The Effect of Dietary Supplementation of Green Tea Catechins on Cardiovascular Disease Risk Markers in Humans: A Systematic Review of Clinical Trials
Abstract
:1. Introduction
2. Methods/Design
2.1. Search Terminology
2.2. Selection Criteria and Data Extraction
2.3. Outcomes
2.4. Data Analysis
3. Results
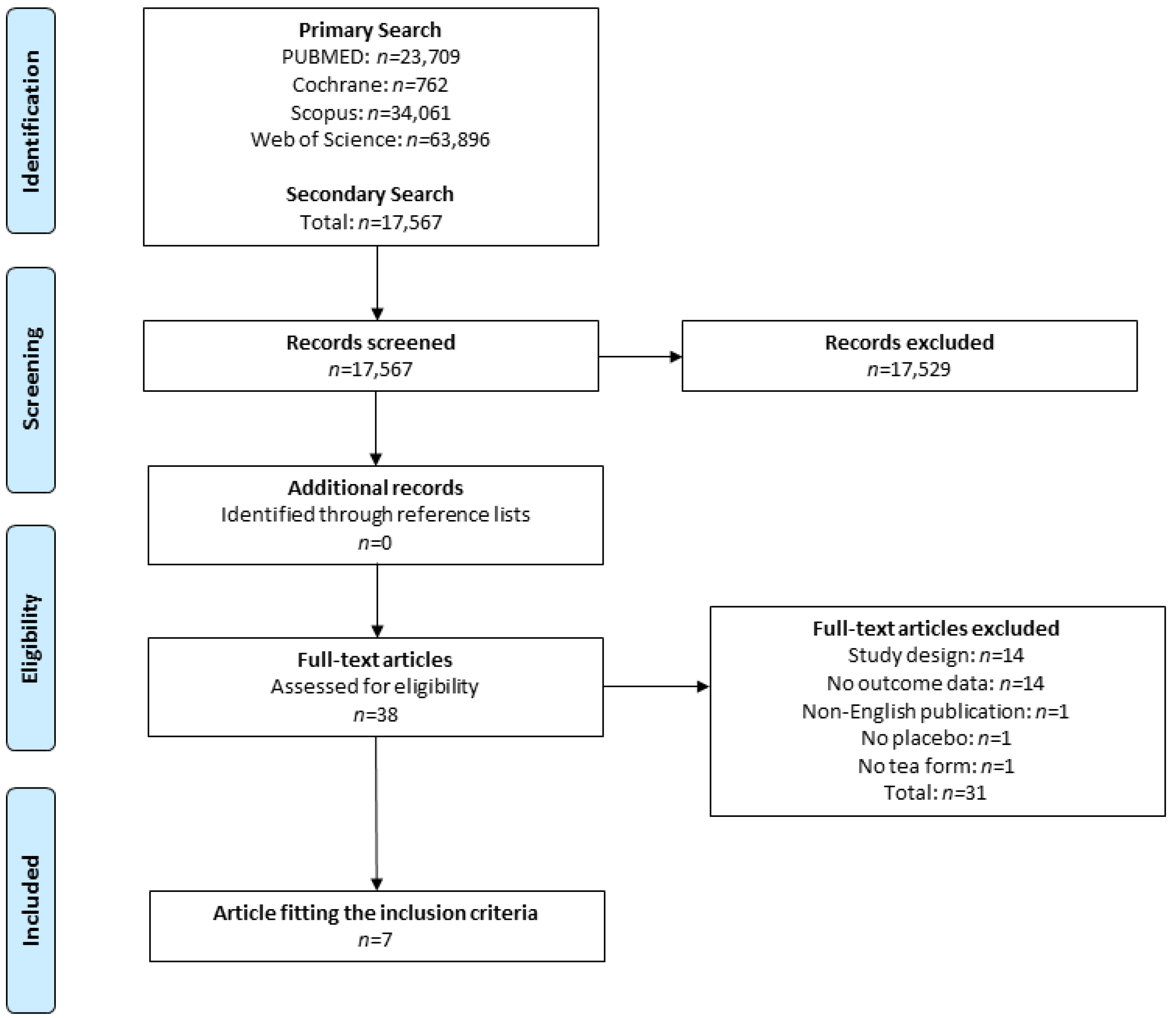
3.1. Search in Literature
3.2. Risk of Bias in Included Studies
3.3. Results of the Selected Studies
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CVD | Cardiovascular Disease |
| BP | Blood Pressure |
| LDL-C | Low Density Lipoprotein Cholesterol |
| HDL-C | High Density Lipoprotein Cholesterol |
| TC | Total Cholesterol |
| WC | Waist Circumference |
| BMI | Body Mass Index |
| GT | Green Tea |
| GTC | Green Tea Catechins |
| EGCG | Epigallocatechin Gallate |
| EGC | Epigallocatechin |
| ECG | Epicatechin Gallate |
| EC | Epicatechin |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| RIA | Immunoradiometric Assay |
| TNF-α | Tumor Necrosis Factor-α |
| CRP | C-Reactive Protein |
| SBP | Systolic Blood Pressure |
| DBP | Diastolic Blood Pressure |
| TG | Triglycerides |
| HOMA-IR | Homeostatic Model Assessment for Insulin Resistance |
| TAS | Total Antioxidant Status |
| NA | Noradrenaline |
| GLP-1 | Glucagon-Like Peptide 1 |
| wt | Weight |
References
- Basu, A.; Lucas, E.A. Mechanisms and effects of green tea on cardiovascular health. Nutr. Rev. 2007, 65, 361–375. [Google Scholar] [CrossRef] [PubMed]
- WHO. Cardiovascular Disease. Available online: http://www.who.int/cardiovascular_diseases/en/ (accessed on 20 April 2016).
- Poirier, P.; Giles, T.D.; Bray, G.A.; Hong, Y.; Stern, J.S.; Pi-Sunyer, F.X.; Eckel, R.H. Obesity and cardiovascular disease: Pathophysiology, evaluation, and effect of weight loss. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Board, J. Joint British Societies’ consensus recommendations for the prevention of cardiovascular disease (jbs3). Heart 2014, 100, ii1–ii67. [Google Scholar]
- Parikh, R.M.; Mohan, V. Changing definitions of metabolic syndrome. Indian J. Endocrinol. Metab. 2012, 16, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Joffres, M.; Falaschetti, E.; Gillespie, C.; Robitaille, C.; Loustalot, F.; Poulter, N.; McAlister, F.A.; Johansen, H.; Baclic, O.; Campbell, N. Hypertension prevalence, awareness, treatment and control in national surveys from england, the USA and Canada, and correlation with stroke and ischaemic heart disease mortality: A cross-sectional study. BMJ Open 2013, 3, e003423. [Google Scholar] [CrossRef] [PubMed]
