Validation of Psychophysiological Measures for Caffeine Oral Films Characterization by Machine Learning Approaches
Abstract
:1. Introduction
2. Methods
2.1. Participants
2.2. Data Collection
2.3. Procedures
- -
- -
- -
- Perform the Test of variables of attention (T.O.V.A.), which is a continuous performance test that assesses attention and impulsivity [33].
2.4. Experimental Indicies
- -
- ECG
- -
- RF
- -
- EDA
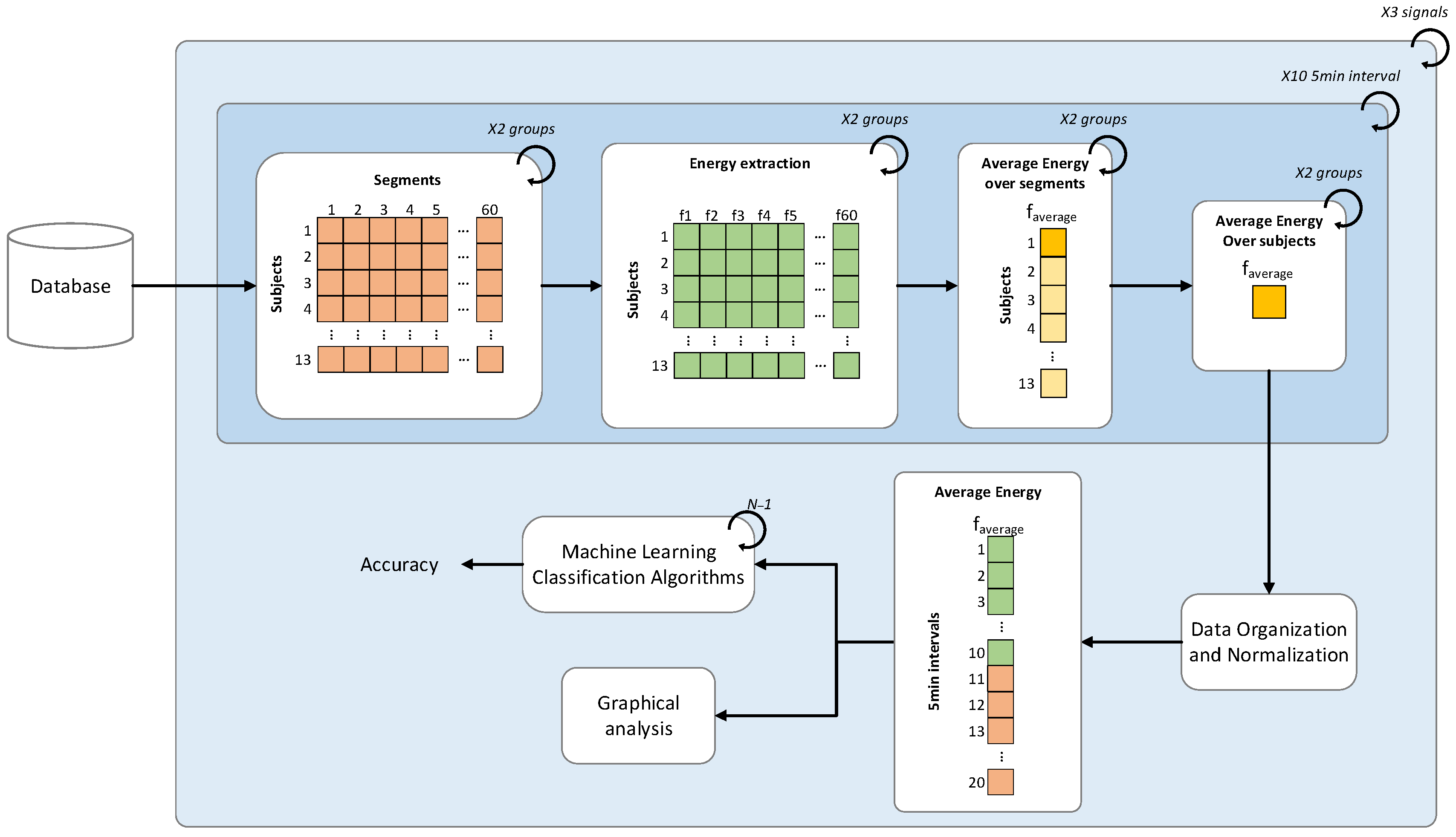
2.5. Time Signal Analysis
2.6. Statistical Analysis
3. Results
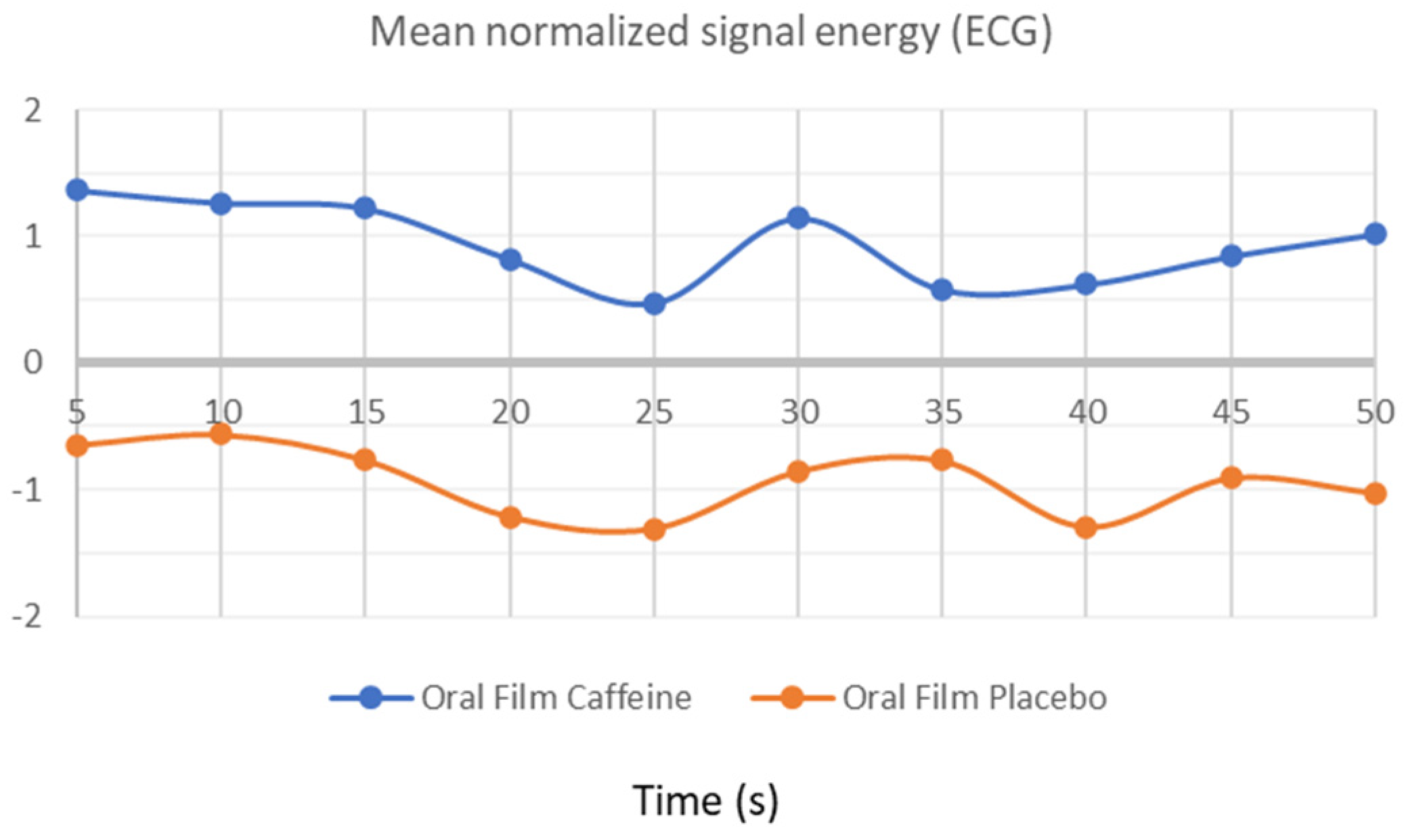
3.1. ECG Activity
3.2. EDA Activity
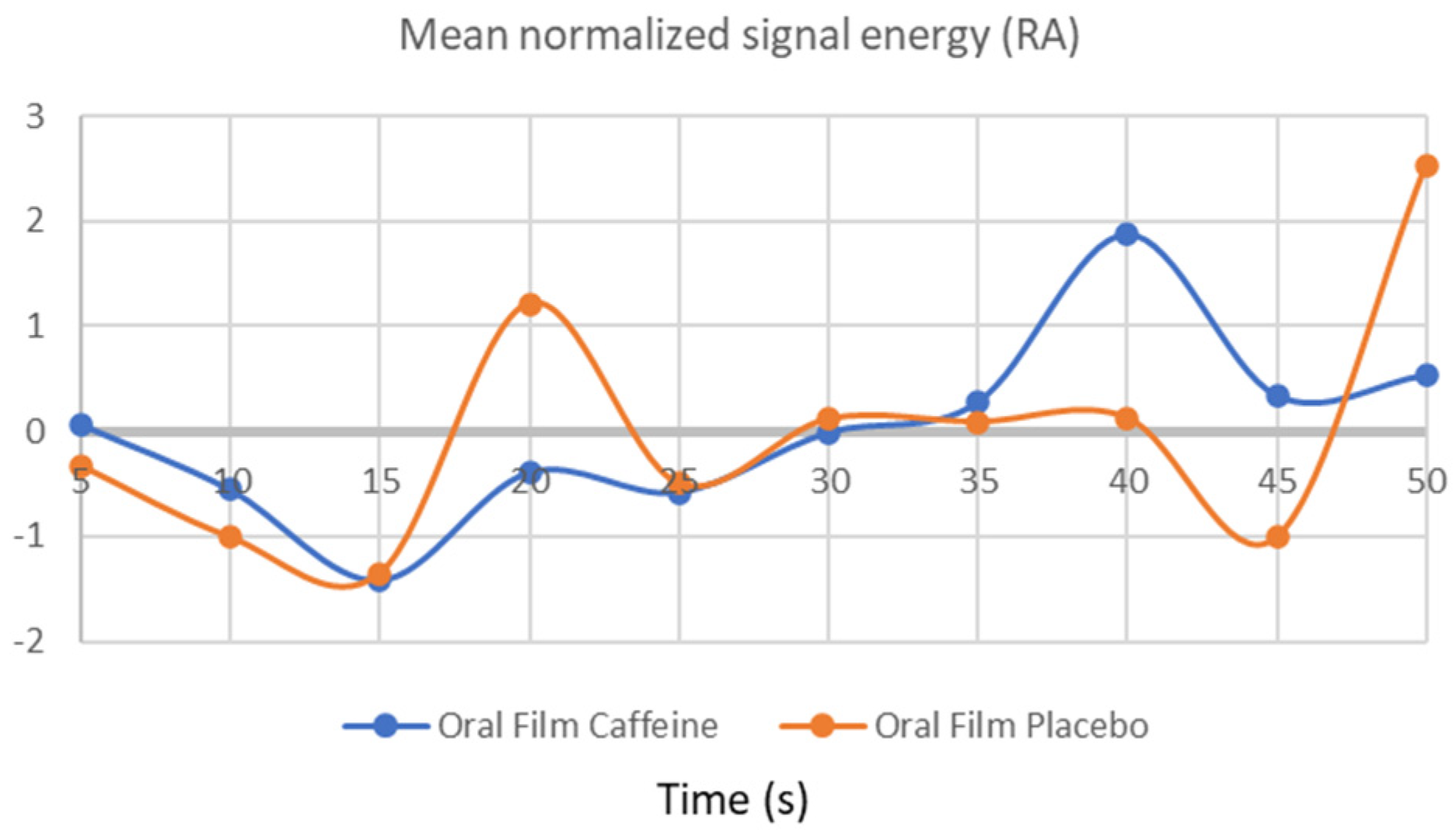
3.3. Respiratory Frequency
3.4. Machine Learning Groups Discrimination
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khalid, G.M.; Selmin, F.; Musazzi, U.M.; Gennari, C.G.; Minghetti, P.; Cilurzo, F. Trends in the Characterization Methods of Orodispersible Films. Curr. Drug Deliv. 2021, 18, 935–946. [Google Scholar] [CrossRef]
- Özakar, R.S.; Özakar, E. Current Overview of Oral Thin Films. Turk. J. Pharm. Sci. 2021, 18, 111. [Google Scholar] [CrossRef] [PubMed]
- Dixit, R.P.; Puthli, S.P. Oral strip technology: Overview and future potential. J. Control. Release 2009, 139, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, E.M.; Breitenbach, A.; Breitkreutz, J. Advances in orodispersible films for drug delivery. Expert Opin. Drug Deliv. 2011, 8, 299–316. [Google Scholar] [CrossRef]
- Tian, Y.; Orlu, M.; Woerdenbag, H.J.; Scarpa, M.; Kiefer, O.; Kottke, D.; Sjöholm, E.; Öblom, H.; Sandler, N.; Hinrichs, W.L.J.; et al. Oromucosal films: From patient centricity to production by printing techniques. Expert Opin. Drug Deliv. 2019, 16, 981–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, M.S.; Kumar, T.P.; Davidson, R.; Kuppu, G.R.; Pathak, K.; Gowda, D.V. Printing Methods in the Production of Orodispersible Films. AAPS PharmSciTech 2021, 22, 129. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Zhu, L.; Yang, N.; Li, H.; Yang, Q. Recent advances of oral film as platform for drug delivery. Int. J. Pharm. 2021, 604, 120759. [Google Scholar] [CrossRef] [PubMed]
