The study design presented in this section begins with an overview of the animal study that supplied preliminary data and an explanation of the scheme used for informing PCLC tuning, followed by descriptions of each main controller type and their respective tuning processes. Next, the test platform is introduced along with the simulated patient scenarios used for performance testing. Concluding the section is a summary of the standard, modified, and aggregated performance metrics used for relative ranking of the PCLC designs.
2.1. Overview of Animal Study
The Institutional Animal Care and Use Committee (IACUC) at the United States Army Institute of Surgical Research approved all research conducted for this study. This proof-of-concept animal study exposed swine (sus scrofa domestica) to hemorrhagic shock and performed fluid resuscitation, as has been described previously [
6]. A total of
N = 10 female Yorkshire crossbred swine were included in this study, approximately four months in age and an average of 40 kg in weight. All animals were maintained at a surgical plane of anesthesia throughout the study, first by continuous inhalation of isoflurane (0–5%), followed by total intravenous anesthesia (TIVA) using ketamine (0–10 mg/kg/h) and midazolam (0–2 mg/kg/h); buprenorphine SR (0.24 mg/kg) was given once for analgesia.
For this study, the data used was captured during infusion of norepinephrine vasopressor (dosage shown on
Table 1) from a state of hemorrhagic shock. This occurred after an initial hemorrhage to 35 mmHg and resuscitation with whole blood using an adaptive resuscitation controller (ARC) to return animals to a target MAP, which was based on the starting blood pressure of the swine prior to hemorrhage [
7]. ARC maintained swine at target MAP for one hour, followed by a secondary hemorrhage event to 35 mmHg that was held for at least 10 min. Next, norepinephrine (NE) infusion alongside a constant 10 mL/min crystalloid infusion rate was started. Since a single concentration of NE solution (4.0 mcg/mL) was used, the delivered dosage was solely dependent on the infusion rate in mL/min and was how “dosage” was defined throughout controller development. Dosing was incrementally increased after MAP stabilized at each dose until reaching the target MAP. At this point, NE dosing was reduced by one step in the dosing table to observe the effect on MAP. After allowing MAP to stabilize, swine were humanely euthanized.
The data captured from this animal study were used to incorporate techniques and approaches for developing the Adaptive Neural Fuzzy Inference System (AN-FIS), step fuzzy inference system (Step-FIS), and patient-following controller (PFC) described in more detail below.
2.2. Overview of V-ARC Test Platform
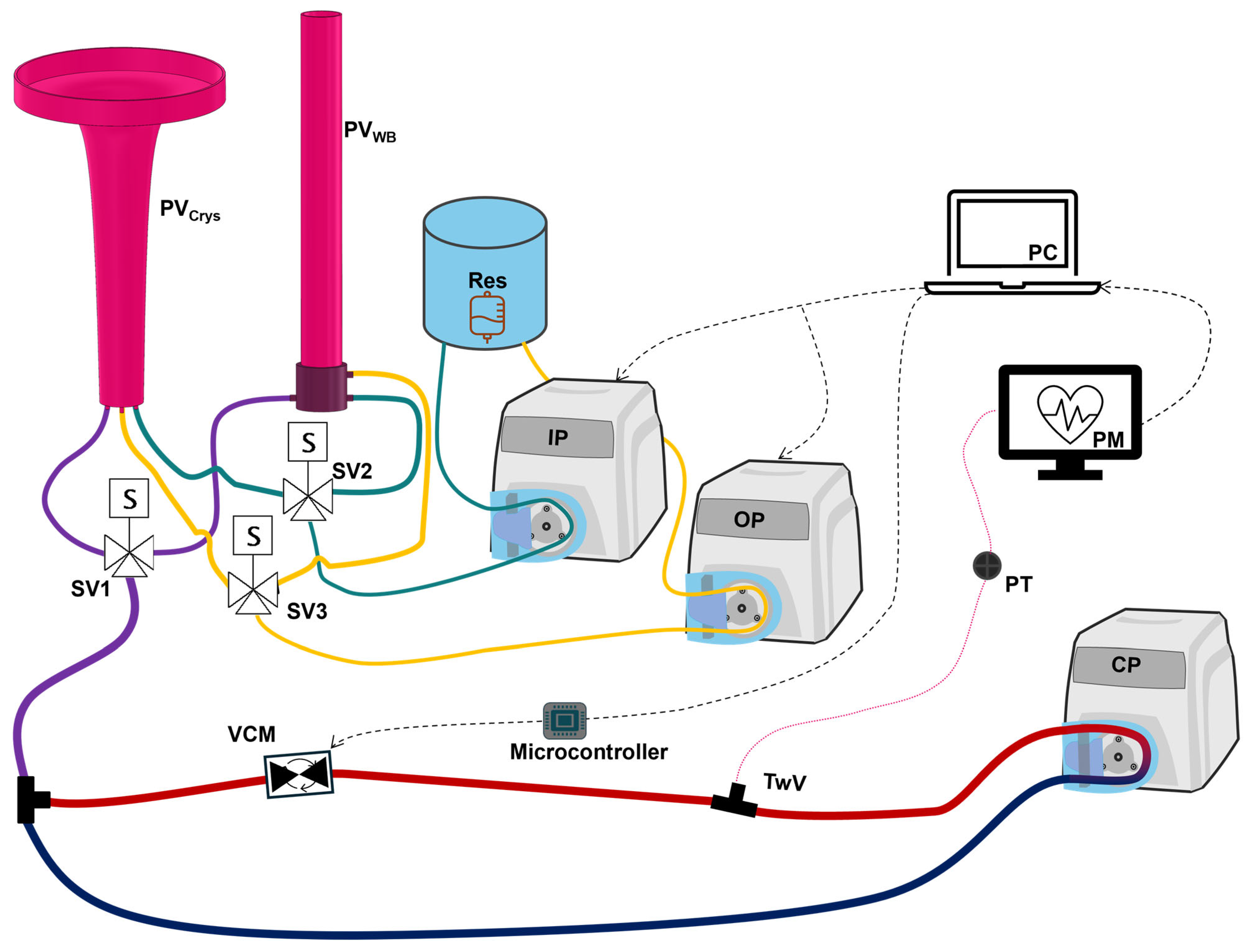
For testing, the hardware-in-loop automated testbed for resuscitation controllers (HATRC) with an integrated Vasopressor Control Module (VCM) was used (
Figure 1). This system and its development using large-animal study datasets have been described previously [
22,
23], as well as its successful use to tune hemorrhage resuscitation controllers [
6,
7]. Briefly, a circulating fluid loop with a peristaltic pump-driven (MasterFlex, Gelsenkirchen, Germany) pulsatile flow produces a pressure signal that is detected via arterial line by a patient monitor (Drager, Lübeck, Germany). This pulsatile pressure signal is used to calculate the MAP which can also be down-sampled, reflecting constraints such as relying on non-invasive blood pressure measurements [
