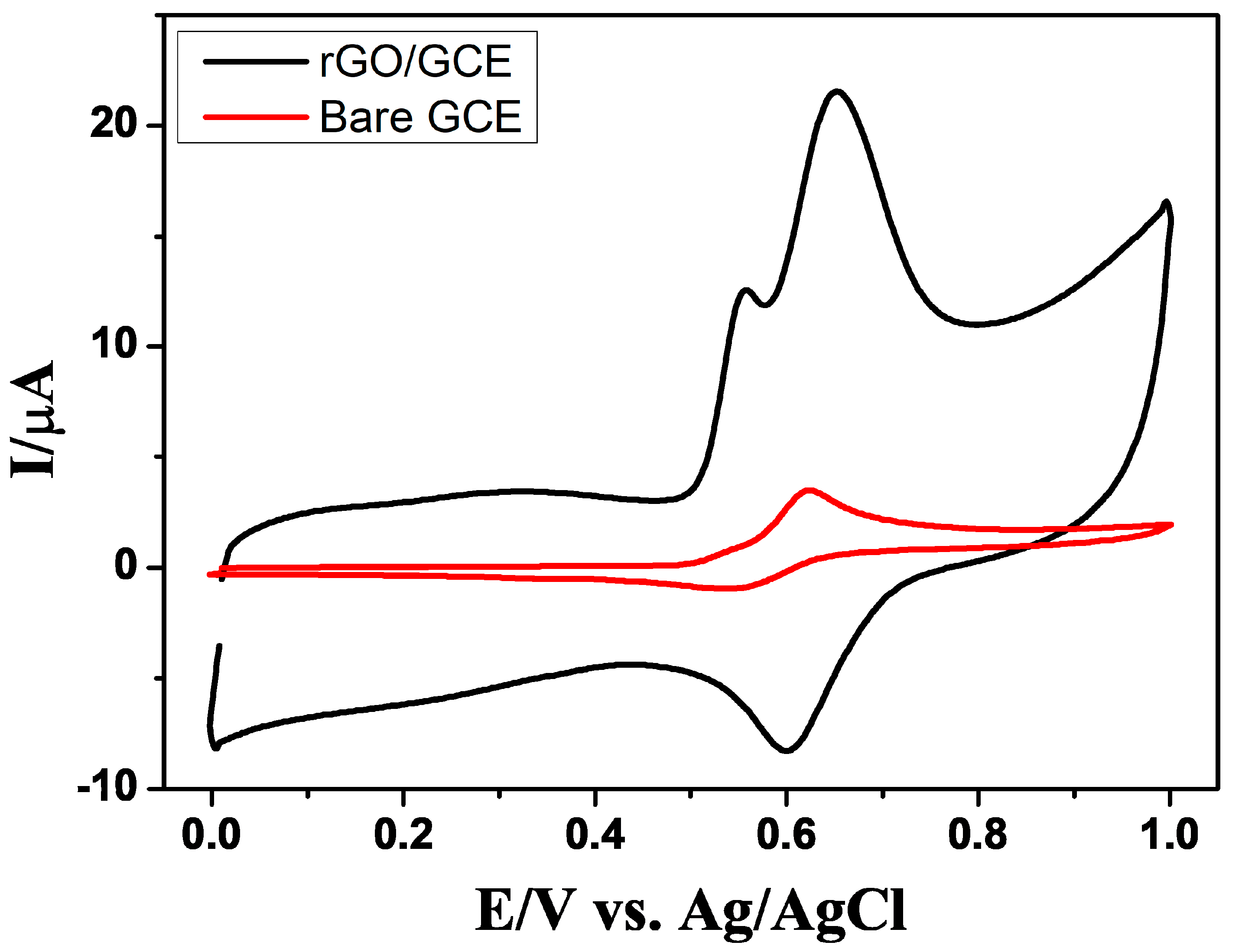
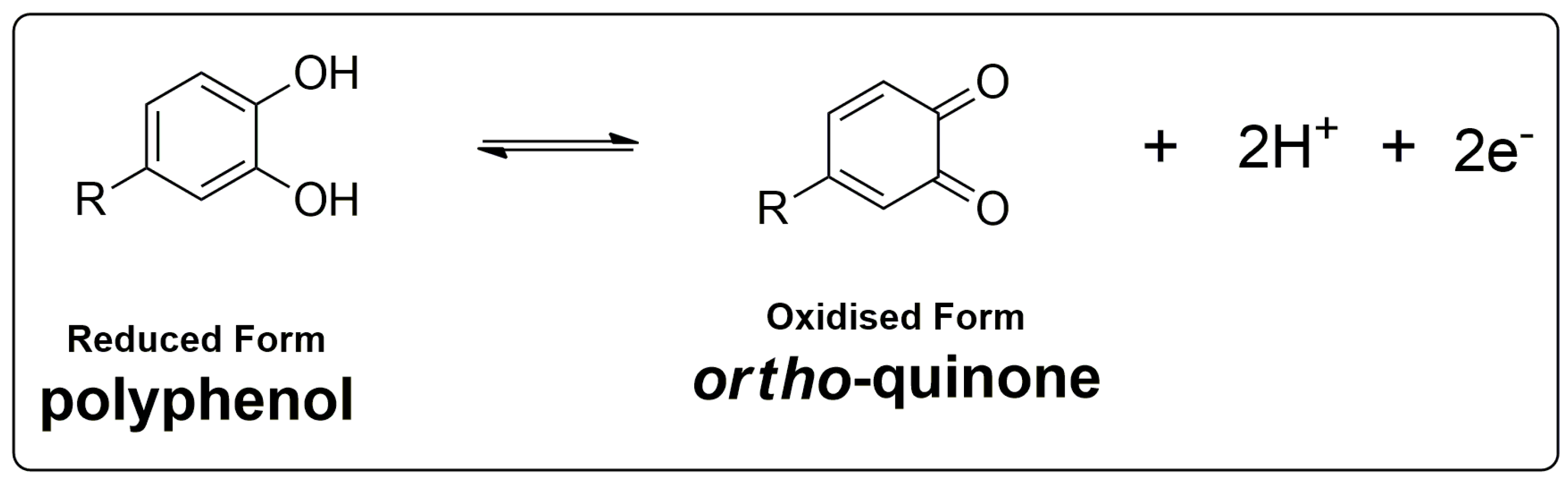
3.2. Electrochemical Oxidation of GA and PA
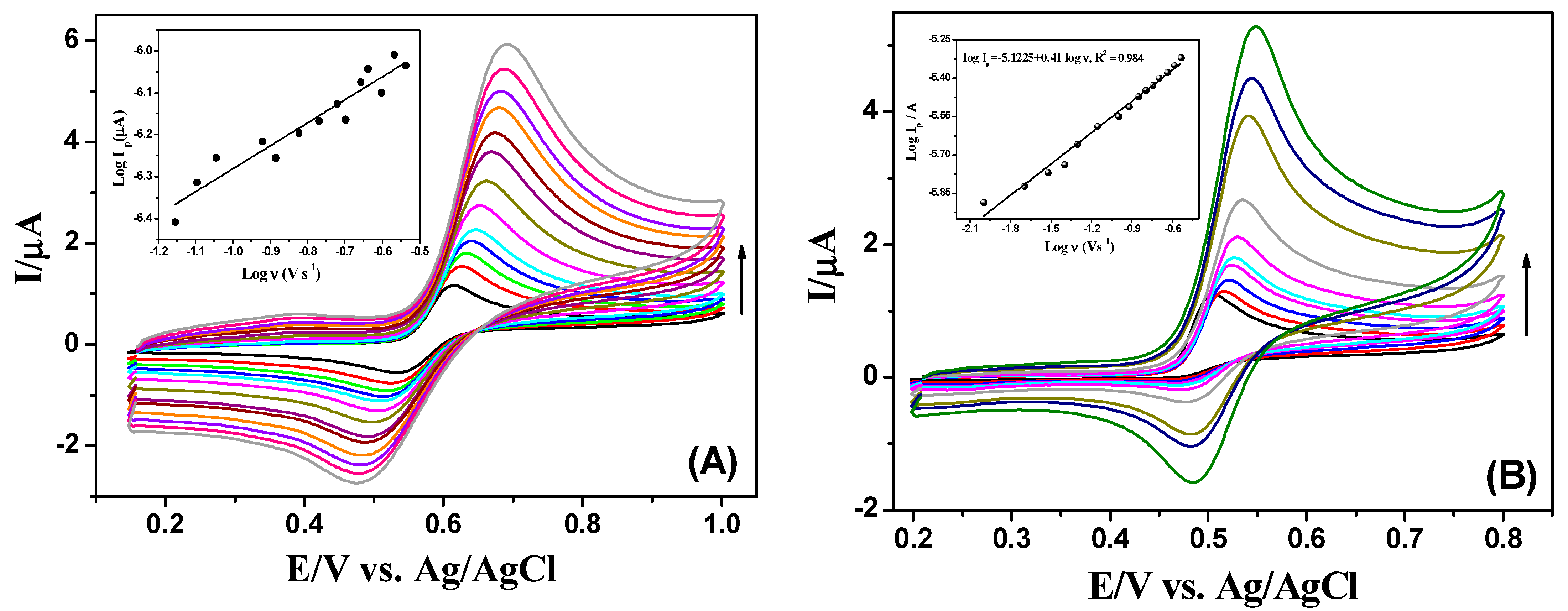
3.3. Effects of pH and Scan Rate on the Electrochemistry of GA and PA
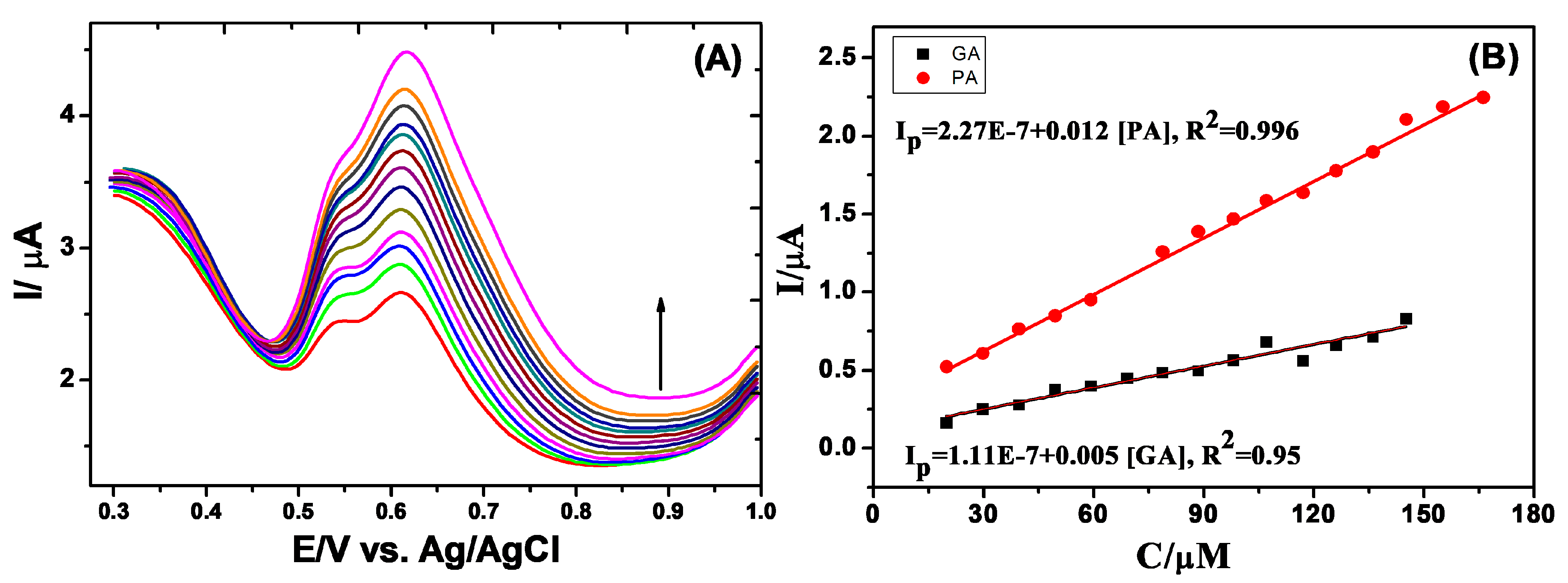
3.4. Simultaneous Determination of GA and PA
3.5. Interference, Repeatability and Stability Studies
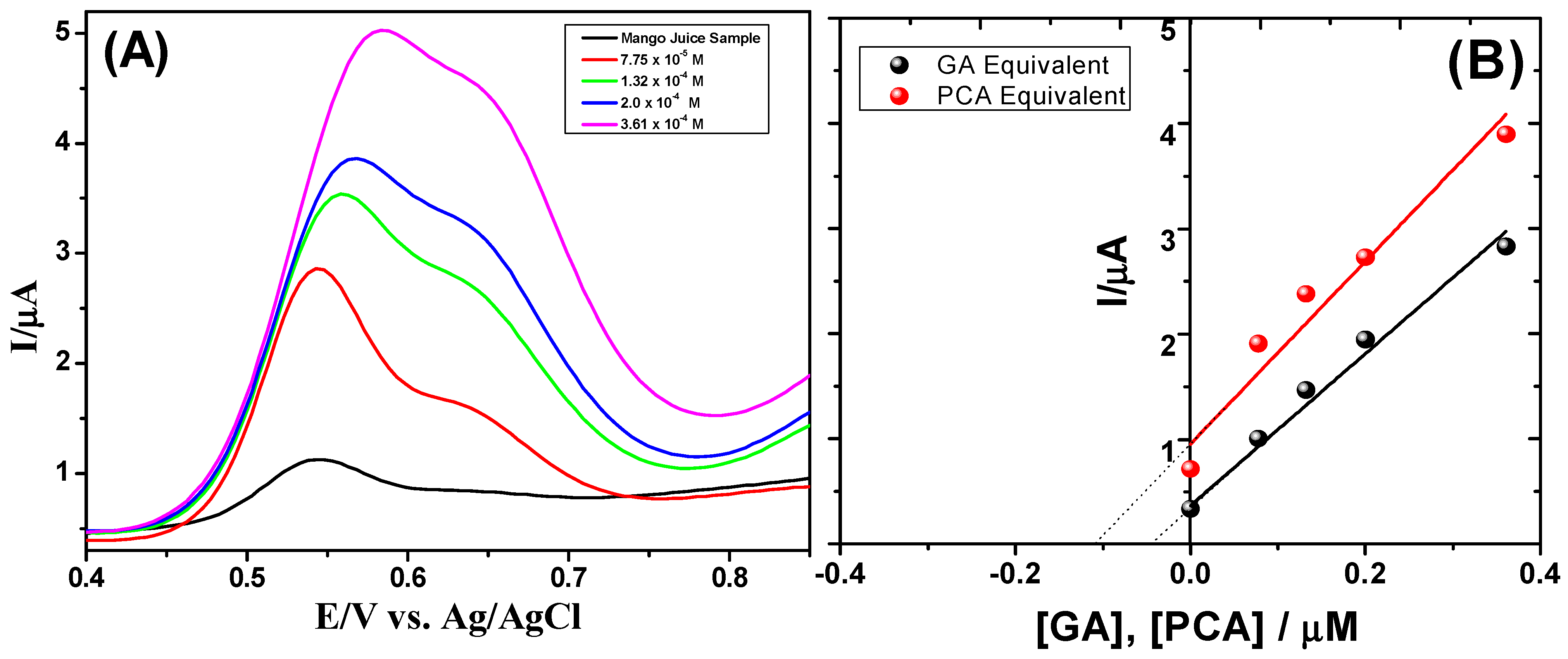
3.6. Analysis of Real Samples
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ghaani, M.; Nasirizadeh, N.; Ardakani, S.A.Y.; Mehrjardi, F.Z.; Scampicchiod, M.; Farris, S. Development of an electrochemical nanosensor for the determination of gallic acid in food. J. Anal. Methods 2016, 8, 1103–1110. [Google Scholar] [CrossRef]
- Kumar, S.; Singha, A.; Kumar, B.; Pharmaceut, J. Identification and characterization of phenolics and terpenoids from ethanolic extracts of Phyllanthus species by HPLC-ESI-QTOF-MS/MS. J. Pharm. Anal. 2017, 7, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Mudnic, I.; Modun, D.; Rastija, V.; Vukovic, J.; Brizic, I.; Katalinic, V.; Kozina, B.; Medic-Saric, M.; Boban, M. Antioxidative and vasodilatory effects of phenolic acids in wine. Food Chem. 2010, 119, 1205–1210. [Google Scholar] [CrossRef]
- Sarjit, A.; Wang, Y.; Dykes, G.A. Antimicrobial activity of gallic acid against thermophilic Campylobacter is strain specific and associated with a loss of calcium Ions. Food Microbiol. 