- Batista, P.; Duque, V.; Luzio-Vaz, A.; Pereira, A. Anxiety impact during COVID-19: A systematic review. J. Infect. Dev. Ctries. 2021, 15, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Bharti, K.; Mittal, P.; Mishra, B. Formulation and characterization of fast dissolving oral films containing buspirone hydrochloride nanoparticles using design of experiment. J. Drug Deliv. Sci. Technol. 2019, 49, 420–432. [Google Scholar] [CrossRef]
- Han, X.; Yan, J.; Ren, L.; Xue, M.; Yuan, Z.; Wang, T.; Yan, Z.; Yin, L.; Yang, L.; Qin, C. Preparation and evaluation of orally disintegrating film containing donepezil for Alzheimer disease. J. Drug Deliv. Sci. Technol. 2019, 54, 101321. [Google Scholar] [CrossRef]
- Łyszczarz, E.; Hofmanová, J.; Szafraniec-Szczęsny, J.; Jachowicz, R. Orodispersible films containing ball milled aripiprazole-poloxamer®407 solid dispersions. Int. J. Pharm. 2020, 575, 118955. [Google Scholar] [CrossRef]
- Batista, P.; Castro, P.; Madureira, A.R.; Sarmento, B.; Pintado, M. Development and characterization of chitosan microparticles-in-films for buccal delivery of bioactive peptides. Pharmaceuticals 2019, 12, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batista, P.; Castro, P.M.; Madureira, A.R.; Sarmento, B.; Pintado, M. Recent insights in the use of nanocarriers for the oral delivery of bioactive proteins and peptides. Peptides 2018, 101, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Castro, P.M.; Sousa, F.; Magalhães, R.; Ruiz-Henestrosa, V.M.P.; Pilosof, A.M.; Madureira, A.R.; Sarmento, B.; Pintado, M.E. Incorporation of beads into oral films for buccal and oral delivery of bioactive molecules. Carbohydr. Polym. 2018, 194, 411–421. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Yang, Z.; Teng, L. Nanotechnology and microtechnology in drug delivery systems. Dose-Response 2020, 18, 1559325820907810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borges, A.F.; Silva, C.; Coelho, J.F.; Simões, S. Outlining critical quality attributes (CQAs) as guidance for the development of orodispersible films. Pharm. Dev. Technol. 2017, 22, 237–245. [Google Scholar] [CrossRef]
