The Platelet Concentrates Therapy: From the Biased Past to the Anticipated Future
Abstract
1. Introduction
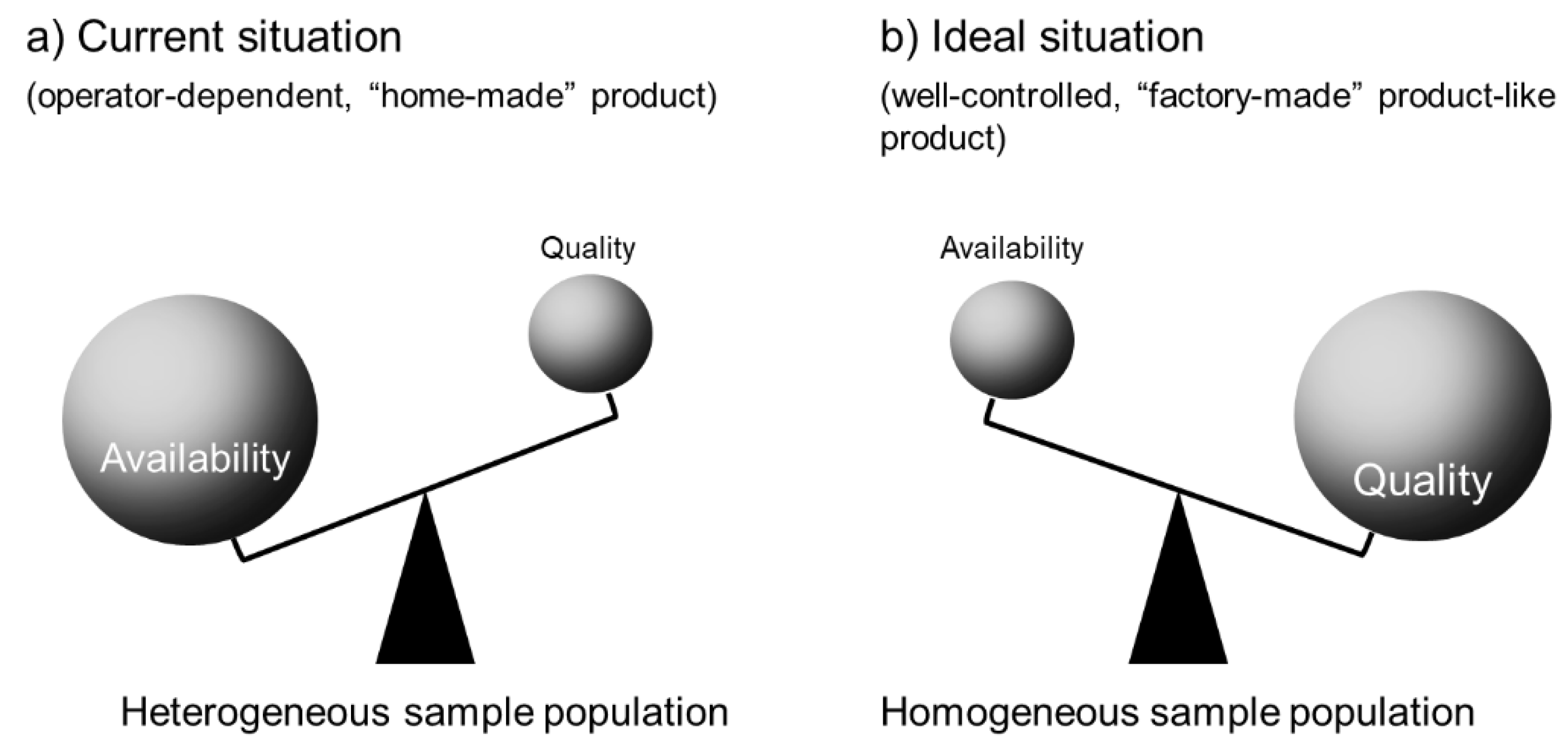
2. Unstable Positioning of PC Therapy in Regenerative Medicine
3. History of PC Study
3.1. Direction and Goal of Initial Generations
3.2. Development of Automated PRP Preparation Devices
3.3. Brand Competition and Consumer Report-Like Study in the Subsequent Generation
3.3.1. Specificity of Centrifuge Types
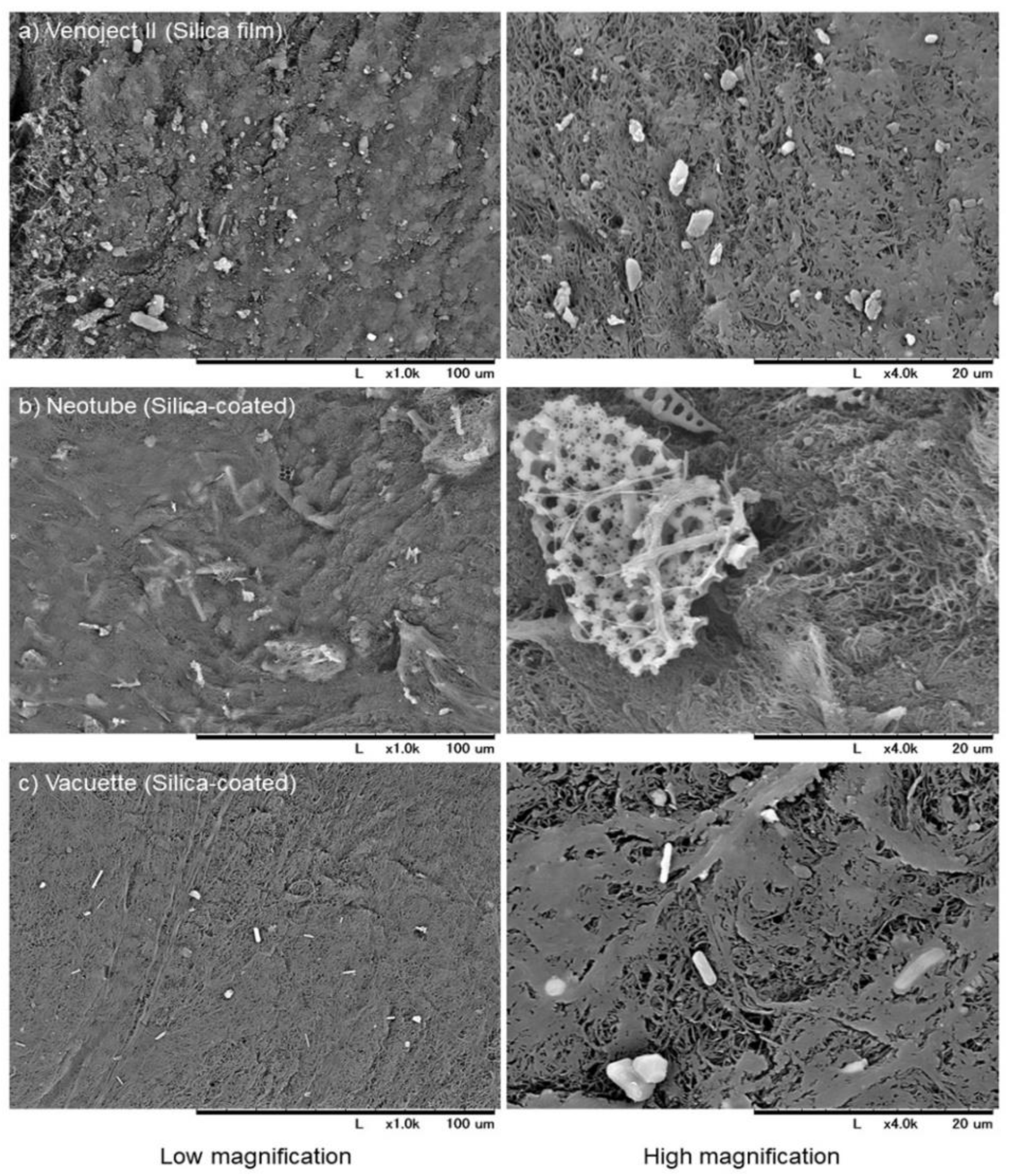
3.3.2. Specificity of Blood Collection Tube Types
3.4. Proposed Mechanisms or Modes of Action of PC in PC Therapy
3.5. RNA Delivery System
3.6. Controlled Release of Growth Factors Retained in PRF Matrices
3.7. Lack of Biological Consideration of Platelets
4. Overlooked Clinical Studies
5. Prioritized Research Investigations
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Marx, R.E.; Carlson, E.R.; Eichstaedt, R.M.; Schimmele, S.R.; Strauss, J.E.; Georgeff, K.R. Platelet-rich plasma: Growth factor enhancement for bone grafts. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1998, 85, 638–646. [Google Scholar] [CrossRef]
- Wang, H.-L.; Avila, G. Platelet rich plasma: Myth or reality? Eur. J. Dent. 2007, 1, 192–194. [Google Scholar] [CrossRef] [PubMed]
- Jacob, N.T. Drug promotion practices: A review. Br. J. Clin. Pharmacol. 2018, 84, 1659–1667. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T. Platelet-rich plasma and its derivatives as promising bioactive materials for regenerative medicine: Basic principles and concepts underlying recent advances. Odontology 2015, 103, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Boyapati, L.; Wang, H.L. The role of platelet-rich plasma in sinus augmentation: A critical review. Implant. Dent. 2006, 15, 160–170. [Google Scholar] [CrossRef]
- Caspi, O.; Sechrest, L.; Pitluk, H.C.; Marshall, C.L.; Bell, I.R.; Nichter, M. On the definition of complementary, alternative, and integrative medicine: Societal mega-stereotypes vs. the patients’ perspectives. Altern. Ther. Health Med. 2003, 9, 58–62. [Google Scholar]
- Kawase, T.; Takahashi, A.; Watanabe, T.; Tsujino, T. Proposal for point-of-care testing of platelet-rich plasma quality. Int. J. Growth Factors Stem Cells Dent. 2019, 2, 13–17. [Google Scholar] [CrossRef]
- Sánchez, M.; Garate, A.; Delgado, D.; Padilla, S. Platelet-rich plasma, an adjuvant biological therapy to assist peripheral nerve repair. Neural Regen. Res. 2017, 12, 47–52. [Google Scholar]
- Yu, P.; Yang, X.; Qi, Z. Efficacy of Autologous Platelet Concentrates as Adjuvant Therapy to Surgical Excision in the Treatment of Keloid Scars Refractory to Conventional Treatments: A Pilot Prospective Study. Ann. Plast. Surg. 2018, 81, 741. [Google Scholar] [CrossRef]
- Apostólico Jde, S.; Lunardelli, V.A.; Coirada, F.C.; Boscardin, S.B.; Rosa, D.S. Adjuvants: Classification, Modus Operandi, and Licensing. J. Immunol. Res. 2016, 2016, 1459394. [Google Scholar] [CrossRef]
- Sándor, G.K. Tissue engineering: Propagating the wave of change. Ann. Maxillofac. Surg. 2013, 3, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Okuda, K.; Nagata, M.; Yoshie, H. The Cell-Multilayered Periosteal Sheet—A Promising Osteogenic and Osteoinductive Grafting Material. In New Trends in Tissue Engineering and Regenerative Medicine—Official Book of the Japanese Society for Regenerative Medicine; Hideharu, H., Ed.; IntechOpen: London, UK, 2014; pp. 19–35. [Google Scholar]
- Xie, X.; Zhang, C.; Tuan, R.S. Biology of platelet-rich plasma and its clinical application in cartilage repair. Arthritis Res. Ther. 2014, 16, 204. [Google Scholar] [CrossRef] [PubMed]
- Utomo, D.N.; Mahyudin, F.; Hernugrahanto, K.D.; Suroto, H.; Chilmi, M.Z.; Rantam, F.A. Implantation of platelet rich fibrin and allogenic mesenchymal stem cells facilitate the healing of muscle injury: An experimental study on animal. Int. J. Surg. Open 2018, 11, 4–9. [Google Scholar] [CrossRef]
- Fernandes, G.; Yang, S. Application of platelet-rich plasma with stem cells in bone and periodontal tissue engineering. Bone Res. 2016, 4, 16036. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudian-Sani, M.R.; Rafeei, F.; Amini, R.; Saidijam, M. The effect of mesenchymal stem cells combined with platelet-rich plasma on skin wound healing. J. Cosmet. Dermatol. 2018, 17, 650–659. [Google Scholar] [CrossRef]
- Landesberg, R.; Roy, M.; Glickman, R.S. Quantification of growth factor levels using a simplified method of platelet-rich plasma gel preparation. J. Oral Maxillofac. Surg. 2000, 58, 297–300, discussion 300-1. [Google Scholar] [CrossRef]