- Nathan, D.M. Diabetes: Advances in diagnosis and treatment. JAMA 2015, 314, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Yudkin, J.S.; Stehouwer, C.; Emeis, J.; Coppack, S. C-reactive protein in healthy subjects: Associations with obesity, insulin resistance, and endothelial dysfunction a potential role for cytokines originating from adipose tissue? Arterioscler. Thromb. Vasc. Biol. 1999, 19, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Bogdanski, P.; Suliburska, J.; Szulinska, M.; Stepien, M.; Pupek-Musialik, D.; Jablecka, A. Green tea extract reduces blood pressure, inflammatory biomarkers, and oxidative stress and improves parameters associated with insulin resistance in obese, hypertensive patients. Nutr. Res. 2012, 32, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.J.; Liu, C.Y.; Chiu, J.P.; Hsu, C.H. Therapeutic effect of high-dose green tea extract on weight reduction: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. 2015, 35, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Diepvens, K.; Kovacs, E.M.; Vogels, N.; Westerterp-Plantenga, M.S. Metabolic effects of green tea and of phases of weight loss. Physiol. Behav. 2006, 87, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.H.; Liao, Y.L.; Lin, S.C.; Tsai, T.H.; Huang, C.J.; Chou, P. Does supplementation with green tea extract improve insulin resistance in obese type 2 diabetics? A randomized, double-blind, and placebo-controlled clinical trial. Altern. Med. Rev. J. Clin. Ther. 2011, 16, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.H.; Tsai, T.H.; Kao, Y.H.; Hwang, K.C.; Tseng, T.Y.; Chou, P. Effect of green tea extract on obese women: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. 2008, 27, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.Y.; Huang, C.J.; Huang, L.H.; Chen, I.J.; Chiu, J.P.; Hsu, C.H. Effects of green tea extract on insulin resistance and glucagon-like peptide 1 in patients with type 2 diabetes and lipid abnormalities: A randomized, double-blinded, and placebo-controlled trial. PLoS ONE 2014, 9, e91163. [Google Scholar] [CrossRef] [PubMed]
- Nagao, T.; Meguro, S.; Hase, T.; Otsuka, K.; Komikado, M.; Tokimitsu, I.; Yamamoto, T.; Yamamoto, K. A catechin-rich beverage improves obesity and blood glucose control in patients with type 2 diabetes. Obesity 2009, 17, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, B. Glutamate as a neurotransmitter in the brain: Review of physiology and pathology. J. Nutr. 2000, 130 (Suppl. S4), 1007S–1015S. [Google Scholar] [PubMed]
- Cabrera, C.; Artacho, R.; Gimenez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Bryant, S.; Huntley, A.L. Green tea and green tea catechin extracts: An overview of the clinical evidence. Maturitas 2012, 73, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Walchuk, C.; Suh, M.; Jones, P.J. Green tea catechins and cardiovascular disease risk factors: Should a health claim be made by the united states food and drug administration? Trends Food Sci. Technol. 2015, 41, 188–197. [Google Scholar] [CrossRef]