24], and the system can operate at physiologically relevant pressures for hemorrhagic shock resuscitation. Additional pressure disturbances and noise can be simulated by the platform as well, though noise profiles specific to the desired setting need to be characterized. Fluid reservoirs, called PhysioVessels, provide hydrostatic pressure to the system that is responsive to changes in volume. The PhysioVessels were developed based on pressure response to fluid volume data from animals to simulate the effects of fluid resuscitation on the system at an MAP level. Two additional pumps are plumbed directly to the PhysioVessels, allowing addition or removal of fluid from the system without disrupting the dynamic pressure signal from the circulating portion of the loop. The integrated VCM provides a way for system pressure (i.e., MAP) to be affected independently of volume and was designed to mimic four characteristic features extracted from in vivo MAP signals during vasopressor dosing changes, including Lag Time, the time delay from dosage change to pressure response; overshoot, how much the pressure peaks before settling; Real Response, the steady-state pressure difference; and responsiveness, the time-rate of change in pressure during the initial rise. This module comprises a stepper motor-driven needle valve and a microcontroller. When a vasopressor dosage is administered, the microcontroller calculates the anticipated pressure change, determines the required amount of occlusion by the needle valve to induce said pressure change, and actuates the motor until the required position is reached. The functions used for these calculations and the characterization of the module are further detailed in previous work [
21].
2.3. Physiological Closed-Loop Controller Development
The five controller architectures evaluated in this study were selected to span the range of approaches used or proposed for automated drug delivery in the literature, from well-established methods to data-driven and hybrid designs. PID control was included as a standard reference benchmark, given its long history in control systems and more recent multi-drug hemodynamic management systems [
25]. While PID remains widely used due to its simplicity and well-understood tuning procedures, prior work in our group on the adaptive resuscitation controller (ARC) for whole-blood and crystalloid fluid resuscitation demonstrated that fuzzy logic-based designs could match or outperform PID in hemorrhage scenarios where nonlinear patient responses and variable drug sensitivity complicated linear control assumptions [
6,
7]. This motivated the inclusion of two fuzzy-based architectures: Step-FIS, a custom rule-based Sugeno controller paired with adaptive step logic, and AN-FIS, which uses the learning capabilities of neural networks to tune fuzzy rule parameters from animal study data [
22]. The choice to include both a manually designed (Step-FIS) and a data-trained (AN-FIS) fuzzy architecture allowed us to compare expert knowledge-driven design against data-driven design parameter optimization for the same general class of controller. ADRC was selected because it is model-agnostic and estimates disturbances in real time, a feature well-suited to the patient-to-patient variability that makes vasopressor titration difficult. More broadly, recent work on pressure regulation in ventilator systems has demonstrated that hybrid fuzzy–PID controllers can outperform conventional PID when dealing with uncertain or time-varying plant dynamics [
26], reinforcing the value of non-PID architectures for physiological control problems. Finally, the patient-following controller (PFC) was developed as a fully custom design inspired by direct observation of vasopressor response dynamics in the animal data, with the goal of capturing physiological behavior that pre-existing controller frameworks may not accommodate. Together, these five architectures form a deliberate gradient from purely model-free classical control (PID), through hybrid intelligent designs (Step-FIS, AN-FIS, ADRC), to a physiology-inspired custom controller (PFC), and provide a systematic basis for identifying the most suitable approach for automated vasopressor management in hemorrhagic shock.