2015, 46, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Phakthong, W.; Liawruangrath, B.; Liawruangrath, S. Determination of gallic acid with rhodanine by reverse flow injection analysis using simplex optimization. Talanta 2014, 130, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, D.; Wel, Y.; Chang, Y.; Zhao, J. A Simple and Sensitive Assay of Gallic Acid Based on Localized Surface Plasmon Resonance Light Scattering of Silver Nanoparticles through Modified Tollens Process. Anal. Sci. 2011, 27, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Narumi, K.; Sonoda, J.I.; Shiotani, K.; Shigeru, M.; Shibata, M.; Kawachi, A.; Tomishige, E.; Sato, K.; Motoya, T. Simultaneous detection of green tea catechins and gallic acid in human serum after ingestion of green tea tablets using ion-pair high-performance liquid chromatography with electrochemical detection. J. Chromatogr. B 2014, 945, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hamid, R.; Newair, F.E. Adsorptive stripping voltammetric determination of gallic acid using an electrochemical sensor based on polyepinephrine/glassy carbon electrode and its determination in black tea sample. J. Electroanal. Chem. 2013, 704, 32–37. [Google Scholar] [CrossRef]
- Sundaram, S.; Jagannathan, M.; Kadir, M.R.A.; Palanivel, S.; Hadibarata, T.; Yusoff, A.R.M. A new electro-generated o-dianisidine derivative stabilized MWCNT-modified GCE for low potential gallic acid detection. RSC Adv. 2015, 5, 45996–46006. [Google Scholar] [CrossRef]
- Sum, Y.L.; Chen, S.H. Sensitive and selective determination of gallic acid in green tea samples based on an electrochemical platform of poly(melamine) film. Anal. Chim. Acta 2015, 901, 41–50. [Google Scholar] [CrossRef]