- International Coffee Organization. Coffee Development Report 2020. Available online: https://www.ico.org/ (accessed on 22 February 2020).
- Grgic, J.; Diaz-Lara, F.J.; Del Coso, J.; Duncan, M.J.; Tallis, J.; Pickering, C.; Schoenfeld, B.J.; Mikulic, P. The effects of caffeine ingestion on measures of rowing performance: A systematic review and meta-analysis. Nutrients 2020, 12, 434. [Google Scholar] [CrossRef] [Green Version]
- Batista, P.; Oliveira-Silva, P.; Heym, N.; Pintado, M.E. Neuropsychophysiological measurements as a tool for neuromodulator oral films evaluation. Int. J. Pharm. Res. 2021, 13, 1051–1062. [Google Scholar] [CrossRef]
- O’Donnell, J.; Smith, R.; Turinske, C.; Young, L. Effects of Caffeine Consumption on Physiological Markers of Stress. J. Adv. Stud. Sci. 2019, 1–19. [Google Scholar]
- Alsabri, S.G.; Mari, W.O.; Younes, S.; Alsadawi, M.A.; Oroszi, T.L. Kinetic and dynamic description of caffeine. J. Caffeine Adenosine Res. 2018, 8, 3–9. [Google Scholar] [CrossRef]
- Oliveira-Silva, P.; Gonçalves, Ó.F. Responding empathically: A question of heart, not a question of skin. Appl. Psychophysiol. Biofeedback 2011, 36, 201–207. [Google Scholar] [CrossRef] [Green Version]
- Grassmann, M.; Vlemincx, E.; Von Leupoldt, A.; Mittelstädt, J.M.; Van den Bergh, O. Respiratory changes in response to cognitive load: A systematic review. Neural Plast. 2016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cacioppo, J.T.; Tassinary, L.G.; Berntson, G. Handbook of Psychophysiology; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Galbraith, G.C.; Kietzman, M.L.; Donchin, E. Neurophysiology and Psychophysiology: Experimental and Clinical Applications; Routledge: London, UK, 2021. [Google Scholar]
- Castro, P.M.; Fonte, P.; Oliveira, A.; Madureira, A.R.; Sarmento, B.; Pintado, M.E. Optimization of two biopolymer-based oral films for the delivery of bioactive molecules. Mater. Sci. Eng. C 2017, 76, 171–180. [Google Scholar] [CrossRef] [PubMed]
- de Castro, P.J.N.M. Association of Micro and Nanocarriers with Thin Films for Buccal Delivery of Bioactive Molecules; Universidade Catolica Portugesa: Porto, Portugal, 2018. [Google Scholar]
