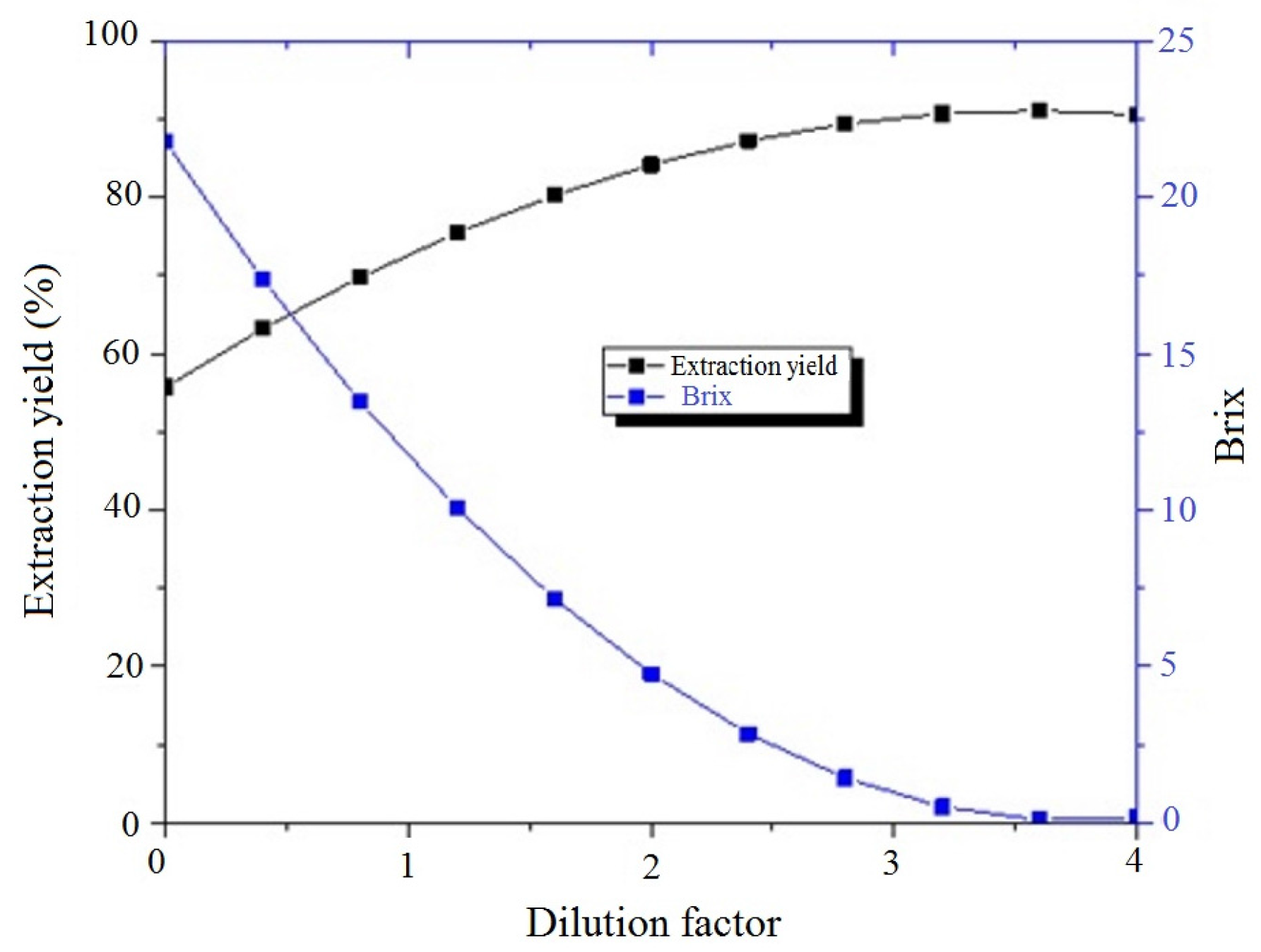
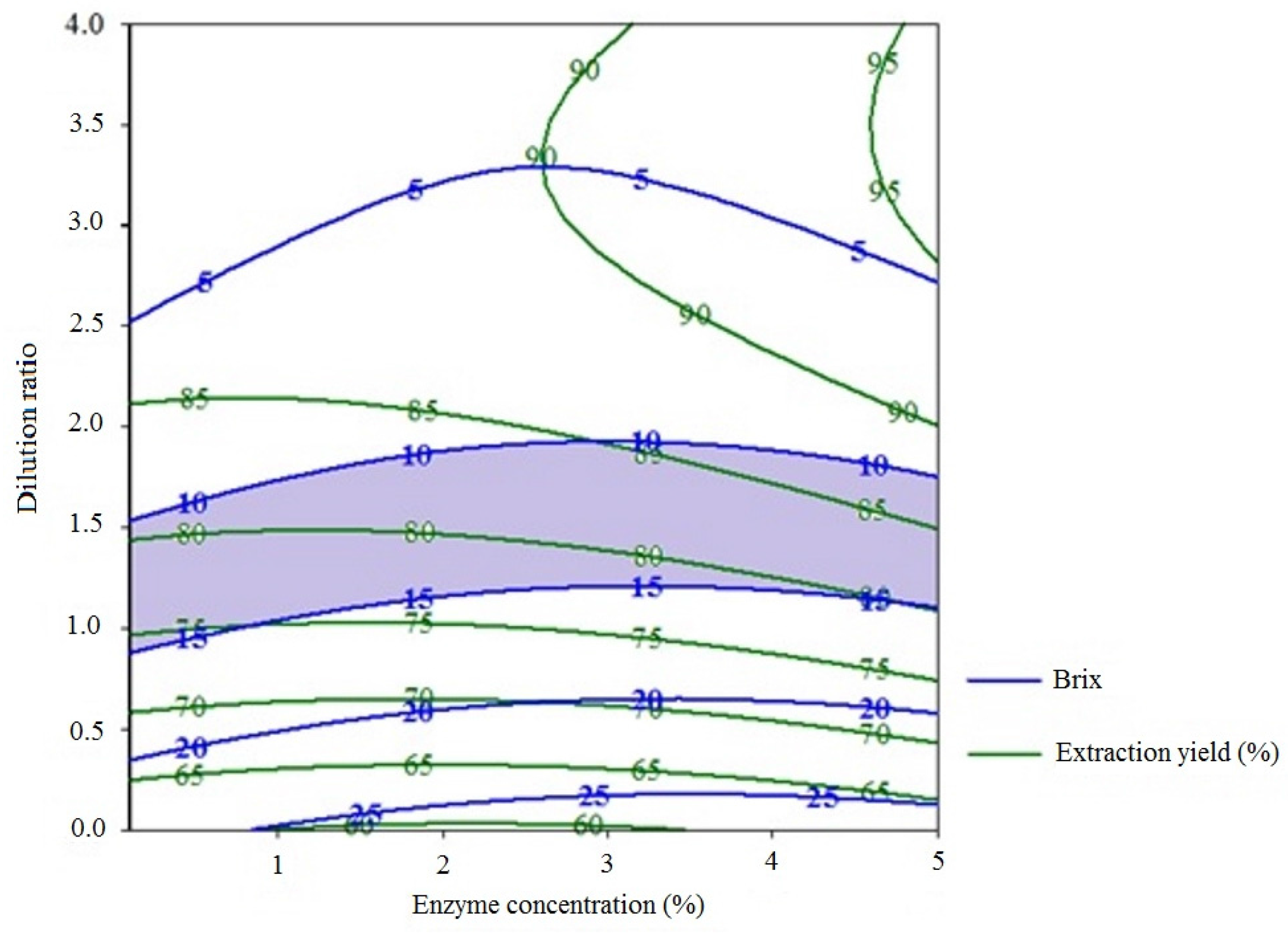
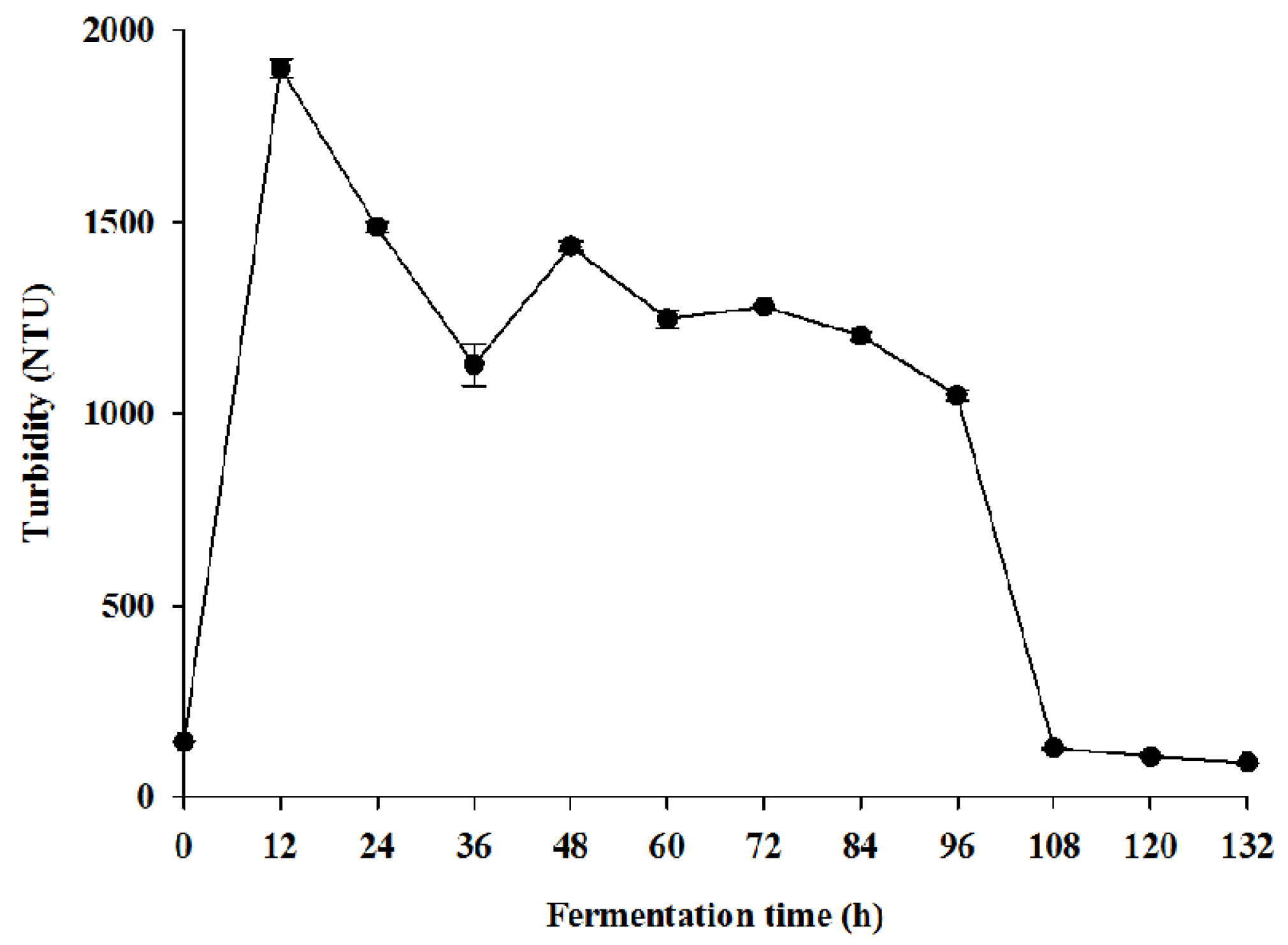
2.2. Methods
2.2.1. Sample Ripening