- Hur, J.M.; Park, J.G.; Yang, K.H.; Park, J.C.; Park, J.R.; Chun, S.S.; Choi, J.S.; Choi, J.W. Effect of Methanol Extract of Zanthoxylum piperitum Leaves and of Its Compound, Protocatechuic Acid, on Hepatic Drug Metabolizing Enzymes and Lipid Peroxidation in Rats. Biosci. Biotechnol. Biochem. 2003, 67, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Vitaglione, P.; Donnarumma, G.; Napolitano, A.; Galvano, F.; Gallo, A.; Scalfi, L.; Fogliano, V. Protocatechuic acid is the major human metabolite of cyanidin-glucosides. J. Nutr. 2007, 137, 2043–2048. [Google Scholar] [CrossRef] [PubMed]
- Kakkar, S.; Bais, S. A Review on Protocatechuic Acid and Its Pharmacological Potential. ISRN Pharmacol. 2014, 2014, 952943. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, X.; Chen, D.; Chen, S. Antioxidant Activity and Mechanism of Protocatechuic Acid in vitro. Funct. Foods Health Dis. 2011, 1, 232–244. [Google Scholar]
- Vari, R.; D’Archivio, M.; Filesi, C.; Carotenuto, S.; Scazzocchio, B.; Santangelo, C.; Giovannini, C.; Masella, R. Protocatechuic acid induces antioxidant/detoxifying enzyme expression through JNK-mediated Nrf2 activation in murine macrophages. J. Nutr. Biochem. 2011, 22, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.Y.; Yin, M.C. Antibacterial effects of roselle calyx extracts and protocatechuic acid in ground beef and apple juice. Foodborne Pathog. Dis. 2009, 6, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Scazzocchio, B.; Var, R.; Filesi, C.; D’Archivio, M.; Santangelo, C.; Giovannini, C.; Iacovelli, A.; Silecchia, G.; Volti, G.L.; Galvano, F. Cyanidin-3-O-β-glucoside and protocatechuic acid exert insulin-like effects by upregulating PPARγ activity in human omental adipocytes. Diabetes 2011, 60, 2234–2244. