Postharvest Operations of Cannabis and Their Effect on Cannabinoid Content: A Review
Abstract
:1. Introduction
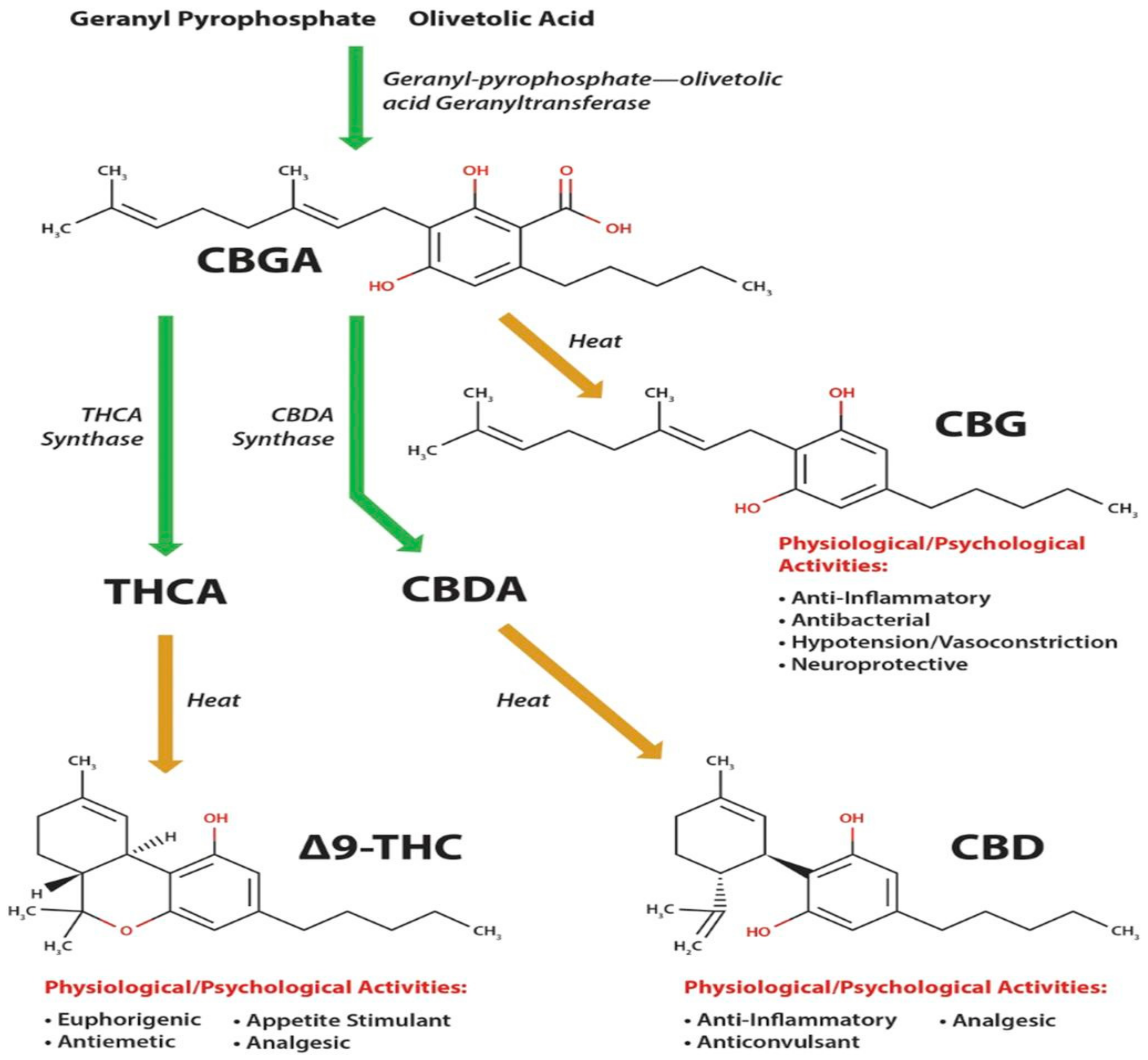
2. Phytocannabinoids and Decarboxylation
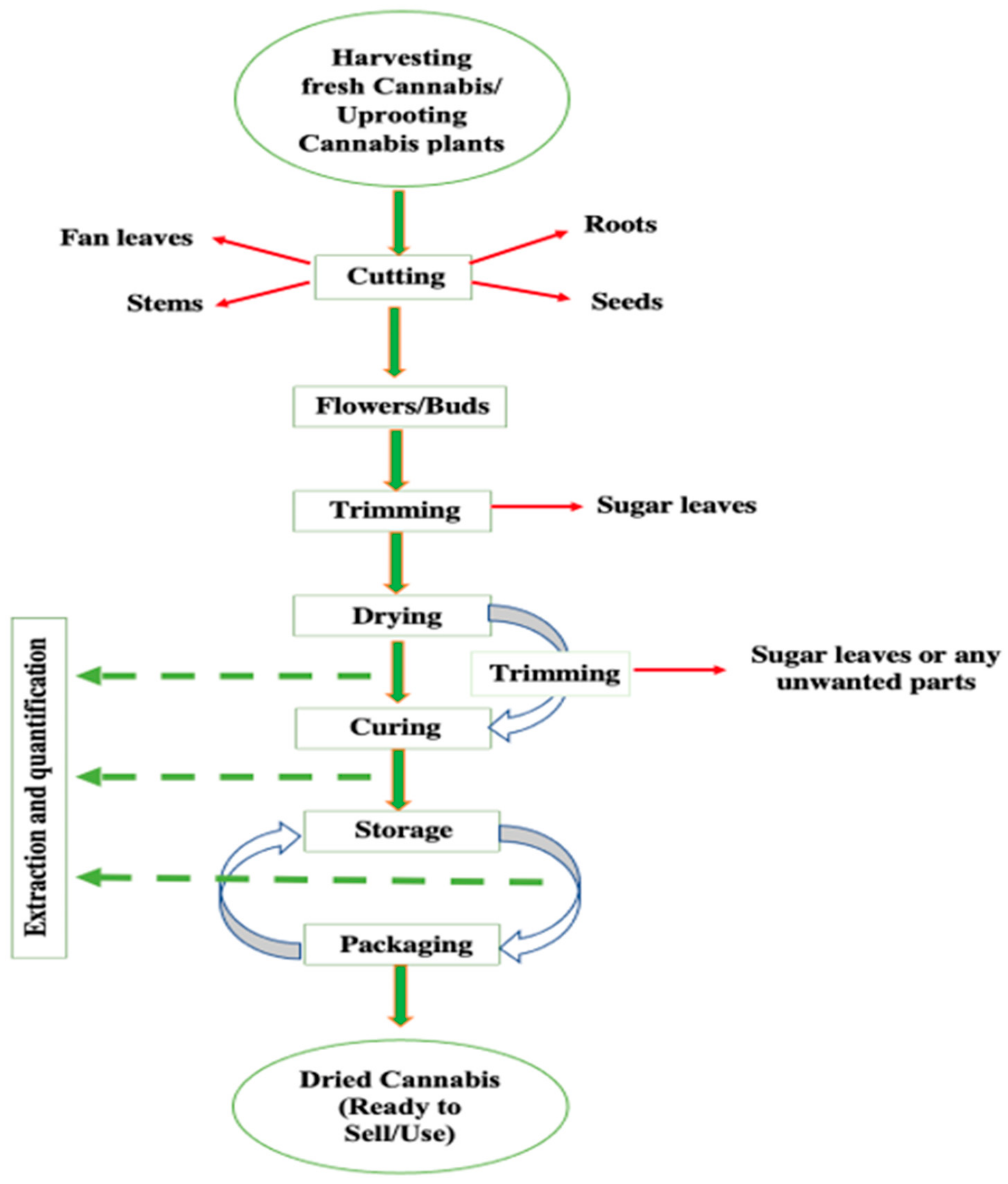
3. Postharvest Operations Involved in Cannabis
3.1. Trimming
3.2. Drying of Cannabis
3.2.1. Mechanism of Drying
3.2.2. Current Industrial Drying Practices for Cannabis
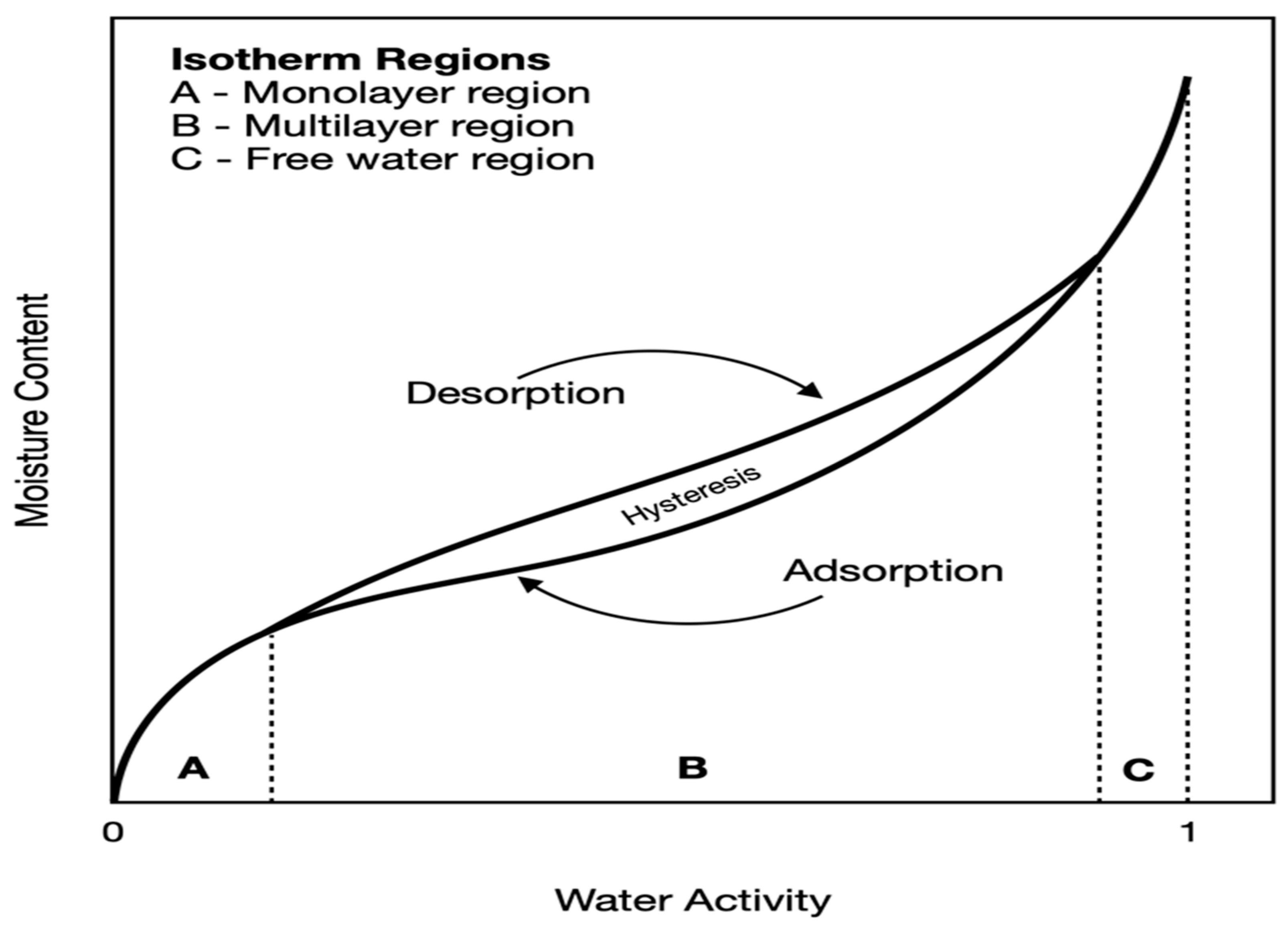
3.2.3. Equilibrium Moisture and Sorption Isotherms
Sorption Isotherm Models
- Modified Henderson equation:
- Modified Chung–Pfost equation:
- Modified Halsey equation:
- Modified Oswin equation:
- Guggenheim–Anderson–de Boer (GAB) equation:where ERH = equilibrium relative humidity (decimal value); T = temperature (°C); MCD = dry-basis moisture content (decimal value); and A, B, and C = equation constants, and the related data specifications based on which these constants were obtained are summarized in Table 2 of ASABE Standards ASAE D245.7 [44].
3.2.4. Possible Pre-Treatments for Improvement of Drying of Cannabis
Microwave Heating
Cold Plasma
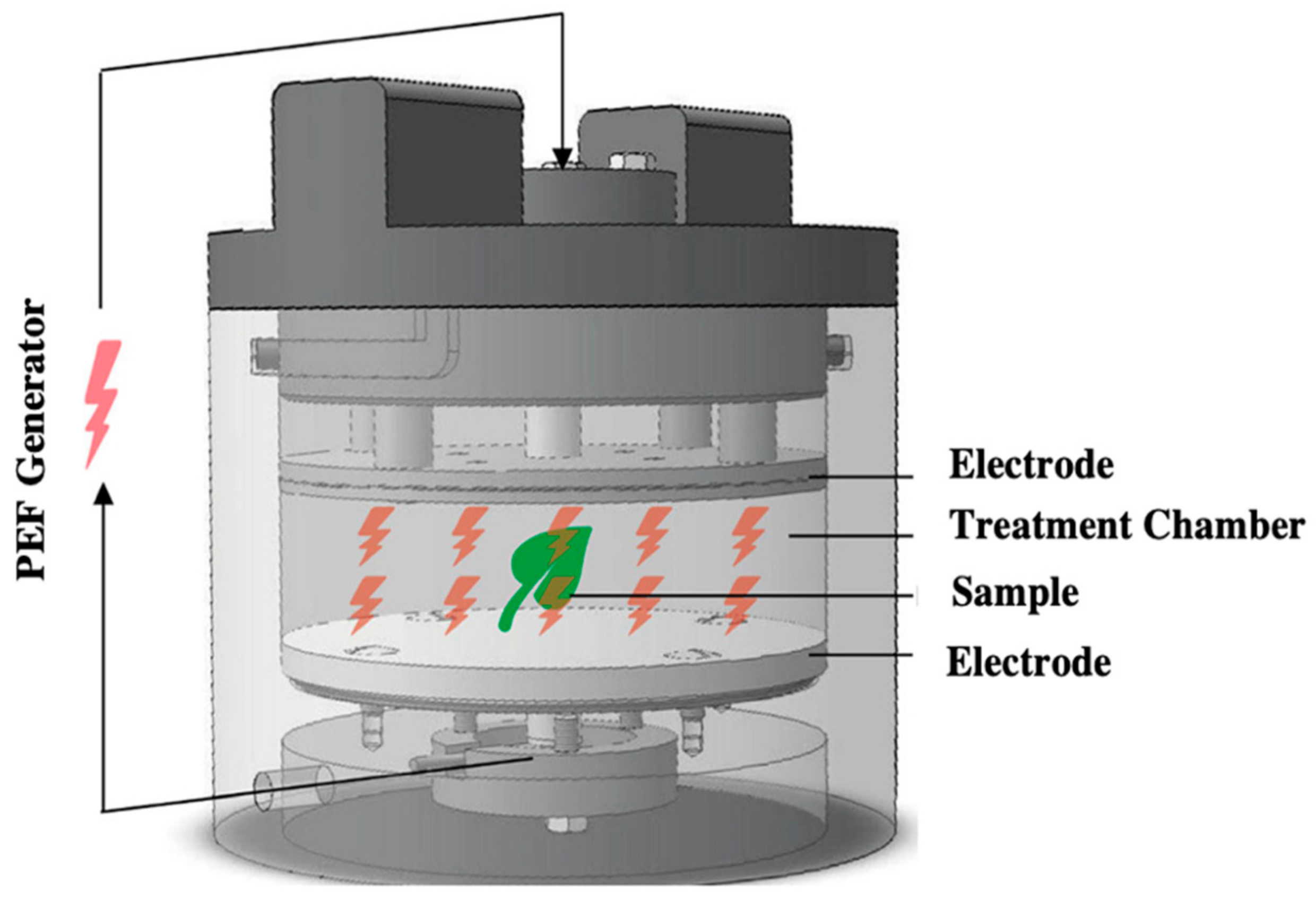
Pulsed Electric Field
Ultrasound
Irradiation
3.3. Curing
3.4. Packaging and Storage
4. Effect of Postharvest Processing on Phytoconstituents of Cannabis
| Sample | Moisture Level (wb) | Experimental Details | Findings | Source |
|---|---|---|---|---|
| Inflorescence and leaves of hemp (three varieties: Pipeline, Maverick and Queen Dream CBD) | Initial: 75–78%, Final: 9–13% | Freeze drying, Ambient drying, Hot air drying, and Sequential infrared and hot air (SIRHA) drying |
| [34] |
| Hemp buds | Initial: 65%, Final: 10% | Freeze drying, Hot air drying, Non-isothermal (stepwise) drying |
| [131] |
| Inflorescences of medicinal cannabis (Cannabis sativa) | Not mentioned | Steam sterilization for 10s at 62.5°C, 15 s at 65 °C and 20 s at 70 °C |
| [134] |
| Powder of Cannabis sativa seeds | Initial: Not mentioned, Final: 3.5–5.1% | In situ decarboxylation using pressurized hot water extraction technique at temperature (80 to 150 °C) for 5 to 60 min |
| [135] |
| Inflorescences of hemp cv Felina 32 | Not mentioned | Steam distillation (SD) or hydro distillation (HD) of fresh sample; HD of ambiently dried inflorescences; HD of blended and powdered inflorescences; HD of powdered and heated (120 °C for 1, 3, or 6 min) inflorescences; HD of powdered and microwaved (900 and 450 W) inflorescences |
| [49] |
| Cannabis sativa inflorescence collected from upper, middle, and lower portion of stem | Not mentioned | Solvent extraction (ethanol, n-Hexane, mixture of hexane and ethanol (7:3, v:v)) of undried and dried (using gentle stream of nitrogen, vacuum dryer and rotary evaporator) |
| [132] |
| Inflorescences of Cannabis sativa (cultivars: Pink, RBS, RMS, and GSC) | Not mentioned | Dried and irradiated with 5 kGy emitted from a 10 MeV accelerator |
| [110] |
| Two Cannabis sativa strains combined together, sieved through a 355-mm sieve, and homogenized (one strain contained primarily THCA/THC and the other contained CBDA/CBD). | Not mentioned | Stored in 66-L microbiological incubators with ±0.2 °C consistency for up to 52 weeks at different temperature (20 °C, +4 °C, +20 °C, +32 °C, +37 °C, and +40 °C). |
