Grape Juice Consumption with or without High Fat Diet during Pregnancy Reduced the Weight Gain and Improved Lipid Profile and Oxidative Stress Levels in Liver and Serum from Wistar Rats
Abstract
:1. Introduction
2. Methodology
2.1. Grape Juice
2.2. Analysis of Phenolic Compounds
2.3. Diets
2.4. Animals
2.5. Treatment
2.6. Biochemical Parameters Evaluated in Serum
2.7. Oxidative Stress
2.8. Protein Determination
2.9. Statistical Analysis
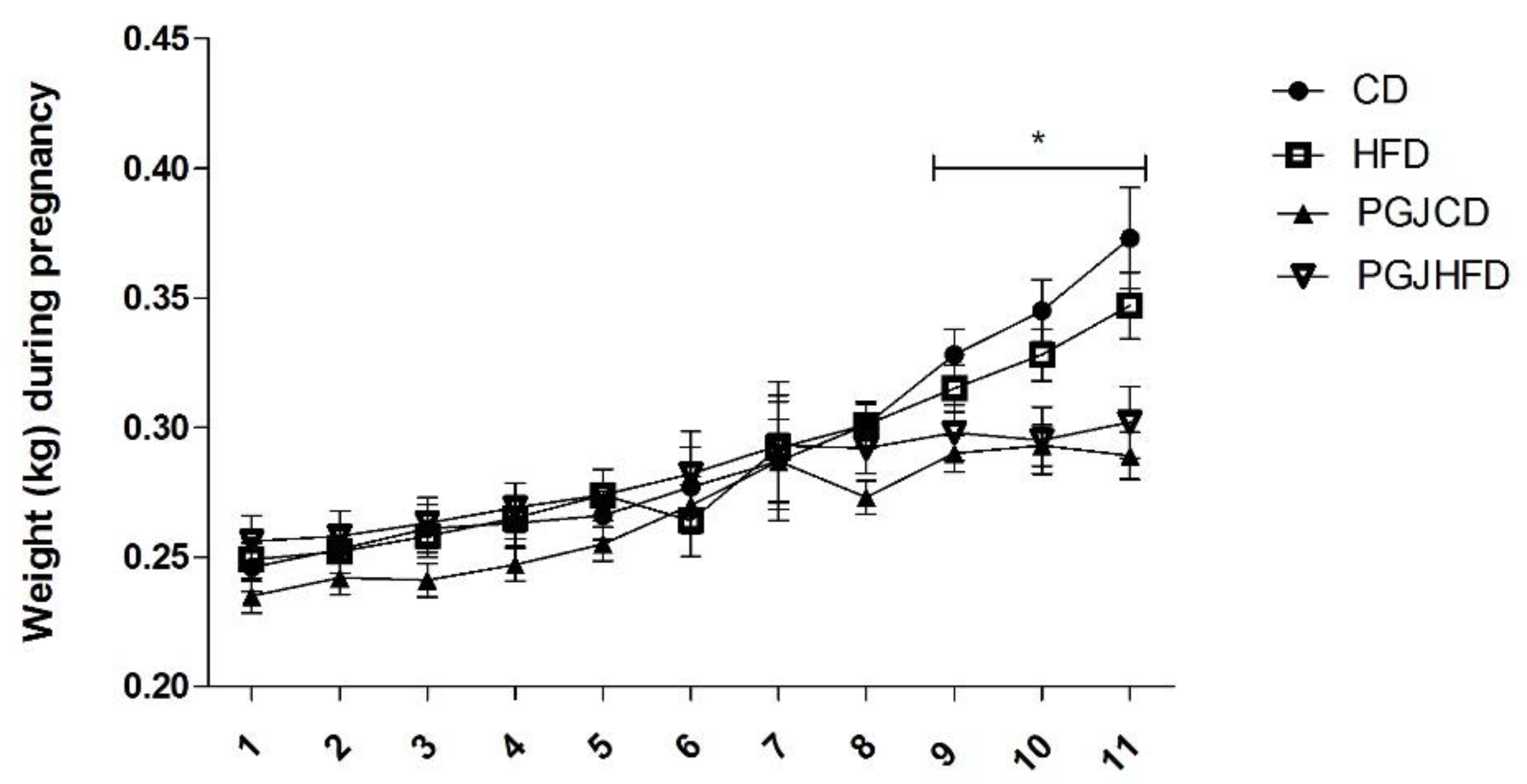
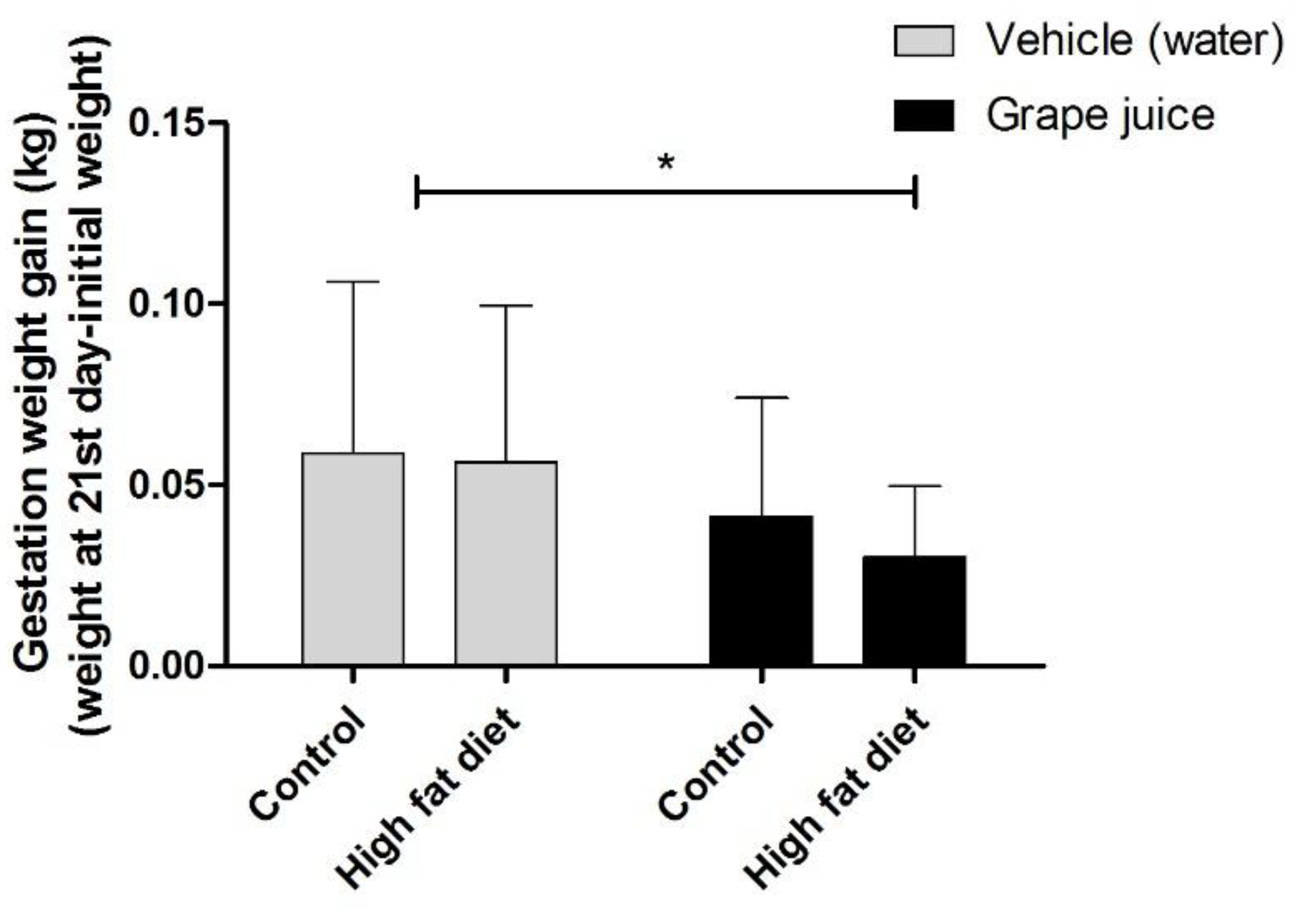
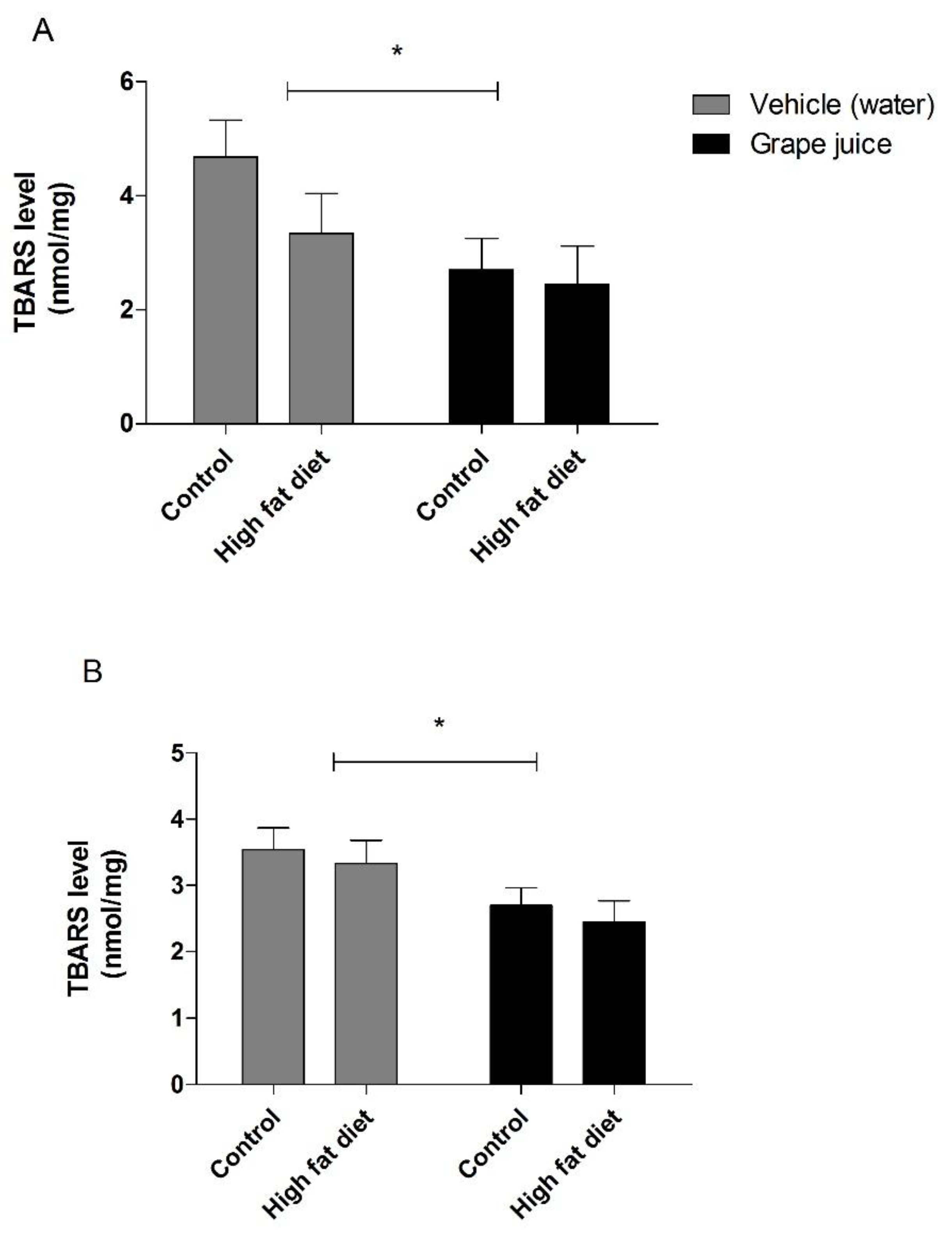
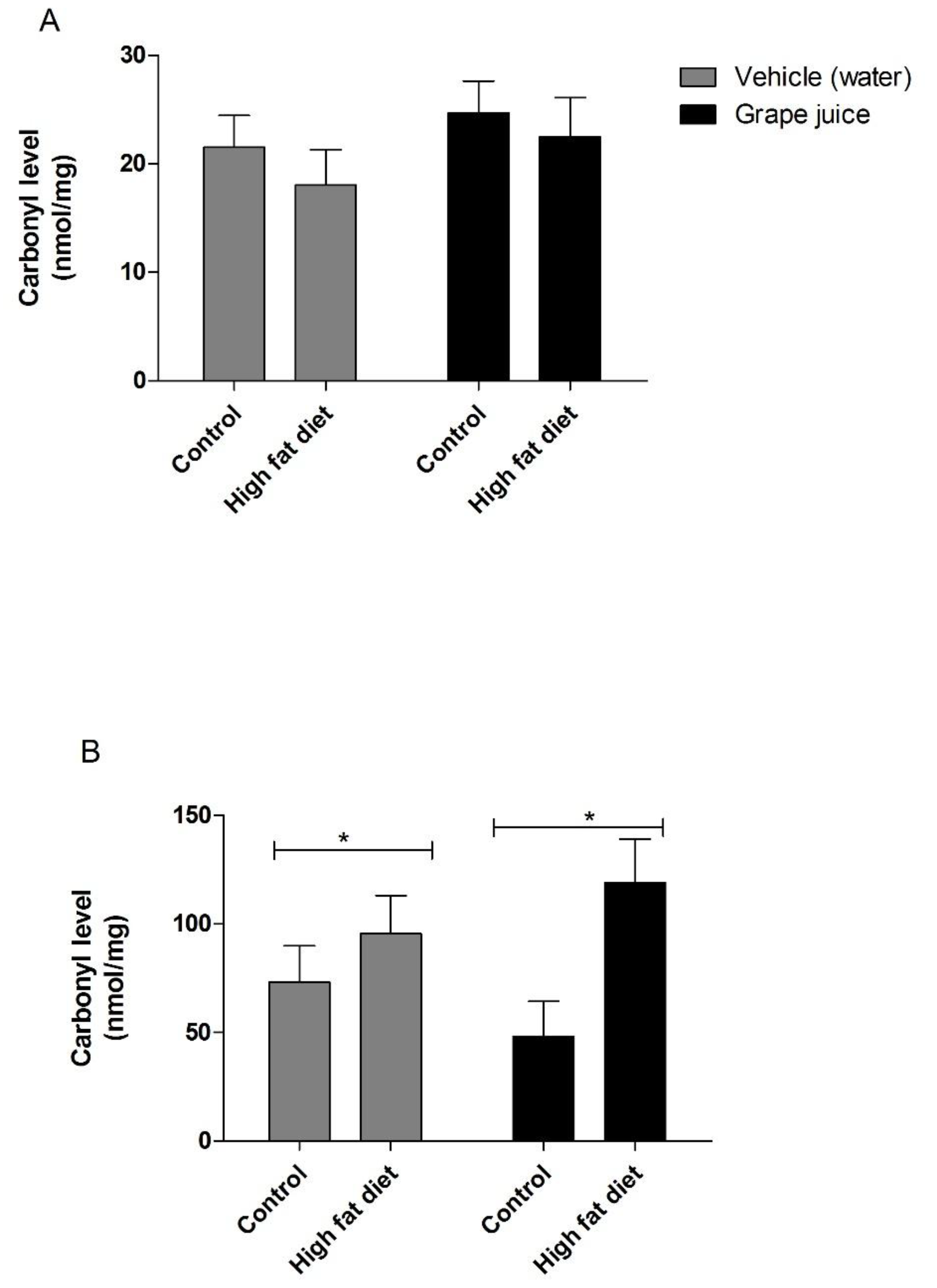
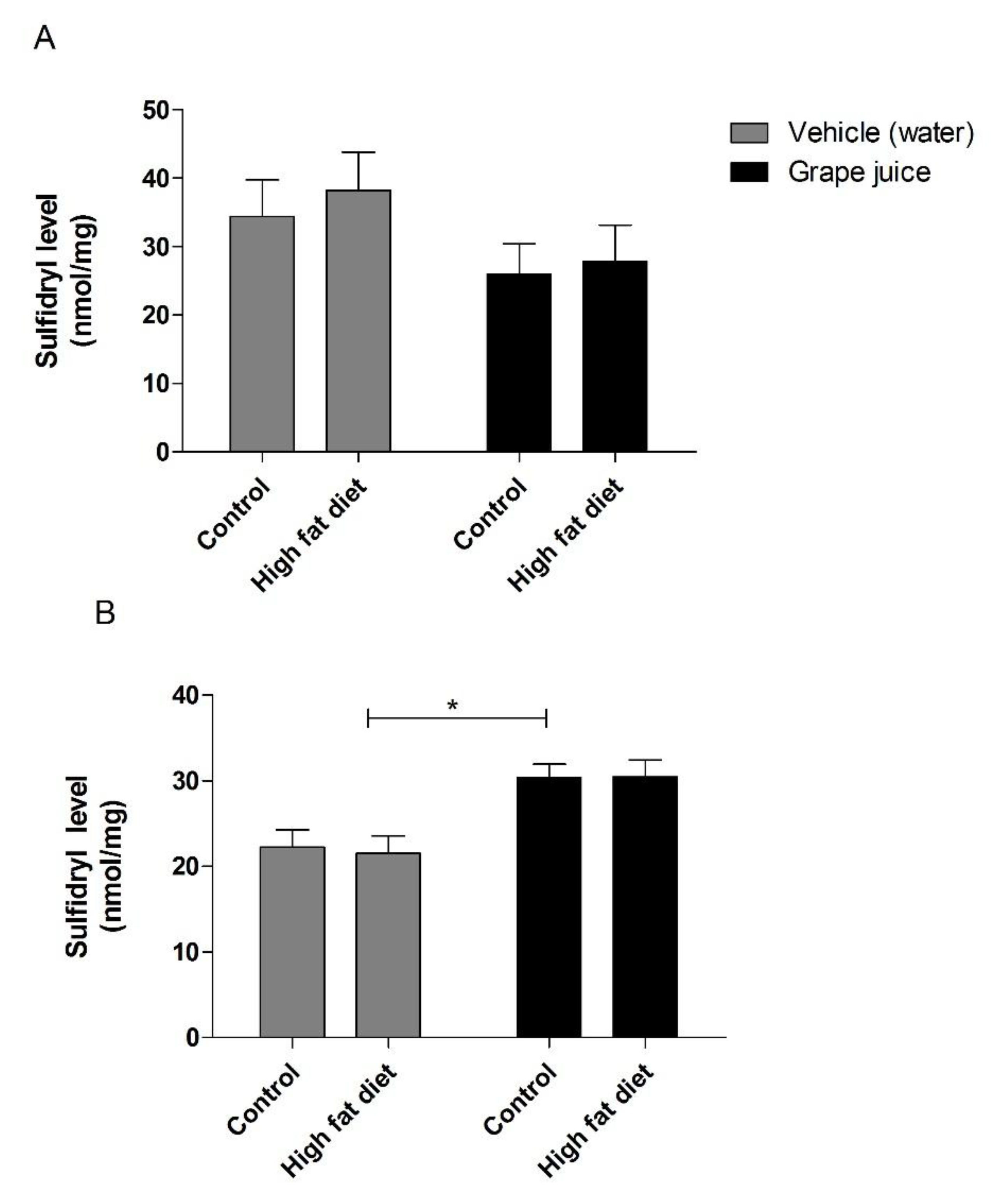
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Souza, A.I. De Nutrição em obstetrícia e pediatria. Rev. Bras. Saúde Matern. Infant. 2004, 4, 203–204. [Google Scholar] [CrossRef]
- Kennedy, E.; Bowman, S.; Powell, R. Dietary-fat intake in the US population. J. Am. Coll. Nutr. 1999, 18, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kwak, H.-B. Role of adiponectin in metabolic and cardiovascular disease. J. Exerc. Rehabil. 2014, 10, 54–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruse, M.; Seki, Y.; Vuguin, P.M.; Du, X.Q.; Fiallo, A.; Glenn, A.S.; Singer, S.; Breuhahn, K.; Katz, E.B.; Charron, M.J. High-Fat Intake During Pregnancy and Lactation Exacerbates High-Fat Diet-Induced Complications in Male Offspring in Mice. Endocrinology 2013, 154, 3565–3576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ziegler, E.E.; Filer, L.J. Conocimientos actuales sobre nutrición. Rev. Esp. Salud Publica 1998, 72, 379–380. [Google Scholar] [CrossRef]
- Fontelles, C.C.; Guido, L.N.; Rosim, M.P.; Andrade, F.O.; Jin, L.; Inchauspe, J.; Pires, V.C.; de Castro, I.A.; Hilakivi-Clarke, L.; de Assis, S.; et al. Paternal programming of breast cancer risk in daughters in a rat model: Opposing effects of animal- and plant-based high-fat diets. Breast Cancer Res. 2016, 18, 71. [Google Scholar] [CrossRef] [PubMed]
- Fontelles, C.C.; da Cruz, R.S.; Hilakivi-Clarke, L.; de Assis, S.; Ong, T.P. Investigation of Paternal Programming of Breast Cancer Risk in Female Offspring in Rodent Models. In Methods in Molecular Biology (Clifton, N.J.); Humana Press: New York, NY, USA, 2018; Volume 1735, pp. 207–220. [Google Scholar]
