In Situ “Humanization” of Porcine Bioprostheses: Demonstration of Tendon Bioprostheses Conversion into Human ACL and Possible Implications for Heart Valve Bioprostheses
Abstract
1. Introduction
2. Anti-Porcine Antibody Response in Humans Implanted with Porcine Bioprostheses
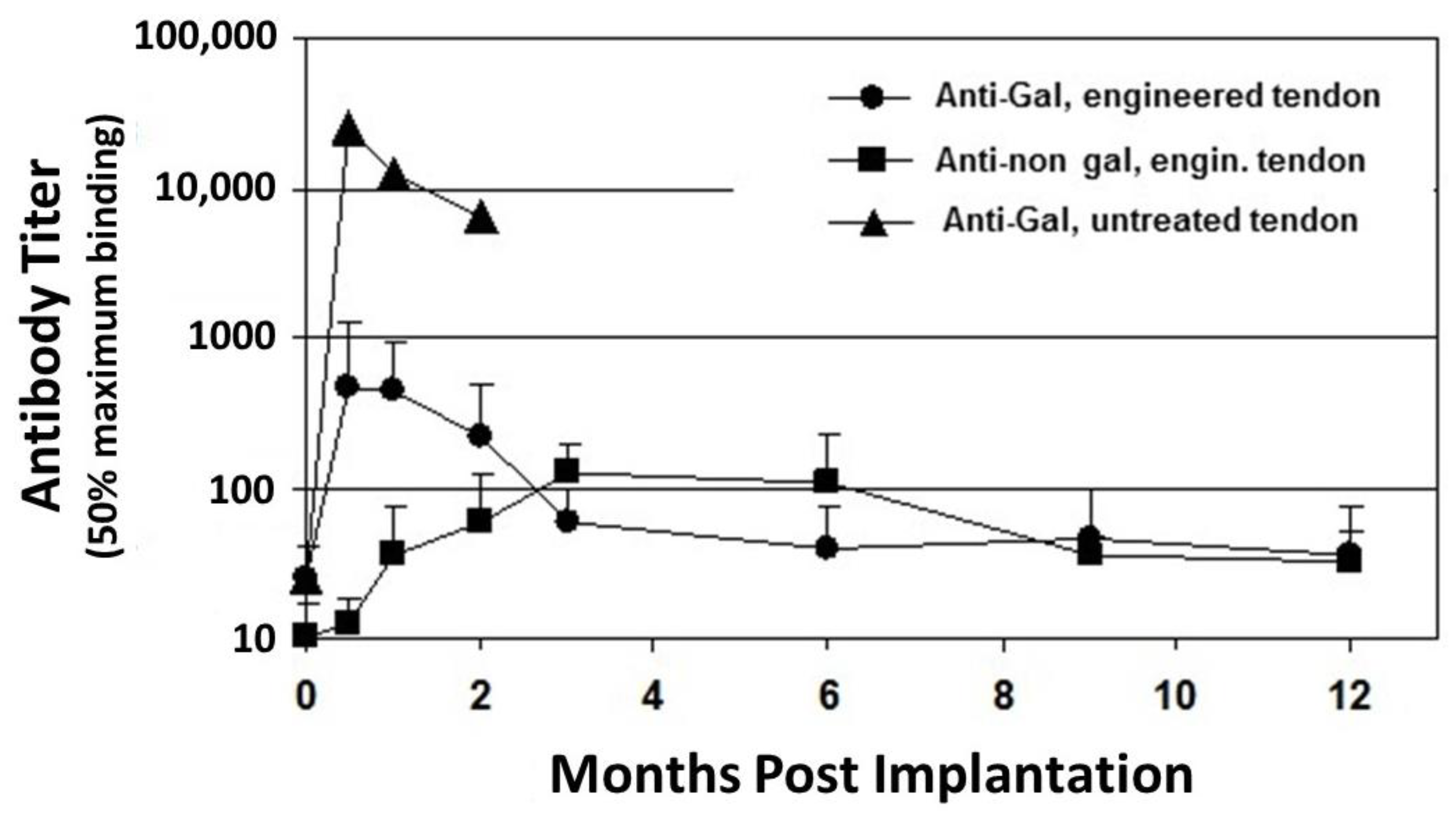
2.1. Anti-Gal Antibody
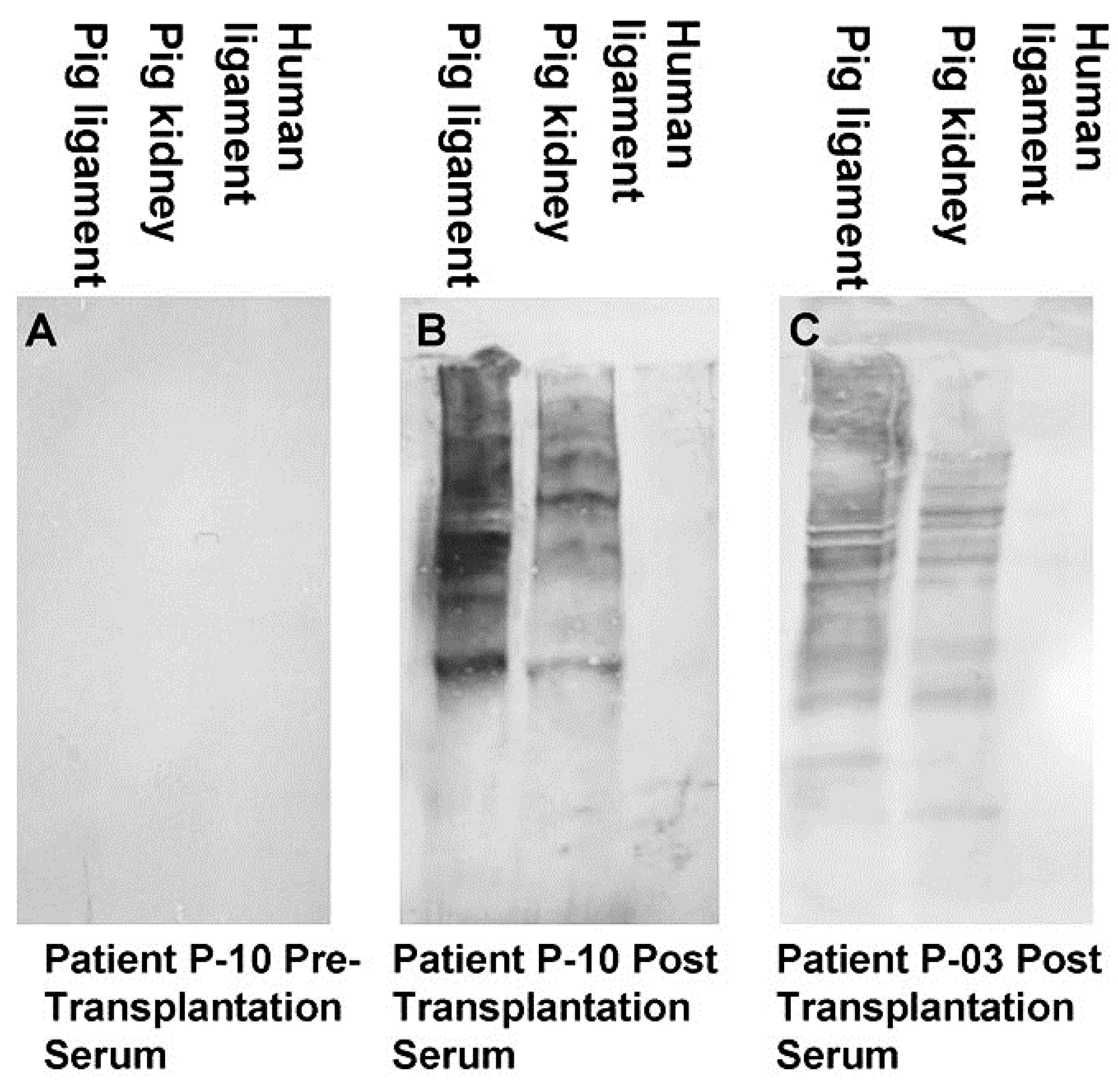
2.2. Anti-Non Gal Antibodies
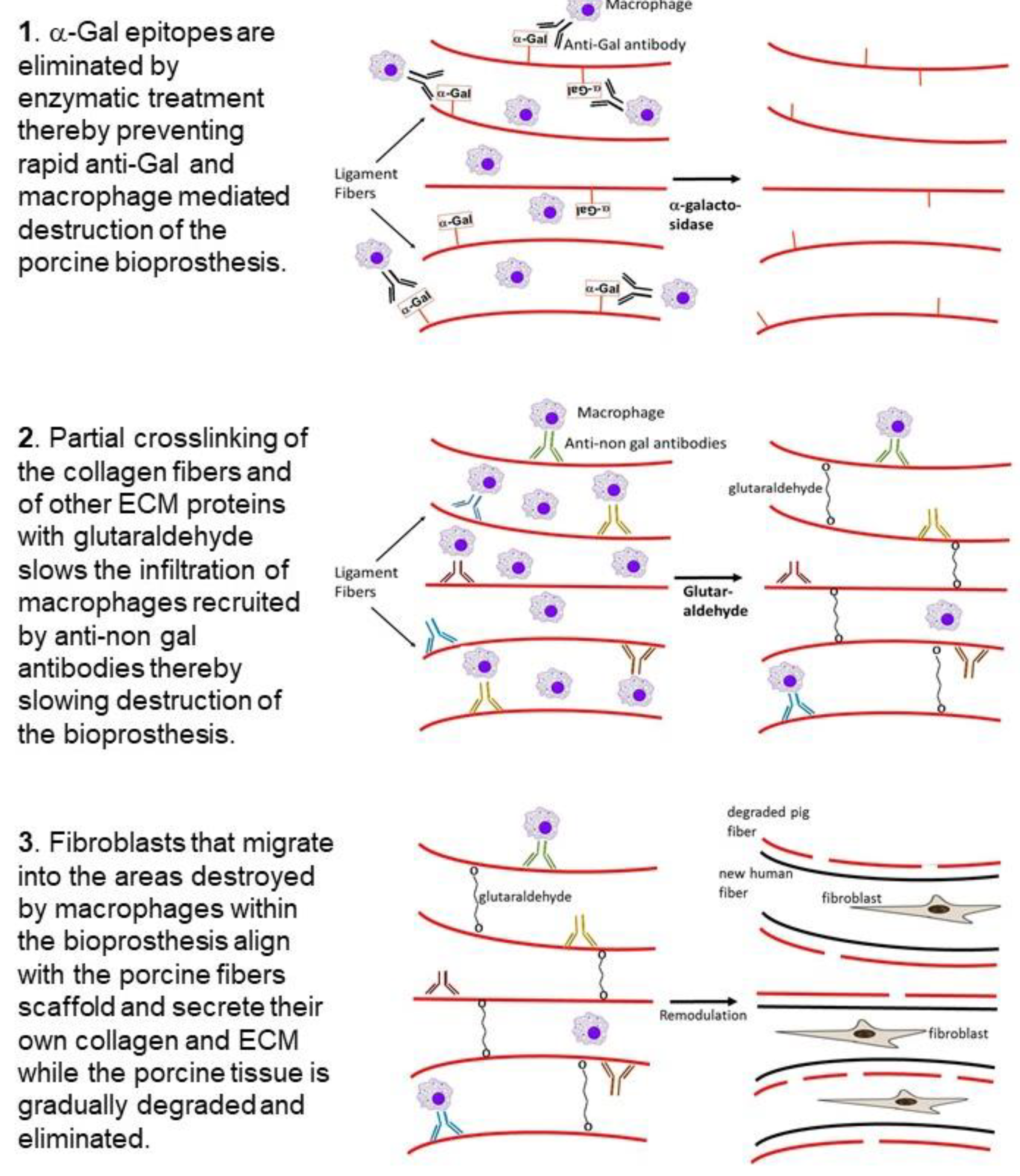
3. Hypothesis on Humanization of Porcine Bioprostheses
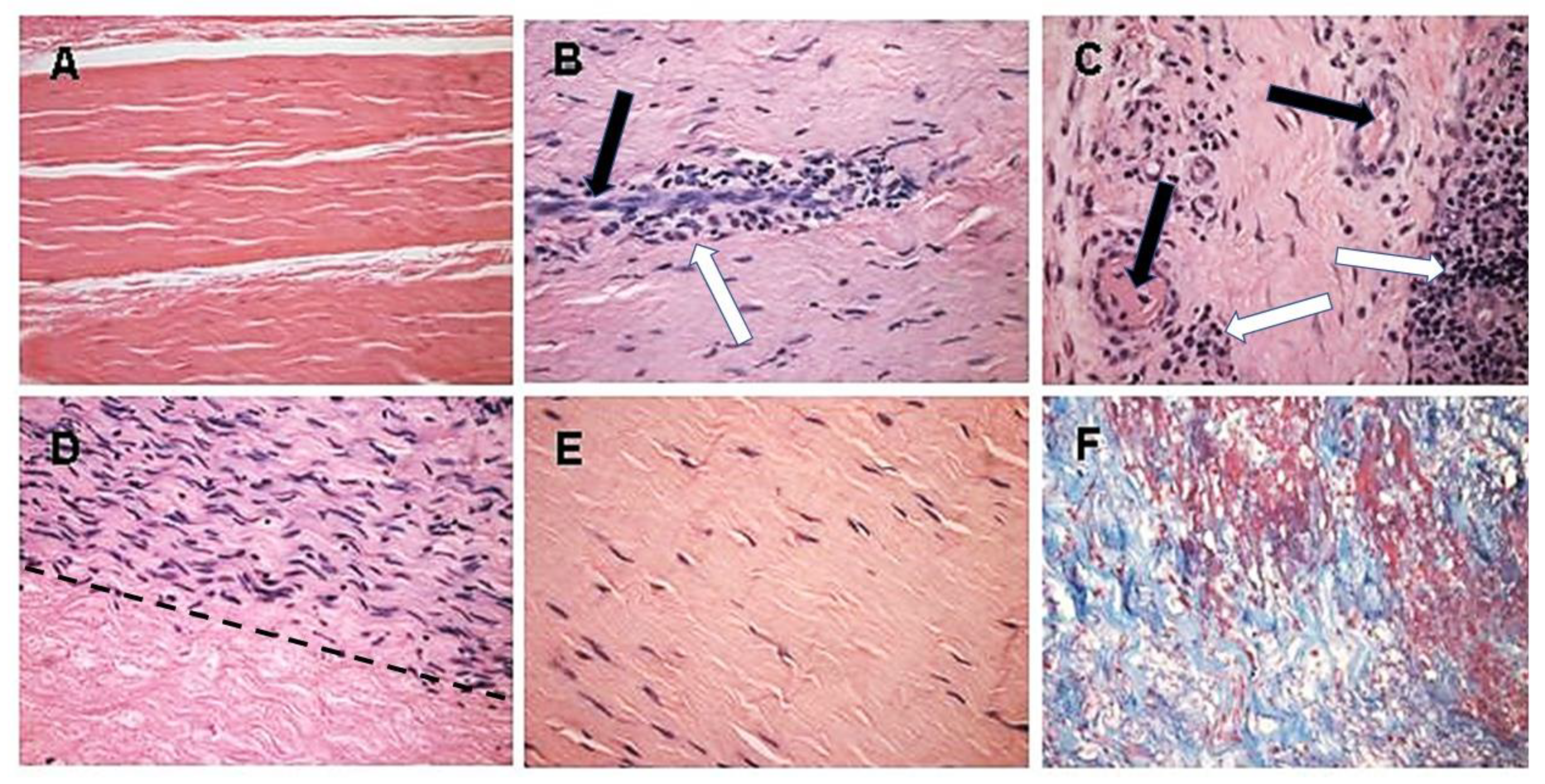
4. Processing of Porcine Patellar-Tendon into Bioprostheses, and Pre-Clinical Studies in Monkeys
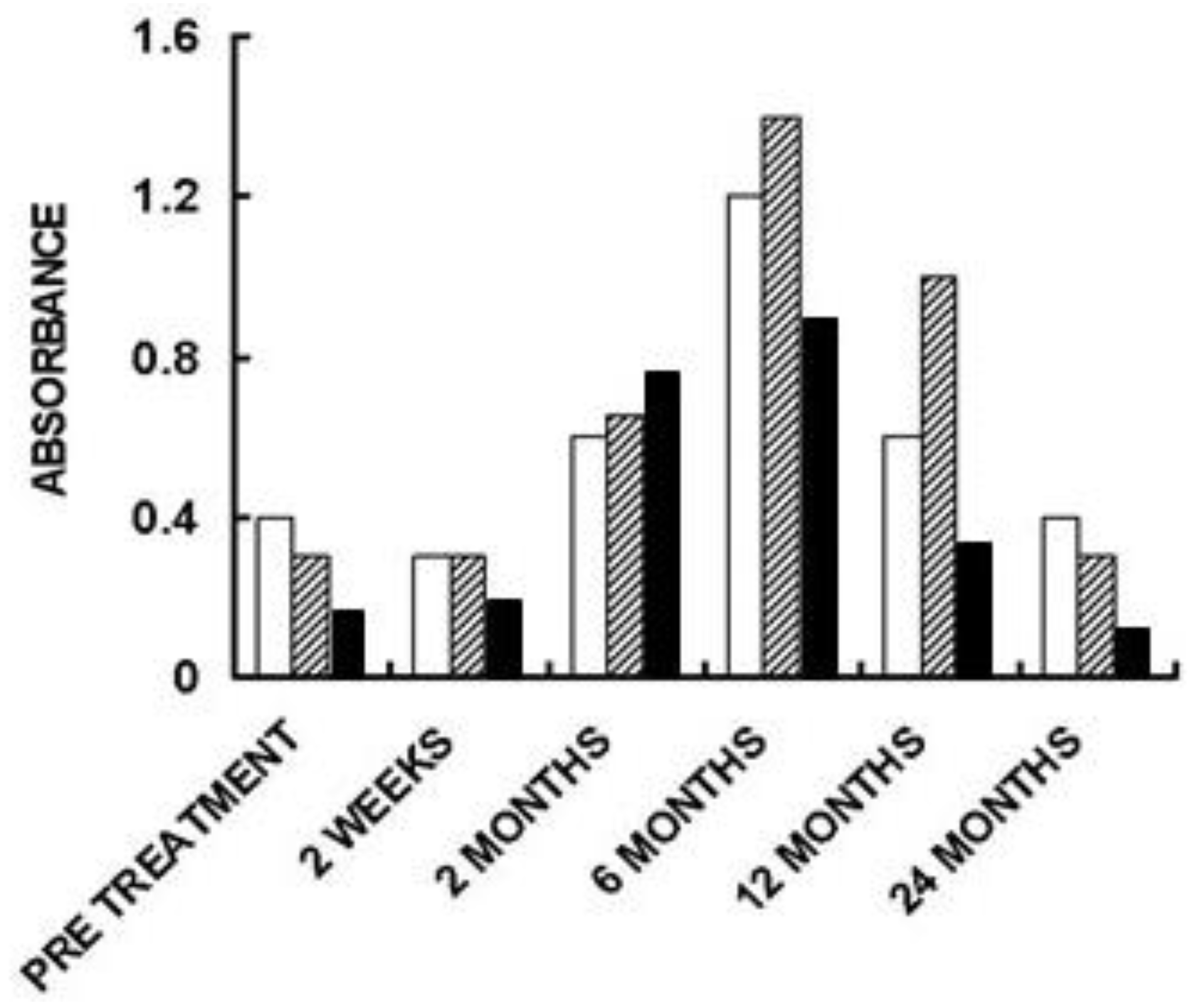
5. Implantation of Porcine BTB Bioprosthesis in Patients with Torn ACL
6. Potential Translation of ACL Studies to Porcine BHV Implants
6.1. Removal of the α-Gal Epitope from the Porcine BHV
6.2. Glutaraldehyde Crosslinking of the Porcine BHV
6.3. Monitoring Humanization of the Implanted Porcine BHV
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Manji, R.A.; Lee, W.; Cooper, D.K.C. Xenograft bioprosthetic heart valves: Past, present and future. Int. J. Surg. 2015, 23 Pt B, 280–284. [Google Scholar] [CrossRef]
- Soares, J.S.; Feaver, K.R.; Zhang, W.; Kamensky, D.; Aggarwal, A.; Sacks, M.S. Biomechanical behavior of bioprosthetic heart valve heterograft tissues: Characterization, simulation, and performance. Cardiovasc. Eng. Technol. 2016, 7, 309–351. [Google Scholar] [CrossRef] [PubMed]
- Goldstone, A.B.; Chiu, P.; Baiocchi, M.; Lingala, B.; Patrick, W.L.; Fischbein, M.P.; Woo, Y.J. Mechanical or biologic prostheses for aortic-valve and mitral-valve replacement. N. Engl. J. Med. 2017, 377, 1847–1857. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Furnary, A.P.; Li, H.F.; Grunkemeier, G.L. Bioprosthetic aortic valve durability: A meta-regression of published studies. Ann. Thorac. Surg. 2017, 104, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Head, S.J.; Çelik, M.; Kappetein, A.P. Mechanical versus bioprosthetic aortic valve replacement. Eur. Heart J. 2017, 38, 2183–2191. [Google Scholar] [CrossRef] [PubMed]
- Stein, P.D.; Wang, C.H.; Riddle, J.M.; Magilligan, D.J., Jr. Leukocytes, platelets, and surface microstructure of spontaneously degenerated porcine bioprosthetic valves. J. Card. Surg. 1988, 3, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Grabenwöger, M.; Fitzal, F.; Gross, C.; Hutschala, D.; Böck, P.; Brucke, P.; Wolner, E. Different modes of degeneration in autologous and heterologous heart valve prostheses. J. Heart Valve Dis. 2000, 9, 104–111. [Google Scholar]
- Manji, R.A.; Zhu, L.F.; Nijjar, N.K.; Rayner, D.C.; Korbutt, G.S.; Churchill, T.A.; Rajotte, R.V.; Koshal, A.; Ross, D.B. Glutaraldehyde-fixed bioprosthetic heart valve conduits calcify and fail from xenograft rejection. Circulation 2006, 114, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmi, M.H.; Mertsching, H.; Wilhelmi, M.; Leyh, R.; Haverich, A. Role of inflammation in allogeneic and xenogeneic heart valve degeneration: Immunohistochemical evaluation of inflammatory endothelial cell activation. J. Heart Valve Dis. 2003, 12, 520–526. [Google Scholar]