- Soares, A.P.; Pinheiro, A.P.; Costa, A.; Frade, C.S.; Comesaña, M.; Pureza, R. Adaptation of the international affective picture system (IAPS) for European Portuguese. Behav. Res. Methods 2015, 47, 1159–1177. [Google Scholar] [CrossRef] [Green Version]
- Adan, A.; Prat, G.; Fabbri, M.; Sànchez-Turet, M. Early effects of caffeinated and decaffeinated coffee on subjective state and gender differences. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2008, 32, 1698–1703. [Google Scholar] [CrossRef]
- Delgado, D.A.; Lambert, B.S.; Boutris, N.; McCulloch, P.C.; Robbins, A.B.; Moreno, M.R.; Harris, J.D. Validation of digital visual analog scale pain scoring with a traditional paper-based visual analog scale in adults. J. Am. Acad. Orthop. Surg. Glob. Res. Rev. 2018, 2, e088. [Google Scholar] [CrossRef]
- Fan, J.; McCandliss, B.D.; Sommer, T.; Raz, A.; Posner, M.I. Testing the efficiency and independence of attentional networks. J. Cogn. Neurosci. 2002, 14, 340–347. [Google Scholar] [CrossRef]
- McDonough, I.M.; Wood, M.M.; Miller, W.S., Jr. Focus: Attention Science: A Review on the Trajectory of Attentional Mechanisms in Aging and the Alzheimer’s Disease Continuum through the Attention Network Test. Yale J. Biol. Med. 2019, 92, 37. [Google Scholar]
- Hunt, M.G.; Momjian, A.J.; Wong, K.K. Effects of diurnal variation and caffeine consumption on Test of Variables of Attention (TOVA) performance in healthy young adults. Psychol. Assess. 2011, 23, 226. [Google Scholar] [CrossRef] [PubMed]
- Ágoston, C.; Urbán, R.; Király, O.; Griffiths, M.D.; Rogers, P.J.; Demetrovics, Z. Why do you drink caffeine? The development of the Motives for Caffeine Consumption Questionnaire (MCCQ) and its relationship with gender, age and the types of caffeinated beverages. Int. J. Ment. Health Addict. 2018, 16, 981–999. [Google Scholar] [CrossRef] [Green Version]
- Andreassi, J.L. Psychophysiology: Human Behavior and Physiological Response; Psychology Press: New York, NY, USA, 2010. [Google Scholar]