- Chacko, S.M.; Thambi, P.T.; Kuttan, R.; Nishigaki, I. Beneficial effects of green tea: A literature review. Chin. Med. 2010. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, S.; Shimazu, T.; Ohmori, K.; Kikuchi, N.; Nakaya, N.; Nishino, Y.; Tsubono, Y.; Tsuji, I. Green tea consumption and mortality due to cardiovascular disease, cancer, and all causes in Japan: The ohsaki study. JAMA 2006, 296, 1255–1265. [Google Scholar] [CrossRef] [PubMed]
- Keating, B.; Lindstrom, A.; Lynch, M.-E.; Blumenthal, M. Sales of tea & herbal tea increase 5.9% in united states in 2013. In 2014 Tea Sales in Track to Set a Record; HerbalEGram: Austin, TX, USA, 2014. [Google Scholar]
- Legeay, S.; Rodier, M.; Fillon, L.; Faure, S.; Clere, N. Epigallocatechin gallate: A review of its beneficial properties to prevent metabolic syndrome. Nutrients 2015, 7, 5443–5468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watson, R.R.; Preedy, V.R.; Zibadi, S. Polyphenols in Human Health and Disease; Elsevier: London, UK, 2014; p. 3. [Google Scholar]
- Naumovski, N. Bioactive composition of plants and plant foods. In Plant Bioactive Compounds for Pancreatic Cancer Prevention and Treatment; Scarlett, C.J., Vuong, Q.V., Eds.; Nova Publishers: New York, NY, USA, 2015; p. 322. [Google Scholar]
- Kim, S.; Thiessen, P.A.; Bolton, E.E.; Chen, J.; Fu, G.; Gindulyte, A.; Han, L.; He, J.; He, S.; Shoemaker, B.A.; et al. Pubchem substance and compound databases. Nucleic Acids Res. 2016, 44, D1202–D1213. [Google Scholar] [CrossRef] [PubMed]
- Hartley, L.; Flowers, N.; Holmes, J.; Clarke, A.; Stranges, S.; Hooper, L.; Rees, K. Green and black tea for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef] [Green Version]
- Engler, M.B.; Engler, M.M.; Chen, C.Y.; Malloy, M.J.; Browne, A.; Chiu, E.Y.; Kwak, H.K.; Milbury, P.; Paul, S.M.; Blumberg, J.; et al. Flavonoid-rich dark chocolate improves endothelial function and increases plasma epicatechin concentrations in healthy adults. J. Am. Coll. Nutr. 2004, 23, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Klaunig, J.E.; Xu, Y.; Han, C.; Kamendulis, L.M.; Chen, J.; Heiser, C.; Gordon, M.S.; Mohler, E.R., 3rd. The effect of tea consumption on oxidative stress in smokers and nonsmokers. Proc. Soc. Exp. Biol. Med. 1999, 220, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Princen, H.M.; van Duyvenvoorde, W.; Buytenhek, R.; Blonk, C.; Tijburg, L.B.; Langius, J.A.; Meinders, A.E.; Pijl, H. No effect of consumption of green and black tea on plasma lipid and antioxidant levels and on ldl oxidation in smokers. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Curtis, P.J.; Sampson, M.; Potter, J.; Dhatariya, K.; Kroon, P.A.; Cassidy, A. Chronic ingestion of flavan-3-ols and isoflavones improves insulin sensitivity and lipoprotein status and attenuates estimated 10-year cvd risk in medicated postmenopausal women with type 2 diabetes: A 1-year, double-blind, randomized, controlled trial. Diabetes Care 2012, 35, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Inami, S.; Takano, M.; Yamamoto, M.; Murakami, D.; Tajika, K.; Yodogawa, K.; Yokoyama, S.; Ohno, N.; Ohba, T.; Sano, J.; et al. Tea catechin consumption reduces circulating oxidized low-density lipoprotein. Int. Heart J. 2007, 48, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Mi, X.N.; Zheng, X.X.; Xu, Y.L.; Lu, J.; Huang, X.H. Effects of tea intake on blood pressure: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2014, 112, 1043–1054. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, L.P.; Knibel, M.P.; Torres, M.R.; Nogueira Neto, J.F.; Sanjuliani, A.F. Consumption of high-polyphenol dark chocolate improves endothelial function in individuals with stage 1 hypertension and excess body weight. Int. J. Hypertens. 2012. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Khanna, D. Green tea catechins: Defensive role in cardiovascular disorders. Chin. J. Nat. Med. 2013, 11, 345–353. [Google Scholar] [CrossRef]
- Nichols, M.; Townsend, N.; Scarborough, P.; Rayner, M. Cardiovascular disease in Europe 2014: Epidemiological update. Eur. Heart J. 2014, 35, 2950–2959. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Babu, P.V.; Liu, D. Green tea catechins and cardiovascular health: An update. Curr. Med. Chem. 2008, 15, 1840–1850. [Google Scholar] [CrossRef] [PubMed]
- Naumovski, N.; Blades, B.L.; Roach, P.D. Food inhibits the oral bioavailability of the major green tea antioxidant epigallocatechin gallate in humans. Antioxidants 2015, 4, 373–393. [Google Scholar] [CrossRef] [PubMed]
- Nakachi, K.; Matsuyama, S.; Miyake, S.; Suganuma, M.; Imai, K. Preventive effects of drinking green tea on cancer and cardiovascular disease: Epidemiological evidence for multiple targeting prevention. BioFactors 2000, 13, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Maki, K.C.; Reeves, M.S.; Farmer, M.; Yasunaga, K.; Matsuo, N.; Katsuragi, Y.; Komikado, M.; Tokimitsu, I.; Wilder, D.; Jones, F.; et al. Green tea catechin consumption enhances exercise-induced abdominal fat loss in overweight and obese adults. J. Nutr. 2009, 139, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Borchardt, R.T.; Huber, J.A. Catechol o-methyltransferase. 5. Structure-activity relationships for inhibition by flavonoids. J. Med. Chem. 1975, 18, 120–122. [Google Scholar] [CrossRef] [PubMed]
