Effect of Different Desensitizers on Shear Bond Strength of Self-Adhesive Resin Cements to Dentin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
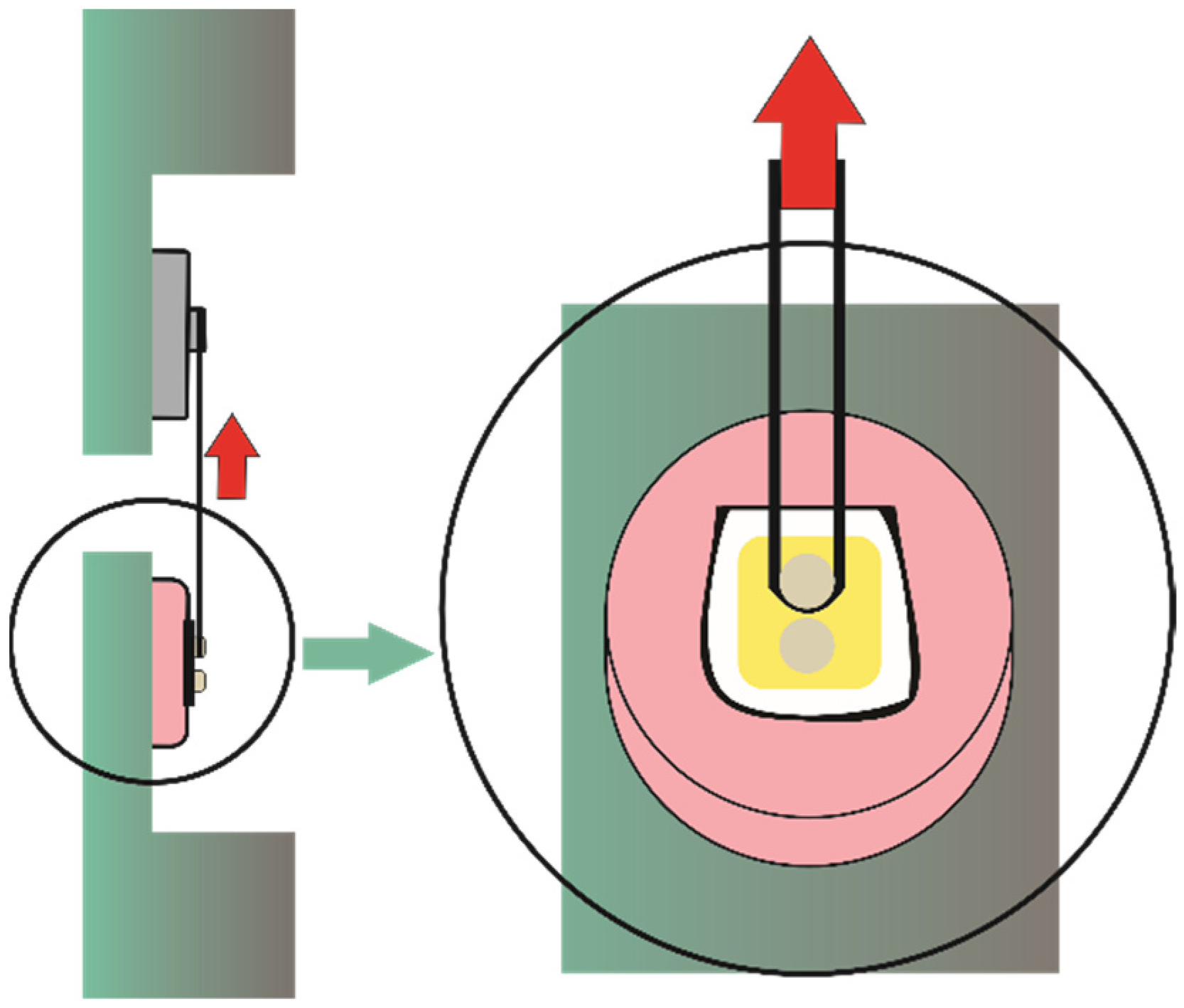
2.2. Specimen Preparation
2.3. Shear Bond Strength Test
2.4. Contact Angle
2.5. Statistical Analysis
3. Results
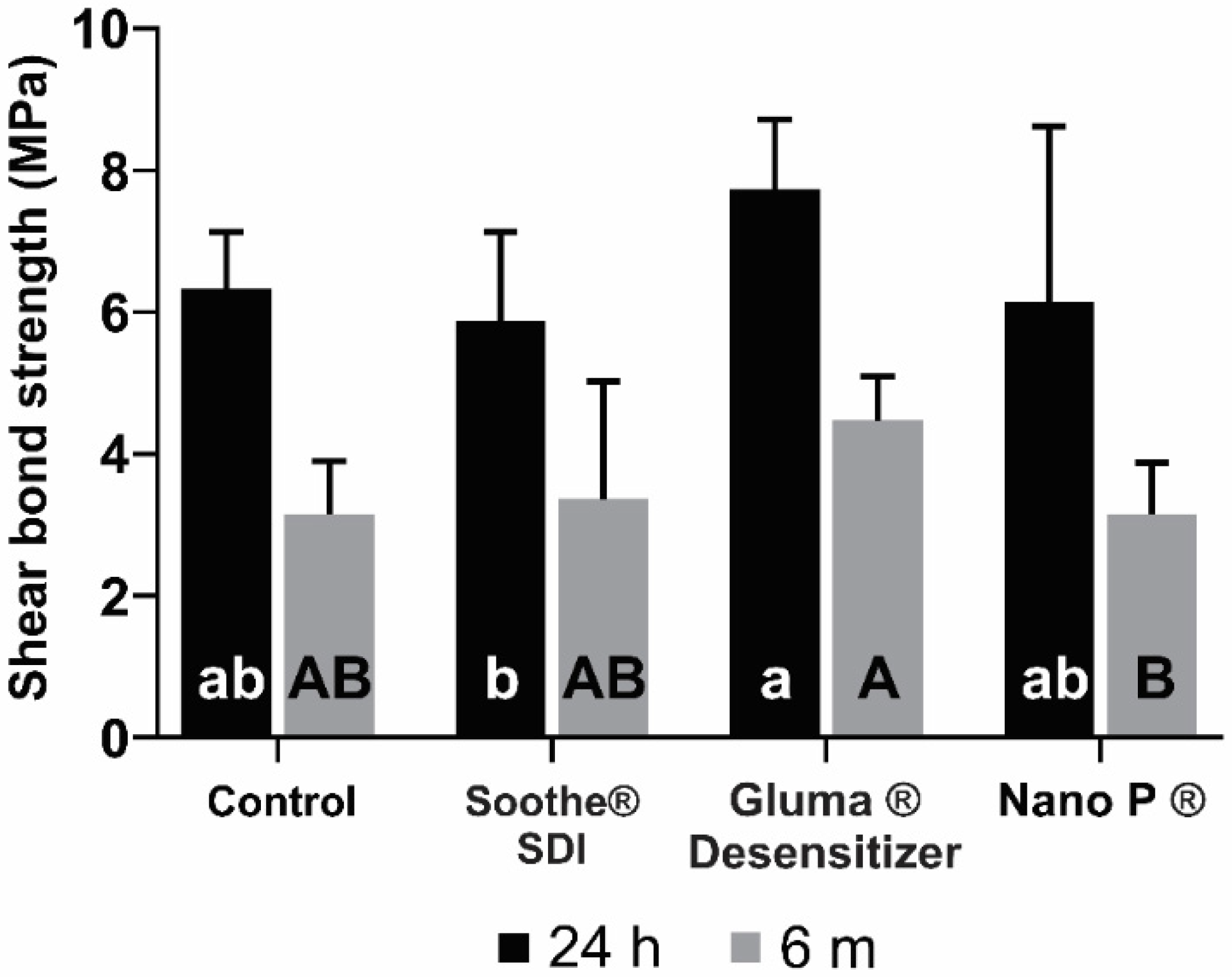
3.1. Shear Bond Strength and Failure Mode
3.2. Contact Angle
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Demirtag, Z.; Uzgur, R.; Turkal, M.; Uzgur, Z.; Colak, H.; Özcan, M. A Survey on Prevalence, Causes and Prevention of Postcementation Hypersensitivity. Eur. J. Prosthodont. Restor. Dent. 2016, 24, 158–163. [Google Scholar] [PubMed]
- Liu, X.-X.; Tenenbaum, H.C.; Wilder, R.S.; Quock, R.; Hewlett, E.R.; Ren, Y.-F. Pathogenesis, Diagnosis and Management of Dentin Hypersensitivity: An Evidence-Based Overview for Dental Practitioners. BMC Oral Health 2020, 20, 220. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Park, J.-C. Dentin Hypersensitivity and Emerging Concepts for Treatments. J. Oral Biosci. 2017, 59, 211–217. [Google Scholar] [CrossRef]
- Brännström, M. The Hydrodynamic Theory of Dentinal Pain: Sensation in Preparations, Caries, and the Dentinal Crack Syndrome. J. Endod. 1986, 12, 453–457. [Google Scholar] [CrossRef]
- Orsini, G.; Procaccini, M.; Manzoli, L.; Giuliodori, F.; Lorenzini, A.; Putignano, A. A double-blind randomized-controlled trial comparing the desensitizing efficacy of a new dentifrice containing carbonate/hydroxyapatite nanocrystals and a sodium fluoride/potassium nitrate dentifrice. J. Clin. Periodontol. 2010, 37, 510–517. [Google Scholar] [CrossRef]