| [136] |
| Inflorescences of medicinal cannabis (one is ∆9-THC-rich and another is CBD rich) | Initial: Not mentioned Final: 9 ± 0.3% | Samples stored in the dark condition for 12 months at 4 distinct temperatures (−80, −30 °C, 4 °C, and 25 °C) and in 2 physical forms (whole or ground). |
| [137] |
| Inflorescences and leaves of hemp | Initial: 65.7% Final: up to constant moisture ratio | Convective drying at constant (40, 50 and 60 °C) and time varying temperature rise (1.5, 2.5 and 4 °C/h) at temperature in the 40–60 °C range |
| [138] |
5. Summary and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mansouri, H.; Bagheri, M. Induction of Polyploidy and Its Effect on Cannabis sativa L. In Cannabis sativa L.-Botany and Biotechnology; Chandra, S., Lata, H., ElSohly, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 365–383. [Google Scholar]
- Cohen, K.; Weizman, A.; Weinstein, A. Positive and Negative Effects of Cannabis and Cannabinoids on Health. Clin. Pharmacol. Ther. 2019, 105, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Morales, P.; Hurst, D.P.; Reggio, P.H. Molecular Targets of the Phytocannabinoids: A Complex Picture. In Phytocannabinoids: Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa; Kinghorn, A.D., Falk, H., Gibbons, S., Kobayashi, J., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 103–131. [Google Scholar]
- Addo, P.W.; Desaulniers Brousseau, V.; Morello, V.; MacPherson, S.; Paris, M.; Lefsrud, M. Cannabis Chemistry, Post-Harvest Processing Methods and Secondary Metabolite Profiling: A Review. Ind. Crops Prod. 2021, 170, 113743. [Google Scholar] [CrossRef]
- Cox-Georgian, D.; Ramadoss, N.; Dona, C.; Basu, C. Therapeutic and Medicinal Uses of Terpenes. Med. Plants 2019, 333–359. [Google Scholar] [CrossRef]
- Salentijn, E.M.J.; Zhang, Q.; Amaducci, S.; Yang, M.; Trindade, L.M. New Developments in Fiber Hemp (Cannabis sativa L.) Breeding. Ind. Crops Prod. 2015, 68, 32–41. [Google Scholar] [CrossRef]
- Tramèr, M.R.; Carroll, D.; Campbell, F.A.; Reynolds, D.J.M.; Moore, R.A.; McQuay, H.J. Cannabinoids for Control of Chemotherapy Induced Nausea and Vomiting: Quantitative Systematic Review. BMJ 2001, 323, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pertwee, R.G. Targeting the Endocannabinoid System with Cannabinoid Receptor Agonists: Pharmacological Strategies and Therapeutic Possibilities. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 3353–3363. [Google Scholar] [CrossRef]
- Wartenberg, A.C.; Holden, P.A.; Bodwitch, H.; Parker-Shames, P.; Novotny, T.; Harmon, T.C.; Hart, S.C.; Beutel, M.; Gilmore, M.; Hoh, E.; et al. Cannabis and the Environment: What Science Tells Us and What We Still Need to Know. Environ. Sci. Technol. Lett. 2021, 8, 98–107. [Google Scholar] [CrossRef]
- Challa, S.K.R.; Misra, N.N.; Martynenko, A. Drying of Cannabis—State of the Practices and Future Needs. Dry. Technol. 2021, 39, 2055–2064. [Google Scholar] [CrossRef]