- Okuda, K.; Kawase, T.; Momose, M.; Murata, M.; Saito, Y.; Suzuki, H.; Wolff, L.F.; Yoshie, H. Platelet-rich plasma contains high levels of platelet-derived growth factor and transforming growth factor-beta and modulates the proliferation of periodontally related cells in vitro. J. Periodontol. 2003, 74, 849–857. [Google Scholar] [CrossRef]
- Weibrich, G.; Kleis, W.K. Curasan PRP kit vs. PCCS PRP system. Collection efficiency and platelet counts of two different methods for the preparation of platelet-rich plasma. Clin. Oral Implants Res. 2002, 13, 437–443. [Google Scholar] [CrossRef]
- Dhurat, R.; Sukesh, M. Principles and Methods of Preparation of Platelet-Rich Plasma: A Review and Author’s Perspective. J. Cutan. Aesthetic Surg. 2014, 7, 189–197. [Google Scholar] [CrossRef]
- Fitzpatrick, J.; Bulsara, M.K.; McCrory, P.R.; Richardson, M.D.; Zheng, M.H. Analysis of Platelet-Rich Plasma Extraction: Variations in Platelet and Blood Components Between 4 Common Commercial Kits. Orthop. J. Sports Med. 2017, 5. [Google Scholar] [CrossRef]
- Magalon, J.; Bausset, O.; Serratrice, N.; Giraudo, L.; Aboudou, H.; Veran, J.; Magalon, G.; Dignat-Georges, F.; Sabatier, F. Characterization and comparison of 5 platelet-rich plasma preparations in a single-donor model. Arthroscopy 2014, 30, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Everts, P.A.M.; Knape, J.T.A.; Weibrich, G.; Schönberger, J.P.A.M.; Hoffmann, J.; Overdevest, E.P.; Box, H.A.M.; van Zundert, A. Platelet-rich plasma and platelet gel: A review. J. Extra-Corpor. Technol. 2006, 38, 174–187. [Google Scholar] [PubMed]
- Marx, R.E. Platelet-rich plasma (PRP): What is PRP and what is not PRP? Implant. Dent. 2001, 10, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Dohan, D.M.; Del Corso, M.; Charrier, J.-B. Cytotoxicity analyses of Choukroun’s platelet-rich fibrin (PRF) on a wide range of human cells: The answer to a commercial controversy. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2007, 103, 587–593. [Google Scholar] [CrossRef]
- Horimizu, M.; Kawase, T.; Nakajima, Y.; Okuda, K.; Nagata, M.; Wolff, L.F.; Yoshie, H. An improved freeze-dried PRP-coated biodegradable material suitable for connective tissue regenerative therapy. Cryobiology 2013, 66, 223–232. [Google Scholar] [CrossRef]
- Nakajima, Y.; Kawase, T.; Kobayashi, M.; Okuda, K.; Wolff, L.F.; Yoshie, H. Bioactivity of freeze-dried platelet-rich plasma in an adsorbed form on a biodegradable polymer material. Platelets 2012, 23, 594–603. [Google Scholar] [CrossRef]
- Eto, K. Platelets using iPS cell technology; large scale manufacturing. J. Stem Cells Regen. Med. 2019, 15, 52. [Google Scholar]
- Sugimoto, N.; Eto, K. Platelet production from induced pluripotent stem cells. J. Thromb. Haemost. 2017, 15, 1717–1727. [Google Scholar] [CrossRef]
- Dohan Ehrenfest, D.M.; Pinto, N.R.; Pereda, A.; Jimenez, P.; Corso, M.D.; Kang, B.S.; Nally, M.; Lanata, N.; Wang, H.L.; Quirynen, M. The impact of the centrifuge characteristics and centrifugation protocols on the cells, growth factors, and fibrin architecture of a leukocyte- and platelet-rich fibrin (L-PRF) clot and membrane. Platelets 2018, 29, 171–184. [Google Scholar] [CrossRef]
- El Bagdadi, K.; Kubesch, A.; Yu, X.; Al-Maawi, S.; Orlowska, A.; Dias, A.; Booms, P.; Dohle, E.; Sader, R.; Kirkpatrick, C.J.; et al. Reduction of relative centrifugal forces increases growth factor release within solid platelet-rich-fibrin (PRF)-based matrices: A proof of concept of LSCC (low speed centrifugation concept). Eur J. Trauma Emerg. Surg. 2019, 45, 467–479. [Google Scholar] [CrossRef]
- Ghanaati, S.; Booms, P.; Orlowska, A.; Kubesch, A.; Lorenz, J.; Rutkowski, J.; Landes, C.; Sader, R.; Kirkpatrick, C.; Choukroun, J. Advanced platelet-rich fibrin: A new concept for cell-based tissue engineering by means of inflammatory cells. J. Oral Implantol. 2014, 40, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Chai, J.; Zhang, P.; Li, Y.; Wang, Y.; Mourao, C.; Sculean, A.; Fujioka Kobayashi, M.; Zhang, Y. A novel method for harvesting concentrated platelet-rich fibrin (C-PRF) with a 10-fold increase in platelet and leukocyte yields. Clin. Oral Investig. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Kawase, T.; Horimizu, M.; Okuda, K.; Wolff, L.F.; Yoshie, H. A proposed protocol for the standardized preparation of PRF membranes for clinical use. Biologicals 2012, 40, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Fedato, R.A.; Francisco, J.C.; Sliva, G.; de Noronha, L.; Olandoski, M.; Faria Neto, J.R.; Ferreira, P.E.; Simeoni, R.B.; Abdelwahid, E.; de Carvalho, K.A.T.; et al. Stem Cells and Platelet-Rich Plasma Enhance the Healing Process of Tendinitis in Mice. Stem Cells Int. 2019, 2019, 1497898. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; Ku, S.K.; Kwon, Y.S. Adipose-derived mesenchymal stem cells and platelet-rich plasma synergistically ameliorate the surgical-induced osteoarthritis in Beagle dogs. J. Orthop. Surg. Res. 2016, 11, 9. [Google Scholar] [CrossRef]