Two unique PCLC tunings of each controller type were produced using conservative and aggressive tuning schemas, where conservative tuning favored longer rise times to avoid overshooting the target, and aggressive tuning prioritized reduced rise times at the cost of increased overshooting risk. The methods to achieve these performance biases were specific for each controller type and will be covered in their respective subsections. Controller development leveraged vasopressor pressure response dynamic datasets captured in large-animal studies to aid in tuning controller parameters [
17]. An initial test scenario for debugging purposes (different to the scenarios that are mentioned in
Section 2.4) was used for every controller using the HATRC platform to confirm real-time functionality of each controller.
Aggressive and conservative tuning schemas were prospectively defined based on clinically relevant constraints. Aggressive configurations targeted a rise time of 4–6 min to minimize hypotensive duration, while conservative configurations targeted 7–10 min to reduce overshoot and oscillations [
27]. Although these specific rise time targets are engineering design choices, their rationale was grounded in physiological considerations. Prior studies emphasize the dual need to restore arterial pressure promptly to avoid secondary organ injury, while simultaneously avoiding excessive vasoconstriction, masked hypovolemia, and ischemic complications associated with rapid or high-dose vasopressor administration. Clinical guidance commonly targets an MAP near 65 mmHg during damage control resuscitation, and highlights the risks of overly aggressive vasopressor use in hypovolemic states [
27].
2.3.1. Proportional–Integral–Derivative Controller
The Proportional–Integral–Derivative (PID)-based controller modulates infusion rates to maintain MAP near the specified target. The control law is derived from a standard PID structure, where the control signal is formed by the weighted sum of the current MAP error, its time integral, and its time derivative. Specifically, the error term is computed as the difference between a delayed estimate of MAP and the target MAP, with the delay modeled as a first-order low-pass filter that captures the physiological latency between drug delivery and hemodynamic response. This filter evolves according to the differential equation, Equation (1):
where
s, representing the characteristic time constant of the delay.
To improve clinical realism and numerical robustness, the integral term is subject to anti-windup constraints that attenuate accumulation near the setpoint. Specifically, when the absolute MAP error falls below 5 mmHg, the integral accumulator is decayed to reduce excessive gain buildup. The derivative term is computed using a backward difference approximation based on the most recent error value.
In addition, the controller incorporates a rate limiter that bounds the change in infusion rate per time step, ensuring smoother transitions and reducing the risk of abrupt hemodynamic fluctuations. The aggressive PID configuration permits a higher maximum rate of change, favoring faster correction of MAP deviations, and the conservative configuration yields a slower, more gradual response.
Parameter tuning was initially conducted using physiological simulation data from the Pulse Physiology Engine [
28] to establish gain stability and responsiveness. Final adjustments and performance verification were performed on the HATRC platform, where the controller was evaluated under a range of hemorrhagic shock conditions to ensure consistent target tracking and minimal overshoot.
2.3.2. Step Fuzzy Inference System
The proposed step fuzzy inference system (Step-FIS) employs a hybrid architecture that integrates a type-2 Sugeno fuzzy logic controller with unit step logic to precisely regulate vasopressor infusion rates. Initially, the measured blood pressure signal is processed through a first-order Butterworth filter to attenuate high-frequency noise and smooth out transient fluctuations. From the resulting filtered signal, the error relative to the target pressure is computed without taking the absolute value; this preserves the sign of the error to clearly indicate whether the current pressure is above or below the target pressure. Concurrently, the derivative of the filtered error is determined to capture the rate at which the error is changing, thereby providing dynamic information on the system’s fluctuating behavior.