- Rains, T.M.; Agarwal, S.; Maki, K.C. Antiobesity effects of green tea catechins: A mechanistic review. J. Nutr. Biochem. 2011, 22, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.L.; Lane, J.; Coverly, J.; Stocks, J.; Jackson, S.; Stephen, A.; Bluck, L.; Coward, A.; Hendrickx, H. Effects of dietary supplementation with the green tea polyphenol epigallocatechin-3-gallate on insulin resistance and associated metabolic risk factors: Randomized controlled trial. Br. J. Nutr. 2009, 101, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Negishi, H.; Xu, J.W.; Ikeda, K.; Njelekela, M.; Nara, Y.; Yamori, Y. Black and green tea polyphenols attenuate blood pressure increases in stroke-prone spontaneously hypertensive rats. J. Nutr. 2004, 134, 38–42. [Google Scholar] [PubMed]
- Taubert, D.; Roesen, R.; Schomig, E. Effect of cocoa and tea intake on blood pressure: A meta-analysis. Arch. Int. Med. 2007, 167, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Osada, K.; Takahashi, M.; Hoshina, S.; Nakamura, M.; Nakamura, S.; Sugano, M. Tea catechins inhibit cholesterol oxidation accompanying oxidation of low density lipoprotein in vitro. Comp. Biochem. Physiol. Toxicol. Pharmacol. 2001, 128, 153–164. [Google Scholar] [CrossRef]
- Yang, T.T.; Koo, M.W. Hypocholesterolemic effects of chinese tea. Pharmacol. Res. 1997, 35, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Maron, D.J.; Lu, G.P.; Cai, N.S.; Wu, Z.G.; Li, Y.H.; Chen, H.; Zhu, J.Q.; Jin, X.J.; Wouters, B.C.; Zhao, J. Cholesterol-lowering effect of a theaflavin-enriched green tea extract: A randomized controlled trial. Arch. Int. Med. 2003, 163, 1448–1453. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.A.; Abbey, M.; Roach, P.D. A green tea extract lowers plasma cholesterol by inhibiting cholesterol synthesis and upregulating the ldl receptor in the cholesterol-fed rabbit. Atherosclerosis 2007, 193, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.A.; Roach, P.D. A green tea catechin extract upregulates the hepatic low-density lipoprotein receptor in rats. Lipids 2007, 42, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.T.; Fong, W.P.; Cheung, Y.L.; Huang, Y.; Ho, W.K.; Chen, Z.Y. Jasmine green tea epicatechins are hypolipidemic in hamsters (mesocricetus auratus) fed a high fat diet. J. Nutr. 1999, 129, 1094–1101. [Google Scholar] [PubMed]