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Tanaka, T.; Tanaka, M. Potential cancer chemopreventive activity of protocatechuic acid. J. Exp. Clin. Med. 2011, 3, 27–33. [Google Scholar] [CrossRef]
- Li, C.; Jiang, W.; Zhu, H.; Hou, J. Antifibrotic effects of protocatechuic aldehyde on experimental liver fibrosis. Pharm. Biol. 2012, 50, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Ou, C.; Shi, N.; Yang, Q.; Zhang, Y.; Wu, Z.; Wang, B.; Compans, R.W.; He, C. Protocatechuic acid, a novel active substance against avian Influenza virus H9N2 infection. PLoS ONE 2014, 9, e111004. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Guo, J.; Zhang, Y.; Shi, S. Efficient Determination of Protocatechuic Acid in Fruit Juices by Selective and Rapid Magnetic Molecular Imprinted Solid Phase Extraction Coupled with HPLC. J. Agric. Food Chem. 2014, 62, 8221–8228. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hu, X.; Zhang, Y.; Shi, S.; Jiang, X.; Chen, X. High-capacity magnetic hollow porous molecularly imprinted polymers for specific extraction of protocatechuic acid. J. Chromatogr. A 2015, 1404, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Jiang, Q.; Xi, M.; Jiao, K. Determination of 3,4-dihydroxybenzoic acid by electrocatalytic oxidation at an ionic liquid modified electrode. Microchim. Acta 2009, 166, 343–348. [Google Scholar] [CrossRef]