- Patel, B.; Wene, D.; Fan, Z.T. Qualitative and Quantitative Measurement of Cannabinoids in Cannabis Using Modified HPLC/DAD Method. J. Pharm. Biomed. Anal. 2017, 146, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Radwan, M.M.; Wanas, A.S.; Chandra, S.; ElSohly, M.A. Natural Cannabinoids of Cannabis and Methods of Analysis. In Cannabis sativa L. -Botany and Biotechnology; Chandra, S., Lata, H., ElSohly, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 161–182. [Google Scholar]
- Wang, M.; Wang, Y.-H.; Avula, B.; Radwan, M.M.; Wanas, A.S.; van Antwerp, J.; Parcher, J.F.; ElSohly, M.A.; Khan, I.A. Decarboxylation Study of Acidic Cannabinoids: A Novel Approach Using Ultra-High-Performance Supercritical Fluid Chromatography/Photodiode Array-Mass Spectrometry. Cannabis Cannabinoid Res. 2016, 1, 262–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gülck, T.; Møller, B.L. Phytocannabinoids: Origins and Biosynthesis. Trends Plant Sci. 2020, 25, 985–1004. [Google Scholar] [CrossRef] [PubMed]
- Nachnani, R.; Raup-Konsavage, W.M.; Vrana, K.E. The Pharmacological Case for Cannabigerol. J. Pharmacol. Exp. Ther. 2021, 376, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.E.; Elsohly, M.A. Constituents of Cannabis sativa L. XVI. A Possible Decomposition Pathway of Δ9-Tetrahydrocannabinol to Cannabinol. J. Heterocycl. Chem. 1979, 16, 1667–1668. [Google Scholar] [CrossRef]
- Hecksel, R.; LaVigne, J.; Streicher, J.M. In Defense of the Entourage Effect: Terpenes Found in Cannabis sativa Activate the Cannabinoid Receptor 1 In Vitro. FASEB J. 2020, 34, 1. [Google Scholar] [CrossRef]
- Mudge, E.M.; Brown, P.N.; Murch, S.J. The Terroir of Cannabis: Terpene Metabolomics as a Tool to Understand Cannabis sativa Selections. Planta Med. 2019, 85, 781–796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, E.S. Cannabinoid Synthesis and Accumulation in Glandular Trichomes of Cannabis sativa. J. Acupunct. Meridian Stud. 2020, 13, 77. [Google Scholar] [CrossRef]
- Tanney, C.A.S.; Backer, R.; Geitmann, A.; Smith, D.L. Cannabis Glandular Trichomes: A Cellular Metabolite Factory. Front. Plant Sci. 2021, 12, 721986. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.C.; Hemphill, J.K.; Mahlberg, P.G. Trichomes and Cannabinoid Content of Developing Leaves and Bracts of Cannabis sativa L. (Cannabaceae). Am. J. Bot. 1980, 67, 1397–1406. [Google Scholar] [CrossRef]
- Henry, P. Cannabis Chemovar Classification: Terpenes Hyper-Classes and Targeted Genetic Markers for Accurate Discrimination of Flavours and Effects. PeerJ Prepr. 2017, 5, e3307v1. [Google Scholar]
- Kader, A.A.; Kasmire, R.F.; Mitchell, F.G.; Reid, M.S.; Sommer, N.F.; Thompson, J.F. Postharvest Technology of Horticultural Crops; University of California Agriculture & Natural Resources: Davis, CA, USA, 1985. [Google Scholar]
- Available online: https://Weedmaps.Com/Learn/the-Plant/When-to-Harvest-Cannabis (accessed on 3 March 2022).
- Geankoplis, C.J. Transport Processes and Separation Process Principles: (Includes Unit Operations), 4th ed.; Prentice Hall Professional Technical Reference: Upper Saddle River, NJ, USA, 2003. [Google Scholar]
