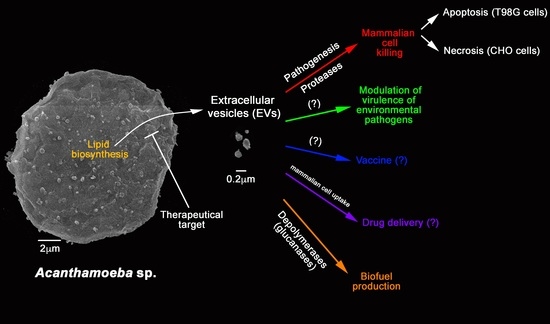
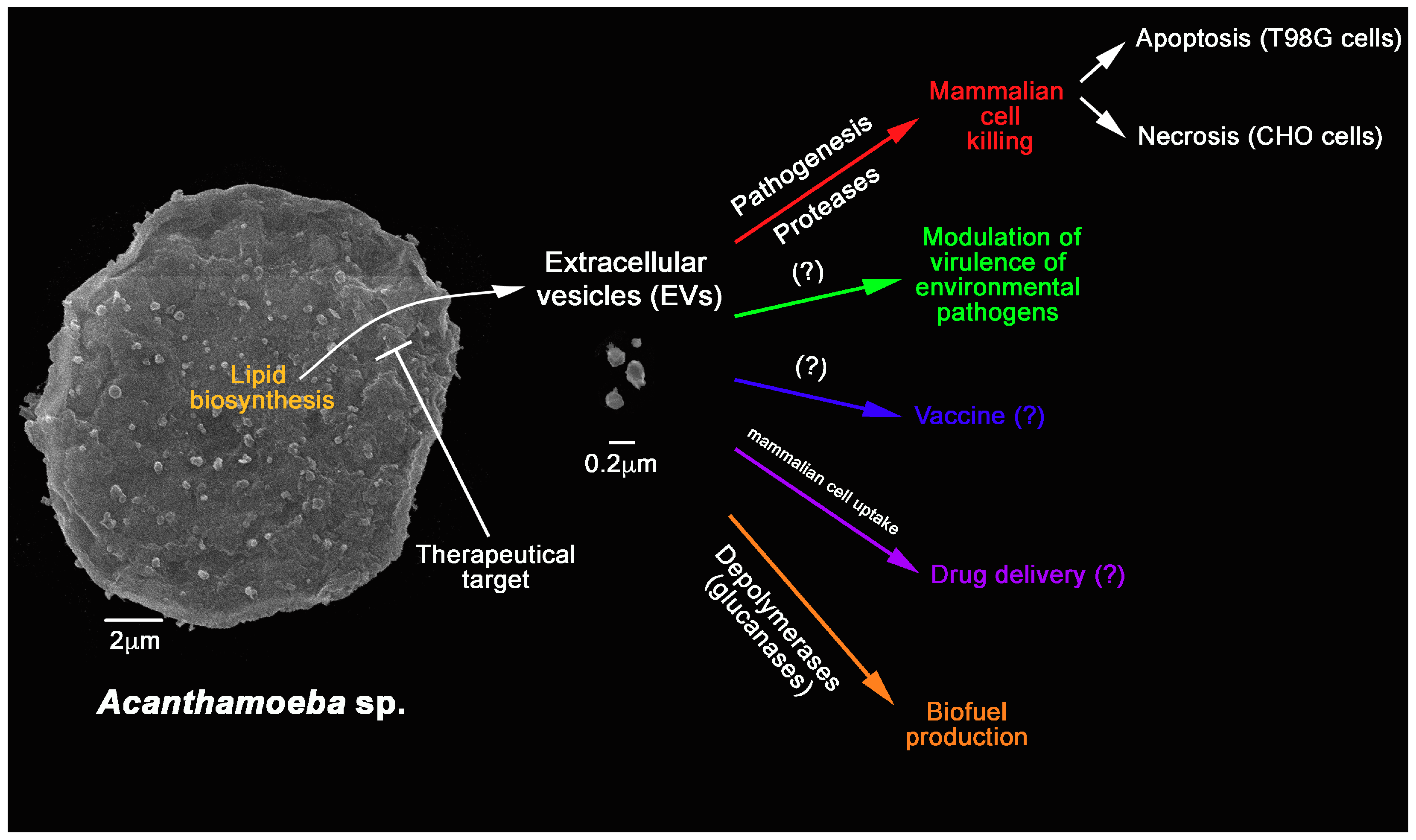
Extracellular Vesicles from the Protozoa Acanthamoeba castellanii: Their Role in Pathogenesis, Environmental Adaptation and Potential Applications
Abstract
1. Introduction
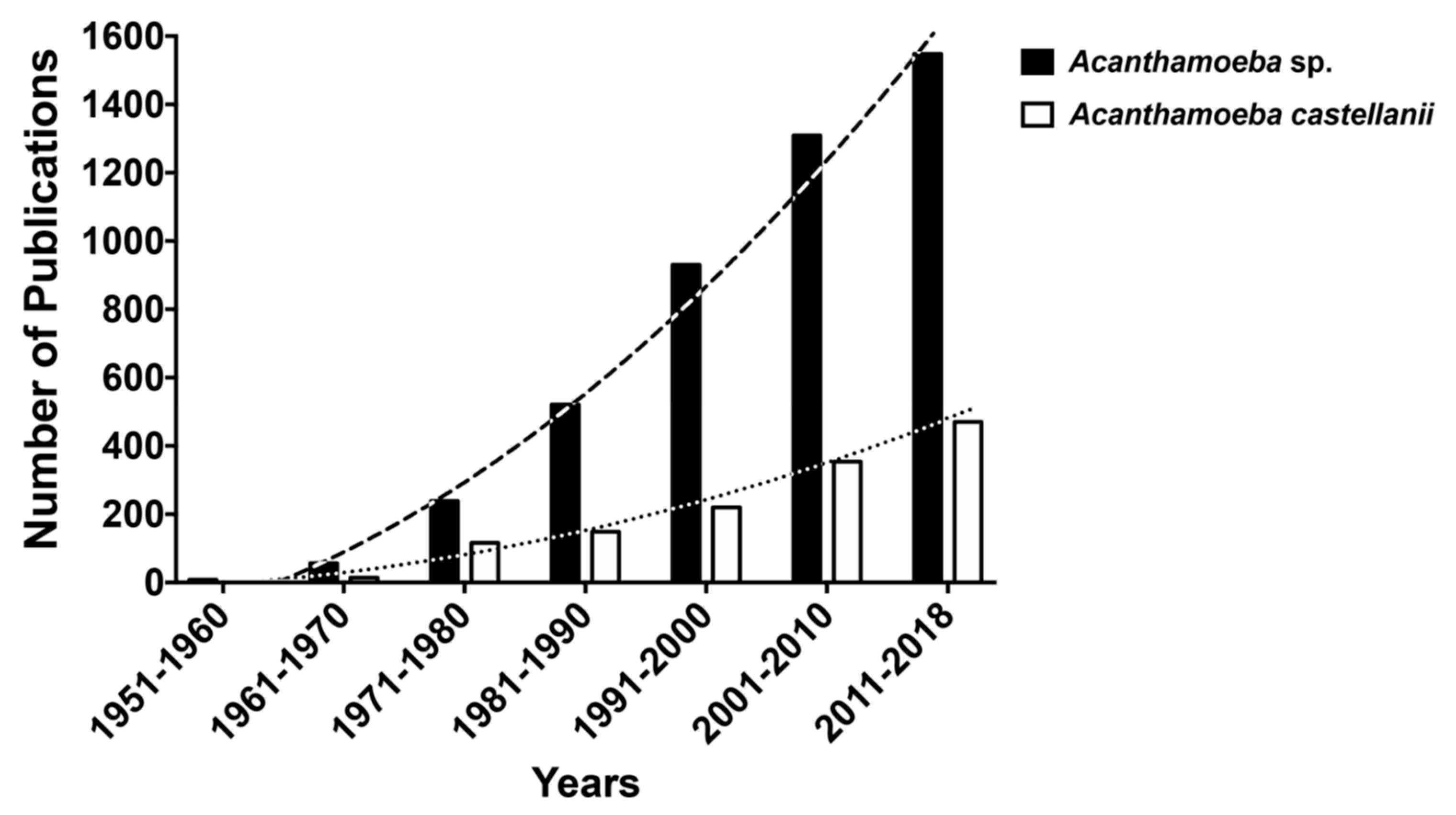
1.1. Free Living Amoebae (FLA)
1.2. FLAs and the Genus Acanthamoeba
2. Extracellular Vesicles (EVs) and Their Role in Microorganism Adaptation
3. Characterized EVs Throughout Other Kingdoms and Their Role in Microbial Pathogenesis
4. Protozoa EVs and Pathogenesis
5. Recently Characterized EVs of FLAs and Their Impact in Pathogenesis
Lipids in A. castellanii EVs
6. Further Potential Applications of FLAs EVs
7. Conclusion Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Thomas, J.M.; Ashbolt, N.J. Do free-living amoebae in treated drinking water systems present an emerging health risk? Environ. Sci. Technol. 2011, 45, 860–869. [Google Scholar] [CrossRef] [PubMed]
- Auran, J.D.; Starr, M.B.; Jakobiec, F.A. Acanthamoeba keratitis. A review of the literature. Cornea 1987, 6, 2–26. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.G., Jr.; Coster, D.J.; Badenoch, P.R. Acanthamoeba keratitis. Resolution after epithelial debridement. Cornea 1994, 13, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Falchi, R.L.R. Contaminação por protozoários potencialmente patogênicos ao homem na água de diferentes pontos da Laguna dos Patos, Rio Grande, RS; Departamento de Microbiologia e Parasitologia, Universidade Federal de Pelotas: Pelotas, Brazil, 2006; p. 90. [Google Scholar]
- Magnet, A.; Galvan, A.L.; Fenoy, S.; Izquierdo, F.; Rueda, C.; Fernandez Vadillo, C.; Perez-Irezabal, J.; Bandyopadhyay, K.; Visvesvara, G.S.; da Silva, A.J.; et al. Molecular characterization of Acanthamoeba isolated in water treatment plants and comparison with clinical isolates. Parasitol. Res. 2012, 111, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Sriram, R.; Shoff, M.; Booton, G.; Fuerst, P.; Visvesvara, G.S. Survival of Acanthamoeba cysts after desiccation for more than 20 years. J. Clin. Microbiol. 2008, 46, 4045–4048. [Google Scholar] [CrossRef] [PubMed]
- Stehr-Green, J.K.; Bailey, T.M.; Visvesvara, G.S. The epidemiology of Acanthamoeba keratitis in the United States. Am. J. Ophthalmol. 1989, 107, 331–336. [Google Scholar] [CrossRef]
- Walker, C.W. Acanthamoeba: Ecology, pathogenicity and laboratory detection. Br. J. Biomed. Sci. 1996, 53, 146–151. [Google Scholar]
- Marciano-Cabral, F.; Cabral, G. Acanthamoeba spp. as agents of disease in humans. Clin. Microbiol. Rev. 2003, 16, 273–307. [Google Scholar] [CrossRef]
- Coulon, C.; Collignon, A.; McDonnell, G.; Thomas, V. Resistance of Acanthamoeba cysts to disinfection treatments used in health care settings. J. Clin. Microbiol. 2010, 48, 2689–2697. [Google Scholar] [CrossRef]
- Taravaud, A.; Ali, M.; Lafosse, B.; Nicolas, V.; Feliers, C.; Thibert, S.; Levi, Y.; Loiseau, P.M.; Pomel, S. Enrichment of free-living amoebae in biofilms developed at upper water levels in drinking water storage towers: An inter- and intra-seasonal study. Sci. Total Environ. 2018, 633, 157–166. [Google Scholar] [CrossRef]
- Ashbolt, N.J. Environmental (Saprozoic) Pathogens of Engineered Water Systems: Understanding Their Ecology for Risk Assessment and Management. Pathogens 2015, 4, 390–405. [Google Scholar] [CrossRef] [PubMed]