- Friedrich, M.; Petzke, K.J.; Raederstorff, D.; Wolfram, S.; Klaus, S. Acute effects of epigallocatechin gallate from green tea on oxidation and tissue incorporation of dietary lipids in mice fed a high-fat diet. Int. J. Obes. 2012, 36, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, N.; Yamda, N.; Mori, M. Powdered green tea has antilipogenic effect on zucker rats fed a high-fat diet. Phytother. Res. 2003, 17, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Grove, K.A.; Sae-tan, S.; Kennett, M.J.; Lambert, J.D. (−)-Epigallocatechin-3-gallate inhibits pancreatic lipase and reduces body weight gain in high fat-fed obese mice. Obesity 2012, 20, 2311–2313. [Google Scholar] [CrossRef] [PubMed]
- Long, A.N.; Dagogo-Jack, S. Comorbidities of diabetes and hypertension: Mechanisms and approach to target organ protection. J. Clin. Hypertens. 2011, 13, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka-Tojo, M.; Tojo, T.; Takahira, N.; Matsunaga, A.; Aoyama, N.; Masuda, T.; Izumi, T. Elevated circulating levels of an incretin hormone, glucagon-like peptide-1, are associated with metabolic components in high-risk patients with cardiovascular disease. Cardiovasc. Diabetol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.; Roach, P.D.; Bottema, C.D.; Pal, S. Green tea upregulates the low-density lipoprotein receptor through the sterol-regulated element binding protein in hepg2 liver cells. J. Agric. Food Chem. 2001, 49, 5639–5645. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.A.; Roach, P.D. Modulation of cholesterol metabolism by the green tea polyphenol (−)-epigallocatechin gallate in cultured human liver (hepg2) cells. J. Agric. Food Chem. 2006, 54, 1621–1626. [Google Scholar] [CrossRef] [PubMed]
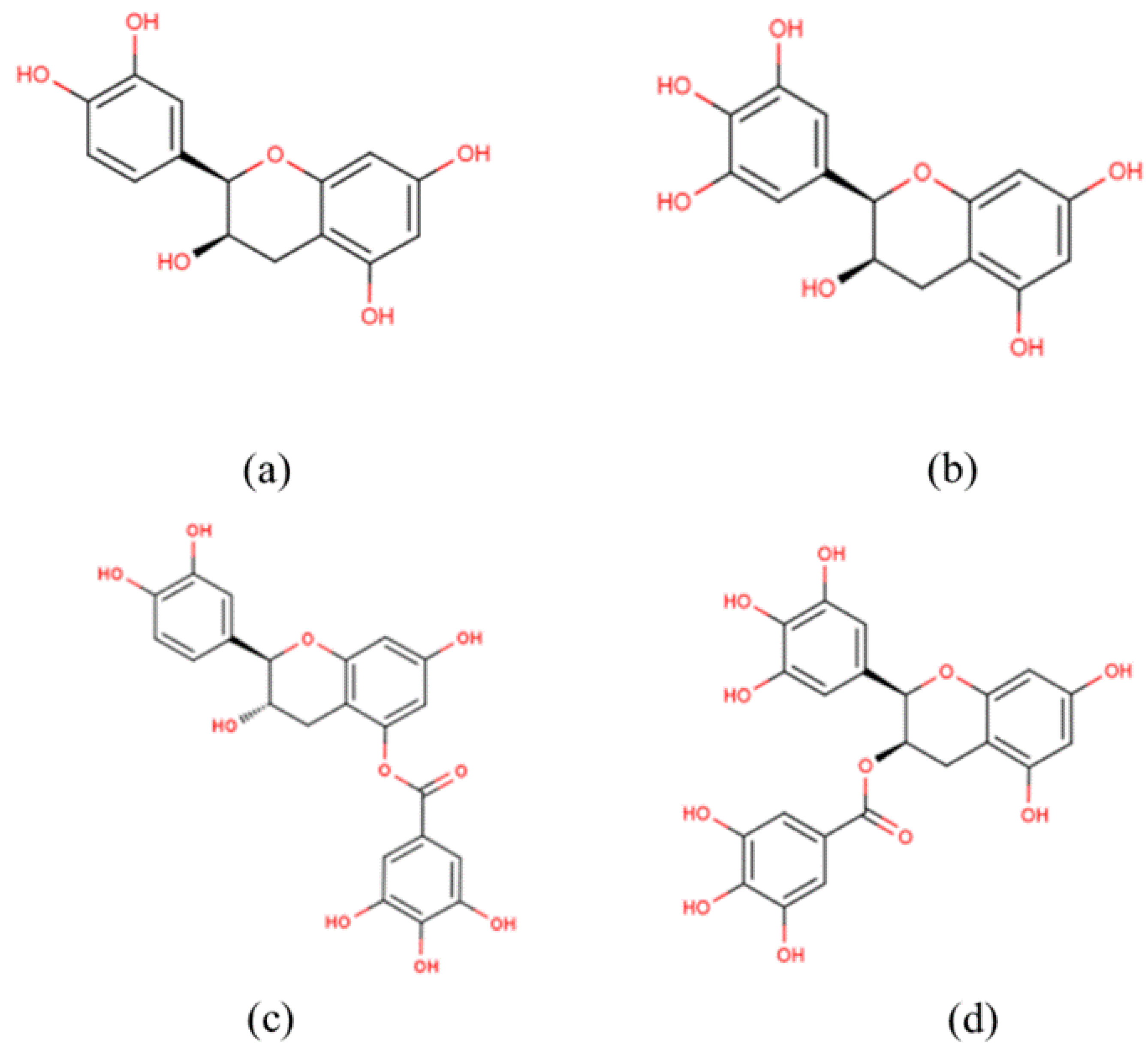
| Author (Year) | Participants, Sample Size | Aim | Intervention | Outcomes (Treatment Group vs. Placebo) |
|---|---|---|---|---|
| Bogdanski et al. (2012) [9] | All (n = 56) (28 Male and 28 Female, 30–60 years old) Age: 50.4 ± 8.1 years Treatment (n = 28), Placebo (n = 28) Obese, hypertensive adults | Effect of GT supplementation on insulin resistance and associated cardiovascular risk factors in obese, hypertensive subjects. | Treatment (capsules 379 mg GTE/day) or placebo for three months. Total GTCs = n.r. EGCG = 208 mg/day | A: (↔BMI, WC: p > 0.05) ↓BP (SBP: p = 0.04, DBP: p < 0.001) Biochem: (↔TC: p = 0.09, ↓LDL-C: p = 0.011, ↑HDL-C: p < 0.01, ↓TG: p = 0.004) |
| Chen et al. (2015) [10] | Female only (n = 77, 20–60 years old) Treatment (n = 39) Placebo (n = 38), Age: 44.5 ± 11.5 years) abdominal obesity with BMI ≥ 27 kg/m2 and WC ≥ 80 cm | Effect and safety of high-dose GTE on weight reduction and changes of lipid profile in women with abdominal obesity. | Treatment (opaque capsules 1500 mg GTE/day) or placebo for 12 weeks. Total GTCs = 1344 mg EGCG = 856.8 mg, ECG = 236.1 mg EGC = 115.5 mg, EC = 71.9 mg, GCG = 637 mg | A: (↓wt: p = 0.025, ↓BMI: p = 0.018, ↓WC: p = 0.023) ↔BP (SBP: p = 0.795, DBP: p = 0.929) Biochem: (↓TC: p = 0.005, ↓LDL-C: p = 0.006, ↔HDL-C: p = 0.208, ↔TG: p = 0.778) |