- Sassoli, C.; Vallone, L.; Tani, A.; Chellini, F.; Nosi, D.; Zecchi-Orlandini, S. Combined use of bone marrow-derived mesenchymal stromal cells (BM-MSCs) and platelet rich plasma (PRP) stimulates proliferation and differentiation of myoblasts in vitro: New therapeutic perspectives for skeletal muscle repair/regeneration. Cell Tissue Res. 2018, 372, 549–570. [Google Scholar] [CrossRef]
- Mehranfar, S.; Abdi Rad, I.; Mostafav, E.; Akbarzadeh, A. The use of stromal vascular fraction (SVF), platelet-rich plasma (PRP) and stem cells in the treatment of osteoarthritis: An overview of clinical trials. Artif. Cells Nanomed. Biotechnol. 2019, 47, 882–890. [Google Scholar] [CrossRef]
- Rodas, G.; Soler, R.; Balius, R.; Alomar, X.; Peirau, X.; Alberca, M.; Sánchez, A.; Sancho, J.G.; Rodellar, C.; Romero, A.; et al. Autologous bone marrow expanded mesenchymal stem cells in patellar tendinopathy: Protocol for a phase I/II, single-centre, randomized with active control PRP, double-blinded clinical trial. J. Orthop. Surg. Res. 2019, 14, 441. [Google Scholar] [CrossRef]
- Nagata, M.; Hoshina, H.; Li, M.; Arasawa, M.; Uematsu, K.; Ogawa, S.; Yamada, K.; Kawase, T.; Suzuki, K.; Ogose, A.; et al. A clinical study of alveolar bone tissue engineering with cultured autogenous periosteal cells: Coordinated activation of bone formation and resorption. Bone 2012, 50, 1123–1129. [Google Scholar] [CrossRef]
- Ogawa, S.; Hoshina, H.; Nakata, K.; Yamada, K.; Uematsu, K.; Kawase, T.; Takagi, R.; Nagata, M. High-Resolution Three-Dimensional Computed Tomography Analysis of the Clinical Efficacy of Cultured Autogenous Periosteal Cells in Sinus Lift Bone Grafting. Clin. Implant. Dent. Relat. Res. 2016, 18, 707–716. [Google Scholar] [CrossRef]
- Araki, J.; Jona, M.; Eto, H.; Aoi, N.; Kato, H.; Suga, H.; Doi, K.; Yatomi, Y.; Yoshimura, K. Optimized preparation method of platelet-concentrated plasma and noncoagulating platelet-derived factor concentrates: Maximization of platelet concentration and removal of fibrinogen. Tissue Eng. Part. C Methods 2012, 18, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Söderström, A.C.; Nybo, M.; Nielsen, C.; Vinholt, P.J. The effect of centrifugation speed and time on pre-analytical platelet activation. Clin. Chem. Lab. Med. 2016, 54, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- Zellner, M.; Oehler, R. Sample Preparation Variables in Platelet Proteomics for Biomarker Research in Platelet Proteomics: Principles, Analysis, and Applications; García-Alonso, Á., Senis, Y., Eds.; Wiley: Hoboken, NJ, USA, 2011; pp. 67–88. [Google Scholar]
- Hechler, B.; Dupuis, A.; Mangin, P.H.; Gachet, C. Platelet preparation for function testing in the laboratory and clinic: Historical and practical aspects. Res. Pr. Thromb. Haemost. 2019, 3, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, K.; Okudera, T.; Watanabe, T.; Isobe, K.; Suzuki, M.; Masuki, H.; Okudera, H.; Uematsu, K.; Nakata, K.; Kawase, T. Basic characteristics of plasma rich in growth factors (PRGF): Blood cell components and biological effects. Clin. Exp. Dent. Res. 2016. [Google Scholar] [CrossRef]
- Davis, V.L.; Abukabda, A.B.; Radio, N.M.; Witt-Enderby, P.A.; Clafshenkel, W.P.; Cairone, J.V.; Rutkowski, J.L. Platelet-rich preparations to improve healing. Part II: Platelet activation and enrichment, leukocyte inclusion, and other selection criteria. J. Oral Implantol. 2014, 40, 511–521. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Saita, Y.; Nishio, H.; Ikeda, H.; Takazawa, Y.; Nagao, M.; Takaku, T.; Komatsu, N.; Kaneko, K. Leukocyte concentration and composition in platelet-rich plasma (PRP) influences the growth factor and protease concentrations. J. Orthop. Sci. 2016, 21, 683–689. [Google Scholar] [CrossRef]
- Shin, K.H.; Lee, H.; Kang, S.; Ko, Y.-J.; Lee, S.-Y.; Park, J.-H.; Bae, J.-H. Effect of Leukocyte-Rich and Platelet-Rich Plasma on Healing of a Horizontal Medial Meniscus Tear in a Rabbit Model. BioMed Res. Int. 2015, 2015, 179756. [Google Scholar] [CrossRef]