- Berry, D.; Xi, C.; Raskin, L. Microbial ecology of drinking water distribution systems. Curr. Opin. Biotechnol. 2006, 17, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Miller, H.C.; Wylie, J.T.; Kaksonen, A.H.; Sutton, D.; Puzon, G.J. Competition between Naegleria fowleri and Free Living Amoeba Colonizing Laboratory Scale and Operational Drinking Water Distribution Systems. Environ. Sci. Technol. 2018, 52, 2549–2557. [Google Scholar] [CrossRef] [PubMed]
- Gargano, J.W.; Adam, E.A.; Collier, S.A.; Fullerton, K.E.; Feinman, S.J.; Beach, M.J. Mortality from selected diseases that can be transmitted by water—United States, 2003–2009. J. Water Health 2017, 15, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A. Cards of Virulence and the Global Virulome for Humans. Microbe 2006, 1, 359–364. [Google Scholar] [CrossRef]
- Casadevall, A. Amoeba provide insight into the origin of virulence in pathogenic fungi. Adv. Exp. Med. Biol. 2012, 710, 1–10. [Google Scholar] [PubMed]
- Guimaraes, A.J.; Gomes, K.X.; Cortines, J.R.; Peralta, J.M.; Peralta, R.H. Acanthamoeba spp. as a universal host for pathogenic microorganisms: One bridge from environment to host virulence. Microbiol. Res. 2016, 193, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Mylonakis, E.; Casadevall, A.; Ausubel, F.M. Exploiting amoeboid and non-vertebrate animal model systems to study the virulence of human pathogenic fungi. PLoS Pathog. 2007, 3, e101. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Pirofski, L.A. Accidental virulence, cryptic pathogenesis, martians, lost hosts, and the pathogenicity of environmental microbes. Eukaryot. Cell 2007, 6, 2169–2174. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Pirofski, L. Host-pathogen interactions: The attributes of virulence. J. Infect. Dis. 2001, 184, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Chrisman, C.J.; Albuquerque, P.; Guimaraes, A.J.; Nieves, E.; Casadevall, A. Phospholipids trigger Cryptococcus neoformans capsular enlargement during interactions with amoebae and macrophages. PLoS Pathog. 2011, 7, e1002047. [Google Scholar] [CrossRef] [PubMed]
- Steenbergen, J.N.; Shuman, H.A.; Casadevall, A. Cryptococcus neoformans interactions with amoebae suggest an explanation for its virulence and intracellular pathogenic strategy in macrophages. Proc. Natl. Acad. Sci. USA 2001, 98, 15245–15250. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.J.; Sotelo-Avila, C.; Garcia-Tamayo, J.; Moron, J.T.; Willaert, E.; Stamm, W.P. Meningoencephalitis due to Acanthamoeba SP. Pathogenesis and clinico-pathological study. Acta Neuropathol. 1977, 37, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Duma, R.J.; Helwig, W.B.; Martinez, A.J. Meningoencephalitis and brain abscess due to a free-living amoeba. Ann. Intern. Med. 1978, 88, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Bhagwandeen, S.B.; Carter, R.F.; Naik, K.G.; Levitt, D. A case of hartmannellid amebic meningoencephalitis in Zambia. Am. J. Clin. Pathol. 1975, 63, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Ofori-Kwakye, S.K.; Sidebottom, D.G.; Herbert, J.; Fischer, E.G.; Visvesvara, G.S. Granulomatous brain tumor caused by Acanthamoeba. Case report. J. Neurosurg. 1986, 64, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Ringsted, J.; Jager, B.V.; Suk, D.; Visvesvara, G.S. Probable acanthamoeba meningoencephalitis in a Korean child. Am. J. Clin. Pathol. 1976, 66, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Singhal, T.; Bajpai, A.; Kalra, V.; Kabra, S.K.; Samantaray, J.C.; Satpathy, G.; Gupta, A.K. Successful treatment of Acanthamoeba meningitis with combination oral antimicrobials. Pediatr. Infect. Dis. J. 2001, 20, 623–627. [Google Scholar] [CrossRef]