- Sparks, B.D. The Latest Updates in Cannabis Trimming Innovation. Available online: https://www.greenhousegrower.com/production/the-latest-updates-in-cannabis-trimming-innovations/ (accessed on 5 March 2022).
- Cresp, H. Best Bud Trimmers—Top Trimming Machines of 2020. Grow Light Central. Available online: https://growlightcentral.com/blogs/news/best-bud-trimmers (accessed on 16 March 2022).
- Jin, D.; Jin, S.; Chen, J. Cannabis Indoor Growing Conditions, Management Practices, and Post-Harvest Treatment: A Review. Am. J. Plant Sci. 2019, 10, 925–946. [Google Scholar] [CrossRef] [Green Version]
- Lazarjani, M.P.; Young, O.; Kebede, L.; Seyfoddin, A. Processing and Extraction Methods of Medicinal Cannabis: A Narrative Review. J. Cannabis Res. 2021, 3, 32. [Google Scholar] [CrossRef] [PubMed]
- Brooker, D.B. Drying and Storage of Grains and Oilseeds; Van Nostrand Reinhold: New York, NY, USA, 1992. [Google Scholar]
- van ’t Land, C.M. Drying in the Process Industry; WILEY: Hoboken, NJ, USA, 2011. [Google Scholar]
- Stroshine, R.L. Physical Properties of Agricultural Materials and Food Products; R. Stroshine: West Lafayette, IN, USA, 2004. [Google Scholar]
- Srikiatden, J.; Roberts, J.S. Moisture Transfer in Solid Food Materials: A Review of Mechanisms, Models, and Measurements. Int. J. Food Prop. 2007, 10, 739–777. [Google Scholar] [CrossRef]
- Chen, C.; Wongso, I.; Putnam, D.; Khir, R.; Pan, Z. Effect of Hot Air and Infrared Drying on the Retention of Cannabidiol and Terpenes in Industrial Hemp (Cannabis sativa L.). Ind. Crops Prod. 2021, 172, 114051. [Google Scholar] [CrossRef]
- AL Ubeed, H.M.; Wills, R.B.H.; Chandrapala, J. Post-Harvest Operations to Generate High-Quality Medicinal Cannabis Products: A Systemic Review. Molecular 2022, 27, 1719. [Google Scholar] [CrossRef] [PubMed]
- Balmer, R.T. Chapter 12-Mixtures of Gases and Vapors. In Modern Engineering Thermodynamics; Balmer, R.T., Ed.; Academic Press: Boston, MA, USA, 2011; pp. 405–446. [Google Scholar]
- Dincer, I.; Rosen, M.A. Chapter 6-Exergy Analyses of Psychrometric Processes. In Exergy, 3rd ed.; Dincer, I., Rosen, M.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 101–123. [Google Scholar]
- Hall, M.R.; Allinson, D. 1—Heat and Mass Transport Processes in Building Materials. In Materials for Energy Efficiency and Thermal Comfort in Buildings; Hall, M.R., Ed.; Woodhead Publishing Series in Energy; Elsevier: Amsterdam, The Netherlands, 2010; pp. 3–53. [Google Scholar]
- Andrade, R.D.; Lemus, R.; Perez, C.E. Models of Sorption Isotherms for Food: Uses and Limitations. Vitae 2011, 18, 325–334. [Google Scholar]
- Mathlouthi, M.; Rogé, B. Water Vapour Sorption Isotherms and the Caking of Food Powders. Food Chem. 2003, 82, 61–71. [Google Scholar] [CrossRef]
- Troller, J.A.; Christian, J.H.B. Water Activity-1. Basic Concepts. In Water Activity and Food; Troller, J.A., Christian, J.H.B., Eds.; Academic Press: Cambridge, MA, USA, 1978; pp. 1–12. [Google Scholar]
- U.S. Food and Drug Administration. Water Activity (Aw) in Foods; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2014.