- Siddiqui, R.F.; Abraham, J.R.; Butany, J. Bioprosthetic heart valves: Modes of failure. Histopathology 2009, 55, 135–144. [Google Scholar] [CrossRef]
- Nair, V.; Law, K.B.; Li, A.Y.; Phillips, K.R.; David, T.E.; Butany, J. Characterizing the inflammatory reaction in explanted Medtronic Freestyle stentless porcine aortic bioprosthesis over a 6-year period. Cardiovasc. Pathol. 2012, 21, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Vesely, I. Heart valve tissue engineering. Circ. Res. 2005, 97, 743–755. [Google Scholar] [CrossRef] [PubMed]
- Bloch, O.; Golde, P.; Dohmen, P.M.; Posner, S.; Konertz, W.; Erdbrügger, W. Immune response in patients receiving a bioprosthetic heart valve: Lack of response with decellularized valves. Tissue Eng. Part A 2011, 17, 2399–2405. [Google Scholar] [CrossRef] [PubMed]
- Ramm, R.; Hilfiker, A. After decellularization of porcine heart valves: Non Gal antigenic epitopes detectable by non-conditioned human sera. Xenotransplantation 2014, 21, 193. [Google Scholar] [CrossRef]
- Nam, J.; Choi, S.Y.; Sung, S.C.; Lim, H.G.; Park, S.S.; Kim, S.H.; Kim, Y.J. Changes of the structural and biomechanical properties of the bovine pericardium after the removal of α-gal epitopes by decellularization and α-galactosidase treatment. Korean J. Thorac. Cardiovasc. Surg. 2012, 45, 380–389. [Google Scholar] [CrossRef]
- Erdbrügger, W.; Konertz, W.; Dohmen, P.M.; Posner, S.; Ellerbrok, H.; Brodde, O.E.; Robenek, H.; Modersohn, D.; Pruss, A.; Holinski, S.; et al. Decellularized xenogenic heart valves reveal remodeling and growth potential in vivo. Tissue Eng. 2006, 12, 2059–2068. [Google Scholar] [CrossRef]
- Bastian, F.; Stelzmuller, M.E.; Kratochwill, K.; Kasimir, M.T.; Simon, P.; Weigel, G. IgG deposition and activation of the classical complement pathway involvement in the activation of human granulocytes by decellularized porcine heart valve tissue. Biomaterials 2008, 29, 1824–1832. [Google Scholar] [CrossRef]
- Helder, M.R.K.; Stoyles, N.J.; Tefft, B.J.; Hennessy, R.S.; Hennessy, R.R.C.; Dyer, R.; Witt, T.; Simari, R.D.; Lerman, A. Xenoantigenicity of porcine decellularized valves. J. Cardiothorac. Surg. 2017, 12, 56. [Google Scholar] [CrossRef]
- Ruffer, A.; Purbojo, A.; Cicha, I.; Glöckler, M.; Potapov, S.; Dittrich, S.; Cesnjevar, R.A. Early failure of xenogenous de-cellularised pulmonary valve conduits—A word of caution! Eur. J. Cardiothorac. Surg. 2010, 38, 78–85. [Google Scholar] [CrossRef]
- Liao, J.; Joyce, E.M.; Sacks, M.S. Effects of decellularization on the mechanical and structural properties of the porcine aortic valve leaflet. Biomaterials 2008, 29, 1065–1074. [Google Scholar] [CrossRef]
- Choi, S.Y.; Jeong, H.J.; Lim, H.G.; Park, S.S.; Kim, S.H.; Kim, Y.J. Elimination of α-gal xenoreactive epitope: α-galactosidase treatment of porcine heart valves. J. Heart Valve Dis. 2012, 21, 387–397. [Google Scholar] [PubMed]