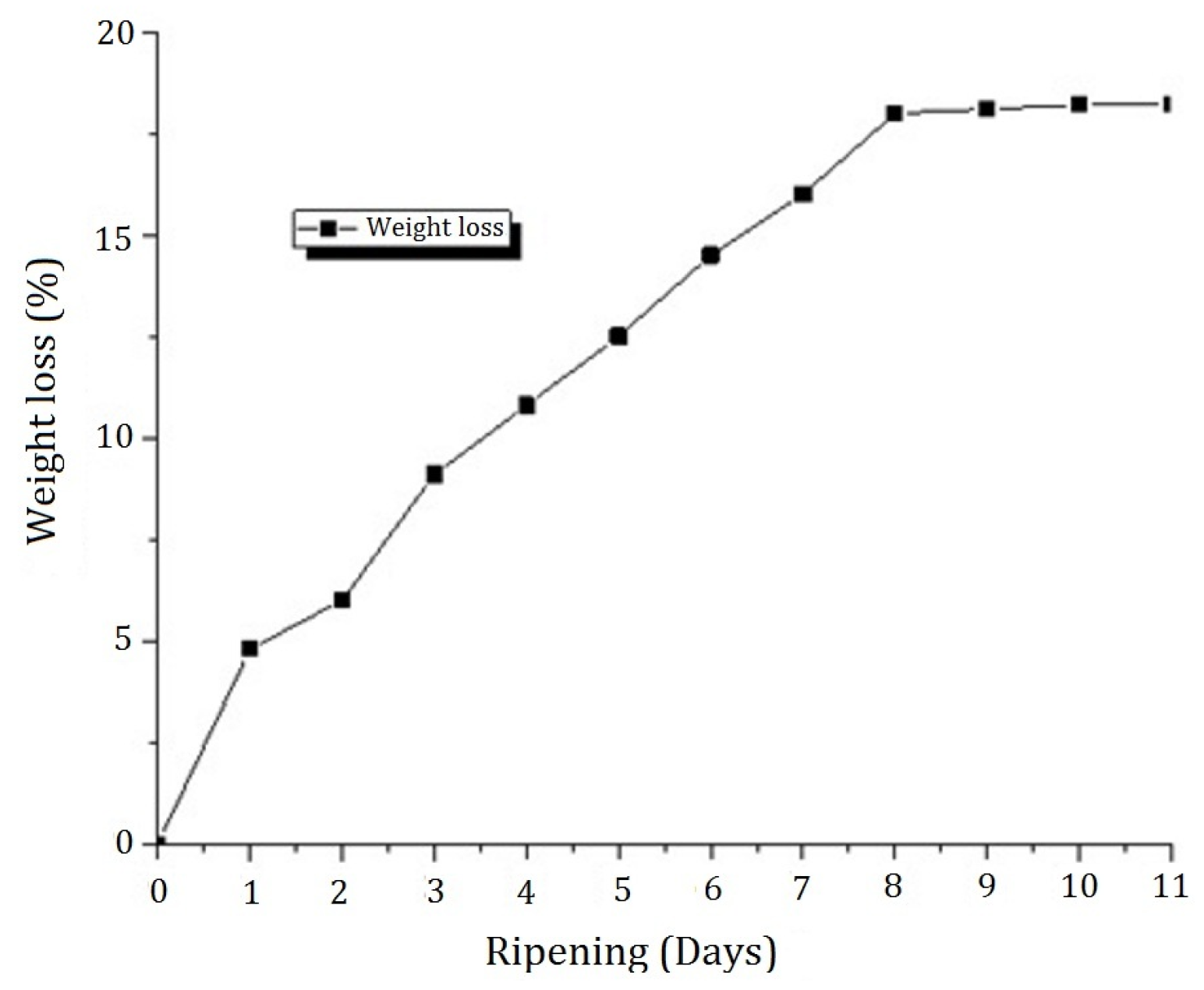
Unripe mature plantains were over-ripened for 11 days at 25 ± 2 °C utilizing a Heraeus-type incubator (D-63450 Hanau, Germany) without the use of ethylene until they passed Stage 7 (over-ripe). After that, samples were used immediately for wine production.