- Labuza, T.P.; Acott, K.; TATiNl, S.R.; Lee, R.Y.; Flink, J.; McCALL, W. Water Activity Determination: A Collaborative Study of Different Methods. J. Food Sci. 2008, 41, 910–917. [Google Scholar] [CrossRef]
- ASABE (American Society of Agricultural and Biological Engineers). D245.7 (R2021): Moisture Relationships of Plant Based Agricultural Products; ASABE: St. Joseph, MI, USA, 2017. [Google Scholar]
- Deng, L.-Z.; Mujumdar, A.S.; Zhang, Q.; Yang, X.-H.; Wang, J.; Zheng, Z.-A.; Gao, Z.-J.; Xiao, H.-W. Chemical and Physical Pretreatments of Fruits and Vegetables: Effects on Drying Characteristics and Quality Attributes–A Comprehensive Review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1408–1432. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, S.; Ramanathan, S.; Basak, T. Microwave Food Processing—A Review. Food Res. Int. 2013, 52, 243–261. [Google Scholar] [CrossRef]
- Rahath Kubra, I.; Kumar, D.; Jagan Mohan Rao, L. Emerging Trends in Microwave Processing of Spices and Herbs. Emerg. Trends Microw. Process. Spices Herbs 2016, 56, 2160–2173. [Google Scholar] [CrossRef] [PubMed]
- Gunasekaran, S. Pulsed Microwave-Vacuum Drying of Food Materials. Dry. Technol. 1999, 17, 395–412. [Google Scholar] [CrossRef]
- Fiorini, D.; Molle, A.; Nabissi, M.; Santini, G.; Benelli, G.; Maggi, F. Valorizing Industrial Hemp (Cannabis sativa L.) by-Products: Cannabidiol Enrichment in the Inflorescence Essential Oil Optimizing Sample Pre-Treatment Prior to Distillation. Ind. Crops Prod. 2019, 128, 581–589. [Google Scholar] [CrossRef]
- Sui, X.; Liu, T.; Ma, C.; Yang, L.; Zu, Y.; Zhang, L.; Wang, H. Microwave Irradiation to Pretreat Rosemary (Rosmarinus officinalis L.) for Maintaining Antioxidant Content during Storage and to Extract Essential Oil Simultaneously. Food Chem. 2012, 131, 1399–1405. [Google Scholar] [CrossRef]
- Drinić, Z.; Vladić, J.; Koren, A.; Zeremski, T.; Stojanov, N.; Kiprovski, B.; Vidović, S. Microwave-Assisted Extraction of Cannabinoids and Antioxidants from Cannabis sativa Aerial Parts and Process Modeling. J. Chem. Technol. Biotechnol. 2020, 95, 831–839. [Google Scholar] [CrossRef]
- Matešić, N.; Jurina, T.; Benković, M.; Panić, M.; Valinger, D.; Gajdoš Kljusurić, J.; Jurinjak Tušek, A. Microwave-Assisted Extraction of Phenolic Compounds from Cannabis sativa L.: Optimization and Kinetics Study. Sep. Sci. Technol. 2021, 56, 2047–2060. [Google Scholar] [CrossRef]
- Radoiu, M.; Kaur, H.; Bakowska-Barczak, A.; Splinter, S. Microwave-Assisted Industrial Scale Cannabis Extraction. Technol. 2020, 8, 45. [Google Scholar] [CrossRef]
- Teh, S.-S.; Niven, B.E.; Bekhit, A.E.-D.A.; Carne, A.; Birch, E.J. The Use of Microwave and Pulsed Electric Field as a Pretreatment Step in Ultrasonic Extraction of Polyphenols from Defatted Hemp Seed Cake (Cannabis sativa) Using Response Surface Methodology. Food Bioprocess Technol. 2014, 7, 3064–3076. [Google Scholar] [CrossRef]
- Muhammad, A.I.; Li, Y.; Liao, X.; Liu, D.; Ye, X.; Chen, S.; Hu, Y.; Wang, J.; Ding, T. Effect of Dielectric Barrier Discharge Plasma on Background Microflora and Physicochemical Properties of Tiger Nut Milk. Food Control. 2019, 96, 119–127. [Google Scholar] [CrossRef]
- Muhammad, A.I.; Liao, X.; Cullen, P.J.; Liu, D.; Xiang, Q.; Wang, J.; Chen, S.; Ye, X.; Ding, T. Effects of Nonthermal Plasma Technology on Functional Food Components. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1379–1394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amini, M.; Ghoranneviss, M. Effects of Cold Plasma Treatment on Antioxidants Activity, Phenolic Contents and Shelf Life of Fresh and Dried Walnut (Juglans regia L.) Cultivars during Storage. LWT 2016, 73, 178–184. [Google Scholar] [CrossRef]
- Shishir, M.R.I.; Karim, N.; Bao, T.; Gowd, V.; Ding, T.; Sun, C.; Chen, W. Cold Plasma Pretreatment–A Novel Approach to Improve the Hot Air Drying Characteristics, Kinetic Parameters, and Nutritional Attributes of Shiitake Mushroom. Dry. Technol. 2020, 38, 2134–2150. [Google Scholar] [CrossRef]
- Domonkos, M.; Tichá, P.; Trejbal, J.; Demo, P. Applications of Cold Atmospheric Pressure Plasma Technology in Medicine, Agriculture and Food Industry. Appl. Sci. 2021, 11, 4809. [Google Scholar] [CrossRef]
- Braný, D.; Dvorská, D.; Halašová, E.; Škovierová, H. Cold Atmospheric Plasma: A Powerful Tool for Modern Medicine. Int. J. Mol. Sci. 2020, 21, 2932. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.-L.; Zhong, C.-S.; Mujumdar, A.S.; Yang, X.-H.; Deng, L.-Z.; Wang, J.; Xiao, H.-W. Cold Plasma Pretreatment Enhances Drying Kinetics and Quality Attributes of Chili Pepper (Capsicum annuum L.). J. Food Eng. 2019, 241, 51–57. [Google Scholar] [CrossRef]
- Li, S.; Chen, S.; Han, F.; Xv, Y.; Sun, H.; Ma, Z.; Chen, J.; Wu, W. Development and Optimization of Cold Plasma Pretreatment for Drying on Corn Kernels. J. Food Sci. 2019, 84, 2181–2189. [Google Scholar] [CrossRef]
- Zhou, Y.-H.; Vidyarthi, S.K.; Zhong, C.-S.; Zheng, Z.-A.; An, Y.; Wang, J.; Wei, Q.; Xiao, H.-W. Cold Plasma Enhances Drying and Color, Rehydration Ratio and Polyphenols of Wolfberry via Microstructure and Ultrastructure Alteration. LWT 2020, 134, 110173. [Google Scholar] [CrossRef]
- Bao, T.; Hao, X.; Shishir, M.R.I.; Karim, N.; Chen, W. Cold Plasma: An Emerging Pretreatment Technology for the Drying of Jujube Slices. Food Chem. 2021, 337, 127783. [Google Scholar] [CrossRef]
- Tabibian, S.A.; Labbafi, M.; Askari, G.H.; Rezaeinezhad, A.R.; Ghomi, H. Effect of Gliding Arc Discharge Plasma Pretreatment on Drying Kinetic, Energy Consumption and Physico-Chemical Properties of Saffron (Crocus sativus L.). J. Food Eng. 2020, 270, 109766. [Google Scholar] [CrossRef]
- Miraei Ashtiani, S.-H.; Rafiee, M.; Mohebi Morad, M.; Khojastehpour, M.; Khani, M.R.; Rohani, A.; Shokri, B.; Martynenko, A. Impact of Gliding Arc Plasma Pretreatment on Drying Efficiency and Physicochemical Properties of Grape. Innov. Food Sci. Emerg. Technol. 2020, 63, 102381. [Google Scholar] [CrossRef]