- Lim, H.-G.; Choi, S.Y.; Yoon, E.J.; Kim, S.H.; Kim, Y.J. In vivo efficacy of α-galactosidase as possible promise for prolonged durability of bioprosthetic heart valve using α1,3-galactosyltransferase knockout mouse. Tissue Eng. A 2013, 19, 2339–2348. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.P.; Sulzenbacher, G.; Yuan, H.; Bennett, E.P.; Pietz, G.; Saunders, K.; Spence, J.; Nudelman, E.; Levery, S.B.; White, T.; et al. Bacterial glycosidases for the production of universal red blood cells. Nat. Biotechnol. 2007, 25, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Smood, B.; Hara, H.; Cleveland, D.C.; Cooper, D.K.C. In Search of the ideal valve: Optimizing genetic modifications to prevent bioprosthetic degeneration. Ann. Thorac. Surg. 2019, 108, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Wang, Y.; Chen, L.; Wang, R.; Li, C.; Li, X.; Fang, B.; Ren, X.; Ruan, M.; Liu, J.; et al. Reducing immunoreactivity of porcine bioprosthetic heart valves by genetically-deleting three major glycan antigens, GGTA1/β4GalNT2/CMAH. Acta Biomater. 2018, 72, 196–205. [Google Scholar] [CrossRef]
- Lutz, A.J.; Li, P.; Estrada, J.L.; Sidner, R.A.; Chihara, R.K.; Downey, S.M.; Burlak, C.; Wang, Z.Y.; Reyes, L.M.; Ivary, B.; et al. Double knockout pigs deficient in N-glycolylneuraminic acid and galactose α-1,3-galactose reduce the humoral barrier to xenotransplantation. Xenotransplantation 2013, 20, 27–35. [Google Scholar] [CrossRef]
- Wagner, F.; Holzapfel, B.M.; McGovern, J.A.; Shafiee, A.; Baldwin, J.G.; Martine, L.C.; Lahr, C.A.; Wunner, F.M.; Friis, T.; Bas, O.; et al. Humanization of bone and bone marrow in an orthotopic site reveals new potential therapeutic targets in osteosarcoma. Biomaterials 2018, 171, 230–246. [Google Scholar] [CrossRef]
- Galili, U.; Rachmilewitz, E.A.; Peleg, A.; Flechner, I. A unique natural human IgG antibody with anti-α-galactosyl specificity. J. Exp. Med. 1984, 160, 1519–1531. [Google Scholar] [CrossRef]
- Towbin, H.; Rosenfelder, G.; Wieslander, J.; Avila, J.L.; Rojas, M.; Szarfman, A.; Esser, K.; Nowack, H.; Timpl, R. Circulating antibodies to mouse laminin in Chagas disease, American cutaneous leishmaniasis, and normal individuals recognize terminal galactosyl [α1-3]-galactose epitopes. J. Exp. Med. 1987, 166, 419–432. [Google Scholar] [CrossRef]
- Parker, W.; Lin, S.S.; Yu, P.B.; Sood, A.; Nakamura, Y.C.; Song, A.; Everett, M.L.; Platt, J.L. Naturally occurring anti-α-galactosyl antibodies: Relationship to xenoreactive anti-α-galactosyl antibodies. Glycobiology 1999, 9, 865–873. [Google Scholar] [CrossRef]
- McMorrow, I.M.; Comrack, C.A.; Sachs, D.H.; DerSimonian, H. Heterogeneity of human anti-pig natural antibodies cross-reactive with the Gal [α1,3] Galactose epitope. Transplantation 1997, 64, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Galili, U.; Macher, B.A.; Buehler, J.; Shohet, S.B. Human natural anti-α-galactosyl IgG. II. The specific recognition of α(1 → 3)-linked galactose residues. J. Exp. Med. 1985, 162, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Galili, U.; Clark, M.R.; Shohet, S.B.; Buehler, J.; Macher, B.A. Evolutionary relationship between the anti-Gal antibody and the Galα1→3Gal epitope in primates. Proc. Natl. Acad. Sci. USA 1987, 84, 1369–1373. [Google Scholar] [CrossRef] [PubMed]