- Nguyen, N.M.; de Oliveira Andrade, F.; Jin, L.; Zhang, X.; Macon, M.; Cruz, M.I.; Benitez, C.; Wehrenberg, B.; Yin, C.; Wang, X.; et al. Maternal intake of high n-6 polyunsaturated fatty acid diet during pregnancy causes transgenerational increase in mammary cancer risk in mice. Breast Cancer Res. 2017, 19, 77. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Allamaneni, S.S.R. Role of free radicals in female reproductive diseases and assisted reproduction. Reprod. Biomed. Online 2004, 9, 338–347. [Google Scholar] [CrossRef]
- Markesbery, W.R. The Role of Oxidative Stress in Alzheimer Disease. Arch. Neurol. 1999, 56, 1449–1452. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Gupta, S.; Sikka, S. The role of free radicals and antioxidants in reproduction. Curr. Opin. Obstet. Gynecol. 2006, 18, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Saleh, M.M.; Abdo, K.R. Intrahepatic cholestasis of pregnancy: Review of the literature and evaluation of current evidence. J. Womens Health (Larchmt) 2007, 16, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Oliboni, L.S.; Vanderlinde, R.; Bonatto, D.; Salvador, M.; Henriques, J.A.P. Phenolic content and antioxidant activities of white and redred juices manufactured with organically- or conventionally-produced grapes. Food Chem. Toxicol. 2007, 45, 2574–2580. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Pasquali, M.A.B.; Oliveira, M.R.; Umezu, F.M.; Salvador, M.; Henriques, J.A.P.; Moreira, J.C.F. Protective Effects of RedPurple grape Juice on Carbon Tetrachloride-Induced Oxidative Stress in Brains of Adult Wistar Rats. J. Med. Food 2008, 11, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Oliboni, L.S.; Pasquali, M.A.B.; Oliveira, M.R.; Umezu, F.M.; Salvador, M.; Moreira, J.C.F.; Henriques, J.A.P. Intake of RedPurple grape Juice as a Hepatoprotective Agent in Wistar Rats. J. Med. Food 2008, 11, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Saleh, M.M.; Abdo, K.R. Intrahepatic Cholestasis of Pregnancy: Review of the Literature and Evaluation of Current Evidence. J. Womens Health 2007, 16, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Arrese, M.; Reyes, H. Intrahepatic cholestasis of pregnancy: A past and present riddle. Ann. Hepatol. 2006, 5, 202–205. [Google Scholar] [PubMed]