- Ebadi, M.-T.; Abbasi, S.; Harouni, A.; Sefidkon, F. Effect of Cold Plasma on Essential Oil Content and Composition of Lemon Verbena. Food Sci. Nutr. 2019, 7, 1166–1171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mildaziene, V.; Pauzaite, G.; Naucienė, Z.; Malakauskiene, A.; Zukiene, R.; Januskaitiene, I.; Jakstas, V.; Ivanauskas, L.; Filatova, I.; Lyushkevich, V. Pre-Sowing Seed Treatment with Cold Plasma and Electromagnetic Field Increases Secondary Metabolite Content in Purple Coneflower (Echinacea purpurea) Leaves. Plasma Process. Polym. 2018, 15, 1700059. [Google Scholar] [CrossRef]
- Jerushalmi, S.; Maymon, M.; Dombrovsky, A.; Freeman, S. Effects of Cold Plasma, Gamma and e-Beam Irradiations on Reduction of Fungal Colony Forming Unit Levels in Medical Cannabis Inflorescences. J. Cannabis Res. 2020, 2, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knorr, D.; Angersbach, A.; Eshtiaghi, M.N.; Heinz, V.; Lee, D.-U. Processing Concepts Based on High Intensity Electric Field Pulses. Trends Food Sci. Technol. 2001, 12, 129–135. [Google Scholar] [CrossRef]
- Rastogi, N.K.; Eshtiaghi, M.N.; Knorr, D. Accelerated Mass Transfer During Osmotic Dehydration of High Intensity Electrical Field Pulse Pretreated Carrots. J. Food Sci. 1999, 64, 1020–1023. [Google Scholar] [CrossRef]
- Wiktor, A.; Nowacka, M.; Dadan, M.; Rybak, K.; Lojkowski, W.; Chudoba, T.; Witrowa-Rajchert, D. The Effect of Pulsed Electric Field on Drying Kinetics, Color, and Microstructure of Carrot. Dry. Technol. 2016, 34, 1286–1296. [Google Scholar] [CrossRef]
- Saulis, G. Electroporation of Cell Membranes: The Fundamental Effects of Pulsed Electric Fields in Food Processing. Food Eng. Rev. 2010, 2, 52–73. [Google Scholar] [CrossRef]
- Liu, Z.; Esveld, E.; Vincken, J.-P.; Bruins, M.E. Pulsed Electric Field as an Alternative Pre-Treatment for Drying to Enhance Polyphenol Extraction from Fresh Tea Leaves. Food Bioprocess Technol. 2019, 12, 183–192. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Guo, Y.; Zhang, D. Study of the Effect of High-Pulsed Electric Field Treatment on Vacuum Freeze-Drying of Apples. Dry. Technol. 2011, 29, 1714–1720. [Google Scholar] [CrossRef]
- Alam, M.R.; Lyng, J.G.; Frontuto, D.; Marra, F.; Cinquanta, L. Effect of Pulsed Electric Field Pretreatment on Drying Kinetics, Color, and Texture of Parsnip and Carrot. J. Food Sci. 2018, 83, 2159–2166. [Google Scholar] [CrossRef] [PubMed]
- Gachovska, T.K.; Simpson, M.V.; Ngadi, M.O.; Raghavan, G. Pulsed Electric Field Treatment of Carrots before Drying and Rehydration. J. Sci. Food Agric. 2009, 89, 2372–2376. [Google Scholar] [CrossRef]
- Lebovka, N.I.; Shynkaryk, N.V.; Vorobiev, E. Pulsed Electric Field Enhanced Drying of Potato Tissue. J. Food Eng. 2007, 78, 606–613. [Google Scholar] [CrossRef]
- Ostermeier, R.; Giersemehl, P.; Siemer, C.; Töpfl, S.; Jäger, H. Influence of Pulsed Electric Field (PEF) Pre-Treatment on the Convective Drying Kinetics of Onions. J. Food Eng. 2018, 237, 110–117. [Google Scholar] [CrossRef]
- Thamkaew, G.; Gómez Galindo, F. Influence of Pulsed and Moderate Electric Field Protocols on the Reversible Permeabilization and Drying of Thai Basil Leaves. Innov. Food Sci. Emerg. Technol. 2020, 64, 102430. [Google Scholar] [CrossRef]
- Ade-Omowaye, B.I.O.; Rastogi, N.K.; Angersbach, A.; Knorr, D. Combined Effects of Pulsed Electric Field Pre-Treatment and Partial Osmotic Dehydration on Air Drying Behaviour of Red Bell Pepper. J. Food Eng. 2003, 60, 89–98. [Google Scholar] [CrossRef]
- Donsì, F.; Ferrari, G.; Pataro, G. Applications of Pulsed Electric Field Treatments for the Enhancement of Mass Transfer from Vegetable Tissue. Food Eng. Rev. 2010, 2, 109–130. [Google Scholar] [CrossRef]
- Goettel, M.; Eing, C.; Gusbeth, C.; Straessner, R.; Frey, W. Pulsed Electric Field Assisted Extraction of Intracellular Valuables from Microalgae. Algal Res. 2013, 2, 401–408. [Google Scholar] [CrossRef]
- Fincan, M.; DeVito, F.; Dejmek, P. Pulsed Electric Field Treatment for Solid–Liquid Extraction of Red Beetroot Pigment. J. Food Eng. 2004, 64, 381–388. [Google Scholar] [CrossRef]
- Puértolas, E.; Cregenzán, O.; Luengo, E.; Álvarez, I.; Raso, J. Pulsed-Electric-Field-Assisted Extraction of Anthocyanins from Purple-Fleshed Potato. Food Chem. 2013, 136, 1330–1336. [Google Scholar] [CrossRef]
- Delsart, C.; Ghidossi, R.; Poupot, C.; Cholet, C.; Grimi, N.; Vorobiev, E.; Milisic, V.; Mietton Peuchot, M. Enhanced Extraction of Phenolic Compounds from Merlot Grapes by Pulsed Electric Field Treatment. Am. J. Enol. Vitic. 2012, 63, 205–211. [Google Scholar] [CrossRef]
- Takaki, K.; Hatayama, H.; Koide, S.; Kawamura, Y. Improvement of Polyphenol Extraction from Grape Skin by Pulse Electric Field. In Proceedings of the 2011 IEEE Pulsed Power Conference, Chicago, IL, USA, 19–23 June 2011; pp. 1262–1265. [Google Scholar]
- Haji-Moradkhani, A.; Rezaei, R.; Moghimi, M. Optimization of Pulsed Electric Field-Assisted Oil Extraction from Cannabis Seeds. J. Food Process Eng. 2019, 42, e13028. [Google Scholar] [CrossRef]
- Musielak, G.; Mierzwa, D.; Kroehnke, J. Food Drying Enhancement by Ultrasound—A Review. Trends Food Sci. Technol. 2016, 56, 126–141. [Google Scholar] [CrossRef]
- Chemat, F.; Zill-e-Huma; Khan, M. K. Applications of Ultrasound in Food Technology: Processing, Preservation and Extraction. Ultrason. Sonochemis 2011, 18, 813–835. [Google Scholar] [CrossRef]
- Chen, Z.-G.; Guo, X.-Y.; Wu, T. A Novel Dehydration Technique for Carrot Slices Implementing Ultrasound and Vacuum Drying Methods. Ultrason. Sonochemis 2016, 30, 28–34. [Google Scholar] [CrossRef]
- Nowacka, M.; Wiktor, A.; Śledź, M.; Jurek, N.; Witrowa-Rajchert, D. Drying of Ultrasound Pretreated Apple and Its Selected Physical Properties. J. Food Eng. 2012, 113, 427–433. [Google Scholar] [CrossRef]
- Cárcel, J.A.; García-Pérez, J.V.; Riera, E.; Mulet, A. Influence of High-Intensity Ultrasound on Drying Kinetics of Persimmon. Dry. Technol. 2007, 25, 185–193. [Google Scholar] [CrossRef]
- Gallego-Juarez, J.A.; Rodriguez-Corral, G.; Gálvez Moraleda, J.C.; Yang, T.S. A New High-Intensity Ultrasonic Technology for Food Dehydration. Dry. Technol. 1999, 17, 597–608. [Google Scholar] [CrossRef]
- Tekin, Z.H.; Baslar, M. The Effect of Ultrasound-Assisted Vacuum Drying on the Drying Rate and Quality of Red Peppers. J. Therm. Anal. Calorim. 2018, 132, 1131–1143. [Google Scholar] [CrossRef]
- Vieira da Silva Júnior, E.; Lins de Melo, L.; Batista de Medeiros, R.A.; Pimenta Barros, Z.M.; Azoubel, P.M. Influence of Ultrasound and Vacuum Assisted Drying on Papaya Quality Parameters. LWT 2018, 97, 317–322. [Google Scholar] [CrossRef]