- Caruana, A.; Savina, D.; Macedo, J.P.; Soares, S.C. From Platelet-Rich Plasma to Advanced Platelet-Rich Fibrin: Biological Achievements and Clinical Advances in Modern Surgery. Eur. J. Dent. 2019, 13, 280–286. [Google Scholar] [CrossRef]
- Anitua, E.; Zalduendo, M.; Troya, M.; Padilla, S.; Orive, G. Leukocyte Inclusion within a Platelet Rich Plasma-Derived Fibrin Scaffold Stimulates a More Pro-Inflammatory Environment and Alters Fibrin Properties. PLoS ONE 2015, 10, e0121713. [Google Scholar] [CrossRef]
- Xu, Z.; Yin, W.; Zhang, Y.; Qi, X.; Chen, Y.; Xie, X.; Zhang, C. Comparative evaluation of leukocyte- and platelet-rich plasma and pure platelet-rich plasma for cartilage regeneration. Sci. Rep. 2017, 7, 43301. [Google Scholar] [CrossRef]
- Jia, J.; Wang, S.-Z.; Ma, L.-Y.; Yu, J.-B.; Guo, Y.-D.; Wang, C. The Differential Effects of Leukocyte-Containing and Pure Platelet-Rich Plasma on Nucleus Pulposus-Derived Mesenchymal Stem Cells: Implications for the Clinical Treatment of Intervertebral Disc Degeneration. Stem Cells Int. 2018, 2018, 7162084. [Google Scholar] [CrossRef] [PubMed]
- Masuki, H.; Okudera, T.; Watanabe, T.; Suzuki, M.; Nishiyama, K.; Okudera, H.; Nakata, K.; Uematsu, K.; Su, C.Y.; Kawase, T. Growth factor and pro-inflammatory cytokine contents in PRP, plasma rich in growth factors (PRGF), advanced-platelet-rich fibrin (A-PRF) and concentrated growth factors (CGF). Int. J. Implant. Dent. 2016, 2, 19. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Chai, J.; Zheng, S.; Feng, M.; Sculean, A.; Zhang, Y. A novel method for evaluating and quantifying cell types in platelet rich fibrin and an introduction to horizontal centrifugation. J. Biomed. Mater. Res. A 2019, 107, 2257–2271. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, E.S.; Mourão, C.; Leite, P.E.C.; Granjeiro, J.M.; Calasans-Maia, M.D.; Alves, G.G. The in vitro release of cytokines and growth factors from fibrin membranes produced through horizontal centrifugation. J. Biomed. Mater. Res. A 2018, 106, 1373–1380. [Google Scholar] [CrossRef]
- Miron, R.J.; Xu, H.; Chai, J.; Wang, J.; Zheng, S.; Feng, M.; Zhang, X.; Wei, Y.; Chen, Y.; Mourão, C.F.D.A.B.; et al. Comparison of platelet-rich fibrin (PRF) produced using 3 commercially available centrifuges at both high (~ 700 g) and low (~ 200 g) relative centrifugation forces. Clin. Oral Investig. 2020, 24, 1171–1182. [Google Scholar] [CrossRef]
- Tsujino, T.; Masuki, H.; Nakamura, M.; Isobe, K.; Kawabata, H.; Aizawa, H.; Watanabe, T.; Kitamura, Y.; Okudera, H.; Okuda, K.; et al. Striking Differences in Platelet Distribution between Advanced-Platelet-Rich Fibrin and Concentrated Growth Factors: Effects of Silica-Containing Plastic Tubes. J. Funct. Biomater. 2019, 10, 43. [Google Scholar] [CrossRef]
- Aizawa, H.; Tsujino, T.; Watanabe, T.; Isobe, K.; Kitamura, Y.; Sato, A.; Yamaguchi, S.; Okudera, H.; Okuda, K.; Kawase, T. Quantitative Near-Infrared Imaging of Platelets in Platelet-Rich Fibrin (PRF) Matrices: Comparative Analysis of Bio-PRF, Leukocyte-Rich PRF, Advanced-PRF and Concentrated Growth Factors. Int. J. Mol. Sci. 2020, 21, 4426. [Google Scholar] [CrossRef]
- Kawase, T.; Tanaka, T. An updated proposal for terminology and classification of platelet-rich fibrin. Regen. Ther. 2017, 7, 80–81. [Google Scholar] [CrossRef]
- Laboratory Centrifuges: When to Choose Swing Out or Fixed Angle Centrifuge Rotors. Available online: https://www.labnetinternational.com/blog/2018/02/laboratory-centrifuges-when-choose-swing-out-or-fixed-angle-centrifuge-rotors#:~:text=Swing%20Out%20Rotors&text=When%20spinning%20starts%2C%20these%20buckets,effective%20separation%20of%20individual%20particles (accessed on 23 June 2020).
- Masuki, H.; Isobe, K.; Kawabata, H.; Tsujino, T.; Yamaguchi, S.; Watanabe, T.; Sato, A.; Aizawa, H.; Mourao, C.F.; Kawase, T. Acute cytotoxic effects of silica microparticles used for coating of plastic blood-collection tubes on human periosteal cells. Odontology 2020, in press. [Google Scholar] [CrossRef]
- Tsujino, T.; Takahashi, A.; Yamaguchi, S.; Watanabe, T.; Isobe, K.; Kitamura, Y.; Tanaka, T.; Nakata, K.; Kawase, T. Evidence for Contamination of Silica Microparticles in Advanced Platelet-Rich Fibrin Matrices Prepared Using Silica-Coated Plastic Tubes. Biomedicines 2019, 7, 45. [Google Scholar] [CrossRef]