- Wasmuth, H.E.; Glantz, A.; Keppeler, H.; Simon, E.; Bartz, C.; Rath, W.; Mattsson, L.-A.; Marschall, H.-U.; Lammert, F. Intrahepatic cholestasis of pregnancy: The severe form is associated with common variants of the hepatobiliary phospholipid transporter ABCB4 gene. Gut 2007, 56, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Buchner, I.; Medeiros, N.; Lacerda, D.D.S.; Normann, C.A.B.M.; Gemelli, T.; Rigon, P.; Wannmacher, C.M.D.; Henriques, J.A.P.; Dani, C.; Funchal, C. Hepatoprotective and Antioxidant Potential of Organic and Conventional Grape Juices in Rats Fed a High-Fat Diet. Antioxidants 2014, 3, 323–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farias, M.; Gonçalves, L.K.; Schaffer, T.K.; Hilger, D.K.; Dario, R.; Neto, B.; Antunes, C.; Frusciante, M.; Rodrigues, A.; Funchal, C.; et al. Effect of grape juice on some biochemical and oxidative stress parameters in serum and liver enzymes of pregnant and lactating rats. Issues Biol. Sci. Pharm. Res. 2015, 3, 37–46. [Google Scholar] [CrossRef]
- Marchi, J.; Berg, M.; Dencker, A.; Olander, E.K.; Begley, C. Risks associated with obesity in pregnancy, for the mother and baby: A systematic review of reviews. Obes. Rev. 2015, 16, 621–638. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Andrade, F.; Fontelles, C.C.; Rosim, M.P.; de Oliveira, T.F.; de Melo Loureiro, A.P.; Mancini-Filho, J.; Rogero, M.M.; Moreno, F.S.; de Assis, S.; et al. Exposure to lard-based high-fat diet during fetal and lactation periods modifies breast cancer susceptibility in adulthood in rats. J. Nutr. Biochem. 2014, 25, 613–622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wills, E.D. Mechanisms of lipid peroxide formation in animal tissues. Biochem. J. 1966, 99, 667–676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levine, R.L.; Garland, D.; Oliver, C.N.; Amici, A.; Climent, I.; Lenz, A.G.; Ahn, B.W.; Shaltiel, S.; Stadtman, E.R. Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol. 1990, 186, 464–478. [Google Scholar] [PubMed]