- Teneberg, S.; Lönnroth, I.; Torres Lopez, J.F.; Galili, U.; Halvarsson, M.O.; Angström, J.; Karlsson, K.A. Molecular mimicry in the recognition of glycosphingolipids by Galα3Galß4GlcNAcß-binding Clostridium difficile toxin A, human natural anti-α-galactosyl IgG and the monoclonal antibody Gal-13: Characterization of a binding-active human glycosphingolipid, non-identical with the animal receptor. Glycobiology 1996, 6, 599–609. [Google Scholar] [PubMed]
- Galili, U.; Shohet, S.B.; Kobrin, E.; Stults, C.L.M.; Macher, B.A. Man, apes, and Old World monkeys differ from other mammals in the expression of α-galactosyl epitopes on nucleated cells. J. Biol. Chem. 1988, 263, 17755–17762. [Google Scholar] [CrossRef]
- Oriol, R.; Candelier, J.J.; Taniguchi, S.; Balanzino, L.; Peters, L.; Niekrasz, M.; Hammer, C.; Cooper, D.K. Major carbohydrate epitopes in tissues of domestic and African wild animals of potential interest for xenotransplantation research. Xenotransplantation 1999, 6, 79–89. [Google Scholar] [CrossRef]
- Teranishi, K.; Mañez, R.; Awwad, M.; Cooper, D.K. Anti-Gal α1-3Gal IgM and IgG antibody levels in sera of humans and Old World non-human primates. Xenotransplantation 2002, 9, 148–154. [Google Scholar] [CrossRef]
- Good, A.H.; Cooper, D.C.K.; Malcolm, A.J.; Ippolito, R.M.; Koren, E.; Neethling, F.A.; Ye, Y.; Zuhdi, N.; Lamontagne, L.R. Identification of carbohydrate structures which bind human anti-porcine antibodies: Implication for discordant xenografting in man. Transplant. Proc. 1992, 24, 559–562. [Google Scholar]
- Galili, U. Interaction of the natural anti-Gal antibody with α-galactosyl epitopes: A major obstacle for xenotransplantation in humans. Immunol. Today 1993, 14, 480–482. [Google Scholar] [CrossRef]
- Sandrin, M.S.; Vaughan, H.A.; Dabkowski, P.L.; McKenzie, I.F.C. Anti-pig IgM antibodies in human serum react predominantly with Gal (αl-3)Gal epitopes. Proc. Natl. Acad. Sci. USA 1993, 90, 11391–11395. [Google Scholar] [CrossRef]
- Cooper, D.K.C.; Good, A.H.; Koren, E.; Oriol, R.; Malcolm, A.J.; Ippolito, R.M.; Neethling, F.A.; Ye, Y.; Romano, E.; Zuhdi, N. Identification of α-galactosyl and other carbohydrate epitopes that are bound by human anti-pig antibodies: Relevance to discordant xenografting in man. Transpl. Immunol. 1993, 1, 198–205. [Google Scholar] [CrossRef]
- Collins, B.H.; Cotterell, A.H.; McCurry, K.R.; Alvarado, C.G.; Magee, J.C.; Parker, W.; Platt, J.L. Cardiac xenografts between primate species provide evidence for the importance of the a-galactosyl. determinant in hyperacute rejection. J. Immunol. 1995, 154, 5500–5510. [Google Scholar]
- Xu, Y.; Lorf, T.; Sablinski, T.; Gianello, P.; Bailin, M.; Monroy, R.; Kozlowski, T.; Awwad, M.; Cooper, D.K.; Sachs, D.H. Removal of anti-porcine natural antibodies from human and nonhuman primate plasma in vitro and in-vivo by a Galα1-3Galβ1-4βGlc-X immunoaffinity column. Transplantation 1998, 65, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Simon, P.M.; Neethling, F.A.; Taniguchi, S.; Goode, P.L.; Zopf, D.; Hancock, W.W.; Cooper, D.K. Intravenous infusion of Galα-1-3Gal oligosaccharides in baboons delays hyperacute rejection of porcine heart xenografts. Transplantation 1998, 65, 346–353. [Google Scholar] [CrossRef]