- Kawase, T.; Okuda, K.; Wolff, L.F.; Yoshie, H. Platelet-rich plasma-derived fibrin clot formation stimulates collagen synthesis in periodontal ligament and osteoblastic cells in vitro. J. Periodontol. 2003, 74, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Okuda, K.; Saito, Y.; Yoshie, H. In vitro evidence that the biological effects of platelet-rich plasma on periodontal ligament cells is not mediated solely by constituent transforming-growth factor-beta or platelet-derived growth factor. J. Periodontol. 2005, 76, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-González, D.J.; Méndez-Bolaina, E.; Trejo-Bahena, N.I. Platelet-Rich Plasma Peptides: Key for Regeneration. Int. J. Pept. 2012, 2012, 532519. [Google Scholar]
- Della Porta, G.; Nguyen, B.N.; Campardelli, R.; Reverchon, E.; Fisher, J.P. Synergistic effect of sustained release of growth factors and dynamic culture on osteoblastic differentiation of mesenchymal stem cells. J. Biomed. Mater. Res. A. 2015, 103, 2161–2171. [Google Scholar] [CrossRef]
- Li, B.; Wang, H.; Qiu, G.; Su, X.; Wu, Z. Synergistic Effects of Vascular Endothelial Growth Factor on Bone Morphogenetic Proteins Induced Bone Formation In Vivo: Influencing Factors and Future Research Directions. BioMed Res. Int. 2016, 2016, 2869572. [Google Scholar] [CrossRef]
- Oneto, P.; Zubiry, P.R.; Schattner, M.; Etulain, J. Anticoagulants Interfere With the Angiogenic and Regenerative Responses Mediated by Platelets. Front. Bioeng. Biotechnol. 2020, 8, 223. [Google Scholar] [CrossRef]
- Modi, P.K.; Prabhu, A.; Bhandary, Y.P.; Shenoy, P.S.; Hegde, A.; Es, S.P.; Johnson, R.P.; Das, S.P.; Vazirally, S.; Rekha, P.-D. Effect of calcium glucoheptonate on proliferation and osteogenesis of osteoblast-like cells in vitro. PLoS ONE 2019, 14, e0222240. [Google Scholar] [CrossRef]
- do Amaral, R.J.F.C.; da Silva, N.P.; Haddad, N.F.; Lopes, L.S.; Ferreira, F.D.; Filho, R.B.; Cappelletti, P.A.; de Mello, W.; Cordeiro-Spinetti, E.; Balduino, A. Platelet-Rich Plasma Obtained with Different Anticoagulants and Their Effect on Platelet Numbers and Mesenchymal Stromal Cells Behavior In Vitro. Stem Cells Int. 2016, 2016, 7414036. [Google Scholar] [CrossRef]
- Aizawa, H.; Kawabata, H.; Sato, A.; Masuki, H.; Watanabe, T.; Tsujino, T.; Isobe, K.; Nakamura, M.; Nakata, K.; Kawase, T. A Comparative Study of The Effects of Anticoagulants on Pure Platelet-Rich Plasma Quality and Potency. Biomedicines 2020, 8, 42. [Google Scholar] [CrossRef]
- Fager, G. Thrombin and proliferation of vascular smooth muscle cells. Circ. Res. 1995, 77, 645–650. [Google Scholar] [CrossRef]
- Wolf, P. The nature and significance of platelet products in human plasma. Br. J. Haematol. 1967, 13, 269–288. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.C.; Guo, S.C.; Zhang, C.Q. Platelet-derived Extracellular Vesicles: An Emerging Therapeutic Approach. Int. J. Biol. Sci. 2017, 13, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Torreggiani, E.; Perut, F.; Roncuzzi, L.; Zini, N.; Baglìo, S.R.; Baldini, N. Exosomes: Novel effectors of human platelet lysate activity. Eur. Cell Mater. 2014, 28, 137–151. [Google Scholar] [CrossRef]
- Ribeiro, M.F.; Zhu, H.; Millard, R.W.; Fan, G.C. Exosomes Function in Pro- and Anti-Angiogenesis. Curr. Angiogenes. 2013, 2, 54–59. [Google Scholar]
- Varon, D.; Hayon, Y.; Dashevsky, O.; Shai, E. Involvement of platelet derived microparticles in tumor metastasis and tissue regeneration. Thromb. Res. 2012, 130 (Suppl. 1), S98–S99. [Google Scholar] [CrossRef]
- Kobayashi, E.; Fluckiger, L.; Fujioka-Kobayashi, M.; Sawada, K.; Sculean, A.; Schaller, B.; Miron, R.J. Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin. Oral Investig. 2016, 20, 2353–2360. [Google Scholar] [CrossRef]
- Schär, M.O.; Diaz-Romero, J.; Kohl, S.; Zumstein, M.A.; Nesic, D. Platelet-rich concentrates differentially release growth factors and induce cell migration in vitro. Clin. Orthop. Relat. Res. 2015, 473, 1635–1643. [Google Scholar] [CrossRef]
- Isobe, K.; Watanebe, T.; Kawabata, H.; Kitamura, Y.; Okudera, T.; Okudera, H.; Uematsu, K.; Okuda, K.; Nakata, K.; Tanaka, T.; et al. Mechanical and degradation properties of advanced platelet-rich fibrin (A-PRF), concentrated growth factors (CGF), and platelet-poor plasma-derived fibrin (PPTF). Int. J. Implant. Dent. 2017, 3, 17. [Google Scholar] [CrossRef]
- Hartshone, J.; Gluckman, H. A comprehensive clinical review of Platelet Rich Fibrin (PRF) and its role in promoting tissue healing and regeneration in dentistry. Part II: Preparation, optimization, handling and application, benefits and limitations of PRF. Int. Dent. 2016, 6, 34–48. [Google Scholar]