- Marto, C.M.; Baptista Paula, A.; Nunes, T.; Pimenta, M.; Abrantes, A.M.; Pires, A.S.; Laranjo, M.; Coelho, A.; Donato, H.; Botelho, M.F. Evaluation of the Efficacy of Dentin Hypersensitivity Treatments—A Systematic Review and Follow-up Analysis. J. Oral Rehabil. 2019, 46, 952–990. [Google Scholar] [CrossRef]
- Douglas-de-Oliveira, D.W.; Vitor, G.P.; Silveira, J.O.; Martins, C.C.; Costa, F.O.; Cota, L.O.M. Effect of Dentin Hypersensitivity Treatment on Oral Health Related Quality of Life—A Systematic Review and Meta-Analysis. J. Dent. 2018, 71, 1–8. [Google Scholar] [CrossRef]
- Moraschini, V.; da Costa, L.S.; Dos Santos, G.O. Effectiveness for Dentin Hypersensitivity Treatment of Non-Carious Cervical Lesions: A Meta-Analysis. Clin. Oral Investig. 2018, 22, 617–631. [Google Scholar] [CrossRef]
- Kielbassa, A.M.; Attin, T.; Hellwig, E.; Schade-Brittinger, C. In Vivo Study on the Effectiveness of a Lacquer Containing CaF2/NaF in Treating Dentine Hypersensitivity. Clin. Oral Investig. 1997, 1, 95–99. [Google Scholar] [CrossRef]
- Yilmaz, H.G.; Kurtulmus-Yilmaz, S.; Cengiz, E. Long-Term Effect of Diode Laser Irradiation Compared to Sodium Fluoride Varnish in the Treatment of Dentine Hypersensitivity in Periodontal Maintenance Patients: A Randomized Controlled Clinical Study. Photomed. Laser Surg. 2011, 29, 721–725. [Google Scholar] [CrossRef]
