Smart-Data-Driven System for Alzheimer Disease Detection through Electroencephalographic Signals
Abstract
:1. Introduction
2. Materials
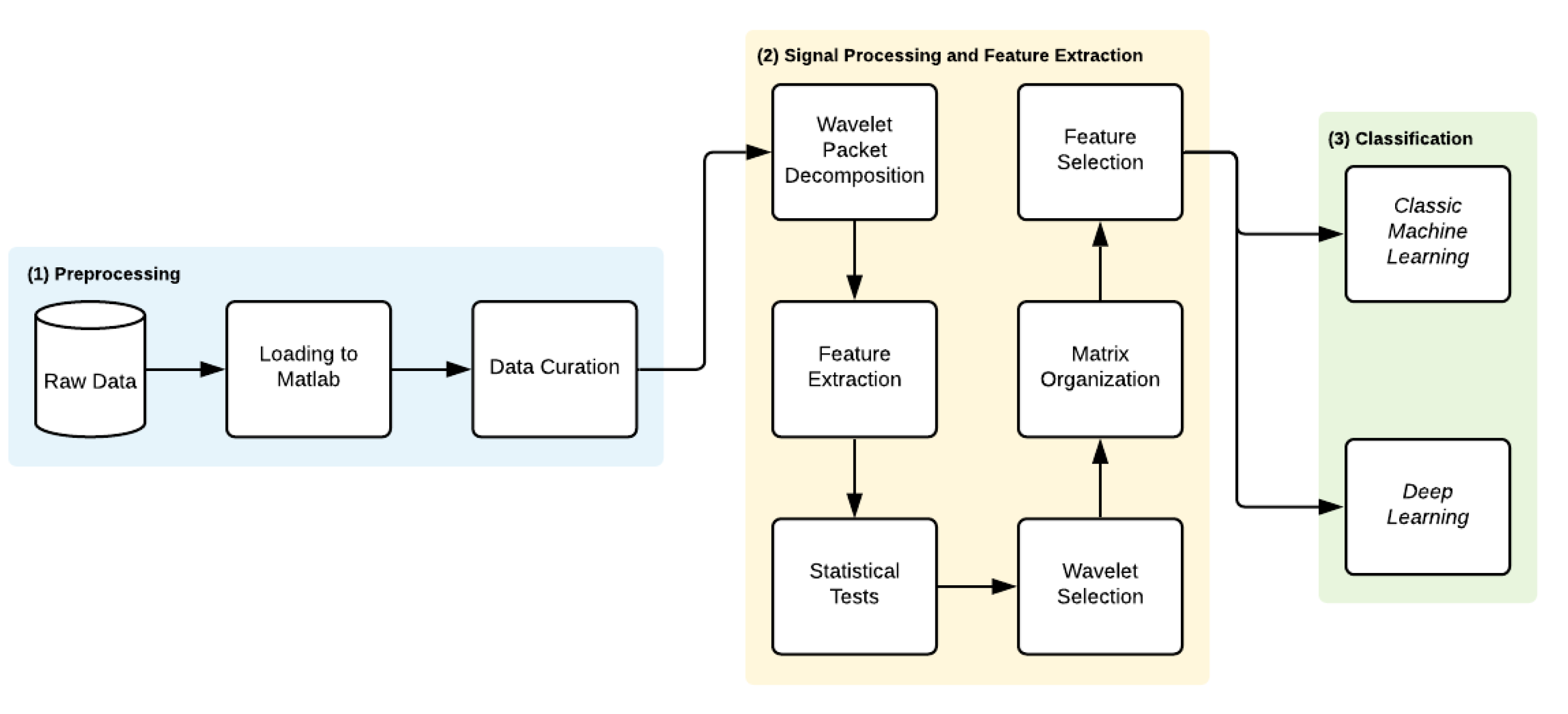
3. Methods
3.1. Preprocessing
3.2. Signal Processing and Feature Extraction
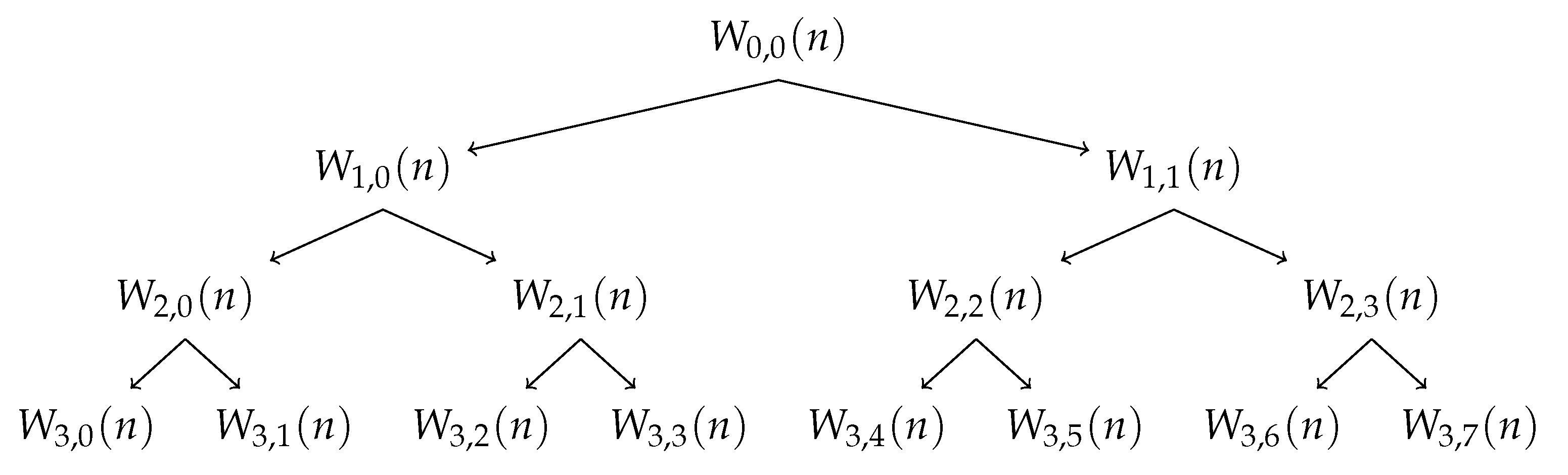
3.2.1. Wavelet Packet Decomposition
3.2.2. Non-Linear Analysis
Energy
Entropy
- Shannon Entropy [27]:
- Logarithmic Entropy [28]:
- Approximate Entropy:where N is the data length (suggested to be 1000 of the signal standard deviation), r is the similar tolerance (between 0.1 and 0.25) and e represents the embedding dimension (between 2 and 3). H is the Heaviside function that results from intermediate calculations [29].
- Sample Entropy:where,andas the factors N, r, e are the same used in Approximate Entropy, the recommended values are similar, and d is the Chebyshev distance between two sets of simultaneous data points [29].
- Permutation Entropy:where e is the embedding dimension and represents the probability of the permutation occurring [30].
Chaos Theory
- Hurst Exponent:where derives from power-law [26].
Fractal Analysis
- Higuchi Exponent:where k indicates the time interval, is the length of the curve in the k time interval and D is the Higuchi Exponent [32].