- Rojas, M.L.; Silveira, I.; Augusto, P.E.D. Ultrasound and Ethanol Pre-Treatments to Improve Convective Drying: Drying, Rehydration and Carotenoid Content of Pumpkin. Food Bioprod. Process. 2020, 119, 20–30. [Google Scholar] [CrossRef]
- Fernandes, F.A.N.; Linhares, F.E.; Rodrigues, S. Ultrasound as Pre-Treatment for Drying of Pineapple. Ultrasonics Sonochemis 2008, 15, 1049–1054. [Google Scholar] [CrossRef]
- Szadzińska, J.; Łechtańska, J.; Pashminehazar, R.; Kharaghani, A.; Tsotsas, E. Microwave- and Ultrasound-Assisted Convective Drying of Raspberries: Drying Kinetics and Microstructural Changes. Dry. Technol. 2019, 37, 1–12. [Google Scholar] [CrossRef]
- Tomšik, A.; Pavlić, B.; Vladić, J.; Ramić, M.; Brindza, J.; Vidović, S. Optimization of Ultrasound-Assisted Extraction of Bioactive Compounds from Wild Garlic (Allium ursinum L.). Ultrason. Sonochem. 2016, 29, 502–511. [Google Scholar] [CrossRef]
- Sharayei, P.; Azarpazhooh, E.; Zomorodi, S.; Ramaswamy, H.S. Ultrasound Assisted Extraction of Bioactive Compounds from Pomegranate (Punica granatum L.) Peel. LWT 2019, 101, 342–350. [Google Scholar] [CrossRef]
- González de Peredo, A.V.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Ferreiro-González, M.; Amores-Arrocha, A.; Palma, M.; Barbero, G.F.; Jiménez-Cantizano, A. Alternative Ultrasound-Assisted Method for the Extraction of the Bioactive Compounds Present in Myrtle (Myrtus communis L.). Molecules 2019, 24, 882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, X.-Y.; Meng, J.-M.; Mao, Q.-Q.; Shang, A.; Li, B.-Y.; Zhao, C.-N.; Tang, G.-Y.; Cao, S.-Y.; Wei, X.-L.; Gan, R.-Y.; et al. Effects of Tannase and Ultrasound Treatment on the Bioactive Compounds and Antioxidant Activity of Green Tea Extract. Antioxidants 2019, 8, 362. [Google Scholar] [CrossRef] [Green Version]
- Banožić, M.; Banjari, I.; Jakovljević, M.; Šubarić, D.; Tomas, S.; Babić, J.; Jokić, S. Optimization of Ultrasound-Assisted Extraction of Some Bioactive Compounds from Tobacco Waste. Molecules 2019, 24, 1611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porto, C.; Decorti, D.; Natolino, A. Ultrasound-Assisted Extraction of Volatile Compounds from Industrial Cannabis sativa L. Inflorescences. Int. J. Appl. Res. Nat. Prod. 2014, 7, 8–14. [Google Scholar]
- Agarwal, C.; Máthé, K.; Hofmann, T.; Csóka, L. Ultrasound-Assisted Extraction of Cannabinoids from Cannabis sativa L. Optimized by Response Surface Methodology. J. Food Sci. 2018, 83, 700–710. [Google Scholar] [CrossRef] [PubMed]
- Esmaeilzadeh Kenari, R.; Dehghan, B. Optimization of Ultrasound-Assisted Solvent Extraction of Hemp (Cannabis sativa L.) Seed Oil Using RSM: Evaluation of Oxidative Stability and Physicochemical Properties of Oil. Food Sci. Nutr. 2020, 8, 4976–4986. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Piazza, L. Ultrasound-Assisted Extraction of Oil from Hempseed (Cannabis sativa L.): Part 1. J. Sci. Food Agric. 2022, 102, 732–739. [Google Scholar] [CrossRef]
- Thakur, B.R.; Singh, R.K. Food Irradiation-chemistry and Applications. Food Rev. Int. 1994, 10, 437–473. [Google Scholar] [CrossRef]
- Kovalchuk, O.; Li, D.; Rodriguez-Juarez, R.; Golubov, A.; Hudson, D.; Kovalchuk, I. The Effect of Cannabis Dry Flower Irradiation on the Level of Cannabinoids, Terpenes and Anti-Cancer Properties of the Extracts. Biocatal. Agric. Biotechnol. 2020, 29, 101736. [Google Scholar] [CrossRef]
- Hazekamp, A. Evaluating the Effects of Gamma-Irradiation for Decontamination of Medicinal Cannabis. Front. Pharmacol. 2016, 7, 108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Chao, Y. Effect of Gamma Irradiation on Quality of Dried Potato. Radiat. Phys. Chem. 2003, 66, 293–297. [Google Scholar] [CrossRef]
- Nayak, C.A.; Suguna, K.; Narasimhamurthy, K.; Rastogi, N.K. Effect of Gamma Irradiation on Histological and Textural Properties of Carrot, Potato and Beetroot. J. Food Eng. 2007, 79, 765–770. [Google Scholar] [CrossRef]
- Ling, Y.; Wang, H.; Fei, X.; Yue, Q.; Huang, T.; Shan, Q.; Hei, D.; Chen, T.; Zhang, X.; Jia, W. Improved Dehydration Performance of Tofu Protein by Ionizing Irradiation Pretreatment. J. Radioanal. Nucl. Chem. 2021, 327, 575–583. [Google Scholar] [CrossRef]
- Sung, Y.J.; Shin, S.-J. Compositional Changes in Industrial Hemp Biomass (Cannabis sativa L.) Induced by Electron Beam Irradiation Pretreatment. Biomass Bioenergy 2011, 35, 3267–3270. [Google Scholar] [CrossRef]
- Anuradha, K.; Shyamala, B.N.; Naidu, M.M. Vanilla- Its Science of Cultivation, Curing, Chemistry, and Nutraceutical Properties. Crit. Rev. Food Sci. Nutr. 2013, 53, 1250–1276. [Google Scholar] [CrossRef] [PubMed]
- Dignum, M.J.W.; Kerler, J.; Verpoorte, R. Vanilla Production: Technological, Chemical, and Biosynthetic Aspects. Food Rev. Int. 2001, 17, 119–120. [Google Scholar] [CrossRef]
- Ko, E.Y.; Sharma, K.; Nile, S.H. Effect of Harvesting Practices, Lifting Time, Curing Methods, and Irrigation on Quercetin Content in Onion (Allium cepa L.) Cultivars. Emir. J. Food Agric. 2016, 28, 594–600. [Google Scholar]
- Mogren, L.M.; Olsson, M.E.; Gertsson, U.E. Quercetin Content in Field-Cured Onions (Allium cepa L.): Effects of Cultivar, Lifting Time, and Nitrogen Fertilizer Level. J. Agric. Food Chem. 2006, 54, 6185–6191. [Google Scholar] [CrossRef]
- Shi, L.; Kim, E.; Yang, L.; Huang, Y.; Ren, N.; Li, B.; He, P.; Tu, Y.; Wu, Y. Effect of a Combined Microwave-Assisted Drying and Air Drying on Improving Active Nutraceutical Compounds, Flavor Quality, and Antioxidant Properties of Camellia sinensis L. (Cv. Longjing 43) Flowers. Food Qual. Saf. 2021, 5, fyaa040. [Google Scholar] [CrossRef]
- Wang, L.; Cheng, B.; Li, Z.; Liu, T.; Li, J. Intelligent tobacco flue-curing method based on leaf texture feature analysis. Optik 2017, 150, 117–130. [Google Scholar] [CrossRef]
- Tabil, L.; Sokhansanj, S. Mechanical and Temperature Effects on Shelf Life Stability of Fruits and Vegetables. In Food Shelf Life Stability; CRC Press Inc.: Boca Raton, FL, USA, 2000; pp. 37–86. [Google Scholar]
- Uline.ca. Uline Products in Stock-Uline.ca. Available online: https://www.uline.ca/cls_uline/Uline-Products (accessed on 14 March 2022).