- Hardan, L.; Devoto, W.; Bourgi, R.; Cuevas-Suárez, C.E.; Lukomska-Szymanska, M.; Fernández-Barrera, M.Á.; Cornejo-Ríos, E.; Monteiro, P.; Zarow, M.; Jakubowicz, N.; et al. Immediate Dentin Sealing for Adhesive Cementation of Indirect Restorations: A Systematic Review and Meta-Analysis. Gels 2022, 8, 175. [Google Scholar] [CrossRef] [PubMed]
- Monticelli, F.; Osorio, R.; Mazzitelli, C.; Ferrari, M.; Toledano, M. Limited Decalcification/Diffusion of Self-Adhesive Cements into Dentin. J. Dent. Res. 2008, 87, 974–979. [Google Scholar] [CrossRef] [PubMed]
- Pedreira, A.P.R.d.V.; D’Alpino, P.H.P.; Pereira, P.N.R.; Chaves, S.B.; Wang, L.; Hilgert, L.; Garcia, F.C.P. Effects of the Application Techniques of Self-Adhesive Resin Cements on the Interfacial Integrity and Bond Strength of Fiber Posts to Dentin. J. Appl. Oral Sci. 2016, 24, 437–446. [Google Scholar] [CrossRef]
- Ferracane, J.L.; Stansbury, J.; Burke, F.J.T. Self-adhesive Resin Cements–Chemistry, Properties and Clinical Considerations. J. Oral Rehabil. 2011, 38, 295–314. [Google Scholar] [CrossRef] [PubMed]
- Gerth, H.U.; Dammaschke, T.; Züchner, H.; Schäfer, E. Chemical Analysis and Bonding Reaction of RelyX Unicem and Bifix Composites—A Comparative Study. Dent. Mater. 2006, 22, 934–941. [Google Scholar] [CrossRef]
- Radovic, I.; Monticelli, F.; Goracci, C.; Vulicevic, Z.R.; Ferrari, M. Self-Adhesive Resin Cements: A Literature Review. J. Adhes. Dent. 2008, 10, 251–258. [Google Scholar]
- ISO/TS 11405; Dental Materials—Testing of Adhesion to Tooth Structure. International Organization for Standardization: London, UK, 2003.
