The Evolution of Lupus Nephritis Therapy from the 1960s to the Present
Abstract
1. Introduction
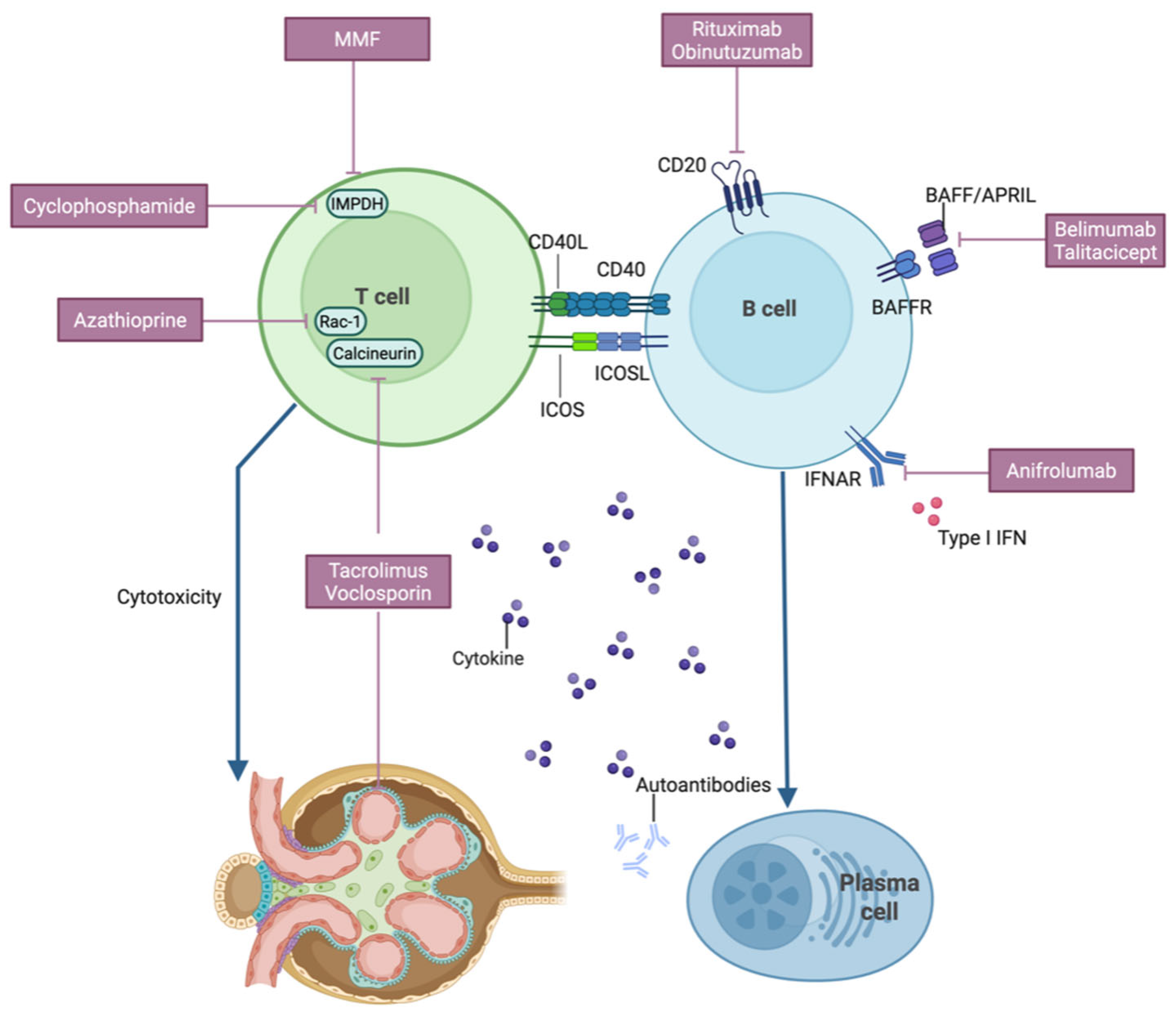
2. Pathogenesis of Lupus Nephritis
2.1. Interferon System
2.2. B-Cells
2.3. T-Cells
2.4. Histopathological Assessment and Its Role in Treatment Decisions
3. A History of Therapeutic Evolution
3.1. The Use of Glucocorticoids
3.2. The Advent of Cytotoxic Agents and Mycophenolate Mofetil
3.3. The Evolution: A New Era of Targeted Treatment
3.3.1. Belimumab
3.3.2. Tacrolimus
3.3.3. Voclosporin
3.3.4. Rituximab
3.3.5. Obinutuzumab
3.3.6. Anifrolumab
3.3.7. Talitacicept
4. A New Standard-of-Care: Combination Therapy
Current KDIGO Guideline Recommendations
5. Special Considerations
5.1. Refractory Disease
5.2. Children and Adolescents
5.3. Lupus Nephritis and Pregnancy
5.4. Racial and Ethnic Difference
6. The Future of LN Management
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, C.H.; Sammaritano, L.R. Systemic lupus erythematosus: A review. JAMA 2024, 331, 1480–1491. [Google Scholar] [CrossRef] [PubMed]
- Yap, D.Y.H.; Tang, C.S.O.; Ma, M.K.M.; Lam, M.F.; Chan, T.M. Survival analysis and causes of mortality in patients with lupus nephritis. Nephrol. Dial. Transplant. 2012, 27, 3248–3254. [Google Scholar] [CrossRef] [PubMed]
- Obrișcă, B.; Sorohan, B.; Tuță, L.; Ismail, G. Advances in lupus nephritis pathogenesis: From bench to bedside. Int. J. Mol. Sci. 2021, 22, 3766. [Google Scholar] [CrossRef] [PubMed]
- Fairhurst, A.-M.; Xie, C.; Fu, Y.; Wang, A.; Boudreaux, C.; Zhou, X.J.; Cibotti, R.; Coyle, A.; Connolly, J.E.; Wakeland, E.K.; et al. Type I interferons produced by resident renal cells may promote end-organ disease in autoantibody-mediated glomerulonephritis. J. Immunol. 2009, 183, 6831–6838. [Google Scholar] [CrossRef]
- Ding, X.; Ren, Y.; He, X. IFN-I mediates lupus nephritis from the beginning to renal fibrosis. Front. Immunol. 2021, 12, 676082. [Google Scholar] [CrossRef]
- Yung, S.; Yap, D.Y.; Chan, T.M. A review of advances in the understanding of lupus nephritis pathogenesis as a basis for emerging therapies. F1000Research 2020, 9, 905. [Google Scholar] [CrossRef]
- Yap, D.Y.H.; Chan, T.M. B cell abnormalities in systemic lupus erythematosus and lupus nephritis—Role in pathogenesis and effect of immunosuppressive treatments. Int. J. Mol. Sci. 2019, 20, 6231. [Google Scholar] [CrossRef]