3.2.3. Feature Extraction Process
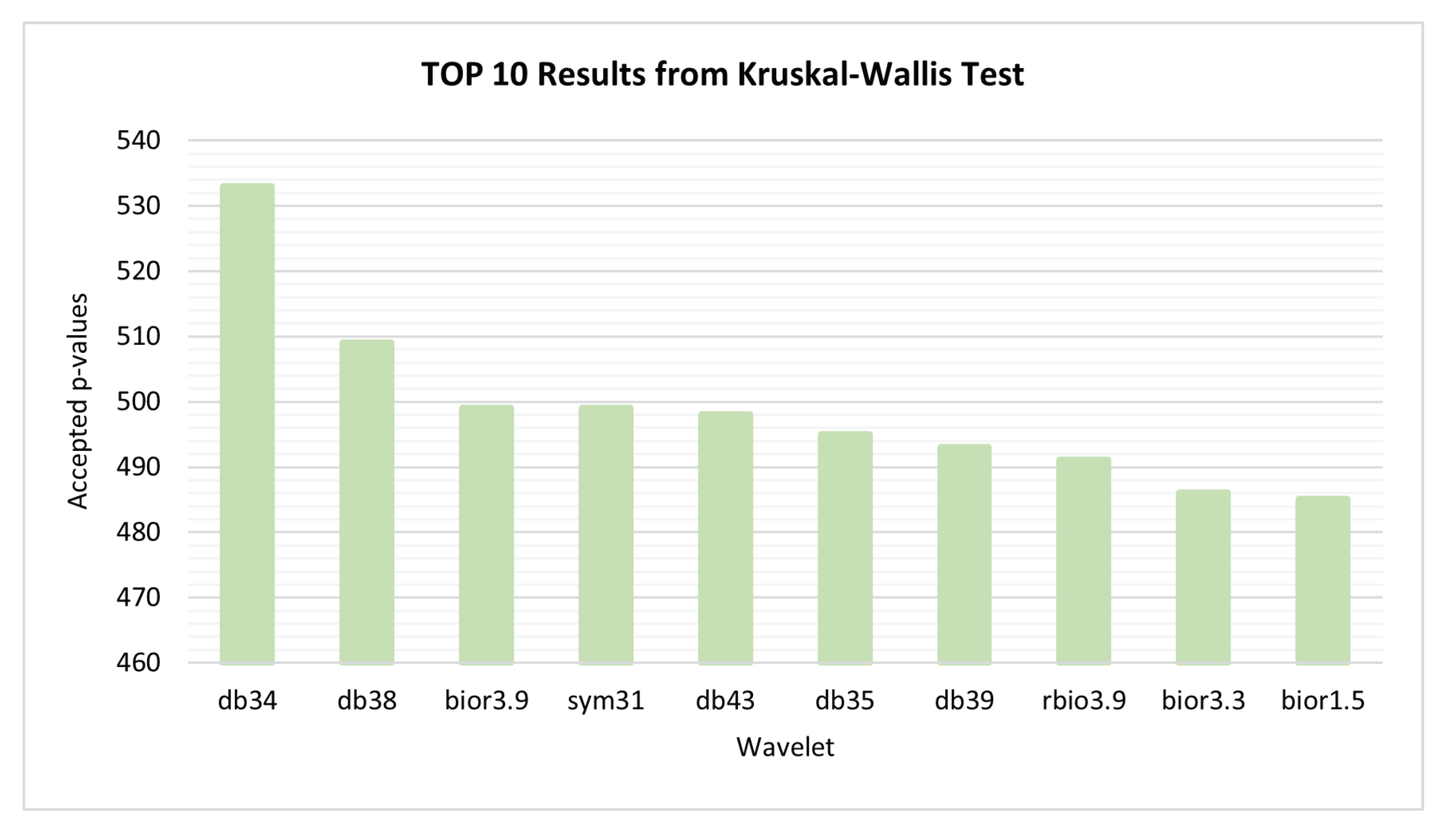
3.3. Wavelet Selection Process
3.4. Feature Selection and Classification Procedure
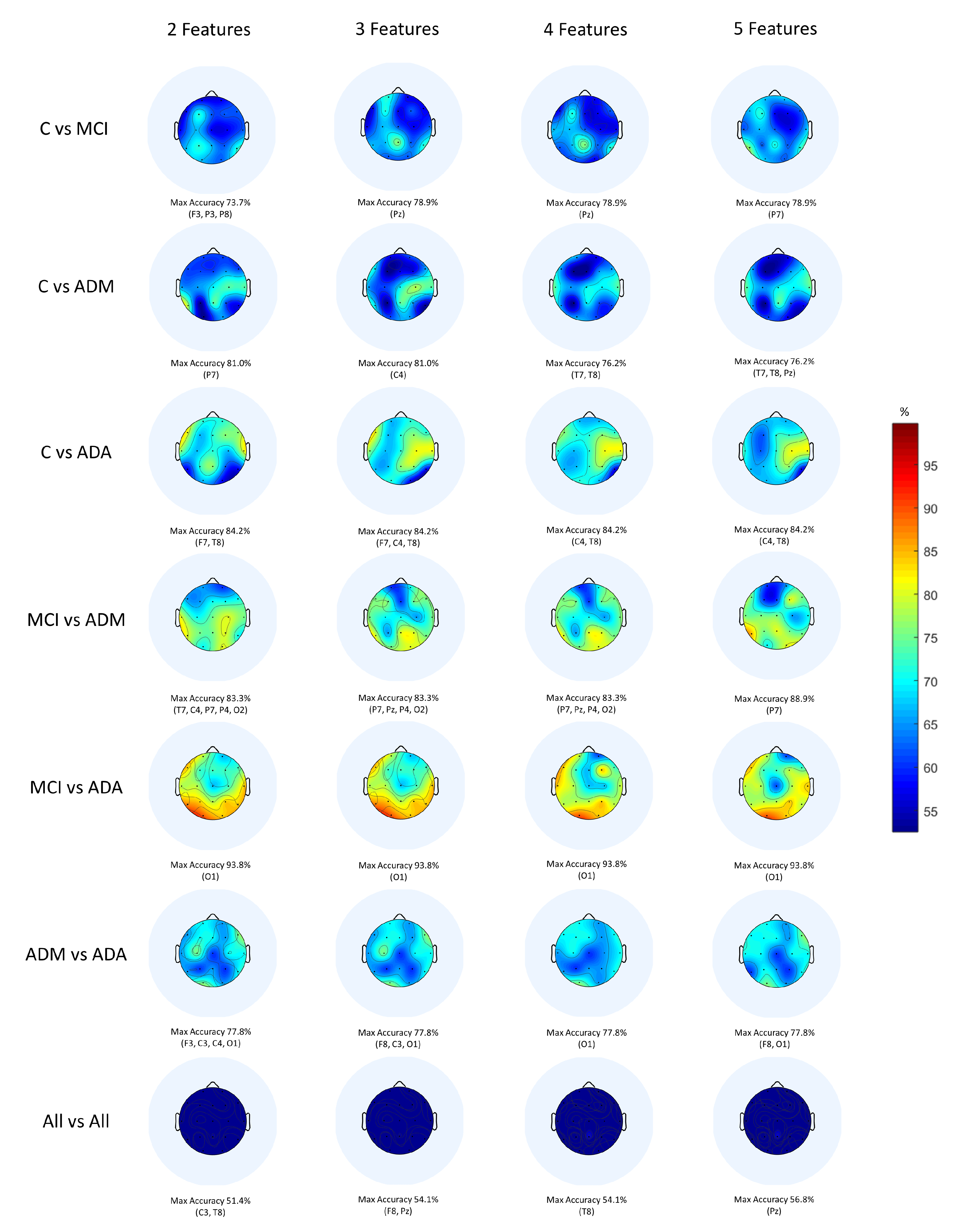
4. Results and Discussion
- 1
- Compared to other methods of diagnosing AD through EEG signals from the same database (Table 5), the proposed method outperformed the study developed by Rodrigues et al. [19] by 2% in the binary comparison MCI vs. ADM. It can be seen that CNNs have never been applied to this dataset, so this work is the first and the only one that follows this approach. Indeed, this works presents added value to the scientific community, as it has the potential to be improved and become a powerful tool for AD diagnosis in all its stages.