| Diepvens et al. (2006) [11] | Female only (n = 46 19–57 years old) Age: 41.6 ± 9.3 years, Treatment (n = 23) Placebo (n = 23), Overweight with 25 kg/m2 <BMI> 35 kg/m2 | Effect of GTE ingestion with a low-energy diet on health-related blood parameters and its relationships with changes in metabolic parameters and weight loss. | Treatment (capsules 1206.9 mg GTE/day) or placebo for 12.4 weeks. Total GTCs = 522 mg, EGCG = 264.8 mg ECG = 94.4 mg, EGC = 106.8 mg EC = 56 mg, GCG = n.r. | A: (↓wt, BMI, WC: p < 0.01) ↓BP (SBP: p < 0.01, DBP: p < 0.05) Biochem: (↓TC: p < 0.05, ↓LDL-C: p > 0.05, ↓HDL-C: p < 0.05, ↔TG: p > 0.05) * no direct test between groups |
| Hsu et al. (2011) [12] | All (n = 68, 20–65 years old) (24 Male and 44 Female), Age: 51.3 ± 9.1 years Obese; type 2 diabetes Treatment (n = 35), Placebo (n = 33) | Effects of decaffeinated GTE on obese individuals with type 2 diabetes. | Treatment decaffeinated GTE (opaque capsules 1500 mg GTE/day) or placebo for 16 weeks. Total GTCs = 1344 mg, EGCG = 856.8 mg, ECG = 236.1 mg, EGC = 115.5 mg EC = 71.9 mg, GCG = 637mg | A:(↔BMI: p = 0.43, ↔WC: p = 0.06) ↔BP (SBP: p = 0.68, DBP: p = 0.86) Biochem: ↔ (TC: p = 0.39, LDL-C: p = 0.24, HDL-C: p = 0.78, TG: p = 0.66) |
| Hsu et al. (2008) [13] | Female only (n = 78, 16–60 years old) Age: 43.5 ± 11.8 years, Treatment (n = 41) Placebo (n = 37), Obese (BMI > 27 kg/m2) | Effects of GTE on obese women and obesity-related peptide hormones. | Treatment (opaque capsules 400 mg GTE/day) or placebo for 12 weeks. Total GTCs = 605.26 mg EGCG = 377.2 mg, ECG = 31.76 mg EGC = 36.91 mg, GC = 61.58 mg, GCG = 27.48 mg | A: (↔wt: p > 0.05, ↔BMI: p > 0.05, ↓WC: p < 0.05) ↔BP (SBP, DBP: p > 0.05) Biochem (↔TC: p > 0.05, ↓LDL-C: p < 0.001, ↔HDL-C: p > 0.05, ↓TG: p < 0.001) |
| Liu et al. (2014) [14] | All (n = 77, 20–65 years old) (32 Male and 45 Female), Age: 54.3 ± 6.8 years Treatment (n = 39), Placebo (n = 38) Dyslipidaemic adults with type 2 diabetes | Effects of GTE on lipid profiles in participants with type 2 diabetes and dyslipidaemia. | Treatment (capsules 1500 mg GTE/day) or placebo for 16 weeks. Total GTCs = 1344 mg EGCG = 856.8 mg, ECG = 236.1 mg EGC = 115.5 mg, EC = 71.9 mg, GCG = 637 mg | A: ↔ (BMI: p = 0.06, WC: p = 0.17, wt: p = 0.09) ↔BP (SBP: p = 0.48, DBP: p = 0.68) Biochem: (↔TC: p = 0.67, ↔LDL-C: p = 0.55, ↑HDL-C: p = 0.04, ↓TG: p = 0.03) |