2.2.2. General Process Overview
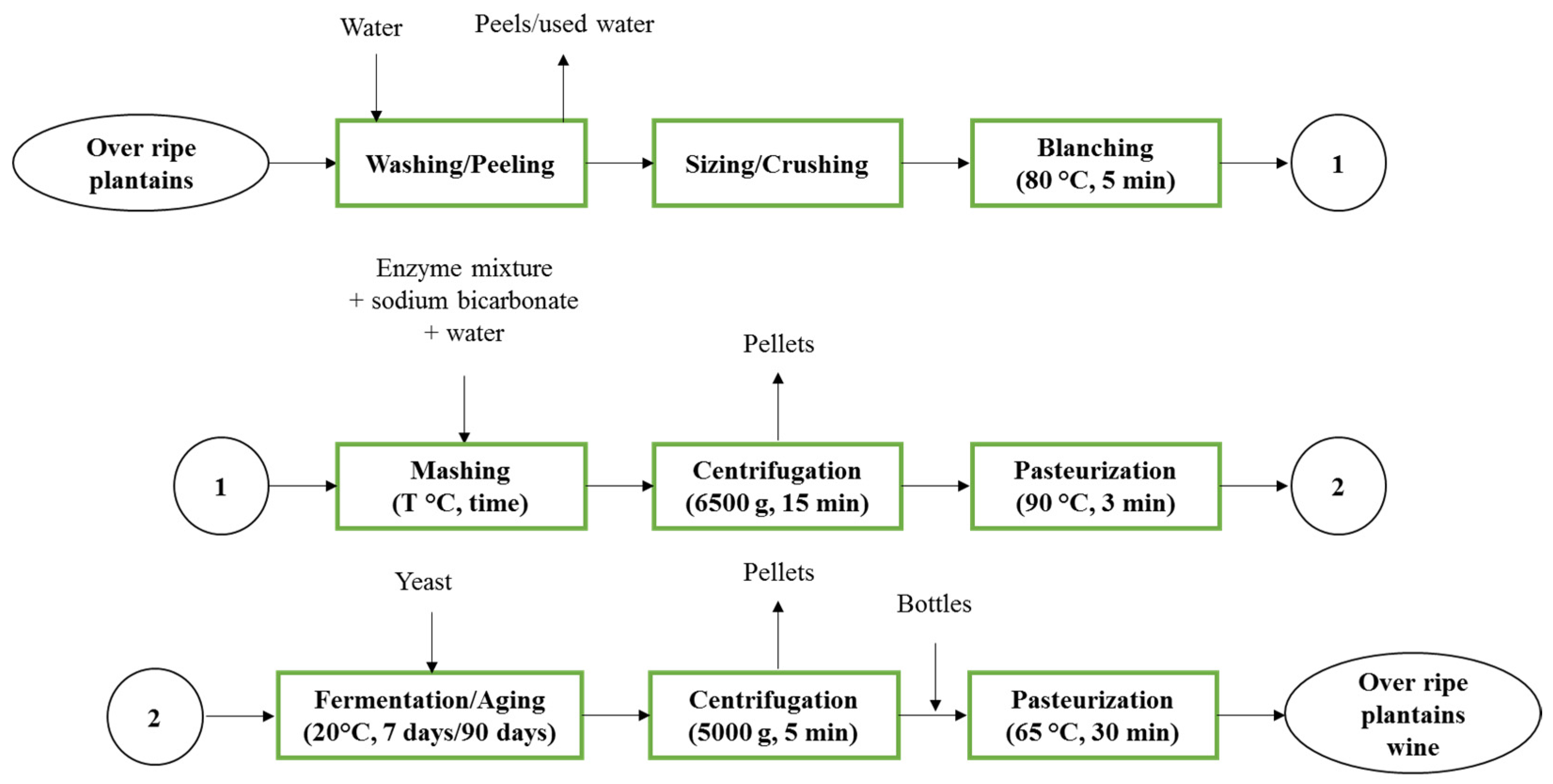
The methods involved in wine production from over-ripe giant horn plantain are given in
Figure 2.