These two key parameters—the filtered error and its derivative—form a state space that serves as the input to the fuzzy logic controller. Each input is represented using three membership functions that encode the qualitative behavior of the system. For both error and rate of change, the membership functions consist of:
- •
Negative—A linear Z-shaped function capturing negative deviations;
- •
Zero—A triangular function centered around the target condition;
- •
Positive—A linear S-shaped function representing positive deviations.
This structured membership function design, shown in
Figure 2, provides smooth transitions in vasopressor rates. Using a type-2 Sugeno framework, the controller effectively addresses uncertainties and variations found in live patient data. A predefined set of IF–THEN rules interprets the state information and generates an output in the range of −1 to 1. This output directs the unit step logic to increase, decrease, or maintain the vasopressor infusion rate, accordingly, ensuring appropriate adjustment in response to both the magnitude and trend of the pressure error.
The complete rule base governing the Step-FIS controller is summarized in
Table 2. Five IF–THEN rules map combinations of error and rate-of-change conditions to one of three output actions: increase, maintain, or decrease the vasopressor infusion rate. The rules were designed to enforce a conservative control philosophy: infusion is only increased when the measured pressure is below target and not already rising (Rules 1 and 2). When pressure is actively climbing—regardless of whether the current error is above or below target—the controller holds the current rate to avoid compounding the ongoing response (Rule 3). If the controller detects that the patient has overshot the target (positive error), the infusion rate is decreased with a reduced weight of 0.75 to prevent abrupt withdrawal of vasopressor support (Rule 4). When the error is near zero, the controller maintains the current rate (5). This asymmetric design reflects a clinical preference for gradual dose reduction over aggressive cutbacks, which aligns with guidance on avoiding rebound hypotension during vasopressor weaning.
The resulting control surface is shown in
Figure 3, which plots the defuzzified output (VasoRate) as a function of both the error and its rate of change. Across the input space, the controller transitions smoothly between increasing, maintaining, and decreasing actions, with the steepest gradients occurring near the boundary between negative and zero error. Both the plateau in the maintain region and the attenuated slope in the decrease region are direct consequences of the rule structure and the reduced weighting of Rule 4, respectively.
Moreover, the control strategy incorporates adaptive step size adjustment to fine-tune the rate adjustments. There are two baseline configurations, aggressive and conservative, with step sizes of 0.035 and 0.025 mL/min, respectively. These initial step sizes were derived from responses observed in the animal data, then used during preliminary controller development to fine-tune the system. To further enhance control performance, the step size was dynamically adjusted: if the filtered error exceeds 30%, the step size is doubled to rapidly counteract significant deviations, whereas if the error is below 3%, the step size is halved to minimize the risk of overshoot. This approach creates a stable response when large corrections are needed while ensuring stability and precision as the pressure converges toward the target. This controller was tested on the HATRC platform to assess performance functionality before fine-tuning and evaluating scenario data.
2.3.3. Adaptive Neural Fuzzy Inference System
Adaptive Neural Fuzzy Inference System (AN-FIS) combines both the rule-based architecture of fuzzy logic (IF–THEN rules) with the learning capabilities of neural networks to adjust the weights of the fuzzy rules based on training data [
29]. AN-FIS controller designs have been leveraged successfully for various applications, supporting their possible utility for this effort [
30,
31,
32].
For AN-FIS, the inputs were determined to be the error and rate of change in the error. Given that MAP may vary across subjects, the rate of change can serve as an indicator of how MAP is acting. The output for AN-FIS would be the appropriate vasopressor dosage the system would dictate based on the observed error from target pressure and the rate of change.
AN-FIS selected appropriate vasopressor dosages based on training and tuning performed using animal study data. The full dataset was first aggregated across studies, and outliers were identified and removed to reduce the influence of nonphysiological measurements and improve model robustness. This cleaned dataset was used as the training data for model development. During training, AN-FIS utilized the training data to generate fuzzy rules with generalized bell-shaped membership functions for the input variables and a linear output function [
29]. With this, the model was designed as a first-order Sugeno network architecture with 9 rules, and 3 membership functions per variable. Parameter optimization was performed using a hybrid learning algorithm combining least squares estimation for consequent parameters and backpropagation for premise parameters [
29]. Training was conducted using the MATLAB R2024b Fuzzy Logic Toolbox, with a maximum number of 100 epochs as a hyperparameter if convergence was not achieved by stabilization of the training error and improvement across successive epochs.
Overfitting was mitigated through multiple strategies, including outlier removal prior to training, careful tuning of the backpropagation learning rate, and study-wise validation using independent animal study datasets not included in the training process. Validation was performed separately using each individual animal study dataset, allowing model generalizability to be assessed across different experimental conditions. Randomization and blinding were not applied during data selection or analysis; instead, all available datasets were used in both training and validation according to the predefined study-wise validation framework. A 70-30 split was applied for training and testing data to evaluate performance on AN-FIS.