- Sangruchi, T.; Martinez, A.J.; Visvesvara, G.S. Spontaneous granulomatous amebic encephalitis: Report of four cases from Thailand. Southeast Asian J. Trop. Med. Public Health 1994, 25, 309–313. [Google Scholar]
- Bacon, A.S.; Frazer, D.G.; Dart, J.K.; Matheson, M.; Ficker, L.A.; Wright, P. A review of 72 consecutive cases of Acanthamoeba keratitis, 1984–1992. Eye 1993, 7 Pt 6, 719–725. [Google Scholar] [CrossRef]
- Moore, M.B.; McCulley, J.P.; Luckenbach, M.; Gelender, H.; Newton, C.; McDonald, M.B.; Visvesvara, G.S. Acanthamoeba keratitis associated with soft contact lenses. Am. J. Ophthalmol. 1985, 100, 396–403. [Google Scholar] [CrossRef]
- Seal, D.V.; Hay, J.; Devonshire, P.; Kirkness, C.M. Acanthamoeba and contact lens disinfection: Should chlorine be discontinued? Br. J. Ophthalmol. 1993, 77, 128. [Google Scholar] [CrossRef] [PubMed]
- Seal, D.V.; Hay, J. Acanthamoeba keratitis. BMJ 1994, 309, 1019. [Google Scholar] [CrossRef] [PubMed]
- Visvesvara, G.S.; Booton, G.C.; Kelley, D.J.; Fuerst, P.; Sriram, R.; Finkelstein, A.; Garner, M.M. In vitro culture, serologic and molecular analysis of Acanthamoeba isolated from the liver of a keel-billed toucan (Ramphastos sulfuratus). Vet. Parasitol. 2007, 143, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Barker, J.; Brown, M.R. Trojan horses of the microbial world: Protozoa and the survival of bacterial pathogens in the environment. Microbiology 1994, 140 Pt 6, 1253–1259. [Google Scholar] [CrossRef]
- Elsheikha, H.M.; Khan, N.A. Protozoa traversal of the blood-brain barrier to invade the central nervous system. FEMS Microbiol. Rev. 2010, 34, 532–553. [Google Scholar] [CrossRef] [PubMed]
- Tosetti, N.; Croxatto, A.; Greub, G. Amoebae as a tool to isolate new bacterial species, to discover new virulence factors and to study the host-pathogen interactions. Microb. Pathog. 2014, 77, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, D.S.; Ferreira, M.D.S.; Liedke, S.C.; Gomes, K.X.; de Oliveira, G.A.; Leao, P.E.L.; Cesar, G.V.; Seabra, S.H.; Cortines, J.R.; Casadevall, A.; et al. Extracellular vesicles and vesicle-free secretome of the protozoa Acanthamoeba castellanii under homeostasis and nutritional stress and their damaging potential to host cells. Virulence 2018, 9, 818–836. [Google Scholar] [CrossRef]
- Mantel, P.Y.; Marti, M. The role of extracellular vesicles in Plasmodium and other protozoan parasites. Cell. Microbiol. 2014, 16, 344–354. [Google Scholar] [CrossRef]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef]
- Trocoli Torrecilhas, A.C.; Tonelli, R.R.; Pavanelli, W.R.; da Silva, J.S.; Schumacher, R.I.; de Souza, W.; E Silva, N.C.; de Almeida Abrahamsohn, I.; Colli, W.; Manso Alves, M.J. Trypanosoma cruzi: Parasite shed vesicles increase heart parasitism and generate an intense inflammatory response. Microbes Infect. 2009, 11, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Ulsamer, A.G.; Smith, F.R.; Korn, E.D. Lipids of Acanthamoeba castellanii. Composition and effects of phagocytosis on incorporation of radioactive precursors. J. Cell Biol. 1969, 43, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Joffe, L.S.; Nimrichter, L.; Rodrigues, M.L.; Del Poeta, M. Potential Roles of Fungal Extracellular Vesicles during Infection. mSphere 2016, 1, e00099-16. [Google Scholar] [CrossRef]