- Yildirim, S.; Kadioğlu, A.; Sağlam, A.; Yaşar, A. Determination of phenolic acids and rutin in Heliotropium thermophilum by high-performance liquid chromatography with photodiode array detection. Instrum. Sci. Technol. 2017, 45, 35–48. [Google Scholar] [CrossRef]
- Zhang, A.; Wan, L.; Wu, C.; Fang, Y.; Han, G.; Li, H.; Zhang, Z.; Wang, H. Simultaneous Determination of 14 Phenolic Compounds in Grape Canes by HPLC-DAD-UV Using Wavelength Switching Detection. Molecules 2013, 18, 14241–14257. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.L.; Pereiraa, J.; Woutera, V.G.; Giro, C.; Câmara, J.S. A fast method using a new hydrophilic-lipophilic balanced sorbent in combination with ultra-high-performance liquid chromatography for quantification of significant bioactive metabolites in wines. Talanta 2011, 86, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Nicoleti, I.; Bello, C.; de Rossi, A.; Corrandini, D. Identification and Quantification of Phenolic Compounds in Grapes by HPLC-PDA-ESI-MS on a Semimicro Separation Scale. J. Agric. Food Chem. 2008, 56, 8801–8808. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Guo, J.; Zhang, Y.; Hua, Y.; You, Q.; Shi, S. Novel molecular imprinted polymers over magnetic mesoporous silica microspheres for selective and efficient determination of protocatechuic acid in Syzygium aromatic. Food Chem. 2015, 178, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Wang, J.; Zhu, L.; Cai, M.; Du, X. Electrochemical determination of catechin, protocatechuic acid, and L-lactic acid based on voltammetric response of ferroceneboronic acid. J. AOAC Int. 2014, 97, 1742–1745. [Google Scholar] [CrossRef] [PubMed]