- Wang, J.; Wang, L.; Zhou, Z.; Lai, H.; Xu, P.; Liao, L.; Wei, J. Biodegradable Polymer Membranes Applied in Guided Bone/Tissue Regeneration: A Review. Polymers 2016, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Kamiya, M.; Kobayashi, M.; Tanaka, T.; Okuda, K.; Wolff, L.F.; Yoshie, H. The heat-compression technique for the conversion of platelet-rich fibrin preparation to a barrier membrane with a reduced rate of biodegradation. J. Biomed. Mater. Res. B Appl. Biomater. 2014, 103, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Mourão, C.F.; Gheno, E.; Lourenço, E.S.; Barbosa, R.L.; Kurtzman, G.; Javid, K.; Mavropoulos, E.; Benedicenti, S.; Calasans-Maia, M.D.; de Mello Machado, R.C.; et al. Characterization of a new membrane from concentrated growth factors associated with denaturized Albumin (Alb-CGF) for clinical applications: A preliminary study. Int. J. Growth Factors Stem Cells Dent. 2018, 1, 64–69. [Google Scholar] [CrossRef]
- Mourão, C.F.; Javid, K. New and improved platelet-rich fibrin membranes. Int. J. Growth Factors Stem Cells Dent. 2020, 3, 1–2. [Google Scholar] [CrossRef]
- Fujioka-Kobayashi, M.; Schaller, B.; Mourão, C.; Zhang, Y.; Sculean, A.; Miron, R.J. Biological characterization of an injectable platelet-rich fibrin mixture consisting of autologous albumin gel and liquid platelet-rich fibrin (Alb-PRF). Platelets 2020, 1–8. [Google Scholar] [CrossRef]
- Gheno, E.; Mourão, C.; Mello-Machado, R.C.; Stellet Lourenço, E.; Miron, R.J.; Catarino, K.F.F.; Alves, A.T.; Alves, G.G.; Calasans-Maia, M.D. In vivo evaluation of the biocompatibility and biodegradation of a new denatured plasma membrane combined with liquid PRF (Alb-PRF). Platelets 2020, 1–13. [Google Scholar] [CrossRef]
- Toyoda, T.; Isobe, K.; Tsujino, T.; Koyata, Y.; Ohyagi, F.; Watanabe, T.; Nakamura, M.; Kitamura, Y.; Okudera, H.; Nakata, K.; et al. Direct activation of platelets by addition of CaCl2 leads coagulation of platelet-rich plasma. Int. J. Implant. Dent. 2018, 4, 23. [Google Scholar] [CrossRef]
- Kitamura, Y.; Suzuki, M.; Tsukioka, T.; Isobe, K.; Tsujino, T.; Watanabe, T.; Watanabe, T.; Okudera, H.; Nakata, K.; Tanaka, T.; et al. Spectrophotometric determination of platelet counts in platelet-rich plasma. Int. J. Implant. Dent. 2018, 4, 29. [Google Scholar] [CrossRef]
- Takahashi, A.; Takahashi, S.; Tsujino, T.; Isobe, K.; Watanabe, T.; Kitamura, Y.; Watanabe, T.; Nakata, K.; Kawase, T. Platelet adhesion on commercially pure titanium plates in vitro I: Effects of plasma components and involvement of the von Willebrand factor and fibronectin. Int. J. Implant. Dent. 2019, 5, 5. [Google Scholar] [CrossRef]
- Tsujino, T.; Isobe, K.; Kawabata, H.; Aizawa, H.; Yamaguchi, S.; Kitamura, Y.; Masuki, H.; Watanabe, T.; Okudera, H.; Nakata, K.; et al. Spectrophotometric Determination of the Aggregation Activity of Platelets in Platelet-Rich Plasma for Better Quality Control. Dent. J. 2019, 7, 61. [Google Scholar] [CrossRef]
- Tsukioka, T.; Hiratsuka, T.; Nakamura, M.; Watanabe, T.; Kitamura, Y.; Isobe, K.; Okudera, T.; Okudera, H.; Azuma, A.; Uematsu, K.; et al. An on-site preparable, novel bone-grafting complex consisting of human platelet-rich fibrin and porous particles made of a recombinant collagen-like protein. J. Biomed. Mater. Res. B Appl. Biomater. 2019, 107, 1420–1430. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, Y.; Isobe, K.; Kawabata, H.; Tsujino, T.; Watanabe, T.; Nakamura, M.; Toyoda, T.; Okudera, H.; Okuda, K.; Nakata, K.; et al. Quantitative evaluation of morphological changes in activated platelets in vitro using digital holographic microscopy. Micron 2018, 113, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tsujino, T.; Takahashi, A.; Watanabe, T.; Isobe, K.; Kitamura, Y.; Okuda, K.; Nakata, K.; Kawase, T. Platelet Adhesion on Commercially Pure Titanium Plates in Vitro II. Immunofluorescence Visualization of PDGF-B, TGFβ1, and PPARγ Released from Activated Adherent Platelets. Dent. J. 2019, 7, 109. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, G.; Golino, P. Platelet biology and receptor pathways. J. Cardiovasc. Transl. Res. 2013, 6, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Matthay, Z.A.; Kornblith, L.Z. Platelet Contributions to Trauma-Induced Coagulopathy: Updates in Post-injury Platelet Biology, Platelet Transfusions, and Emerging Platelet-Based Hemostatic Agents. Curr. Trauma Rep. 2019, 5, 202–209. [Google Scholar] [CrossRef]
- van der Meijden, P.E.J.; Heemskerk, J.W.M. Platelet biology and functions: New concepts and clinical perspectives. Nat. Rev. Cardiol. 2019, 16, 166–179. [Google Scholar] [CrossRef]