- di Fronso, S.; Robazza, C.; Bortoli, L.; Bertollo, M. Performance optimization in sport: A psychophysiological approach. Mot. Rev. Educ. Física 2017, 23, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Elitok, A.; Öz, F.; Panc, C.; Sarıkaya, R.; Sezikli, S.; Pala, Y.; Bugan, Ö.S.; Ateş, M.; Parıldar, H.; Ayaz, M.B. Acute effects of Red Bull energy drink on ventricular repolarization in healthy young volunteers: A prospective study. Anatol. J. Cardiol. 2016, 15, 919. [Google Scholar] [CrossRef] [PubMed]
- Lagast, S.; De Steur, H.; Gadeyne, S.; Hödl, S.; Staljanssens, W.; Vonck, K.; Boon, P.; Gellynck, X.; De Herdt, V. Heart rate, electrodermal responses and frontal alpha asymmetry to accepted and non-accepted solutions and drinks. Food Qual. Prefer. 2020, 82, 103893. [Google Scholar] [CrossRef]
- Ghiasi, S.; Greco, A.; Barbieri, R.; Scilingo, E.P.; Valenza, G. Assessing Autonomic Function from Electrodermal Activity and Heart Rate Variability During Cold-Pressor Test and Emotional Challenge. Sci. Rep. 2020, 10, 5406. [Google Scholar] [CrossRef] [PubMed]

| Continuous Measure | Min | Max | Mean | SD | ||
|---|---|---|---|---|---|---|
| Age | 18 | 46 | 24.15 | 7.71 | ||
| Categorical Measure | % | |||||
| Gender | ||||||
| Females | 61.5 | |||||
| Males | 38.5 | |||||
| Marital status | ||||||
| Single | 92.3 | |||||
| Married/In a relationship | 0 | |||||
| Divorced/separated | 7.7 | |||||
| Widower | 0 | |||||
| Education levels | ||||||
| Elementary School | 7.7 | |||||
| Secondary | 61.5 | |||||
| Higher education (degree) | 23.1 | |||||
| Higher education (master’s, doctorate and post-doc) | 7.7 | |||||
| Profession | ||||||
| Students | 84.6 | |||||
| Other profession | 15.4 |
| Categorical Measure | % | |
|---|---|---|
| Enjoy coffee | ||
| Yes | 84.6 | |
| No | 15.4 | |
| Frequency of coffee drinking per day | ||
| Up to once a day | 38.5 | |
| 2 times a day | 7.7 | |
| 2 to 3 times a day | 23.1 | |
| More than 3 times a day | 7.7 | |
| Rarely | 15.4 | |
| A few times a week | ||
| Reasons for drinking coffee | ||
| Wake up | 15.4 | |
| Socially | 7.7 | |
| Health | 7.7 | |
| Several (no specific) | 69.2 |
| Classifier | Optimal Parameters | Accuracy (%) | ||