The over-ripe plantains coming from the ripening process were washed three times with tap water and peeled. After the peeling process, they were sized and crushed to get the pulp paste. Then, the blanching process was executed at 80 °C for 5 min. Pulp paste was mashed using Doehlert experimental design conditions when the enzyme mixture, sodium bicarbonate and water were added. At the end of the mashing process, the mash was centrifuged (6500× g, 15 min) to remove pellets, and the must was then pasteurized (90 °C, 3 min). After cooling, the must was fermented at 20 °C for 7 days using yeast (4 g/L) and aged for 90 days. At the end of the aging process, the wine was centrifuged (5000× g, 5 min) to remove pellets (trub and yeast) and bottled. The bottles containing wine were then pasteurized at 65 °C for 30 min.
2.2.3. Experimental Design for the Extraction Process
Doehlert’s experimental design [
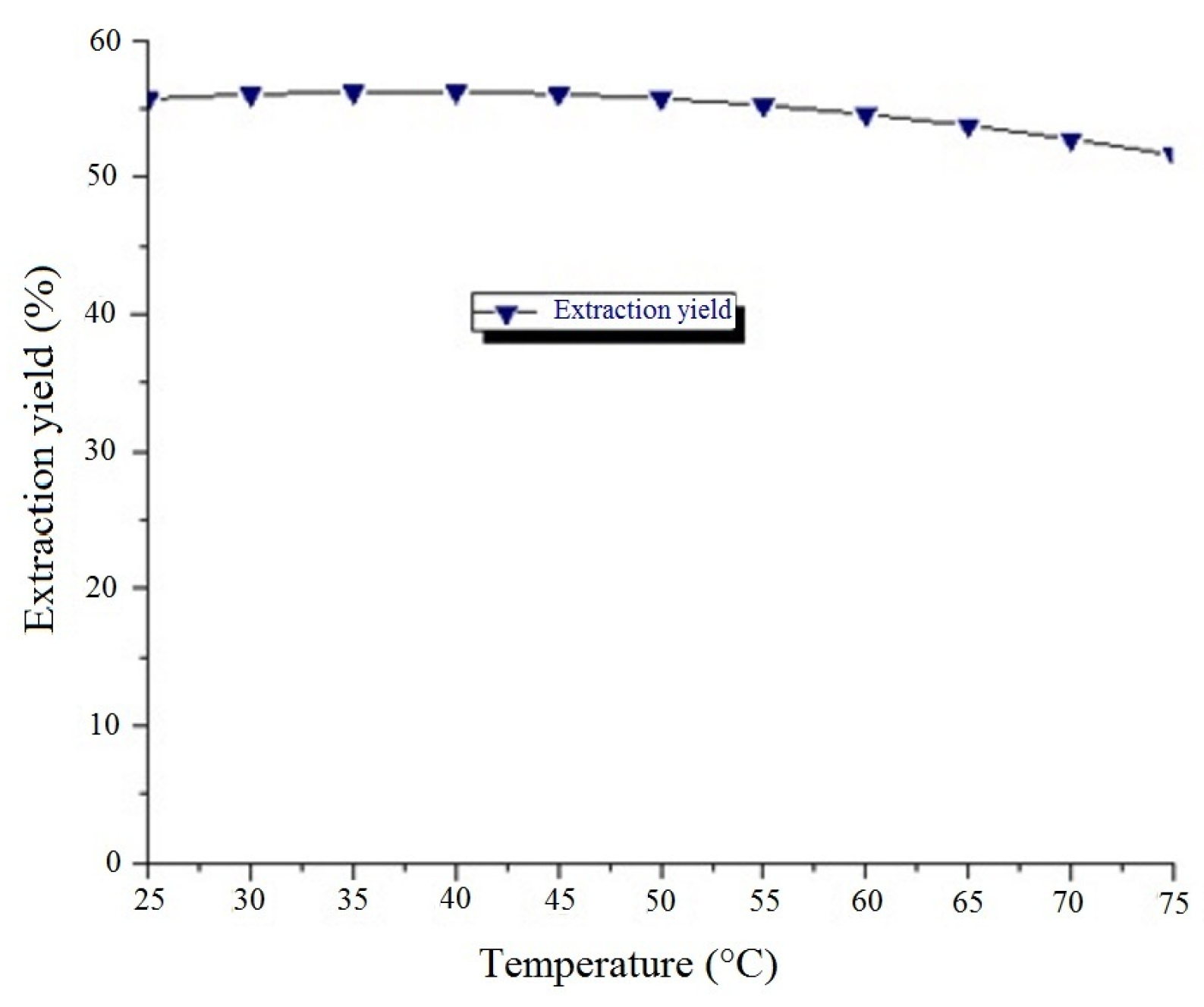
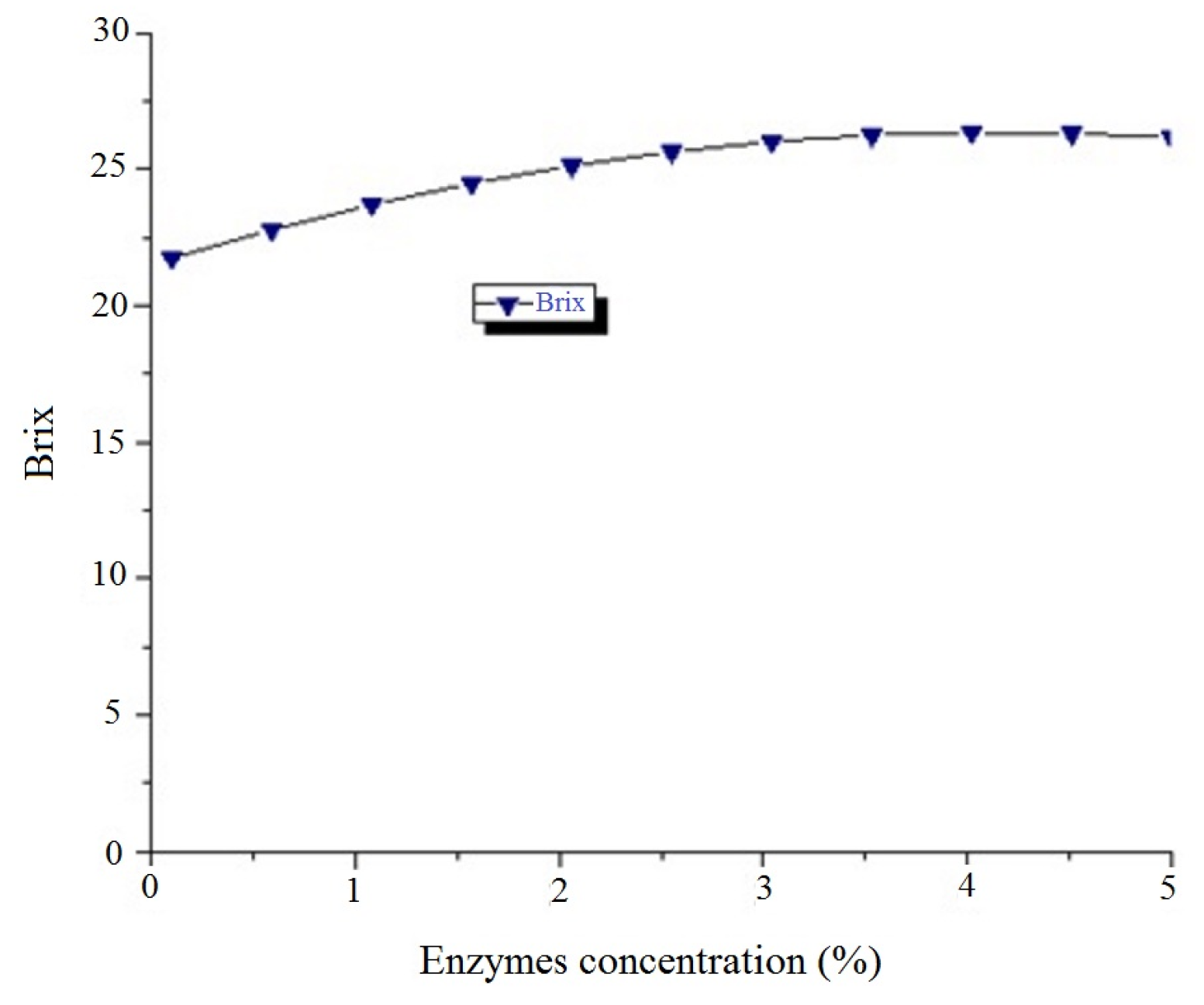
14] was used to model four factors: extraction temperature (°C), enzyme concentration (%), dilution factor and extraction time (h). The experimental domains (
Table 2) over which factors were studied were based on the literature. These independent variables were studied using the response surface methodology (RSM) at three different levels (−1, 0, +1) to give a total of 25 experiments. The conversion of coded values into real values was done according to the literature [
15].
2.2.4. Modelling and Optimizing
The model chosen was an empirical quadratic model, which determined the magnitude of the contributions of each factor in the linear, quadratic and interaction forms. The model used to determine the responses is given below:
where
is the response,
a constant,
the linear coefficients,
the quadratic coefficients,
the coefficients of interaction, and
,
,
,
and
, the linear, interaction and quadratic forms of the experimental variables respectively.
is the error. The determination of the coefficients of the models was done by a multilinear regression in which we had:
the number of experiments in the design matrix,
the number of coefficients in the chosen model,
the vector of experimental responses
yi,
;
the vector of theoretical responses
,
;
the matrix
, matrix of the model.
is the column vector of the parameters to be estimated;
…;
;
is the column vector of estimators
,
is the column vector of experimental error
,
.