- Puangjan, A.; Chaiyasith, S. An efficient ZrO2/Co3O4/reduced graphene oxide nanocomposite electrochemical sensor for simultaneous determination of gallic acid, caffeic acid and protocatechuic acid natural antioxidants. Electrochim. Acta 2014, 211, 273–288. [Google Scholar] [CrossRef]
- Liscio, A.; Veronese, G.P.; Treossi, E.; Suriano, F.; Rosella, F.; Bellani, V.; Rizzoli, R.; Samori, P.; Palermo, V. Charge transport in graphene–polythiophene blends as studied by Kelvin Probe Force Microscopy and transistor characterization. J. Mater. Chem. 2011, 21, 2924–2931. [Google Scholar] [CrossRef]
- Yang, L.; Liu, D.; Huang, J.; You, T. Simultaneous determination of dopamine, ascorbic acid and uric acid at electrochemically reduced graphene oxide modified electrode. Sens. Actuators B 2014, 193, 166–172. [Google Scholar] [CrossRef]
- Roghani-Mamaqani, H.; Haddadi-Asl, V.; Khezri, K.; Salami-Kalajahi, M. Polystyrene-grafted graphene nanoplatelets with various graft densities by atom transfer radical polymerization from the edge carboxyl groups. RSC Adv. 2014, 4, 24439–24452. [Google Scholar] [CrossRef]
- Yang, J.; Deng, S.; Lei, J.; Jub, H.; Gunasekarana, S. Electrochemical synthesis of reduced graphene sheet–AuPd alloy nanoparticle composites for enzymatic biosensing. Biosens. Bioelectron. 2011, 29, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.F.; Xie, F.; Hu, R.F. Electrochemical study of brucine on an electrode modified with magnetic carbon-coated nickel nanoparticles. Anal. Bioanal. Chem. 2007, 387, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hamid, R.; Newair, E.F. Electrochemical behavior of antioxidants: I. Mechanistic study on electrochemical oxidation of gallic acid in aqueous solutions at glassy-carbon electrode. J. Electroanal. Chem. 2011, 657, 107–112. [Google Scholar] [CrossRef]