- 2
- Compared to other techniques of diagnosing AD through EEG signals from different databases (Table 6), it is observed that the present study outperformed the work carried out by Fiscon et al. [36] by 13% in the pair MCI vs. AD. It is noteworthy that the present study has the peculiarity of being the only one that applied the F-score technique, so it may have highly contributed to the good classification results.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations: Department of Economic and Social Affairs: Population Division. World Population Ageing 2015 Highlights; United Nations: New York, NY, USA, 2015. [Google Scholar]
- Hou, Y.; Dan, X.; Babbar, M.; Wei, Y.; Hasselbalch, S.G.; Croteau, D.L.; Bohr, V.A. Ageing as a risk factor for neurodegenerative disease. Nat. Rev. Neurol. 2019, 15, 565–581. [Google Scholar] [CrossRef]
- Alzheimer’s Association. 2021 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2021, 17, 327–406. [Google Scholar] [CrossRef]
- Shirai, K.; Iso, H. Dementia. In Social Determinants of Health in Non-Communicable Diseases; Springer: Singapore, 2020; pp. 105–123. [Google Scholar] [CrossRef]
- Alzheimer’s Association. 2019 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2019, 15, 321–387. [Google Scholar] [CrossRef]
- Revett, T.; Baker, G.; Jhamandas, J.; Kar, S. Glutamate system, amyloid β peptides and tau protein: Functional interrelationships and relevance to Alzheimer disease pathology. J. Psychiatry Neurosci. 2013, 38, 6–23. [Google Scholar] [CrossRef] [Green Version]
- Cassani, R.; Estarellas, M.; San-Martin, R.; Fraga, F.J.; Falk, T.H. Systematic Review on Resting-State EEG for Alzheimer’s Disease Diagnosis and Progression Assessment. Dis. Markers 2018, 2018, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Houmani, N.; Vialatte, F.; Gallego-Jutglà, E.; Dreyfus, G.; Nguyen-Michel, V.H.; Mariani, J.; Kinugawa, K. Diagnosis of Alzheimer’s disease with Electroencephalography in a differential framework. PLoS ONE 2018, 13, e0193607. [Google Scholar] [CrossRef] [Green Version]
- Scheltens, P.; Blennow, K.; Breteler, M.M.B.; de Strooper, B.; Frisoni, G.B.; Salloway, S.; der Flier, W.M.V. Alzheimer’s disease. Lancet 2016, 388, 505–517. [Google Scholar] [CrossRef]
- Fishman, E. Risk of Developing Dementia at Older Ages in the United States. Demography 2017, 54, 1897–1919. [Google Scholar] [CrossRef] [Green Version]
- Eratne, D.; Loi, S.M.; Farrand, S.; Kelso, W.; Velakoulis, D.; Looi, J.C. Alzheimer’s disease: Clinical update on epidemiology, pathophysiology and diagnosis. Australas. Psychiatry 2018, 26, 347–357. [Google Scholar] [CrossRef]
- Kester, M.I.; Scheltens, P. Dementia: The bare essentials. Pract. Neurol. 2009, 9, 241–251. [Google Scholar] [CrossRef]
- Pietrzak, K.; Czarnecka, K.; Mikiciuk-Olasik, E.; Szymanski, P. New Perspectives of Alzheimer Disease Diagnosis—The Most Popular and Future Methods. Med. Chem. 2018, 14, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.M.; Teixeira, J.P. Classification of Electroencephalogram signals using Artificial Neural Networks. In Proceedings of the 2010 3rd International Conference on Biomedical Engineering and Informatics, Yantai, China, 16–18 October 2010; Volume 2, pp. 808–812. [Google Scholar] [CrossRef] [Green Version]
- Blocka, K. EEG (Electroencephalogram): Purpose, Procedure, and Risks. Available online: https://www.healthline.com/health/eeg (accessed on 10 February 2022).