- Kalluri, R. The biology and function of exosomes in cancer. J. Clin. Investig. 2016, 126, 1208–1215. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, D.L.; Freire-de-Lima, C.G.; Nosanchuk, J.D.; Casadevall, A.; Rodrigues, M.L.; Nimrichter, L. Extracellular vesicles from Cryptococcus neoformans modulate macrophage functions. Infect. Immun. 2010, 78, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Schorey, J.S.; Harding, C.V. Extracellular vesicles and infectious diseases: New complexity to an old story. J. Clin. Investig. 2016, 126, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Turpin, D.; Truchetet, M.E.; Faustin, B.; Augusto, J.F.; Contin-Bordes, C.; Brisson, A.; Blanco, P.; Duffau, P. Role of extracellular vesicles in autoimmune diseases. Autoimmun. Rev. 2016, 15, 174–183. [Google Scholar] [CrossRef]
- Reece, J.B.; Urry, L.A.; Cain, M.L.; Wasserman, S.A.; Minorsky, P.V.; Jackson, R.B. Campbell Biology; Pearson: Boston, MA, USA, 2014. [Google Scholar]
- Rodrigues, M.L.; Nakayasu, E.S.; Almeida, I.C.; Nimrichter, L. The impact of proteomics on the understanding of functions and biogenesis of fungal extracellular vesicles. J. Proteomics 2014, 97, 177–186. [Google Scholar] [CrossRef]
- Brown, L.; Wolf, J.M.; Prados-Rosales, R.; Casadevall, A. Through the wall: Extracellular vesicles in Gram-positive bacteria, mycobacteria and fungi. Nat. Rev. Microbiol. 2015, 13, 620–630. [Google Scholar] [CrossRef]
- Zamith-Miranda, D.; Nimrichter, L.; Rodrigues, M.L.; Nosanchuk, J.D. Fungal extracellular vesicles: Modulating host-pathogen interactions by both the fungus and the host. Microbes Infect. 2018, 20, 501–504. [Google Scholar] [CrossRef]
- Deatherage, B.L.; Cookson, B.T. Membrane vesicle release in bacteria, eukaryotes, and archaea: A conserved yet underappreciated aspect of microbial life. Infect. Immun. 2012, 80, 1948–1957. [Google Scholar] [CrossRef] [PubMed]
- Schwechheimer, C.; Kuehn, M.J. Outer-membrane vesicles from Gram-negative bacteria: Biogenesis and functions. Nat. Rev. Microbiol. 2015, 13, 605–619. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, R.; Fernandez, S.; Zayas, C.; Acosta, A.; Sarmiento, M.E.; Ferro, V.A.; Rosenqvist, E.; Campa, C.; Cardoso, D.; Garcia, L.; et al. Bacterial outer membrane vesicles and vaccine applications. Front. Immunol. 2014, 5, 121. [Google Scholar] [CrossRef] [PubMed]
- Arnold, R.; Galloway, Y.; McNicholas, A.; O’Hallahan, J. Effectiveness of a vaccination programme for an epidemic of meningococcal B in New Zealand. Vaccine 2011, 29, 7100–7106. [Google Scholar] [CrossRef] [PubMed]
- Holst, J.; Oster, P.; Arnold, R.; Tatley, M.V.; Naess, L.M.; Aaberge, I.S.; Galloway, Y.; McNicholas, A.; O’Hallahan, J.; Rosenqvist, E.; et al. Vaccines against meningococcal serogroup B disease containing outer membrane vesicles (OMV): Lessons from past programs and implications for the future. Hum. Vaccin. Immunother. 2013, 9, 1241–1253. [Google Scholar] [CrossRef]
- Lee, W.H.; Choi, H.I.; Hong, S.W.; Kim, K.S.; Gho, Y.S.; Jeon, S.G. Vaccination with Klebsiella pneumoniae-derived extracellular vesicles protects against bacteria-induced lethality via both humoral and cellular immunity. Exp. Mol. Med. 2015, 47, e183. [Google Scholar] [CrossRef]
- Shockman, G.D.; Barrett, J.F. Structure, function, and assembly of cell walls of gram-positive bacteria. Annu. Rev. Microbiol. 1983, 37, 501–527. [Google Scholar] [CrossRef]
- Rivera, J.; Cordero, R.J.; Nakouzi, A.S.; Frases, S.; Nicola, A.; Casadevall, A. Bacillus anthracis produces membrane-derived vesicles containing biologically active toxins. Proc. Natl. Acad. Sci. USA 2010, 107, 19002–19007. [Google Scholar] [CrossRef]
- Prados-Rosales, R.; Carreno, L.J.; Batista-Gonzalez, A.; Baena, A.; Venkataswamy, M.M.; Xu, J.; Yu, X.; Wallstrom, G.; Magee, D.M.; LaBaer, J.; et al. Mycobacterial membrane vesicles administered systemically in mice induce a protective immune response to surface compartments of Mycobacterium tuberculosis. mBio 2014, 5, e01921-14. [Google Scholar] [CrossRef]