- Galili, U.; Anaraki, F.; Thall, A.; Hill-Black, C.; Radic, M. One percent of human circulating B lymphocytes are capable of producing the natural anti-Gal antibody. Blood 1993, 82, 2485–2493. [Google Scholar] [CrossRef] [PubMed]
- Galili, U.; Mandrell, R.E.; Hamadeh, R.M.; Shohet, S.B.; Griffiss, J.M. Interaction between human natural anti-α-galactosyl immunoglobulin G and bacteria of the human flora. Infect. Immun. 1988, 56, 1730–1737. [Google Scholar] [CrossRef]
- Posekany, K.J.; Pittman, H.K.; Bradfield, J.F.; Haisch, C.E.; Verbanac, K.M. Induction of cytolytic anti-Gal antibodies in α-1,3-galactosyltransferase gene knockout mice by oral inoculation with Escherichia coli O86, B7 bacteria. Infect. Immun. 2002, 70, 6215–6222. [Google Scholar] [CrossRef]
- Mañez, R.; Blanco, F.J.; Díaz, I.; Centeno, A.; Lopez-Pelaez, E.; Hermida, M.; Davies, H.F.; Katopodis, A. Removal of bowel aerobic gram-negative bacteria is more effective than immunosuppression with cyclophosphamide and steroids to decrease natural α-galactosyl IgG antibodies. Xenotransplantation 2001, 8, 15–23. [Google Scholar] [CrossRef]
- Konakci, K.Z.; Bohle, B.; Blumer, R.; Hoetzenecker, W.; Roth, G.; Moser, B.; Boltz-Nitulescu, G.; Gorlitzer, M.; Klepetko, W.; Wolner, E.; et al. α-Gal on bioprostheses: Xenograft immune response in cardiac surgery. Eur. J. Clin. Investig. 2005, 35, 17–23. [Google Scholar] [CrossRef]
- Mangold, A.; Szerafin, T.; Hoetzenecker, K.; Hacker, S.; Lichtenauer, M.; Niederpold, T.; Nickl, S.; Dworschak, M.; Blumer, R.; Auer, J.; et al. α-Gal specific IgG immune response after implantation of bioprostheses. Thorac. Cardiovasc. Surg. 2009, 57, 191–195. [Google Scholar] [CrossRef]
- Park, C.S.; Oh, S.S.; Kim, Y.E.; Choi, S.Y.; Lim, H.G.; Ahn, H.; Kim, Y.J. Anti-α-Gal antibody response following xenogeneic heart valve implantation in adults. J. Heart Valve Dis. 2013, 22, 222–229. [Google Scholar] [PubMed]
- Park, C.S.; Park, S.S.; Choi, S.Y.; Yoon, S.H.; Kim, W.H.; Kim, Y.J. Anti α-gal immune response following porcine bioprosthesis implantation in children. J. Heart Valve Dis. 2010, 19, 124–130. [Google Scholar]
- Stone, K.R.; Walgenbach, A.W.; Turek, T.J.; Somers, D.L.; Wicomb, W.; Galili, U. Anterior cruciate ligament reconstruction with a porcine xenograft: A serologic, histologic, and biomechanical study in primates. Arthroscopy 2007, 23, 411–419. [Google Scholar] [CrossRef]
- Stone, K.R.; Ayala, G.; Goldstein, J.; Hurst, R.; Walgenbach, A.; Galili, U. Porcine cartilage transplants in the cynomolgus monkey. III. Transplantation of α-galactosidase-treated porcine cartilage. Transplantation 1998, 65, 1577–1583. [Google Scholar] [CrossRef] [PubMed]
- Lila, N.; McGregor, C.G.; Carpentier, S.; Rancic, J.; Byrne, G.W.; Carpentier, A. Gal knockout pig pericardium: New source of material for heart valve bioprostheses. J. Heart Lung Transplant. 2010, 29, 538–543. [Google Scholar] [CrossRef] [PubMed]
- McGregor, C.G.; Carpentier, A.; Lila, N.; Logan, J.S.; Byrne, G.W. Cardiac xenotransplantation technology provides materials for improved bioprosthetic heart valves. J. Thorac. Cardiovasc. Surg. 2011, 141, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Galili, U.; Tibell, A.; Samuelsson, B.; Rydberg, L.; Groth, C.G. Increased anti-Gal activity in diabetic patients transplanted with fetal porcine islet cell clusters. Transplantation 1995, 59, 1549–1556. [Google Scholar] [CrossRef]