- Chan, O.T.M.; Madaio, M.R.; Shlomchik, M.J.; Chan, O.I.M.; Madaio, M.P.; Shlomchik, M.J. The central and multiple roles of B cells in lupus pathogenesis. Immunol. Rev. 1999, 169, 107–121. [Google Scholar] [CrossRef]
- Batten, M.; Groom, J.; Cachero, T.G.; Qian, F.; Schneider, P.; Tschopp, J.; Browning, J.L.; Mackay, F.J. BAFF mediates survival of peripheral immature B lymphocytes. J. Exp. Med. 2000, 192, 1453–1466. [Google Scholar] [CrossRef]
- Mackay, F.; Schneider, P.; Rennert, P.; Browning, J. BAFF and APRIL: A tutorial on B cell survival. Annu. Rev. Immunol. 2003, 21, 231–264. [Google Scholar] [CrossRef]
- Myette, J.R.; Kano, T.; Suzuki, H.; Sloan, S.E.; Szretter, K.J.; Ramakrishnan, B.; Adari, H.; Deotale, K.D.; Engler, F.; Shriver, Z.; et al. A proliferation inducing ligand (APRIL) targeted antibody is a safe and effective treatment of murine IgA nephropathy. Kidney Int. 2019, 96, 104–116. [Google Scholar] [CrossRef]
- Neusser, M.A.; Lindenmeyer, M.T.; Edenhofer, I.; Gaiser, S.; Kretzler, M.; Regele, H.; Segerer, S.; Cohen, C.D. Intrarenal production of B-cell survival factors in human lupus nephritis. Mod. Pathol. 2011, 24, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Botta Gordon-Smith, S.; Urber, S.; Engleman, E.G. Long-lived plasma cells and tissue-resident memory B cells in systemic autoimmunity. Sci. Transl. Med. 2019, 11, eaav1648. [Google Scholar] [CrossRef]
- Schett, G.; Mackensen, A.; Mougiakakos, D. CAR T-cell therapy in autoimmune diseases. Lancet Rheumatol. 2023, 5, e684–e694. [Google Scholar] [CrossRef] [PubMed]
- Koga, T.; Ichinose, K.; Tsokos, G.G. T cells and IL-17 in lupus nephritis. Clin. Immunol. 2017, 185, 95–99. [Google Scholar] [CrossRef]
- Wiechmann, A.; Wilde, B.; Tyczynski, B.; Amann, K.; Abdulahad, W.H.; Kribben, A.; Lang, K.S.; Witzke, O.; Dolff, S. CD107a+ (LAMP-1) cytotoxic CD8+ T-cells in lupus nephritis patients. Front. Med. 2021, 8, 556776. [Google Scholar] [CrossRef]
- Li, Y.; Tang, D.; Yin, L.; Dai, Y. New insights for regulatory T cell in lupus nephritis. Autoimmun. Rev. 2022, 21, 103134. [Google Scholar] [CrossRef]
- Macian, F. NFAT proteins: Key regulators of T-cell development and function. Nat. Rev. Immunol. 2005, 5, 472–484. [Google Scholar] [CrossRef]