- Albuquerque, P.C.; Nakayasu, E.S.; Rodrigues, M.L.; Frases, S.; Casadevall, A.; Zancope-Oliveira, R.M.; Almeida, I.C.; Nosanchuk, J.D. Vesicular transport in Histoplasma capsulatum: An effective mechanism for trans-cell wall transfer of proteins and lipids in ascomycetes. Cell. Microbiol. 2008, 10, 1695–1710. [Google Scholar] [CrossRef]
- Rodrigues, M.L.; Nimrichter, L.; Oliveira, D.L.; Frases, S.; Miranda, K.; Zaragoza, O.; Alvarez, M.; Nakouzi, A.; Feldmesser, M.; Casadevall, A. Vesicular polysaccharide export in Cryptococcus neoformans is a eukaryotic solution to the problem of fungal trans-cell wall transport. Eukaryot. Cell 2007, 6, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.L.; Nimrichter, L.; Oliveira, D.L.; Nosanchuk, J.D.; Casadevall, A. Vesicular Trans-Cell Wall Transport in Fungi: A Mechanism for the Delivery of Virulence-Associated Macromolecules? Lipid Insights 2008, 2, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Vallejo, M.C.; Matsuo, A.L.; Ganiko, L.; Medeiros, L.C.; Miranda, K.; Silva, L.S.; Freymuller-Haapalainen, E.; Sinigaglia-Coimbra, R.; Almeida, I.C.; Puccia, R. The pathogenic fungus Paracoccidioides brasiliensis exports extracellular vesicles containing highly immunogenic alpha-Galactosyl epitopes. Eukaryot. Cell. 2011, 10, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Vargas, G.; Rocha, J.D.; Oliveira, D.L.; Albuquerque, P.C.; Frases, S.; Santos, S.S.; Nosanchuk, J.D.; Gomes, A.M.; Medeiros, L.C.; Miranda, K.; et al. Compositional and immunobiological analyses of extracellular vesicles released by Candida albicans. Cell. Microbiol. 2015, 17, 389–407. [Google Scholar] [CrossRef] [PubMed]
- Rittershaus, P.C.; Kechichian, T.B.; Allegood, J.C.; Merrill, A.H., Jr.; Hennig, M.; Luberto, C.; Del Poeta, M. Glucosylceramide synthase is an essential regulator of pathogenicity of Cryptococcus neoformans. J. Clin. Investig. 2006, 116, 1651–1659. [Google Scholar] [CrossRef] [PubMed]
- Eisenman, H.C.; Frases, S.; Nicola, A.M.; Rodrigues, M.L.; Casadevall, A. Vesicle-associated melanization in Cryptococcus neoformans. Microbiology 2009, 155, 3860–3867. [Google Scholar] [CrossRef] [PubMed]
- Peres da Silva, R.; Puccia, R.; Rodrigues, M.L.; Oliveira, D.L.; Joffe, L.S.; Cesar, G.V.; Nimrichter, L.; Goldenberg, S.; Alves, L.R. Extracellular vesicle-mediated export of fungal RNA. Sci. Rep. 2015, 5, 7763. [Google Scholar] [CrossRef] [PubMed]
- Marti, M.; Johnson, P.J. Emerging roles for extracellular vesicles in parasitic infections. Curr. Opin. Microbiol. 2016, 32, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Silverman, J.M.; Clos, J.; de’Oliveira, C.C.; Shirvani, O.; Fang, Y.; Wang, C.; Foster, L.J.; Reiner, N.E. An exosome-based secretion pathway is responsible for protein export from Leishmania and communication with macrophages. J. Cell Sci. 2010, 123, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Geiger, A.; Hirtz, C.; Becue, T.; Bellard, E.; Centeno, D.; Gargani, D.; Rossignol, M.; Cuny, G.; Peltier, J.B. Exocytosis and protein secretion in Trypanosoma. BMC Microbiol. 2010, 10, 20. [Google Scholar] [CrossRef] [PubMed]