- Galili, U.; Chen, Z.C.; Tanemura, M.; Seregina, T.; Link, C.J. Understanding the induced antibody response (in xenograft recipients). GRAFT 2001, 4, 32–35. [Google Scholar] [CrossRef]
- Galili, U. The Natural Anti-Gal Antibody as Foe Turned Friend in Medicine; Elsevier/Academic Press: London, UK, 2018; p. 14. [Google Scholar]
- Lai, L.; Kolber-Simonds, D.; Park, K.W.; Cheong, H.T.; Greenstein, J.L.; Im, G.S.; Samuel, M.; Bonk, A.; Rieke, A.; Day, B.N.; et al. Production of α-1,3-galactosyltransferase knockout pigs by nuclear transfer cloning. Science 2002, 295, 1089–1092. [Google Scholar] [CrossRef]
- Phelps, C.J.; Koike, C.; Vaught, T.D.; Boone, J.; Wells, K.D.; Chen, S.H.; Ball, S.; Specht, S.M.; Polejaeva, I.A.; Monahan, J.A.; et al. Production of α1,3-galactosyltransferase-deficient pigs. Science 2003, 299, 411–414. [Google Scholar] [CrossRef]
- Merrick, J.M.; Zadarlik, K.; Milgrom, F. Characterization of the Hanganutziu-Deicher [serum-sickness] antigen as gangliosides containing N-glycolylneuraminic acid. Int. Arch. Allergy Appl. Immunol. 1978, 57, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.; Hurst, R. Anti-N-glycolylneuraminic acid antibodies identified in healthy human serum. Xenotransplantation 2002, 9, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.H.; Tangvoranuntakul, P.; Varki, A. Effects of natural human antibodies against a nonhuman sialic acid that metabolically incorporates into activated and malignant immune cells. J. Immunol. 2005, 175, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Muchmore, E.A.; Diaz, S.; Varki, A. A structural difference between the cell surfaces of humans and the great apes. Am. J. Phys. Anthropol. 1998, 107, 187–198. [Google Scholar] [CrossRef]
- Gagneux, P.; Varki, A. Evolutionary considerations in relating oligosaccharide diversity to biological function. Glycobiology 1999, 9, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Varki, A. Colloquium paper: Uniquely human evolution of sialic acid genetics and biology. Proc. Natl. Acad. Sci. USA 2010, 107 (Suppl. S2), 8939–8946. [Google Scholar] [CrossRef]
- Padler-Karavani, V.; Yu, H.; Cao, H.; Chokhawala, H.; Karp, F.; Varki, N.; Chen, X.; Varki, A. Diversity in specificity, abundance, and composition of anti-Neu5Gc antibodies in normal humans: Potential implications for disease. Glycobiology 2008, 18, 818–830. [Google Scholar] [CrossRef]
- Padler-Karavani, V.; Varki, A. Potential impact of the non-human sialic acid N-glycolylneuraminic acid on transplant rejection risk. Xenotransplantation 2011, 18, 1–5. [Google Scholar] [CrossRef]
- Galili, U. Induced anti-non gal antibodies in human xenograft recipients. Transplantation 2012, 93, 11–16. [Google Scholar] [CrossRef]
- Stone, K.R.; Abdel-Motal, U.; Walgenbach, A.W.; Turek, T.J.; Galili, U. Replacement of human anterior cruciate ligaments with pig ligaments: A model for anti-non-gal antibody response in long-term xenotransplantation. Transplantation 2007, 83, 211–219. [Google Scholar] [CrossRef]