- Aksenov, M.Y.; Markesbery, W.R. Changes in thiol content and expression of glutathione redox system genes in the hippocampus and cerebellum in Alzheimer’s disease. Neurosci. Lett. 2001, 302, 141–145. [Google Scholar] [CrossRef]
- Bannister, J.V.; Calabrese, L. Assays for superoxide dismutase. Methods Biochem. Anal. 1987, 32, 279–312. [Google Scholar] [PubMed]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [PubMed]
- Lowry, O.H.; Rosebrouh, N.J.; Lewis-Farr, A.L.; Randall, R.J. Protein Measurement with the Folin phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jin, Y.; Choi, Y.; Park, T. Resveratrol exerts anti-obesity effects via mechanisms involving down-regulation of adipogenic and inflammatory processes in mice. Biochem. Pharmacol. 2011, 81, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- Szkudelska, K.; Nogowski, L.; Szkudelski, T. The inhibitory effect of resveratrol on leptin secretion from rat adipocytes. Eur. J. Clin. Investig. 2009, 39, 899–905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardozo, M.G.; Medeiros, N.; Dos Santos Lacerda, D.; De Almeida, D.C.; Henriques, J.A.P.; Dani, C.; Funchal, C. Effect of chronic treatment with conventional and organic redpurple grape juices (Vitis labrusca) on rats fed with high-fat diet. Cell. Mol. Neurobiol. 2013, 33, 1123–1133. [Google Scholar] [CrossRef] [PubMed]
- Aderimi, F. Effects of Replacement of Wheat Bran with Cassava Root Sieviate Supplemented or Unsupplemented with Enzyme on Hematology and Serum Biochemistry of Pulled Chicks. J. Trop. For. Sci. 2004, 7, 147–153. [Google Scholar]
- Shankar, K.; Singh, S.; Kumar, D.; Varshney, S.; Gupta, A.; Rajan, S.; Srivastava, A.; Beg, M.; Srivastava, A.; Kanojiya, S.; et al. Cucumis melo ssp. Agrestis var. Agrestis Ameliorates High Fat Diet Induced Dyslipidemia in Syrian Golden Hamsters and Inhibits Adipogenesis in 3T3-L1 Adipocytes. Pharmacogn. Mag. 2015, 11, 501–510. [Google Scholar] [CrossRef]