- Szempruch, A.J.; Sykes, S.E.; Kieft, R.; Dennison, L.; Becker, A.C.; Gartrell, A.; Martin, W.J.; Nakayasu, E.S.; Almeida, I.C.; Hajduk, S.L.; et al. Extracellular Vesicles from Trypanosoma brucei Mediate Virulence Factor Transfer and Cause Host Anemia. Cell 2016, 164, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Lavialle, F.; Deshayes, S.; Gonnet, F.; Larquet, E.; Kruglik, S.G.; Boisset, N.; Daniel, R.; Alfsen, A.; Tatischeff, I. Nanovesicles released by Dictyostelium cells: A potential carrier for drug delivery. Int. J. Pharm. 2009, 380, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Tatischeff, I.; Lavialle, F.; Pigaglio-Deshayes, S.; Pechoux-Longin, C.; Chinsky, L.; Alfsen, A. Dictyostelium extracellular vesicles containing hoechst 33342 transfer the dye into the nuclei of living cells: A fluorescence study. J. Fluoresc. 2008, 18, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Marcilla, A.; Trelis, M.; Cortes, A.; Sotillo, J.; Cantalapiedra, F.; Minguez, M.T.; Valero, M.L.; Sanchez del Pino, M.M.; Munoz-Antoli, C.; Toledo, R.; et al. Extracellular vesicles from parasitic helminths contain specific excretory/secretory proteins and are internalized in intestinal host cells. PLoS ONE 2012, 7, e45974. [Google Scholar] [CrossRef] [PubMed]
- Torrecilhas, A.C.; Schumacher, R.I.; Alves, M.J.; Colli, W. Vesicles as carriers of virulence factors in parasitic protozoan diseases. Microbes Infect. 2012, 14, 1465–1474. [Google Scholar] [CrossRef] [PubMed]
- Mugnier, M.R.; Papavasiliou, F.N.; Schulz, D. Vesicles as Vehicles for Virulence. Trends Parasitol. 2016, 32, 435–436. [Google Scholar] [CrossRef] [PubMed]
- Silverman, J.M.; Chan, S.K.; Robinson, D.P.; Dwyer, D.M.; Nandan, D.; Foster, L.J.; Reiner, N.E. Proteomic analysis of the secretome of Leishmania donovani. Genome Biol. 2008, 9, R35. [Google Scholar] [CrossRef] [PubMed]
- Silverman, J.M.; Clos, J.; Horakova, E.; Wang, A.Y.; Wiesgigl, M.; Kelly, I.; Lynn, M.A.; McMaster, W.R.; Foster, L.J.; Levings, M.K.; et al. Leishmania exosomes modulate innate and adaptive immune responses through effects on monocytes and dendritic cells. J. Immunol. 2010, 185, 5011–5022. [Google Scholar] [CrossRef]
- Fernandez-Calero, T.; Garcia-Silva, R.; Pena, A.; Robello, C.; Persson, H.; Rovira, C.; Naya, H.; Cayota, A. Profiling of small RNA cargo of extracellular vesicles shed by Trypanosoma cruzi reveals a specific extracellular signature. Mol. Biochem. Parasitol. 2015, 199, 19–28. [Google Scholar] [CrossRef]
- Du, Q.; Kawabe, Y.; Schilde, C.; Chen, Z.H.; Schaap, P. The Evolution of Aggregative Multicellularity and Cell-Cell Communication in the Dictyostelia. J. Mol. Biol. 2015, 427, 3722–3733. [Google Scholar] [CrossRef]
- Loomis, W.F. Cell signaling during development of Dictyostelium. Dev. Biol. 2014, 391, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Lidell, M.E.; Moncada, D.M.; Chadee, K.; Hansson, G.C. Entamoeba histolytica cysteine proteases cleave the MUC2 mucin in its C-terminal domain and dissolve the protective colonic mucus gel. Proc. Natl. Acad. Sci. USA 2006, 103, 9298–9303. [Google Scholar] [CrossRef] [PubMed]
- Ocadiz, R.; Orozco, E.; Carrillo, E.; Quintas, L.I.; Ortega-Lopez, J.; Garcia-Perez, R.M.; Sanchez, T.; Castillo-Juarez, B.A.; Garcia-Rivera, G.; Rodriguez, M.A. EhCP112 is an Entamoeba histolytica secreted cysteine protease that may be involved in the parasite-virulence. Cell. Microbiol. 2005, 7, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Bouyer, S.; Rodier, M.H.; Guillot, A.; Hechard, Y. Acanthamoeba castellanii: Proteins involved in actin dynamics, glycolysis, and proteolysis are regulated during encystation. Exp. Parasitol. 2009, 123, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Dudley, R.; Alsam, S.; Khan, N.A. The role of proteases in the differentiation of Acanthamoeba castellanii. FEMS Microbiol. Lett. 2008, 286, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, M.; Shah, N.; Zanetti, B.R.; Maugeri, M.; Silvestre, R.N.; Fatima, F.; Neder, L.; Valadi, H. Extracellular Vesicles and Matrix Remodeling Enzymes: The Emerging Roles in Extracellular Matrix Remodeling, Progression of Diseases and Tissue Repair. Cells 2018, 7, 167. [Google Scholar] [CrossRef] [PubMed]