- Siuly, S.; Li, Y.; Zhang, Y. Electroencephalogram (EEG) and Its Background. In Health Information Science; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; pp. 3–21. [Google Scholar] [CrossRef]
- Sanei, S.; Chambers, J. EEG Signal Processing; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2007. [Google Scholar] [CrossRef]
- Rodrigues, P.M.; Freitas, D.R.; Teixeira, J.P.; Alves, D.; Garrett, C. Electroencephalogram Signal Analysis in Alzheimer’s Disease Early Detection. Int. J. Reliab. Qual. E-Healthc. 2018, 7, 40–59. [Google Scholar] [CrossRef]
- Rodrigues, P.M.; Bispo, B.C.; Garrett, C.; Alves, D.; Teixeira, J.P.; Freitas, D. Lacsogram: A New EEG Tool to Diagnose Alzheimer’s Disease. IEEE J. Biomed. Health Inf. 2021, 25, 3384–3395. [Google Scholar] [CrossRef] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [Green Version]
- Galea, M.; Woodward, M. Mini-Mental State Examination (MMSE). Aust. J. Physiother. 2005, 51, 198. [Google Scholar] [CrossRef] [Green Version]
- Bajaj, N. Wavelets for EEG Analysis. In Wavelet Theory; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Misiti, M.; Misiti, Y.; Oppenheim, G.; Poggi, J.M. Wavelet Toolbox User’s Guide Version 3; MathWorks: Natick, MA, USA, 2004. [Google Scholar]
- Sairamya, N.; Subathra, M.; Suviseshamuthu, E.S.; George, S.T. A new approach for automatic detection of focal EEG signals using wavelet packet decomposition and quad binary pattern method. Biomed. Signal Process. Control 2021, 63, 102096. [Google Scholar] [CrossRef]
- Al-Qammaz, A.Y.; Yusof, Y.; Ahamd, F.K. An enhanced Discrete Wavelet Packet Transform for Feature Extraction in Electroencephalogram Signals. In Proceedings of the International Conference on Imaging, Signal Processing and Communication—ICISPC 2017, Penang, Malaysia, 26–28 July 2017; ACM Press: New York, NY, USA, 2017. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, Z. EEG Signal Processing and Feature Extraction; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Das, A.B.; Bhuiyan, M.I.H. Discrimination and classification of focal and non-focal EEG signals using entropy-based features in the EMD-DWT domain. Biomed. Signal Process. Control. 2016, 29, 11–21. [Google Scholar] [CrossRef]
- Rodríguez-Sotelo, J.; Osorio-Forero, A.; Jiménez-Rodríguez, A.; Cuesta-Frau, D.; Cirugeda-Roldán, E.; Peluffo, D. Automatic Sleep Stages Classification Using EEG Entropy Features and Unsupervised Pattern Analysis Techniques. Entropy 2014, 16, 6573–6589. [Google Scholar] [CrossRef] [Green Version]
- Faust, O.; Bairy, M. Nonlinear analysis of physiological signals: A review. J. Mech. Med. Biol. 2012, 12, 1–21. [Google Scholar] [CrossRef]
- Olofsen, E.; Sleigh, J.; Dahan, A. Permutation entropy of the electroencephalogram: A measure of anaesthetic drug effect. Br. J. Anaesth. 2008, 101, 810–821. [Google Scholar] [CrossRef] [Green Version]
- Albertovich, T.D.; Aleksandrovna, R.I. The Fractal Analysis of the Images and Signals in Medical Diagnostics. In Fractal Analysis—Applications in Health Sciences and Social Sciences; InTechOpen: London, UK, 2017. [Google Scholar] [CrossRef] [Green Version]
- Wijayanto, I.; Hartanto, R.; Nugroho, H.A. Higuchi and Katz Fractal Dimension for Detecting Interictal and Ictal State in Electroencephalogram Signal. In Proceedings of the 2019 11th International Conference on Information Technology and Electrical Engineering (ICITEE), Pattaya, Thailand, 10–11 October 2019. [Google Scholar]
- Peck, R. Preliminary Edition of Statistics: Learning from Data (with Printed Access Card for Jmp); Thomson Learning: Boston, MA, USA, 2013. [Google Scholar]