- Külünk, Ş.; Sarac, D.; Külünk, T.; Karakaş, Ö. The Effects of Different Desensitizing Agents on the Shear Bond Strength of Adhesive Resin Cement to Dentin. J. Esthet. Restor. Dent. 2011, 23, 380–387. [Google Scholar] [CrossRef]
- Armstrong, S.; Breschi, L.; Özcan, M.; Pfefferkorn, F.; Ferrari, M.; Van Meerbeek, B. Academy of Dental Materials Guidance on in Vitro Testing of Dental Composite Bonding Effectiveness to Dentin/Enamel Using Micro-Tensile Bond Strength (ΜTBS) Approach. Dent. Mater. 2017, 33, 133–143. [Google Scholar] [CrossRef]
- Tay, F.R.; Pashley, D.H. Resin Bonding to Cervical Sclerotic Dentin: A Review. J. Dent. 2004, 32, 173–196. [Google Scholar] [CrossRef]
- Tay, F.R.; Nawareg, M.A.; Abuelenain, D.; Pashley, D.H. Cervical Sclerotic Dentin: Resin Bonding. In Understanding Dental Caries; Goldberg, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 97–125. [Google Scholar]
- Tan, Y.; Gu, M.; Li, W.; Guo, L. Effect of a Filled Adhesive as the Desensitizer on Bond Strength of “Self-Adhesive Cements to” Differently Severity of Fluorosed Dentin. Microsc. Res. Tech. 2018, 81, 805–815. [Google Scholar] [CrossRef]
- Uğur, M.; Altıntaş, S.H. Evaluation of Different Desensitizing Agents Effect on Shear Bond Strength of Adhesive Resin Cement to Dentin. J. Adhes. Sci. Technol. 2019, 33, 1695–1704. [Google Scholar] [CrossRef]
- Sailer, I.; Oendra, A.E.H.; Stawarczyk, B.; Hämmerle, C.H. The Effects of Desensitizing Resin, Resin Sealing, and Provisional Cement on the Bond Strength of Dentin Luted with Self-Adhesive and Conventional Resincements. J. Prosthet. Dent. 2012, 107, 252–260. [Google Scholar] [CrossRef]
- Stawarczyk, B.; Hartmann, R.; Hartmann, L.; Roos, M.; Özcan, M.; Sailer, I.; Hämmerle, C.H. The Effect of Dentin Desensitizer on Shear Bond Strength of Conventional and Self-Adhesive Resin Luting Cements after Aging. Oper. Dent. 2011, 36, 492–501. [Google Scholar] [CrossRef]
- Acar, O.; Tuncer, D.; Yuzugullu, B.; Celik, C. The Effect of Dentin Desensitizers and Nd: YAG Laser Pre-Treatment on Microtensile Bond Strength of Self-Adhesive Resin Cement to Dentin. J. Adv. Prosthodont. 2014, 6, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Dewan, H.; Sayed, M.E.; Alqahtani, N.M.; Alnajai, T.; Qasir, A.; Chohan, H. The Effect of Commercially Available Desensitizers on Bond Strength Following Cementation of Zirconia Crowns Using Self-Adhesive Resin Cement—An in Vitro Study. Materials 2022, 15, 514. [Google Scholar] [CrossRef] [PubMed]
- Huh, J.-B.; Kim, J.-H.; Chung, M.-K.; Lee, H.; Choi, Y.-G.; Shim, J.-S. The Effect of Several Dentin Desensitizers on Shear Bond Strength of Adhesive Resin Luting Cement Using Self-Etching Primer. J. Dent. 2008, 36, 1025–1032. [Google Scholar] [CrossRef]
- Schupbach, P.; Lutz, F.; Finger, W. Closing of Dentinal Tubules by Gluma Desensitizer. Eur. J. Oral Sci. 1997, 105, 414–421. [Google Scholar] [CrossRef]
- Garcia, R.N.; Giannini, M.; Takagaki, T.; Sato, T.; Matsui, N.; Nikaido, T.; Tagami, J. Effect of Dentin Desensitizers on Resin Cement Bond Strengths. RSBO 2015, 12, 14–22. [Google Scholar] [CrossRef]
- Lawaf, S.; Jalalian, E.; Roshan, R.; Azizi, A. Effect of GLUMA Desensitizer on the Retention of Full Metal Crowns Cemented with Rely X U200 Self-Adhesive Cement. J. Adv. Prosthodont. 2016, 8, 404–410. [Google Scholar] [CrossRef]