|---|---|---|---|---|
| ECG | EDA | RA | ||
| Decision Trees | ||||
| Fine Tree | Maximum number of splits = 150 | 100 | 70 | 20 |
| Medium Tree | Maximum number of splits = 150 | 100 | 70 | 20 |
| SVMs | ||||
| Linear Kernel | Box constraint level = 5 | 100 | 80 | 0 |
| Quadratic Kernel | Box constraint level = 3 | 100 | 70 | 40 |
| Cubic Kernel | Box constraint level = 2 | 100 | 70 | 50 |
| Nearest Neighbor Classifiers | ||||
| Cosine KNN | Number of neighbors = 3 | 100 | 80 | 35 |
| Cubic KNN | Number of neighbors = 3 | 100 | 70 | 50 |
| Discriminant Analysis | ||||
| Linear | Covariance structure: Full | 100 | 75 | 0 |
| Logistic | Covariance structure: Full | 100 | 80 | 0 |
| Quadratic | Covariance structure: Full | 100 | 75 | 40 |
| XROC | - | 100 | 85 | 60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batista, P.; Rodrigues, P.M.; Ferreira, M.; Moreno, A.; Silva, G.; Alves, M.; Pintado, M.; Oliveira-Silva, P. Validation of Psychophysiological Measures for Caffeine Oral Films Characterization by Machine Learning Approaches. Bioengineering 2022, 9, 114. https://doi.org/10.3390/bioengineering9030114
Batista P, Rodrigues PM, Ferreira M, Moreno A, Silva G, Alves M, Pintado M, Oliveira-Silva P. Validation of Psychophysiological Measures for Caffeine Oral Films Characterization by Machine Learning Approaches. Bioengineering. 2022; 9(3):114. https://doi.org/10.3390/bioengineering9030114
Chicago/Turabian StyleBatista, Patrícia, Pedro Miguel Rodrigues, Miguel Ferreira, Ana Moreno, Gabriel Silva, Marco Alves, Manuela Pintado, and Patrícia Oliveira-Silva. 2022. "Validation of Psychophysiological Measures for Caffeine Oral Films Characterization by Machine Learning Approaches" Bioengineering 9, no. 3: 114. https://doi.org/10.3390/bioengineering9030114
APA StyleBatista, P., Rodrigues, P. M., Ferreira, M., Moreno, A., Silva, G., Alves, M., Pintado, M., & Oliveira-Silva, P. (2022). Validation of Psychophysiological Measures for Caffeine Oral Films Characterization by Machine Learning Approaches. Bioengineering, 9(3), 114. https://doi.org/10.3390/bioengineering9030114