Given the following classical matrix:
when
, the least squares method is used to estimate the coefficients of the polynomial.
is an estimator of
To this effect, Minitab software was used to determine the coefficients of the models.
In order to write equations that allow for the prediction of responses in the domain defined for the studies, it was important to validate the models. To that effect, the observed values and the values predicted by the model were compared. Further statistical tools were used in addition to the determination coefficient. Absolute average deviation (AAD) was calculated as follows:
is the experimental response and the response calculated from model i; N is number of experiments.
The bias factor (
) and exactitude factor (
) were also expressed as follows:
with:
Table 3 gives the acceptable values of the different indicators of valid models. The graphs and contour plots were done using OriginPro 2016 b9.3.226 (OriginLab Corporation,
www.originlab.com).
Optimization was done using the software Minitab® 17 (Build 17.3.1, Minitab, Inc.). The conditions were fixed to maximize the extraction yield and Brix. After that, a compromise was made. The use of Sigmaplot (Version 12.5, Systat Software, Inc.) allowed drawing the contour plots and superimpose the graphs in order to determine the interest zone.
The analysis of variance (
Table 4) was used to identify the influence of each factor and also the significance of their effects. This was achieved by comparing the average square of each effect to the experimental error. The significance of each factor was determined by the Fischer test. The significance of the Fischer (
F) ’ratio’ indicates the values necessary to reject the null hypothesis at the 0.05 probability level.
2.2.5. Juice Extraction
Over ripe plantains were washed 3 times, hand peeled and sized. A Panasonic blender (MX-GM1011) was used to crush the plantains, then they were blanched in water at 80 °C for 5 min in a water bath (Memmert). The four enzymes, amyloglucosidase, alpha amylase (BAN 480 L), pectinase and bioglucanase TX, were then introduced into biological reactors (beakers) containing 20 g of the pulp with its respective dilutions, which was then incubated at a temperature and time specified by the Doehlert experimental design in a water bath. After incubation, the pulp was then centrifuged at 6500× g for 15 min using a centrifuge (Heraeus-Kendro Lab products, model: Biofuge primo R, type: D-37520, Hanau, Germany). The supernatant was collected and pasteurized at 90 °C for 1 min in the water bath. Each experiment was conducted in triplicate.
2.2.6. Yeast Preparation and Must Fermentation
The white wine yeast Lalvin ICV D-47 Saccharomyces cerevisiae obtained from The Home Brew Shop (Unit 3, Hawley Lane Business Park, United Kingdom) was used for the fermentation of the giant horn plantain. Yeast, which was kept at 2–4 °C, was rehydrated at 30 °C in tap water (10 mL of water/g of yeast) for about 10 min and stirred for 5 s. After rehydration, yeast was introduced to the giant horn plantain juice extracted utilizing optimal conditions, and the mixture was mixed to ensure must aeration and yeast distribution. The principal fermentation was then done at 20 °C during 7 days and aging for 90 days in an incubator (SHP Biochemical Incubator 250 L, Shanghai, Guangzhou, China).
2.2.7. Determination of Extraction Yield
The percentage of juice yield (%
w/
w) was calculated as the difference between the initial mass and the weight of the pellet after centrifugation divided by the initial mass as follows:
2.2.8. Determination of Total Soluble Solids [19]
Before reading the value of total soluble solids, the Hanna HI 96801 refractometer (Hanna instruments Inc., Woonsocket, RI, USA) was calibrated using distilled water. An equal number of drops from the prepared plantain juice was placed onto the refractometer prism plate. The reading on the prism scale is generated numerically. After each test, the prism plate was cleaned with (distilled) water and wiped dry with a soft tissue.
2.2.9. Determination of pH
The initial pH of each plantain juice was determined using a pH meter (HANNA® Calibration Check™ pH meter, HI 223 Type, Johannesburg, South Africa). Twenty milliliters of each freshly-prepared plantain juice was placed in a glass beaker on a thermostatically-controlled electric hotplate (mark: GMARK) at 25 °C. Before reading its pH, each sample was agitated (using a magnetic stirrer) for 30 s until a stable reading was assessed. Each test was made in triplicate. Between readings, the electrode was rinsed with distilled water for the accuracy of the measurement.
2.2.10. Determination of Turbidity
To the turbidimeter cell, the plantain juice was added up to the horizontal mark. After closing the cell, it was wiped using a tissue and placed in the turbidimeter (Hach, Model 2100 N, Hach Company, Loveland, CO, USA), and the sample was covered. The turbidity value was then assessed when the reading was stable.