- Priddy, K.L.; Keller, P.E. Artificial Neural Networks: An Introduction; SPIE: Bellingham, WA, USA, 2005. [Google Scholar] [CrossRef]
- Sevani, N.; Hermawan, I.; Jatmiko, W. Feature Selection based on F-score for Enhancing CTG Data Classification. In Proceedings of the 2019 IEEE International Conference on Cybernetics and Computational Intelligence (CyberneticsCom), Banda Aceh, Indonesia, 22–24 August 2019. [Google Scholar] [CrossRef]
- Fiscon, G.; Weitschek, E.; Cialini, A.; Felici, G.; Bertolazzi, P.; Salvo, S.D.; Bramanti, A.; Bramanti, P.; Cola, M.C.D. Combining EEG signal processing with supervised methods for Alzheimer’s patients classification. BMC Med. Infor. Decis. Mak. 2018, 18. [Google Scholar] [CrossRef]
- Safi, M.S.; Safi, S.M.M. Early detection of Alzheimer’s disease from EEG signals using Hjorth parameters. Biomed. Signal Process. Control 2021, 65, 102338. [Google Scholar] [CrossRef]
- Esteva, A.; Robicquet, A.; Ramsundar, B.; Kuleshov, V.; DePristo, M.; Chou, K.; Cui, C.; Corrado, G.; Thrun, S.; Dean, J. A guide to deep learning in healthcare. Nat. Med. 2019, 25, 24–29. [Google Scholar] [CrossRef]

| Subjects | C | MCI | ADM | ADA |
|---|---|---|---|---|
| # | 11 | 8 | 11 | 8 |
| Age Average | 74 | 80 | 79 | 79 |
| MMSE Average | 28.68 | 26.29 | 18.89 | 11.50 |
| Classifier | Optimal Parameters | |
|---|---|---|
| Decision Trees | Fine Tree—FT | Maximum number of splits = 150 |
| Medium Tree—MT | Maximum number of splits = 150 | |
| Coarse Tree—CT | Maximum number of splits = 150 | |
| Discriminant Analysis | Linear Discriminant—LD | Covariance structure: Full |
| Quadratic Discriminant—QD | Covariance structure: Full | |
| Logistic Regression - LR | Covariance structure: Full | |
| Naive Bayes | Gaussian Naive Bayes— GNB | - |
| Kernel Naive Bayes—KNB | - | |
| SVM | Linear SVM—LSVM | Box constraint level = 3 |
| Quadratic SVM—QSVM | Box constraint level = 3 | |
| Cubic SVM—CSVM | Box constraint level = 4 | |
| Fine Gaussian SVM—FGSVM | Box constraint level = 3 | |
| Medium Gaussian SVM—MGSVM | Box constraint level = 3 | |
| Coarse Gaussian SVM—CGSVM | Box constraint level = 1 | |
| KNN | Fine KNN—FKNN | Number of neighbors = 3 |
| Medium KNN—MKNN | Number of neighbors = 3 | |
| Coarse KNN—CKNN | Number of neighbors = 3 | |
| Cosine KNN CosKNN | Number of neighbors = 3 | |
| Cubic KNN—CubKNN | Number of neighbors = 3 | |
| Weighted KNN—WKNN | Number of neighbors = 3 | |
| Ensemble | Boosted Trees—BossT | Maximum number of splits = 150 |
| Bagged Trees—Bagt | Maximum number of splits = 150 | |
| Subspace Discriminant—SubD | Covariance structure: Full | |
| Subspace KNN—SubKNN | Number of neighbors = 3 | |
| RUSBoosted Trees—RUSBT | Maximum number of splits = 150 | |
| CNN | imageInputLayer = 1 | |
| convolution2dLayer = 1 | ||
| reluLayer = 1 | ||
| fullyConnectedLayer = 3 | ||
| softmaxLayer = 1 | ||
| classificationLayer = 1 | ||
| Training algorithm = adam | ||
| Max epochs = 1000 | ||
| Features | Maximum Accuracy | |||||
|---|---|---|---|---|---|---|
| C-MCI | C-ADM | C-ADA | MCI-ADM | MCI-ADA | ADM-ADA | |
| 1080 | 73.7% | 76.2% | 68.4% | 83.3% | 87.5% | 72.2% |
| 20% | 73.7% | 76.2% | 78.9% | 88.9% | 93.8% | 72.2% |
| 10% | 78.9% | 76.2% | 78.9% | 88.9% | 93.8% | 72.2% |
| 5% | 78.9% | 76.2% | 78.9% | 83.3% | 93.8% | 77.8% |
| 20 | 73.7% | 81.0% | 78.9% | 88.9% | 93.8% | 77.8% |
| 15 | 78.9% | 81.0% | 78.9% | 88.9% | 93.8% | 77.8% |