- Rocha-Azevedo, B.D.; Jamerson, M.; Cabral, G.A.; Silva-Filho, F.C.; Marciano-Cabral, F. Acanthamoeba interaction with extracellular matrix glycoproteins: Biological and biochemical characterization and role in cytotoxicity and invasiveness. J. Eukaryot. Microbiol. 2009, 56, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Bayer-Santos, E.; Aguilar-Bonavides, C.; Rodrigues, S.P.; Cordero, E.M.; Marques, A.F.; Varela-Ramirez, A.; Choi, H.; Yoshida, N.; da Silveira, J.F.; Almeida, I.C. Proteomic analysis of Trypanosoma cruzi secretome: Characterization of two populations of extracellular vesicles and soluble proteins. J. Proteome Res. 2013, 12, 883–897. [Google Scholar] [CrossRef]
- Mantel, P.Y.; Hoang, A.N.; Goldowitz, I.; Potashnikova, D.; Hamza, B.; Vorobjev, I.; Ghiran, I.; Toner, M.; Irimia, D.; Ivanov, A.R.; et al. Malaria-infected erythrocyte-derived microvesicles mediate cellular communication within the parasite population and with the host immune system. Cell Host Microbe 2013, 13, 521–534. [Google Scholar] [CrossRef]
- Evans-Osses, I.; Reichembach, L.H.; Ramirez, M.I. Exosomes or microvesicles? Two kinds of extracellular vesicles with different routes to modify protozoan-host cell interaction. Parasitol. Res. 2015, 114, 3567–3575. [Google Scholar] [CrossRef]
- Rodrigues, M.L.; Nakayasu, E.S.; Oliveira, D.L.; Nimrichter, L.; Nosanchuk, J.D.; Almeida, I.C.; Casadevall, A. Extracellular vesicles produced by Cryptococcus neoformans contain protein components associated with virulence. Eukaryot. Cell 2008, 7, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Ulsamer, A.G.; Wright, P.L.; Wetzel, M.G.; Korn, E.D. Plasma and phagosome membranes of Acanthamoeba castellanii. J. Cell Biol. 1971, 51, 193–215. [Google Scholar] [CrossRef] [PubMed]
- Korn, E.D.; Dearborn, D.G.; Wright, P.L. Lipophosphonoglycan of the plasma membrance of Acanthamoeba castellanii. Isolation from whole amoebae and identification of the water-soluble products of acid hydrolysis. J. Biol Chem. 1974, 249, 3335–3341. [Google Scholar] [PubMed]
- Bowers, B.; Korn, E.D. Localization of lipophosphonoglycan on both sides of Acanthamoeba plasma membrane. J. Cell Biol. 1974, 62, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Dearborn, D.G.; Smith, S.; Korn, E.D. Lipophosphonoglycan of the plasma membrane of A canthamoeba castellanii. Inositol and phytosphingosine content and general structural features. J. Biol Chem. 1976, 251, 2976–2982. [Google Scholar] [PubMed]
- Dearborn, D.G.; Korn, E.D. Lipophosphonoglycan of the plasma membrane of Acanthamoeba castellanii. Fatty acid composition. J. Biol Chem. 1974, 249, 3342–3346. [Google Scholar]
- Karas, M.A.; Russa, R. New long chain bases in lipophosphonoglycan of Acanthamoeba castellanii. Lipids 2013, 48, 639–650. [Google Scholar] [CrossRef]
- Franco, L.H.; Beverley, S.M.; Zamboni, D.S. Innate immune activation and subversion of Mammalian functions by leishmania lipophosphoglycan. J. Parasitol. Res. 2012, 2012, 165126. [Google Scholar] [CrossRef]
- Cano, A.; Mattana, A.; Woods, S.; Henriquez, F.L.; Alexander, J.; Roberts, C.W. Acanthamoeba Activates Macrophages Predominantly through Toll-Like Receptor 4- and MyD88-Dependent Mechanisms To Induce Interleukin-12 (IL-12) and IL-6. Infect. Immun. 2017, 85, e01054-16. [Google Scholar] [CrossRef]
- Raederstorff, D.; Rohmer, M. Sterol biosynthesis de nova via cycloartenol by the soil amoeba Acanthamoeba polyphaga. Biochem. J. 1985, 231, 609–615. [Google Scholar] [CrossRef]