| Nagao et al. (2009) [15] | All (n = 43) (18 Male and 25 Female, < 65 years old) Treatment (n = 23) Control (n = 20), Age: 63.8 ± 1.9 years Type 2 diabetics | Effects of continuous ingestion of a catechin-rich beverage in type 2 diabetes who were not receiving insulin therapy. | Treatment (beverage 582.8 mg GTCs/day) or Control (96.3 mg GTCs/day) for 12 weeks. Total GTC = 582.8 Individual = n.r. | A: (↔BMI: p > 0.05, ↔wt: p > 0.05, ↓WC: p < 0.05) ↔BP (SBP, DBP: p > 0.05) Biochem: (↓TC: p < 0.05, ↔LDL-C: p > 0.05, ↔HDL-C: p > 0.05, ↔TG: p > 0.05) |
| Bias Category | Bogdanski et al. (2012) [9] | Chen et al. (2015) [10] | Diepvens et al. (2006) [11] | Hsu et al. (2008) [13] | Hsu et al. (2011) [12] | Liu et al. (2014) [14] | Nagao et al. (2009) [15] |
|---|---|---|---|---|---|---|---|
| Random Sequence Generation (selection bias) | Low | Low | Moderate | Low | Low | Moderate | Moderate |
| Blinding of participants and personnel (performance bias). | Low | Low | Low | Low | Low | Low | Low |
| Blinding of outcome assessment (detection bias) (mortality) | Unclear | Unclear | Unclear | Unclear | Unclear | Unclear | Unclear |
| Incomplete data addressed (attrition bias) | Low | Low | Unclear | Moderate | Low | Low | Low |
| Selective reporting (reporting bias) | Low | Low | Low | Low | Low | Low | Low |
| Other bias | Low | Low | Low | Low | Low | Low | Low |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lau, S.O.; Georgousopoulou, E.N.; Kellett, J.; Thomas, J.; McKune, A.; Mellor, D.; Roach, P.D.; Naumovski, N. The Effect of Dietary Supplementation of Green Tea Catechins on Cardiovascular Disease Risk Markers in Humans: A Systematic Review of Clinical Trials. Beverages 2016, 2, 16. https://doi.org/10.3390/beverages2020016
Lau SO, Georgousopoulou EN, Kellett J, Thomas J, McKune A, Mellor D, Roach PD, Naumovski N. The Effect of Dietary Supplementation of Green Tea Catechins on Cardiovascular Disease Risk Markers in Humans: A Systematic Review of Clinical Trials. Beverages. 2016; 2(2):16. https://doi.org/10.3390/beverages2020016
Chicago/Turabian StyleLau, Sarah O., Ekavi N. Georgousopoulou, Jane Kellett, Jackson Thomas, Andrew McKune, Duane Mellor, Paul D. Roach, and Nenad Naumovski. 2016. "The Effect of Dietary Supplementation of Green Tea Catechins on Cardiovascular Disease Risk Markers in Humans: A Systematic Review of Clinical Trials" Beverages 2, no. 2: 16. https://doi.org/10.3390/beverages2020016
APA StyleLau, S. O., Georgousopoulou, E. N., Kellett, J., Thomas, J., McKune, A., Mellor, D., Roach, P. D., & Naumovski, N. (2016). The Effect of Dietary Supplementation of Green Tea Catechins on Cardiovascular Disease Risk Markers in Humans: A Systematic Review of Clinical Trials. Beverages, 2(2), 16. https://doi.org/10.3390/beverages2020016