- Farré, A.L.; Modrego, J.; Zamorano-León, J.J. Effects of hormones on platelet aggregation. Horm. Mol. Biol. Clin. Investig. 2014, 18, 27–36. [Google Scholar] [CrossRef]
- Koltai, K.; Kesmarky, G.; Feher, G.; Tibold, A.; Toth, K. Platelet Aggregometry Testing: Molecular Mechanisms, Techniques and Clinical Implications. Int. J. Mol. Sci. 2017, 18, 1803. [Google Scholar] [CrossRef]
- Broos, K.; Feys, H.B.; De Meyer, S.F.; Vanhoorelbeke, K.; Deckmyn, H. Platelets at work in primary hemostasis. Blood Rev. 2011, 25, 155–167. [Google Scholar] [CrossRef]
- Burkard, P.; Vögtle, T.; Nieswandt, B. Platelets in Thrombo-Inflammation: Concepts, Mechanisms, and Therapeutic Strategies for Ischemic Stroke. Hamostaseologie 2020, 40, 153–164. [Google Scholar] [CrossRef]
- Varga-Szabo, D.; Pleines, I.; Nieswandt, B. Cell adhesion mechanisms in platelets. Arter. Thromb. Vasc. Boil. 2008, 28, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Hanawa, T. Titanium-Tissue Interface Reaction and Its Control with Surface Treatment. Front. Bioeng. Biotechnol. 2019, 7, 170. [Google Scholar] [CrossRef] [PubMed]
- Tomaiuolo, M.; Brass, L.F.; Stalker, T.J. Regulation of Platelet Activation and Coagulation and Its Role in Vascular Injury and Arterial Thrombosis. Interv. Cardiol. Clin. 2017, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Heemskerk, J.W.; Bevers, E.M.; Lindhout, T. Platelet activation and blood coagulation. Thromb. Haemost. 2002, 88, 186–193. [Google Scholar] [PubMed]
- Miron, R.J.; Zhang, Y. Autologous liquid platelet rich fibrin: A novel drug delivery system. Acta Biomater. 2018, 75, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Martínez, C.E.; Smith, P.C.; Palma Alvarado, V.A. The influence of platelet-derived products on angiogenesis and tissue repair: A concise update. Front. Physiol. 2015, 6, 290. [Google Scholar] [CrossRef]
- Ali, R.A.; Wuescher, L.M.; Worth, R.G. Platelets: Essential components of the immune system. Curr. Trends Immunol. 2015, 16, 65–78. [Google Scholar]
- Kraemer, B.F.; Campbell, R.A.; Schwertz, H.; Cody, M.J.; Franks, Z.; Tolley, N.D.; Kahr, W.H.; Lindemann, S.; Seizer, P.; Yost, C.C.; et al. Novel anti-bacterial activities of β-defensin 1 in human platelets: Suppression of pathogen growth and signaling of neutrophil extracellular trap formation. PLoS Pathog. 2011, 7, e1002355. [Google Scholar] [CrossRef]
- Kuffler, D.P. Platelet-rich plasma and the elimination of neuropathic pain. Mol. Neurobiol. 2013, 48, 315–332. [Google Scholar] [CrossRef]
- Araya, N.; Miyatake, K.; Tsuji, K.; Katagiri, H.; Nakagawa, Y.; Hoshino, T.; Onuma, H.; An, S.; Nishio, H.; Saita, Y.; et al. Intra-articular Injection of Pure Platelet-Rich Plasma Is the Most Effective Treatment for Joint Pain by Modulating Synovial Inflammation and Calcitonin Gene-Related Peptide Expression in a Rat Arthritis Model. Am. J. Sports Med. 2020, 48, 2004–2012. [Google Scholar] [CrossRef]
- Periayah, M.H.; Halim, A.S.; Mat Saad, A.Z. Mechanism Action of Platelets and Crucial Blood Coagulation Pathways in Hemostasis. Int. J. Hematol. Stem Cell Res. 2017, 11, 319–327. [Google Scholar]
- Lentz, B.R. Exposure of platelet membrane phosphatidylserine regulates blood coagulation. Prog. Lipid Res. 2003, 42, 423–438. [Google Scholar] [CrossRef]
- Sang, Y.; Roest, M.; de Laat, B.; de Groot, P.G.; Huskens, D. Interplay between platelets and coagulation. Blood Rev. 2020, 100733. [Google Scholar] [CrossRef] [PubMed]
- Cerletti, C.; Tamburrelli, C.; Izzi, B.; Gianfagna, F.; de Gaetano, G. Platelet-leukocyte interactions in thrombosis. Thromb. Res. 2012, 129, 263–266. [Google Scholar] [CrossRef]
- Fu, G.; Deng, M.; Neal, M.D.; Billiar, T.R.; Scott, M.J. Platelet-Monocyte Aggregates: Understanding Mechanisms and Functions in Sepsis. Shock 2020. [Google Scholar] [CrossRef]
- Gato-Calvo, L.; Magalhaes, J.; Ruiz-Romero, C.; Blanco, F.J.; Burguera, E.F. Platelet-rich plasma in osteoarthritis treatment: Review of current evidence. Ther. Adv. Chronic Dis. 2019, 10. [Google Scholar] [CrossRef]
- Stanley, K. Design of randomized controlled trials. Circulation 2007, 115, 1164–1169. [Google Scholar] [CrossRef]
- Deaton, A.; Cartwright, N. Understanding and misunderstanding randomized controlled trials. Soc. Sci. Med. 2018, 210, 2–21. [Google Scholar] [CrossRef]
- Dal-Ré, R.; Janiaud, P.; Ioannidis, J.P.A. Real-world evidence: How pragmatic are randomized controlled trials labeled as pragmatic? BMC Med. 2018, 16, 49. [Google Scholar] [CrossRef]