To address potential bias in the training data and to ensure physiologically safe controller behavior, additional safeguard mechanisms were incorporated into the dosing logic. Specifically, the AN-FIS output was modulated using a performance-error-based scaling term, such that dosing adjustments were proportionally weighted by the deviation of arterial pressure from the target. A bounded multiplier was applied to constrain the magnitude of dose changes, preventing excessively low or high vasopressor administration while preserving controller responsiveness. These constraints acted as mathematical regularization at the control-output level and were independent of the AN-FIS training process.
This iterative training and validation process was repeated until two AN-FIS systems were selected. The first system produced conservative vasopressor dosages, characterized by minimal dosage adjustments, while the second system produced more aggressive vasopressor dosages to rapidly achieve the target arterial pressure. To further tune and validate the performance, AN-FIS was evaluated on the HATRC platform. AN-FIS was later modified to observe more closely the changes in MAP, more specifically, the rate of change, over time when dosage is given more effectively. Validation of the controller was conducted by using different hemorrhage scenarios and evaluating the Effectiveness and timeliness of the controller at reaching the target pressure.
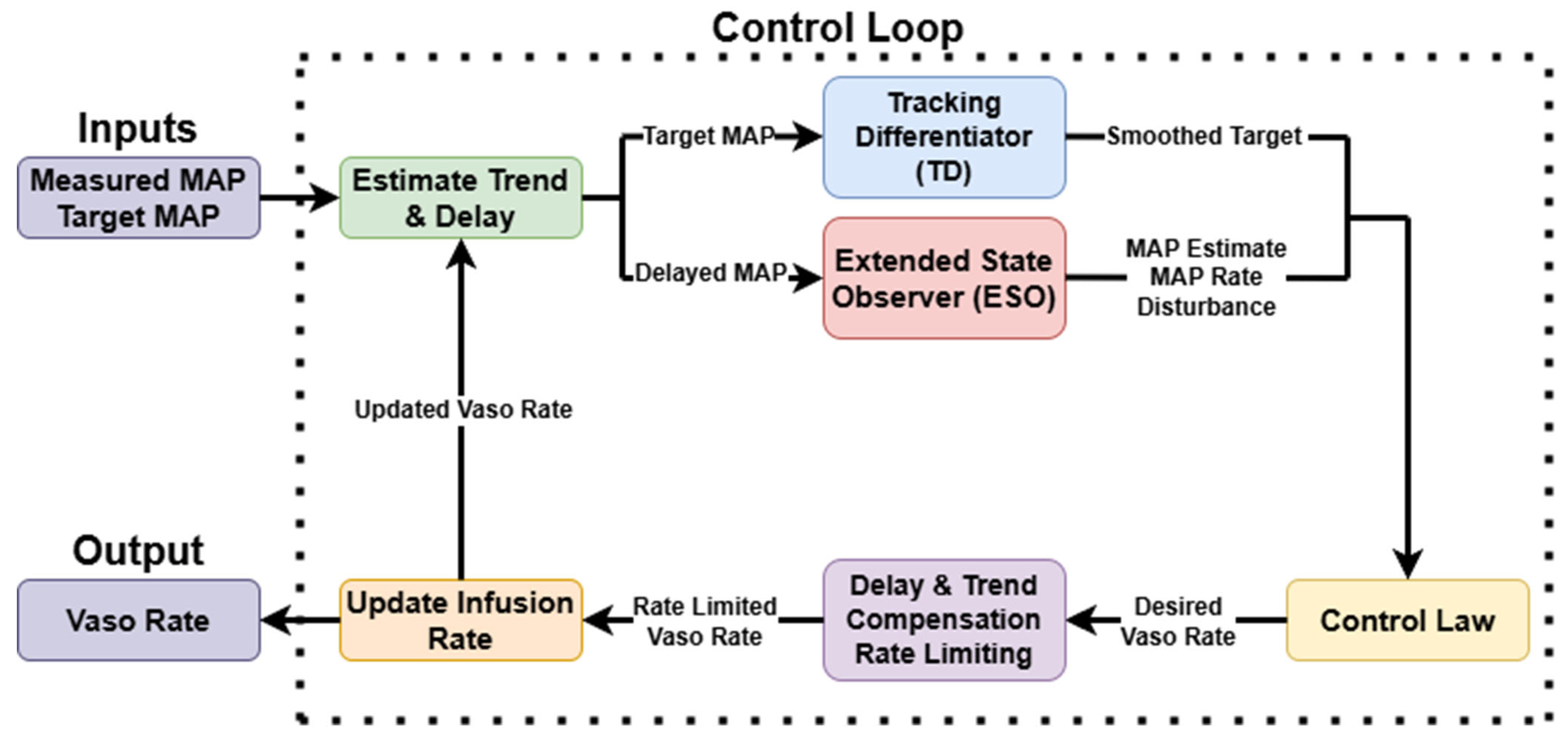
2.3.4. Active Disturbance Rejection Control
Another vasopressor controller was developed using Active Disturbance Rejection Control (ADRC) [
33], a model-agnostic strategy that enables real-time compensation for internal physiological variability and external disturbances. The ADRC architecture comprises three principal components: a tracking differentiator (TD), an extended state observer (ESO), and a state feedback control law (
Figure 4).