- Singer, A.J.; Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef]
- Koh, T.J.; DiPietro, L.A. Inflammation and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef] [PubMed]
- Smigiel, K.S.; Parks, W.C. Macrophages, Wound Healing, and Fibrosis: Recent Insights. Curr. Rheumatol. Rep. 2018, 20, 17I. [Google Scholar] [CrossRef] [PubMed]
- Butler, D.L. Anterior cruciate ligament: Its normal response and replacement. J. Orthop. Res. 1989, 7, 910–921. [Google Scholar] [CrossRef] [PubMed]
- Amiel, S.; Kuiper, S. Experimental studies on anterior cruciate ligament grafts. In Knee Ligaments: Structure, Function, Injury and Repair; Daniel, D., Ed.; Raven Press: Baltimore, MD, USA, 1990; pp. 379–388. [Google Scholar]
- Davarinos, N.; O’Neill, B.J.; Curtin, W.A. Brief History of Anterior Cruciate Ligament Reconstruction. Adv. Orthop. Surg. 2014, 706042. [Google Scholar] [CrossRef]
- Galili, U.; LaTemple, D.C.; Radic, M.Z. A sensitive assay for measuring α-gal epitope expression on cells by a monoclonal anti-Gal antibody. Transplantation 1998, 65, 1129–1132. [Google Scholar] [CrossRef]
- Van Der Merwe, W.; Lind, M.; Faunø, P.; Van Egmond, K.; Zaffagnini, S.; Marcacci, M.; Cugat, R.; Verdonk, R.; Ibañez, E.; Guillen, P.; et al. Xenograft for anterior cruciate ligament reconstruction was associated with high graft processing infection. J. Exp. Orthop. 2020, 7, 79. [Google Scholar] [CrossRef]
- Little, D.; Thompson, J.W.; Dubois, L.G.; Ruch, D.S.; Moseley, M.A.; Guilak, F. Proteomic differences between male and female anterior cruciate ligament and patellar tendon. PLoS ONE 2014, 9, e96526. [Google Scholar] [CrossRef]
- Kodigepalli, K.M.; Thatcher, K.; West, T.; Howsmon, D.P.; Schoen, F.J.; Sacks, M.S.; Breuer, C.K.; Lincoln, J. Biology and Biomechanics of the Heart Valve Extracellular Matrix. J. Cardiovasc. Dev. Dis. 2020, 7, 57. [Google Scholar] [CrossRef]
- Naso, F.; Gandaglia, A.; Bottio, T.; Tarzia, V.; Nottle, M.B.; d’Apice, A.J.; Cowan, P.J.; Cozzi, E.; Galli, C.; Lagutina, I.; et al. First quantification of α-gal epitope in current glutaraldehyde-fixed heart valve bioprostheses. Xenotransplantation 2013, 20, 252–261. [Google Scholar] [CrossRef]
- Zhu, A.; Monahan, C.; Zhang, Z.; Hurst, R.; Leng, L.; Goldstein, J. High-level expression and purification of coffee bean α-galactosidase produced in the yeast Pichia pastoris. Arch. Biochem. Biophys. 1995, 324, 65–70. [Google Scholar] [CrossRef]
- Dor, F.J.; Tseng, Y.L.; Cheng, J.; Moran, K.; Sanderson, T.M.; Lancos, C.J.; Shimizu, A.; Yamada, K.; Awwad, A.; Sachs, D.H.; et al. α1,3-Galactosyltransferase gene-knockout miniature swine produce natural cytotoxic anti-Gal antibodies. Transplantation 2004, 78, 15–20. [Google Scholar] [CrossRef]
- Fang, J.; Walters, A.; Hara, H.; Long, C.; Yeh, P.; Ayares, D.; Cooper, D.K.; Bianchi, J. Anti-gal antibodies in α1,3-galactosyltransferase gene knockout pigs. Xenotransplantation 2012, 19, 305–310. [Google Scholar] [CrossRef]
- Galili, U. α1,3Galactosyltransferase knockout pigs produce the natural anti-Gal antibody and simulate the evolutionary appearance of this antibody in primates. Xenotransplantation 2013, 20, 267–276. [Google Scholar] [CrossRef]