2.2.11. Determination of Total Phenolic Compounds: Folin–Ciocalteu Method [20]
The volume of 0.02 mL of plantain juice before and after fermentation was mixed with 0.1 mL of Folin–Ciocalteu reagent previously diluted with distilled water. A volume of 0.3 mL of 20% sodium carbonate solution was added to the mixture, shaken thoroughly and diluted to 2 mL by adding distilled water. The mixture was allowed to stand for 120 min, and the blue color formed was measured at 760 nm with a spectrophotometer (JENWAY, Model: 7310, Serial No. 39756, JENWAY Limited, Staffordshire, UK). Gallic acid was invoked as a standard for the calibration curve. The concentrations of gallic acid in the solution, utilized for obtaining the calibration curve, were 0, 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10 mg/L (R2 = 0.965). The total polyphenol content was expressed as mg of gallic acid equivalent (GAE) per 100 g of fruit juice. All measurements were carried out in triplicate.
2.2.12. Determination of Titratable Acidity [21]
Titratable acidity was established according to the standardized method, with 0.1 N sodium hydroxide (NaOH) in the presence of the phenolphthalein indicator. Ten milliliters of sample were pipetted into a conical flask and 0.1 mL of phenolphthalein (0.05%) added. Titration is halted when the initial color changes to pink and persisting for at least 30 s. The burette reading is noted. The titratable acidity (TA) is expressed in g/L tartaric acid:
where:
is the volume (mL) of the sodium hydroxide noted at the end point;
is the concentration of the base;
is the volume of titrate.
2.2.13. Determination of Color by the Spectrophotometric Method [22]
The wavelength of the spectrophotometer (JENWAY, Model: 7310, Serial No. 39756, JENWAY Limited, Staffordshire, UK) was set at 430 nm. The cell was filled with water and the absorbance set to read as 0.00. The cell was then rinsed and filled with the sample (must and wine) and the absorbance read. For samples with a turbidity above 1 EBC, a filtration was done before using a 0.45-µm membrane filter (Whatmann, GE Healthcare, Chicago, IL, USA). The results were then expressed as follows:
where:
is absorbance at 430 nm;
is the dilution factor;
EBC is the European Brewery Convention units.
2.2.14. Dry Matter and Water (Moisture) Contents
Crushed plantain pulp (5 g) was weighed using a balance (SCIENTECH, ZSP250 MG Balance, Scientech Inc., Boulder, CO, USA) and placed in a drying dish. The dish was then placed in an oven (Heraeus, model: Kendro laboratory products, D-63450, GmbH, Hanau, Germany) previously set at 105 °C and left there till it attained constant weight. It was removed from the oven at the end of 24 h. After cooling in a desiccator, the dish was then weighed again. The dry matter content represents the difference in mass before and after drying in the oven. The dry matter (DM) content in 100 g of fresh sample was calculated using the following formula:
where:
is mass (in g) of empty drying dish;
is mass (in g) of sample before drying;
is mass (in g) of drying dish + sample after drying.
The water or moisture content (%W) was calculated using the following expression:
2.2.15. Determination of Reducing Sugars by the DNS Method [23,24]
The 3,5-Dinitrosalicylic acid (DNS) reagent was made by mixing in 5 g of dinitrosalicylic acid (Fluka Chemika, Fluka Chemie GmbH, Buchs, Switzerland) in 250 mL of distilled water at 80 °C in a water bath (Memmert, Memmert GmbH + Co., Äußere Rittersbacher Straße 38 D-91126 Schwabach, Germany). When this solution dropped to room temperature (25 °C), 100 mL of NaOH, 2 N (Fluka Chemika, Switzerland) and 150 g of sodium potassium tartrate (Fluka Chemika, Fluka Chemie GmbH, Buchs, Switzerland) were introduced, and the volume was completed with distilled water to 500 mL. The standard calibration curves were on glucose. According to the traditional method, 2 mL of dinitrosalicylic acid reagent and 0.1 mL of sample (must or wine), or distilled water (blank), were added to test tubes. The tubes were plunged in a water bath (100 °C) for 5 min and then cooled in cold water, while 7.9 mL of distilled water were introduced to each tube, resulting in the final reaction mixture. The addition of water while the tubes are plunged in cold water is performed to stop the reaction immediately. The optical density at 540 nm is read in the UV-visible spectrophotometer (JENWAY, Model: 7310, Serial No. 39756, JENWAY Limited, Staffordshire, UK).
2.2.16. Flavonoids
The flavonoid contents in the produced must and wine were determined utilizing spectrophotometric method [
25]. The sample (20 µL) was added to 2 mL of 2% AlCl
3 solution dissolved in methanol. The samples were incubated for 1 h at room temperature (25 °C). The absorbance was assessed using the spectrophotometer at
λmax = 415 nm. The same steps were repeated for the standard solution of rutin, and the calibration line was built. Based on the determined absorbance, the concentration of flavonoids was read (mg/mL) on the calibration curve; and the flavonoid contents of must and wine were expressed in rutin equivalents (µg rutin/g of extract).