- Alexander-Lindo, R.L.; Morrison, E.Y.; Nair, M.G. Hypoglycaemic effect of stigmast-4-en-3-one and its corresponding alcohol from the bark of Anacardium occidentale (cashew). Phytother. Res. 2004, 18, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Thomson, S.; Rice, C.; Zhang, T.; Edrada-Ebel, R.; Henriquez, F.L.; Roberts, C.W. Characterisation of sterol biosynthesis and validation of 14α-demethylase as a drug target in Acanthamoeba. Sci Rep. 2017, 7, 8247. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.L.; Hann, A.C.; Harwood, J.L.; Lloyd, D. Temperature-induced membrane-lipid adaptation in Acanthamoeba castellanii. Biochem. J. 1993, 290 Pt 1, 273–278. [Google Scholar] [CrossRef]
- Liu, H.; Chen, L.; Peng, Y.; Yu, S.; Liu, J.; Wu, L.; Zhang, L.; Wu, Q.; Chang, X.; Yu, X.; et al. Dendritic cells loaded with tumor derived exosomes for cancer immunotherapy. Oncotarget 2018, 9, 2887–2894. [Google Scholar] [CrossRef] [PubMed]
- Peres da Silva, R.; Heiss, C.; Black, I.; Azadi, P.; Gerlach, J.Q.; Travassos, L.R.; Joshi, L.; Kilcoyne, M.; Puccia, R. Extracellular vesicles from Paracoccidioides pathogenic species transport polysaccharide and expose ligands for DC-SIGN receptors. Sci. Rep. 2015, 5, 14213. [Google Scholar] [CrossRef] [PubMed]
- Tatischeff, I. Assets of the non-pathogenic microorganism Dictyostelium discoideum as a model for the study of eukaryotic extracellular vesicles. F1000Res 2013, 2, 73. [Google Scholar] [CrossRef]
- Contesini, F.J.; de Alencar Figueira, J.; Kawaguti, H.Y.; de Barros Fernandes, P.C.; de Oliveira Carvalho, P.; da Graca Nascimento, M.; Sato, H.H. Potential applications of carbohydrases immobilization in the food industry. Int. J. Mol. Sci. 2013, 14, 1335–1369. [Google Scholar] [CrossRef]
- James, J.; Simpson, B.K. Application of enzymes in food processing. Crit. Rev. Food Sci. Nutr. 1996, 36, 437–463. [Google Scholar] [CrossRef]
- de Souza, P.M.; de Oliveira Magalhaes, P. Application of microbial alpha-amylase in industry—A review. Braz. J. Microbiol. 2010, 41, 850–861. [Google Scholar] [CrossRef]
- Divakar, S. Glycosidases; Springer: Bangalore, India, 2013. [Google Scholar]
- Taron, C.; Bielik, A.; Guthrie, E.P.; Shi, X. Redesigning Glycosidase Manufacturing Quality for Pharmaceutical and Clinical Applications; New England Biolabs: Ipswich, MA, USA, 2017. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, D.d.S.; Ferreira, M.d.S.; Guimarães, A.J. Extracellular Vesicles from the Protozoa Acanthamoeba castellanii: Their Role in Pathogenesis, Environmental Adaptation and Potential Applications. Bioengineering 2019, 6, 13. https://doi.org/10.3390/bioengineering6010013
Gonçalves DdS, Ferreira MdS, Guimarães AJ. Extracellular Vesicles from the Protozoa Acanthamoeba castellanii: Their Role in Pathogenesis, Environmental Adaptation and Potential Applications. Bioengineering. 2019; 6(1):13. https://doi.org/10.3390/bioengineering6010013
Chicago/Turabian StyleGonçalves, Diego de Souza, Marina da Silva Ferreira, and Allan J. Guimarães. 2019. "Extracellular Vesicles from the Protozoa Acanthamoeba castellanii: Their Role in Pathogenesis, Environmental Adaptation and Potential Applications" Bioengineering 6, no. 1: 13. https://doi.org/10.3390/bioengineering6010013
APA StyleGonçalves, D. d. S., Ferreira, M. d. S., & Guimarães, A. J. (2019). Extracellular Vesicles from the Protozoa Acanthamoeba castellanii: Their Role in Pathogenesis, Environmental Adaptation and Potential Applications. Bioengineering, 6(1), 13. https://doi.org/10.3390/bioengineering6010013