- Liao, K.; Seifter, E.; Hoffman, D.; Yellin, E.L.; Frater, R.W. Improved postfixation treatment of glutaraldehyde fixed porcine aortic valves by monosodium glutamate. Artif. Organs 1992, 16, 267–272. [Google Scholar] [CrossRef]
- Shang, H.; Claessens, S.M.; Tian, B.; Wright, G.A. Aldehyde reduction in a novel pericardial tissue reduces calcification using rabbit intramuscular model. J. Mater. Sci. Mater. Med. 2017, 28, 16. [Google Scholar] [CrossRef]
- Zabirnyk, A.; Ferrer, M.D.; Bogdanova, M.; Pérez, M.M.; Salcedo, C.; Kaljusto, M.L.; Kvitting, J.P.; Stensløkken, K.O.; Perelló, J.; Vaage, J. SNF472, a novel anti-crystallization agent, inhibits induced calcification in an in vitro model of human aortic valve calcification. Vasc. Pharmacol. 2019, 122–123, 106583. [Google Scholar] [CrossRef]
- Shen, S.H.; Sung, H.W.; Tu, R.; Hata, C.; Lin, D.; Noishiki, Y.; Quijano, R.C. Characterization of a polyepoxy compound fixed porcine heart valve bioprosthesis. J. Appl. Biomater. 1994, 5, 159–162. [Google Scholar] [CrossRef]
- van Wachem, P.B.; Brouwer, L.A.; Zeeman, R.; Dijkstra, P.J.; Feijen, J.; Hendriks, M.; Cahalan, P.T.; van Luyn, M.J. In vivo behavior of epoxy-crosslinked porcine heart valve cusps and walls. J. Biomed. Mater. Res. 2000, 53, 18–27. [Google Scholar] [CrossRef]
- Girardot, J.M.; Girardot, M.N. Amide cross-linking: An alternative to glutaraldehyde fixation. J. Heart Valve Dis. 1996, 5, 518–525. [Google Scholar]
- Leong, J.; Munnelly, A.; Liberio, B.; Cochrane, L.; Vyavahare, N. Neomycin and carbodiimide crosslinking as an alternative to glutaraldehyde for enhanced durability of bioprosthetic heart valves. J. Biomater. Appl. 2013, 27, 948–960. [Google Scholar] [CrossRef] [PubMed]
- Sacks, M.S.; Schoen, F.J. Collagen fiber disruption occurs independent of calcification in clinically explanted bioprosthetic heart valves. J. Biomed. Mater. Res. 2002, 62, 359–371. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galili, U.; Stone, K.R. In Situ “Humanization” of Porcine Bioprostheses: Demonstration of Tendon Bioprostheses Conversion into Human ACL and Possible Implications for Heart Valve Bioprostheses. Bioengineering 2021, 8, 10. https://doi.org/10.3390/bioengineering8010010
Galili U, Stone KR. In Situ “Humanization” of Porcine Bioprostheses: Demonstration of Tendon Bioprostheses Conversion into Human ACL and Possible Implications for Heart Valve Bioprostheses. Bioengineering. 2021; 8(1):10. https://doi.org/10.3390/bioengineering8010010
Chicago/Turabian StyleGalili, Uri, and Kevin R. Stone. 2021. "In Situ “Humanization” of Porcine Bioprostheses: Demonstration of Tendon Bioprostheses Conversion into Human ACL and Possible Implications for Heart Valve Bioprostheses" Bioengineering 8, no. 1: 10. https://doi.org/10.3390/bioengineering8010010
APA StyleGalili, U., & Stone, K. R. (2021). In Situ “Humanization” of Porcine Bioprostheses: Demonstration of Tendon Bioprostheses Conversion into Human ACL and Possible Implications for Heart Valve Bioprostheses. Bioengineering, 8(1), 10. https://doi.org/10.3390/bioengineering8010010