- NDSupplies.ca. All Child Resistant Packaging. ND Supplies—Manufacturing of Packaging Supplies. Available online: https://ndsupplies.ca/product-category/all-child-resistant-packaging/ (accessed on 14 March 2022).
- Popham, N. 8-Resin Infusion for the Manufacture of Large Composite Structures. In Marine Composites; Pemberton, R., Summerscales, J., Graham-Jones, J., Eds.; Woodhead Publishing Series in Composites Science and Engineering; Woodhead Publishing: Sawston, UK, 2019; pp. 227–268. [Google Scholar]
- McKeen, L.W. 6-Polyesters. In Film Properties of Plastics and Elastomers 3rd ed; McKeen, L.W., Ed.; Plastics Design Library: Norwich, NY, USA; William Andrew Publishing, 2012; pp. 91–123. [Google Scholar]
- Trofin, I.G.; Dabija, G.; Vaireanu, I.; Filipescu, L. Long-Term Storage and Cannabis Oil Stability. Rev. Chim. 2012, 63, 293–297. [Google Scholar]
- Trofin, I.G.; Dabija, G.; Vãireanu, I.; Filipescu, L. The Influence of Long-Term Storage Conditions on the Stability of Cannabinoids Derived from Cannabis Resin. Rev. Chim. Bucharest. 2012, 64, 422–427. [Google Scholar]
- Trofin, I.G.; Vlad, C.C.; Dabija, G.; Filipescu, L. Influence of Storage Conditions on the Chemical Potency of Herbal Cannabis. Rev. Chim. 2011, 62, 639–645. [Google Scholar]
- Taschwer, M.; Schmid, M.G. Determination of the Relative Percentage Distribution of THCA and Δ9-THC in Herbal Cannabis Seized in Austria—Impact of Different Storage Temperatures on Stability. Forensic Sci. Int. 2015, 254, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Challa, S.K.R. Drying Kinetics and the Effects of Drying Methods on Quality (CBD, Terpenes and Color) of Hemp (Cannabis sativa L.) Buds. Master’s Thesis, Dalhousie University, Halifax, NS, Canada, 2020. [Google Scholar]
- Namdar, D.; Mazuz, M.; Ion, A.; Koltai, H. Variation in the Compositions of Cannabinoid and Terpenoids in Cannabis sativa Derived from Inflorescence Position along the Stem and Extraction Methods. Ind. Crops Prod. 2018, 113, 376–382. [Google Scholar] [CrossRef]
- Valizadehderakhshan, M.; Shahbazi, A.; Kazem-Rostami, M.; Todd, M.S.; Bhowmik, A.; Wang, L. Extraction of Cannabinoids from Cannabis sativa L. (Hemp)—Review. Agriculture 2021, 11, 384. [Google Scholar] [CrossRef]
- Jerushalmi, S.; Maymon, M.; Dombrovsky, A.; Regev, R.; Schmilovitch, Z.; Namdar, D.; Shalev, N.; Koltai, H.; Freeman, S. Effects of Steam Sterilization on Reduction of Fungal Colony Forming Units, Cannabinoids and Terpene Levels in Medical Cannabis Inflorescences. Sci. Rep. 2021, 11, 13973. [Google Scholar] [CrossRef] [PubMed]
- Nuapia, Y.; Maraba, K.; Tutu, H.; Chimuka, L.; Cukrowska, E. In Situ Decarboxylation-Pressurized Hot Water Extraction for Selective Extraction of Cannabinoids from Cannabis sativa. Chemometric Approach. Molecules 2021, 26, 3343. [Google Scholar] [CrossRef] [PubMed]
- Meija, J.; McRae, G.; Miles, C.O.; Melanson, J.E. Thermal Stability of Cannabinoids in Dried Cannabis: A Kinetic Study. Anal. Bioanal. Chem. 2022, 414, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Milay, L.; Berman, P.; Shapira, A.; Guberman, O.; Meiri, D. Metabolic Profiling of Cannabis Secondary Metabolites for Evaluation of Optimal Postharvest Storage Conditions. Front. Plant Sci. 2020, 11, 583605. [Google Scholar] [CrossRef] [PubMed]
- Chasiotis, V.; Tsakirakis, A.; Termentzi, A.; Machera, K.; Filios, A. Drying and quality characteristics of Cannabis sativa L. inflorescences under constant and time-varying convective drying temperature schemes. Therm. Sci. Eng. Prog. 2022, 28, 101076. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, P.C.; Vista, A.R.; Tabil, L.G.; Baik, O.-D. Postharvest Operations of Cannabis and Their Effect on Cannabinoid Content: A Review. Bioengineering 2022, 9, 364. https://doi.org/10.3390/bioengineering9080364
Das PC, Vista AR, Tabil LG, Baik O-D. Postharvest Operations of Cannabis and Their Effect on Cannabinoid Content: A Review. Bioengineering. 2022; 9(8):364. https://doi.org/10.3390/bioengineering9080364
Chicago/Turabian StyleDas, Pabitra Chandra, Alec Roger Vista, Lope G. Tabil, and Oon-Doo Baik. 2022. "Postharvest Operations of Cannabis and Their Effect on Cannabinoid Content: A Review" Bioengineering 9, no. 8: 364. https://doi.org/10.3390/bioengineering9080364
APA StyleDas, P. C., Vista, A. R., Tabil, L. G., & Baik, O.-D. (2022). Postharvest Operations of Cannabis and Their Effect on Cannabinoid Content: A Review. Bioengineering, 9(8), 364. https://doi.org/10.3390/bioengineering9080364