- Eleftheriades, M.; Pervanidou, P.; Vafaei, H.; Vaggos, G.; Dontas, I.; Skenderi, K.; Sebire, N.J.; Nicolaides, K. Metabolic profiles of adult Wistar rats in relation to prenatal and postnatal nutritional manipulation: The role of birthweight. Hormones (Athens) 2014, 13, 268–279. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Lin, M.; Wang, X.; Guo, K.; Wang, S.; Sun, M.; Wang, J.; Han, X.; Fu, T.; Hu, Y.; et al. Basis of aggravated hepatic lipid metabolism by chronic stress in high-fat diet-fed rat. Endocrine 2015, 48, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Hort, M.A.; Schuldt, E.Z.; Bet, Â.C.; DalBó, S.; Siqueira, J.M.; Ianssen, C.; Abatepaulo, F.; de Souza, H.P.; Veleirinho, B.; Maraschin, M.; et al. Anti-Atherogenic Effects of a Phenol-Rich Fraction from Brazilian Red Wine (Vitis labrusca L.) in Hypercholesterolemic Low-Density Lipoprotein Receptor Knockout Mice. J. Med. Food 2012, 15, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A.; Teufel, K.; Wu, N. Red wine, dealcoholized red wine, and especially grape juice, inhibit atherosclerosis in a hamster model. Atherosclerosis 2001, 156, 67–72. [Google Scholar] [CrossRef]
- ZibaeeNezhad, M.J.; Mohammadi, E.; Babaie Beigi, M.A.; Mirzamohammadi, F.; Salehi, O. The Effects of Unripe Grape Juice on Lipid Profile Improvement. Cholesterol 2012, 2012, 890262. [Google Scholar] [CrossRef] [PubMed]
- Khadem-Ansari, M.H.; Rasmi, Y.; Ramezani, F. Effects of purple grape juice consumption on high density lipoprotein-cholesterol, apolipoprotein AI, apolipoprotein B and homocysteine in healthy human volunteers. Open Biochem. J. 2010, 4, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Castilla, P.; Echarri, R.; Dávalos, A.; Cerrato, F.; Ortega, H.; Teruel, J.L.; Lucas, M.F.; Gómez-Coronado, D.; Ortuño, J.; Lasunción, M.A. Concentrated purple grape juice exerts antioxidant, hypolipidemic, and antiinflammatory effects in both hemodialysis patients and healthy subjects. Am. J. Clin. Nutr. 2006, 84, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Pirinççioğlu, M.; Kızıl, G.; Kızıl, M.; Özdemir, G.; Kanay, Z.; Aydın Ketani, M. Protective effect of Öküzgözü (Vitis vinifera L. cv.) grape juice against carbon tetrachloride induced oxidative stress in rats. Food Funct. 2012, 3, 668–773. [Google Scholar] [CrossRef] [PubMed]