The TD processes the MAP setpoint into a smoothed reference trajectory and its derivative, facilitating smoother convergence and reducing sensitivity to abrupt setpoint changes. The ESO continuously estimates the true MAP, its rate of change, and a lumped disturbance term that aggregates unmodeled dynamics, patient-specific variability, and external perturbations. These states are updated based on a delayed MAP signal using high-gain observer dynamics, with tuning parameters selected to ensure fast convergence and disturbance rejection.
The control input is computed using a nonlinear feedback law that incorporates both state error and estimated disturbance according to the equation , where , , and are the ESO state parameters representing the current estimated MAP value, estimated MAP derivative, and estimated disturbance, respectively; and are the proportional and derivative gains; is the system gain; and is the smoothed reference target. The proportional gain is adapted online as a function of the absolute error magnitude to improve transient performance during large deviations from the target. A delay compensation mechanism modulates the allowable infusion rate adjustment based on the known physiological lag, while trend-aware logic further moderates the rate of change depending on the direction and speed of MAP fluctuations. For instance, when MAP is rapidly rising, the controller restricts further infusion increases to avoid overshooting. Similarly, in cases of steep MAP decline, vasopressor tapering is restrained to preserve perfusion support.
As with the PID controller, ADRC parameters were initially calibrated using simulated data in the Pulse Physiology Engine [
28] and refined through empirical testing on the HATRC platform to ensure robustness and fidelity across diverse shock trajectories.
2.3.5. Patient-Following Controller
The custom controller was developed based on characteristics observed in the MAP waveforms following changes in vasopressor dosage captured from animal data. It should be noted that many waveform features were present whether the dosage was increasing or decreasing, with the main difference being a sign change. However, in the present work, the focus was on the ramping (dosage increasing) phase of treatment, so the evaluation will reflect on the vasopressor infusion phase. Additional details are described in
Section 4 on future work that will address the weaning (dosage decreasing) phase.
Some key features that were common across all subjects irrespective of dose were (1) most of the MAP change in response to vasopressors occurred within a relatively narrow window, (2) the MAP signals in this region can be approximated as smooth and continuous, and (3) there was a maximum change in MAP effected by any given dose, after which the pressure trended towards steady-state unless and until the system was disturbed again. The real-time MAP responsiveness (e.g., in mmHg/min) was therefore approximated using a five-sample sliding window gradient. Following a dosage change, this value would increase until reaching a local maximum (when MAP was rising the fastest), then decrease until MAP stopped rising due to observation 3 above.
Using this understanding of the system, we built a patient-following controller (PFC) that evaluated the trajectory of the signal error using a conditional rule set to determine the appropriate time to increase the dose (
Figure 5). First, a rolling average window was used to smooth the MAP signal, and the smoothed MAP value was converted to a relative error (
) based on the target MAP. When MAP is at the target, this results in an
of 0, and overshoot is indicated by a negative
value. The most recent five samples were used to calculate the error gradient (
). The smallest
value was saved as the current local minimum (
) and was only replaced by a value that was
. After the true local minimum,
gradually approached 0 as the MAP response slowed, which was used as an indication that the maximum potential effect of the current dose had been reached. If the
value crossed a threshold, defined as a certain percentage of
, for at least 5 consecutive samples, and the MAP was not within 1% of the target, then the dosage was increased by a specified increment. Tuning was accomplished by adjusting the size of the moving average window used for smoothing the MAP (conservative = 10; aggressive = 5), the size of the incremental dosage change (conservative = 0.25 mL/min; aggressive = 0.5 mL/min), and the
threshold used for triggering a dosage change (conservative = 0.5; aggressive = 0.9).