| 10 | 78.9% | 76.2% | 78.9% | 88.9% | 87.5% | 77.8% |
| 5 | 78.9% | 76.2% | 84.2% | 88.9% | 93.8% | 77.8% |
| 4 | 78.9% | 76.2% | 84.2% | 83.3% | 93.8% | 77.8% |
| 3 | 78.9% | 81.0% | 84.2% | 83.3% | 93.8% | 77.8% |
| 2 | 73.7% | 81.0% | 84.2% | 83.3% | 93.8% | 77.8% |
| Comparison | Classic ML | Accuracy (Position) | DL | Accuracy (Position) |
|---|---|---|---|---|
| C vs. MCI | FT, MT, & CT | 78.9% (P7 & Pz) | CNN | 78.9% (P8) |
| C vs. ADM | CSVM & FGSVM | 81.0% (C4 & P7) | CNN | 76.2% (Pz) |
| C vs. ADA | LSVM & GNB | 84.2% (F7, C4 & T8) | CNN | 78.9% (F7 & F8) |
| MCI vs. ADM | CosKNN | 88.9% (P7) | CNN | 83.2% (Pz) |
| MCI vs. ADA | FT, MT & CT | 93.8% (O1) | CNN | 93.8 (P4) |
| ADM vs. ADA | FKNN & SubD | 77.8% (F3, F8, C3, C4 & O1) | CNN | 72.2% (Fz, F4 & C4) |
| All vs. All | MGSVM | 56.8% (Pz) | CNN | 51.4% (Pz) |
| Study | Signal Processing | Features | Feature Selection | Best Classifier | Classification Accuracy |
|---|---|---|---|---|---|
| [18] | Multiband Spectral Analysis via DWT | RP, Spectral Ratios, Maxima, Minima and Zero Crossing | KW Test | ANN | C vs. MCI—77% |
| C vs. AD—95% | |||||
| MCI vs. AD—83% | |||||
| All vs. All—90% | |||||
| [19] | Multiband Cepstral and Lacstral Analysis via DWT | Cepstral and Lacstral Distances | Genetic Algorithms | ANN | C vs. MCI—98% |
| C vs. ADM—96% | |||||
| C vs. ADA—96% | |||||
| C vs. ADM-ADA—96% | |||||
| MCI vs. ADM—87% | |||||
| MCI vs. ADA—99% | |||||
| MCI vs. ADM-ADA—94% | |||||
| All vs. All—96% | |||||
| Present Study | Nonlinear and Multiband Analysis via DWPT | Nonlinear and Statistic Parameters | F-score | SVM | C vs. MCI—79% |
| C vs. ADM—81% | |||||
| C vs. ADA—84% | |||||
| MCI vs. ADM—89% | |||||
| MCI vs. ADA—94% | |||||
| ADM vs. ADA—78% | |||||
| All vs. All—57% |
| Study | Signal Processing | Features | Feature Selection | Best Classifier | Classification Accuracy |
|---|---|---|---|---|---|
| [36] | Fourier and Wavelet Analysis via FFT and DWT | Fourier and Wavelet Coefficients | Not applied | DT | C vs. AD—83% |
| C vs. MCI—92% | |||||
| MCI vs. AD—79% | |||||
| [37] | Multiband Analysis via DWT and EMD | Variance, Kurtosis, Skewness, Shannon Entropy, Sure Entropy and Hjorth Parameters | Not applied | KNN | C vs. AD1 vs. AD2—98% |
| Present Study | Nonlinear and Multiband Analysis via DWPT | Nonlinear and Statistic Parameters | F-score | SVM | C vs. MCI—79% |
| C vs. ADM—81% | |||||
| C vs. ADA—84% | |||||
| MCI vs. ADM—89% | |||||
| MCI vs. ADA—94% | |||||
| ADM vs. ADA—78% | |||||
| All vs. All—57% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araújo, T.; Teixeira, J.P.; Rodrigues, P.M. Smart-Data-Driven System for Alzheimer Disease Detection through Electroencephalographic Signals. Bioengineering 2022, 9, 141. https://doi.org/10.3390/bioengineering9040141
Araújo T, Teixeira JP, Rodrigues PM. Smart-Data-Driven System for Alzheimer Disease Detection through Electroencephalographic Signals. Bioengineering. 2022; 9(4):141. https://doi.org/10.3390/bioengineering9040141
Chicago/Turabian StyleAraújo, Teresa, João Paulo Teixeira, and Pedro Miguel Rodrigues. 2022. "Smart-Data-Driven System for Alzheimer Disease Detection through Electroencephalographic Signals" Bioengineering 9, no. 4: 141. https://doi.org/10.3390/bioengineering9040141
APA StyleAraújo, T., Teixeira, J. P., & Rodrigues, P. M. (2022). Smart-Data-Driven System for Alzheimer Disease Detection through Electroencephalographic Signals. Bioengineering, 9(4), 141. https://doi.org/10.3390/bioengineering9040141