- Kolker, J.L.; Vargas, M.A.; Armstrong, S.R.; Dawson, D.V. Effect of Desensitizing Agents on Dentin Permeability and Dentin Tubule Occlusion. J. Adhes. Dent. 2002, 4, 211–222. [Google Scholar]
- Nakamoto, A.; Sato, T.; Matsui, N.; Ikeda, M.; Nikaido, T.; Burrow, M.F.; Tagami, J. Effect of Fluoride Mouthrinse and Fluoride Concentration on Bonding of a One-Step Self-Etch Adhesive to Bovine Root Dentin. J. Oral Sci. 2019, 61, 125–132. [Google Scholar] [CrossRef] [PubMed]
- De Munck, J.; Vargas, M.; Van Landuyt, K.; Hikita, K.; Lambrechts, P.; Van Meerbeek, B. Bonding of an Auto-Adhesive Luting Material to Enamel and Dentin. Dent. Mater. 2004, 20, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Ferracane, J.L. Hygroscopic and Hydrolytic Effects in Dental Polymer Networks. Dent. Mater. 2006, 22, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M. A Review—Micromorphological Evidence of Degradation in Resin-dentin Bonds and Potential Preventional Solutions. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 92, 268–280. [Google Scholar] [CrossRef]
- Takenaka, H.; Ouchi, H.; Sai, K.; Kawamoto, R.; Murayama, R.; Kurokawa, H.; Miyazaki, M. Ultrasonic Measurement of the Effects of Light Irradiation and Presence of Water on the Polymerization of Self-adhesive Resin Cement. Eur. J. Oral Sci. 2015, 123, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Piwowarczyk, A.; Lauer, H.-C. Mechanical Properties of Luting Cements after Water Storage. Oper. Dent. Univ. Wash. 2003, 28, 535–542. [Google Scholar]
- Piwowarczyk, A.; Bender, R.; Ottl, P.; Lauer, H.-C. Long-Term Bond between Dual-Polymerizing Cementing Agents and Human Hard Dental Tissue. Dent. Mater. 2007, 23, 211–217. [Google Scholar] [CrossRef]
- Mendes, L.C.; Matos, I.C.; Miranda, M.S.; Benzi, M.R. Dual-Curing, Self-Adhesive Resin Cement: Influence of the Polymerization Modes on the Degree of Conversion and Microhardness. Mater. Res. 2010, 13, 171–176. [Google Scholar] [CrossRef]
- Hikita, K.; Van Meerbeek, B.; De Munck, J.; Ikeda, T.; Van Landuyt, K.; Maida, T.; Lambrechts, P.; Peumans, M. Bonding Effectiveness of Adhesive Luting Agents to Enamel and Dentin. Dent. Mater. 2007, 23, 71–80. [Google Scholar] [CrossRef]
- Mecholsky Jr, J.J. Modes of Failure of Bonding Interfaces in Dentistry. Fractography Glasses Ceram. VI 2012, 230, 193. [Google Scholar]
- Armstrong, S.; Keller, J.; Boyer, D. Mode of Failure in the Dentin-Adhesive Resin–Resin Composite Bonded Joint as Determined by Strength-Based (ΜTBS) and Fracture-Based (CNSB) Mechanical Testing. Dent. Mater. 2001, 17, 201–210. [Google Scholar] [CrossRef]
- Lührs, A.-K.; Guhr, S.; Günay, H.; Geurtsen, W. Shear Bond Strength of Self-Adhesive Resins Compared to Resin Cements with Etch and Rinse Adhesives to Enamel and Dentin in Vitro. Clin. Oral Investig. 2010, 14, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Wege, H.A.; Aguilar, J.A.; Rodríguez-Valverde, M.Á.; Toledano, M.; Osorio, R.; Cabrerizo-Vílchez, M.Á. Dynamic Contact Angle and Spreading Rate Measurements for the Characterization of the Effect of Dentin Surface Treatments. J. Colloid Interface Sci. 2003, 263, 162–169. [Google Scholar] [CrossRef]
- Asmussen, E.; Peutzfeldt, A.; Sahafi, A. Bonding of Resin Cements to Post Materials: Influence of Surface Energy Characteristics. J. Adhes. Dent. 2005, 7, 231–234. [Google Scholar] [PubMed]