2.5. Controller Comparison Data Analyses
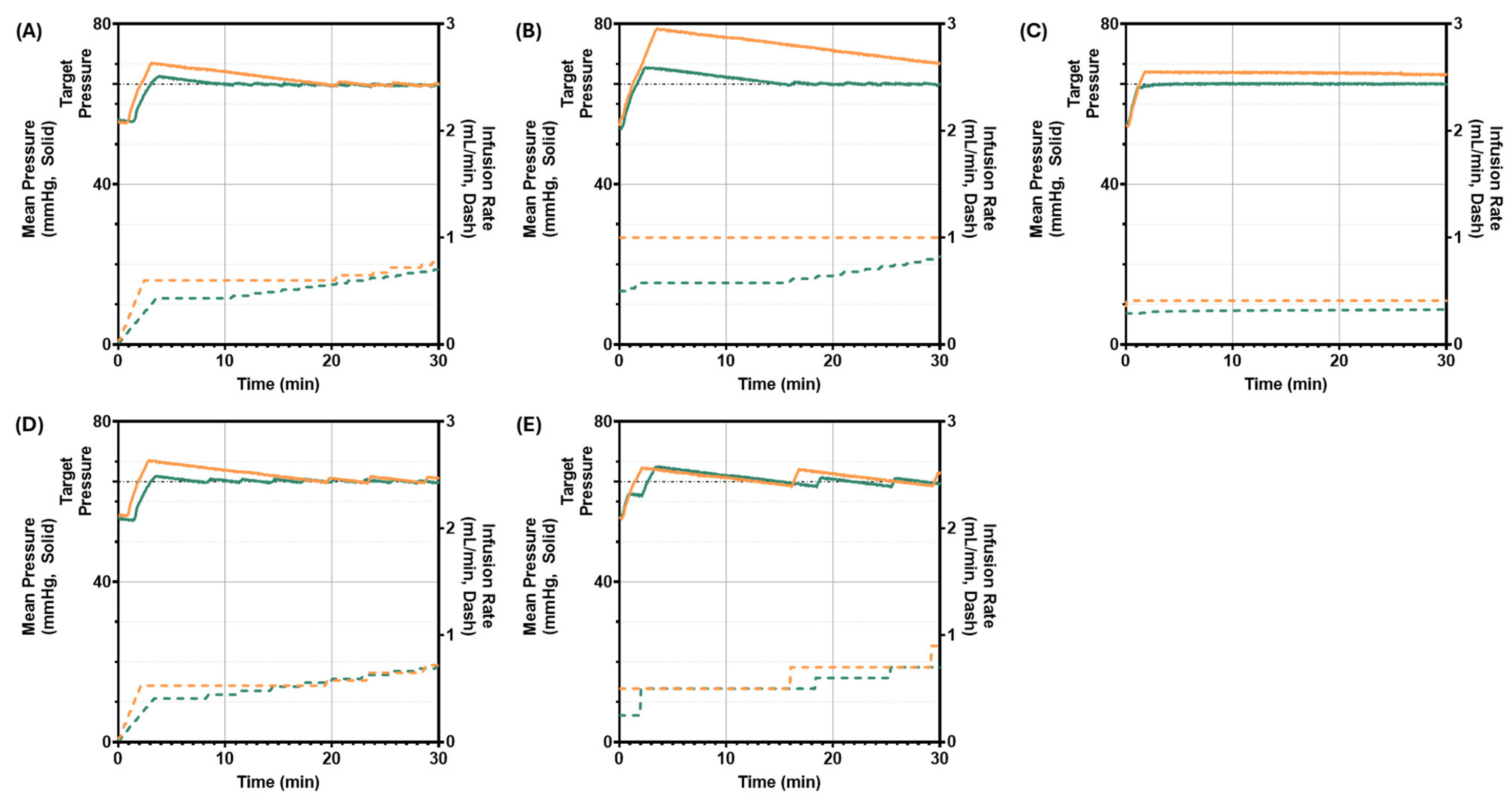
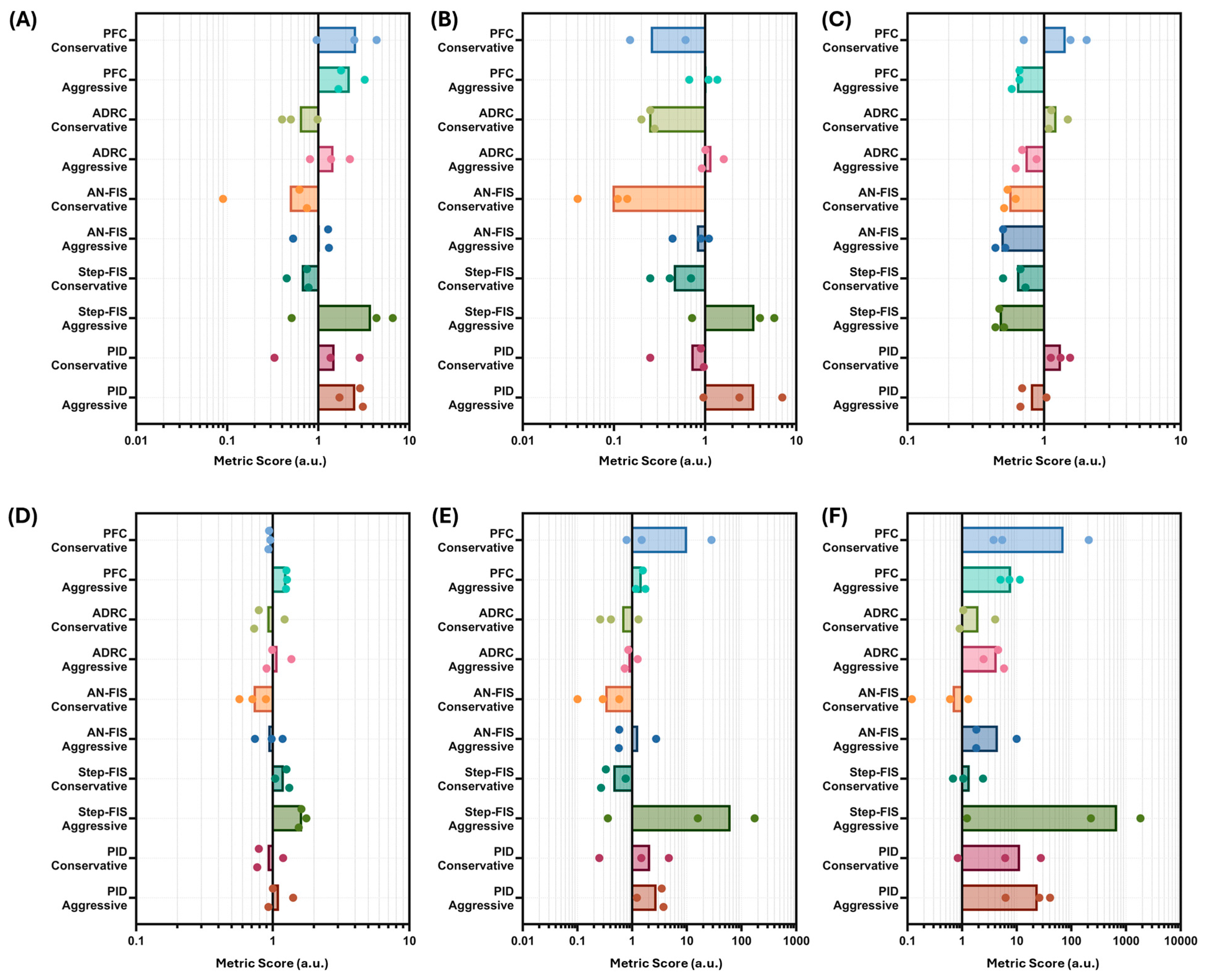
Data from each scenario run (35, 45, and 55 mmHg starting pressure) for each controller type and configuration were compiled across the entire 30 min run. To qualitatively compare controllers, MAP and vasopressor infusion rates were plotted against time for each PCLC. For a more quantitative evaluation, a series of controller performance metrics were calculated for each controller and scenario. These were used to down-select controller designs similar to methodologies used in other recent studies for closed-loop controllers and machine learning modeling methodologies [
22,
36,
37,
38]. These are summarized in
Table 3 and have been thoroughly described in the previous literature, except for Maximum Vasopressor Step Change, which was added to quantify the intensity of dosage change each controller used. Each metric was calculated separately across scenarios and controllers, but an aggregate score was calculated by merging metrics into equation terms (Equations (2)–(6)) based on five controller features: (i) stability, (ii) overshoot, (iii) undershoot, (iv) infusion, and (v) EEffectiveness metrics. The overall aggregate score equaled the sum of the stability, overshoot, undershoot, and infusion terms multiplied by the effectiveness term, where the lowest score indicated best performance as shown in Equation (7).
To have each term be dimensionless and weighted similarly to the overall controller performance score, all metrics were normalized to their median score across all PCLCs for each scenario. Median statistics were used instead of mean values to be less sensitive to large outliers due to poor-performing controller designs. Each term shown in Equation (7) was separately calculated for each scenario and averaged across all three scenarios to best quantify the overall controller designs. As the weights for each term were arbitrarily equal, a sensitivity analysis was performed for the aggregate score calculated with Equation (7) to quantify the effects of different parameter weights on overall controller rankings. Each term of the equation was given 2×, 1×, or 0.5× weights to assess how the scores were impacted. Replicates were taken as the performance scores for each scenario (N = 3), as each scenario represents a different starting pressure, similar to the variability anticipated between live subjects.