- Gamerman, V.; Cai, T.; Elsäßer, A. Pragmatic randomized clinical trials: Best practices and statistical guidance. Health Serv. Outcomes Res. Methodol. 2018, 19, 23–35. [Google Scholar] [CrossRef]
- Kitamura, M.; Akamatsu, M.; Machigashira, M.; Hara, Y.; Sakagami, R.; Hirofuji, T.; Hamachi, T.; Maeda, K.; Yokota, M.; Kido, J.; et al. FGF-2 stimulates periodontal regeneration: Results of a multi-center randomized clinical trial. J. Dent. Res. 2011, 90, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Pannucci, C.J.; Wilkins, E.G. Identifying and avoiding bias in research. Plast. Reconstr. Surg. 2010, 126, 619–625. [Google Scholar] [CrossRef]
- Ghanaati, S.; Herrera-Vizcaino, C.; Al-Maawi, S.; Lorenz, J.; Miron, R.J.; Nelson, K.; Schwarz, F.; Choukroun, J.; Sader, R. Fifteen Years of Platelet Rich Fibrin in Dentistry and Oromaxillofacial Surgery: How High is the Level of Scientific Evidence? J. Oral Implantol. 2018, 44, 471–492. [Google Scholar] [CrossRef] [PubMed]
- Saposnik, G.; Redelmeier, D.; Ruff, C.C.; Tobler, P.N. Cognitive biases associated with medical decisions: A systematic review. BMC Med. Inform. Decis. Mak. 2016, 16, 138. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Oxman, A.D.; Montori, V.; Vist, G.; Kunz, R.; Brozek, J.; Alonso-Coello, P.; Djulbegovic, B.; Atkins, D.; Falck-Ytter, Y.; et al. GRADE guidelines: 5. Rating the quality of evidence—Publication bias. J. Clin. Epidemiol. 2011, 64, 1277–1282. [Google Scholar] [CrossRef] [PubMed]
- Koletsi, D.; Valla, K.; Fleming, P.S.; Chaimani, A.; Pandis, N. Assessment of publication bias required improvement in oral health systematic reviews. J. Clin. Epidemiol. 2016, 76, 118–124. [Google Scholar] [CrossRef]
- Jin, Z.C.; Zhou, X.H.; He, J. Statistical methods for dealing with publication bias in meta-analysis. Stat. Med. 2015, 34, 343–360. [Google Scholar] [CrossRef]
- Welcome to the CONSORT Website. Available online: http://www.consort-statement.org/ (accessed on 28 June 2020).



| Generation | Major Contents |
|---|---|
| 1st |
|
| 2nd |
|
| 3rd |
|
| 4th |
|
| Intra-Spin | Duo Quattro | Horizontal Type | |
|---|---|---|---|
| Angulation (°) | 33.0 | 41.3 | 90.0 |
| Cross-sectional area (mm2) | 183.6π | 151.5π | 100.0π |
| Shape of cross-section | 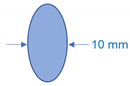 | 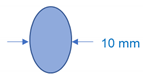 |  |
| Images of angulation and cross-section |  | 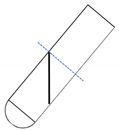 | 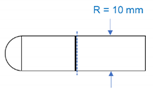 |
| Criteria | Classic RCT | Past and Current PRP/PRFs RCTs | Pragmatic RCT |
|---|---|---|---|
| Required by | Regulatory authorities | – | Clinicians |
| Patients/cases | Homogeneous | Heterogeneous | Heterogeneous |
| Combinational treatments | Controlled | Non- or less-controlled | Less-controlled |
| PC preparation protocol | Highly standardized | Less-standardized | Less-standardized |
| PC quality | Protocol-dependent | Not assured | Not assured |
| Sample size | Large (typically 100–1000; power-test at least 80%) | Small ~ middle | Small ~ large |
| Research center | Preferential multicentric | 1 ~ several | 1 ~ several |
| Primary end points | Narrow (single) | Narrow ~ middle (multi) | Middle (multi)? |
| Clinical outcomes | – | Controversial (negative ≥ positive) | – |
| Type of Bias | How to Avoid |
|---|---|
| |
| Flawed study design |
|
| Selection bias |
|
| channeling bias |
|
| |
| Interview bias |
|
| Chronology bias |
|
| Recall bias |
|
| Transfer bias |
|
| Exposure Misclassification |
|
| Outcome misclassification |
|
| Performance bias |
|
| |
| Citation bias (publication bias) |
|
| Confounding |
|
| Stage | Major Requirements |
|---|---|
| Basic and preclinical |
|
| |
| |
| |
| |
| Clinical |
|
| |
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawase, T.; Mubarak, S.; Mourão, C.F. The Platelet Concentrates Therapy: From the Biased Past to the Anticipated Future. Bioengineering 2020, 7, 82. https://doi.org/10.3390/bioengineering7030082
Kawase T, Mubarak S, Mourão CF. The Platelet Concentrates Therapy: From the Biased Past to the Anticipated Future. Bioengineering. 2020; 7(3):82. https://doi.org/10.3390/bioengineering7030082
Chicago/Turabian StyleKawase, Tomoyuki, Suliman Mubarak, and Carlos Fernando Mourão. 2020. "The Platelet Concentrates Therapy: From the Biased Past to the Anticipated Future" Bioengineering 7, no. 3: 82. https://doi.org/10.3390/bioengineering7030082
APA StyleKawase, T., Mubarak, S., & Mourão, C. F. (2020). The Platelet Concentrates Therapy: From the Biased Past to the Anticipated Future. Bioengineering, 7(3), 82. https://doi.org/10.3390/bioengineering7030082