- Hotta, H.; Ueda, M.; Nagano, S.; Tsujino, Y.; Koyama, J.; Osakai, T. Mechanistic Study of the Oxidation of Caffeic Acid by Digital Simulation of Cyclic Voltammograms. J. Anal. Biochem. 2002, 303, 66–72. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdel-Hamid, R.; Bakr, A.; Newair, E.F.; Garcia, F. Simultaneous Voltammetric Determination of Gallic and Protocatechuic Acids in Mango Juice Using a Reduced Graphene Oxide-Based Electrochemical Sensor. Beverages 2019, 5, 17. https://doi.org/10.3390/beverages5010017
Abdel-Hamid R, Bakr A, Newair EF, Garcia F. Simultaneous Voltammetric Determination of Gallic and Protocatechuic Acids in Mango Juice Using a Reduced Graphene Oxide-Based Electrochemical Sensor. Beverages. 2019; 5(1):17. https://doi.org/10.3390/beverages5010017
Chicago/Turabian StyleAbdel-Hamid, Refat, Ahmed Bakr, Emad F. Newair, and François Garcia. 2019. "Simultaneous Voltammetric Determination of Gallic and Protocatechuic Acids in Mango Juice Using a Reduced Graphene Oxide-Based Electrochemical Sensor" Beverages 5, no. 1: 17. https://doi.org/10.3390/beverages5010017
APA StyleAbdel-Hamid, R., Bakr, A., Newair, E. F., & Garcia, F. (2019). Simultaneous Voltammetric Determination of Gallic and Protocatechuic Acids in Mango Juice Using a Reduced Graphene Oxide-Based Electrochemical Sensor. Beverages, 5(1), 17. https://doi.org/10.3390/beverages5010017