- Orhan, D.D.; Orhan, N.; Ergun, E.; Ergun, F. Hepatoprotective effect of Vitis vinifera L. leaves on carbon tetrachloride-induced acute liver damage in rats. J. Ethnopharmacol. 2007, 112, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Lacerda, D.S.; Santos, C.F.; Oliveira, A.S.; Zimmermann, R.; Schneider, R.; Agostini, F.; Dani, C.; Funchal, C.; Gomez, R. Antioxidant and hepatoprotective effects of an organic grapevine leaf (Vitis labrusca L.) extract in diabetic rats. RSC Adv. 2014, 4, 52611–52619. [Google Scholar] [CrossRef]
- Ahad, A.; Ahsan, H.; Mujeeb, M.; Siddiqui, W.A. Gallic acid ameliorates renal functions by inhibiting the activation of p38 MAPK in experimentally induced type 2 diabetic rats and cultured rat proximal tubular epithelial cells. Chem. Biol. Interact. 2015, 240, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Lin, S.; Zhang, L.; Li, Y. Resveratrol prevents renal lipotoxicity in high-fat diet-treated mouse model through regulating PPAR-α pathway. Mol. Cell. Biochem. 2016, 411, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Safa, J.; Argani, H.; Bastani, B.; Nezami, N.; Rahimi Ardebili, B.; Ghorbanihaghjo, A.; Kalagheichi, H.; Amirfirouzi, A.; Mesgari, M.; Soleimany Rad, J. Protective effect of grape seed extract on gentamicin-induced acute kidney injury. Iran. J. Kidney Dis. 2010, 4, 285–291. [Google Scholar] [PubMed]
- Vannucchi, C.I.; Jordao, A.A.; Vannucchi, H. Antioxidant compounds and oxidative stress in female dogs during pregnancy. Res. Vet. Sci. 2007, 83, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Moses, G.S.; Jensen, M.D.; Lue, L.-F.; Walker, D.G.; Sun, A.Y.; Simonyi, A.; Sun, G.Y. Secretory PLA 2-IIA: A new inflammatory factor for Alzheimer’s disease. J. Neuroinflammation 2006, 3. [Google Scholar] [CrossRef] [PubMed]
- Puglielli, L. Aging of the brain, neurotrophin signaling, and Alzheimer’s disease: Is IGF1-R the common culprit? Neurobiol. Aging 2008, 29, 795–811. [Google Scholar] [CrossRef] [PubMed]
- Barakauskas, V.E.; Ypsilanti, A.R.; Barr, A.M.; Innis, S.M.; Honer, W.G.; Beasley, C.L. Effects of sub-chronic clozapine and haloperidol administration on brain lipid levels. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2010, 34, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, G.F.; Santos, I.B.; de Bem, G.F.; Cordeiro, V.S.C.; da Costa, C.A.; de Carvalho, L.C.R.M.; Ognibene, D.T.; Resende, A.C.; de Moura, R.S. The Beneficial Effect of Anthocyanidin-Rich Vitis vinifera L. Grape Skin Extract on Metabolic Changes Induced by High-Fat Diet in Mice Involves Antiinflammatory and Antioxidant Actions. Phyther. Res. 2017, 31, 1621–1632. [Google Scholar] [CrossRef] [PubMed]
- Van Greevenbroek, M.M.J.; Schalkwijk, C.G.; Stehouwer, C.D.A. Dysfunctional adipose tissue and low-grade inflammation in the management of the metabolic syndrome: Current practices and future advances. F1000Research 2016, 5, 2515. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.D.; Scheffel, T.B.; Scola, G.; dos Santos, M.T.; Fank, B.; Dani, C.; Vanderlinde, R.; Henriques, J.A.P.; Coitinho, A.S.; Salvador, M. RedPurple grape juices prevent pentylenetetrazol-induced oxidative damage in the liver and serum of Wistar rats. Nutr. Res. 2013, 33, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Delwing-de Lima, D.; Ulbricht, A.S.S.F.; Werlang-Coelho, C.; Delwing-Dal Magro, D.; Joaquim, V.H.A.; Salamaia, E.M.; de Quevedo, S.R.; Desordi, L. Effects of two aerobic exercise training protocols on parameters of oxidative stress in the blood and liver of obese rats. J. Physiol. Sci. 2018, 68, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Free radicals, antioxidants, and human disease: Curiosity, cause, or consequence? Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef]
- Andrade, E.R.; Cruz, I.B.M.; Andrade, V.V.R.; Piccoli, J.C.E.; González-Gallego, J.; Barrio, J.P.; González, P. Evaluation of the potential protective effects of ad libitum black grape juice against liver oxidative damage in whole-body acute X-irradiated rats. Food Chem. Toxicol. 2011, 49, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
| Content | Conventional Diet | High Fat Diet | ||
|---|---|---|---|---|
| Calories | Gram | Calories | Gram | |
| Carbohydrates | 240 | 66 | 100 | 25 |
| Protein | 88 | 22 | 80 | 20 |
| Lipids | 36 | 4 | 450 | 50 |
| Total | 364 | 630 | ||
| Phenolic Compounds (mg/L) | Average | Standard Deviation |
|---|---|---|
| Total phenolic compounds | 2796.57 | 11.20 |
| Total flavonoids content | 77.27 | 2.10 |