- Al-Saleh, M.; El-Mowafy, O.; Tam, L.; Fenton, A. Microleakage of Posterior Composite Restorations Lined with Self-Adhesive Resin Cements. Oper. Dent. 2010, 35, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Hardan, L.; Bourgi, R.; Kharouf, N.; Mancino, D.; Zarow, M.; Jakubowicz, N.; Haikel, Y.; Cuevas-Suárez, C.E. Bond Strength of Universal Adhesives to Dentin: A Systematic Review and Meta-Analysis. Polymers 2021, 13, 814. [Google Scholar] [CrossRef]



| Study Group | Desensitizer Agent | Chemical Composition * | Application Protocol |
|---|---|---|---|
| Gluma | Gluma® Desensitizer (Kulzer, Hanau, Germany) | (2-hydroxyethyl) methacrylate glutardialdehyde, purified water | Apply to the dentin for 60 s with a microbrush and dry with dry air until it disappears (observe a non-shiny surface) and then wash with water |
| Nano P | Nano P® (FGM, Joinville, Brazil) | Potassium nitrate and sodium fluoride | Apply with microbrush on the dentin surface, rub the product with a rubber cup for 10 s, leave the product to rest for 5 min and finally remove the excess with a cotton pad |
| Soothe | Soothe® (SDI, Victoria, Australia) | 6% potassium nitrate and 0.1% fluoride gel | Apply on the surface for 2 min |
| Cement used: Rely X U200 | Base paste: Methacrylate monomers containing phosphoric acid groups, methacrylate monomers, silanated fillers, initiator components, stabilizers, rheological additives Catalyst: Methacrylate monomers, Alkaline (basic) fillers, Silanated fillers, Initiator components, Stabilizers, Pigments, Rheological additives | Mix base paste and catalyst paste into a homogenous paste within 20 s. Spread cement within the restoration and apply moderate pressure | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elizalde-Hernández, A.; Hardan, L.; Bourgi, R.; Isolan, C.P.; Moreira, A.G.; Zamarripa-Calderón, J.E.; Piva, E.; Cuevas-Suárez, C.E.; Devoto, W.; Saad, A.; et al. Effect of Different Desensitizers on Shear Bond Strength of Self-Adhesive Resin Cements to Dentin. Bioengineering 2022, 9, 372. https://doi.org/10.3390/bioengineering9080372
Elizalde-Hernández A, Hardan L, Bourgi R, Isolan CP, Moreira AG, Zamarripa-Calderón JE, Piva E, Cuevas-Suárez CE, Devoto W, Saad A, et al. Effect of Different Desensitizers on Shear Bond Strength of Self-Adhesive Resin Cements to Dentin. Bioengineering. 2022; 9(8):372. https://doi.org/10.3390/bioengineering9080372
Chicago/Turabian StyleElizalde-Hernández, Alejandro, Louis Hardan, Rim Bourgi, Cristina Pereira Isolan, Andressa Goicochea Moreira, J. Eliezer Zamarripa-Calderón, Evandro Piva, Carlos Enrique Cuevas-Suárez, Walter Devoto, Ahmed Saad, and et al. 2022. "Effect of Different Desensitizers on Shear Bond Strength of Self-Adhesive Resin Cements to Dentin" Bioengineering 9, no. 8: 372. https://doi.org/10.3390/bioengineering9080372
APA StyleElizalde-Hernández, A., Hardan, L., Bourgi, R., Isolan, C. P., Moreira, A. G., Zamarripa-Calderón, J. E., Piva, E., Cuevas-Suárez, C. E., Devoto, W., Saad, A., Proc, P., & Lukomska-Szymanska, M. (2022). Effect of Different Desensitizers on Shear Bond Strength of Self-Adhesive Resin Cements to Dentin. Bioengineering, 9(8), 372. https://doi.org/10.3390/bioengineering9080372