| Resveratrol | 0.506 | 0.01 |
| Epicatechin | 1.95 | 0.04 |
| Naringin | 3.37 | 0.11 |
| Rutin | 17.41 | 0.25 |
| Chlorogenic acid | 12.37 | 0.12 |
| Parameters | Group | |||
|---|---|---|---|---|
| CD | HFD | PGJCD | PGJHFD | |
| Total cholesterol (mg/dL) | 69.43 ± 3.87 | 81.67 ± 4.17 * | 69.80 ± 4.57 | 97.33 ± 5.90 * |
| Triglycerides (mg/dL) | 65.20 ± 10.59 | 115.67 ± 9.66 * | 79.75 ± 11.83 | 138.67 ± 13.67 * |
| HDL (mg/dL) | 39.71 ± 2.33 | 26.67 ± 2.52 * | 36.80 ± 2.76 | 33.50 ± 3.08 * |
| AST (U/L) | 268.57 ± 11.82 | 264.50 ± 12.77 | 262.80 ± 13.99 | 205.75 ± 15.64 *,# |
| ALT (U/L) | 115.43 ± 6.69 | 74.83 ± 7.43 * | 123.80 ± 8.14 | 69.00 ± 9.10 * |
| Urea (mg/dL) | 79.00 ± 3.30 | 44.50 ± 3.56 * | 75.80 ± 3.90 | 44.25 ± 4.36 * |
| Creatinine (mg/dL) | 0.46 ± 0.030 | 047 ± 0.03 | 0.52 ± 0.03 | 0.52 ± 0.04 |
| Enzymes Actvity | C | HFD | PGJCD | PGJHDF |
|---|---|---|---|---|
| SOD (U SOD/mg) | 45.45 ± 13.35 | 70.13 ± 14.00 | 23.25 ± 12.28 * | 22.38 ± 13.35 * |
| CAT (U CAT/mg) | 3.95 ± 0.76 | 2.91 ± 0.84 | 3.29 ± 0.66 | 3.07 ± 0.80 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, L.K.; Bortolato, G.; Dario Braccini Neto, R.; Rocha Frusciante, M.; Funchal, C.; Dani, C. Grape Juice Consumption with or without High Fat Diet during Pregnancy Reduced the Weight Gain and Improved Lipid Profile and Oxidative Stress Levels in Liver and Serum from Wistar Rats. Beverages 2018, 4, 78. https://doi.org/10.3390/beverages4040078
Gonçalves LK, Bortolato G, Dario Braccini Neto R, Rocha Frusciante M, Funchal C, Dani C. Grape Juice Consumption with or without High Fat Diet during Pregnancy Reduced the Weight Gain and Improved Lipid Profile and Oxidative Stress Levels in Liver and Serum from Wistar Rats. Beverages. 2018; 4(4):78. https://doi.org/10.3390/beverages4040078
Chicago/Turabian StyleGonçalves, Luciana Kneib, Gabrielli Bortolato, Ruben Dario Braccini Neto, Marina Rocha Frusciante, Claudia Funchal, and Caroline Dani. 2018. "Grape Juice Consumption with or without High Fat Diet during Pregnancy Reduced the Weight Gain and Improved Lipid Profile and Oxidative Stress Levels in Liver and Serum from Wistar Rats" Beverages 4, no. 4: 78. https://doi.org/10.3390/beverages4040078