- van Gelder, T.; Lerma, E.; Engelke, K.; Huizinga, R.B. Voclosporin: A novel calcineurin inhibitor for the treatment of lupus nephritis. Expert Rev. Clin. Pharmacol. 2022, 15, 515–529. [Google Scholar] [CrossRef]
- Rovin, B.H.; Ayoub, I.M.; Chan, T.M.; Liu, Z.-H.; Mejía-Vilet, J.M.; Floege, J. KDIGO 2024 clinical practice guideline for the management of lupus nephritis. Kidney Int. 2024, 105, S1–S69. [Google Scholar] [CrossRef]
- Umeda, R.; Ogata, S.; Hara, S.; Takahashi, K.; Inaguma, D.; Hasegawa, M.; Yasuoka, H.; Yuzawa, Y.; Hayashi, H.; Tsuboi, N. Comparison of the 2018 and 2003 International Society of Nephrology/Renal Pathology Society classification in terms of renal prognosis in patients of lupus nephritis: A retrospective cohort study. Arthritis Res. Ther. 2020, 22, 261. [Google Scholar] [CrossRef] [PubMed]
- Moroni, G.; Depetri, F.; Ponticelli, C. Lupus nephritis: When and how often to biopsy and what does it mean? J. Autoimmun. 2016, 74, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Renaudineau, Y.; Brooks, W.; Belliere, J. Lupus Nephritis Risk Factors and Biomarkers: An Update. Int. J. Mol. Sci. 2023, 24, 14526. [Google Scholar] [CrossRef] [PubMed]
- Zickert, A.; Sundelin, B.; Svenungsson, E.; Gunnarsson, I. Role of early repeated renal biopsies in lupus nephritis. Lupus Sci. Med. 2014, 1, e000018. [Google Scholar] [CrossRef]
- Parodis, I.; Tamirou, F.; Houssiau, F.A. Prediction of prognosis and renal outcome in lupus nephritis. Lupus Sci. Med. 2022, 9, e000689. [Google Scholar] [CrossRef]
- Malvar, A.; Pirruccio, P.; Alberton, V.; Lococo, B.; Recalde, C.; Fazini, B.; Nagaraja, H.; Indrakanti, D.; Rovin, B.H. Histologic versus clinical remission in proliferative lupus nephritis. Nephrol. Dial. Transplant. 2017, 32, 1338–1344. [Google Scholar] [CrossRef]
- Moroni, G.; Porata, G.; Raffiotta, F.; Frontini, G.; Calatroni, M.; Reggiani, F.; Banfi, G.; Ponticelli, C. Predictors of increase in chronicity index and of kidney function impairment at repeat biopsy in lupus nephritis. Lupus Sci. Med. 2022, 9, e000721. [Google Scholar] [CrossRef]
- Parodis, I.; Tamirou, F.; Houssiau, F.A. Treat-to-Target in Lupus Nephritis. What is the Role of the Repeat Kidney Biopsy? Arch. Immunol. Ther. Exp. 2022, 70, 15. [Google Scholar] [CrossRef]
- Querin, V.; Palazzo, L.; Fogo, A.B.; Haas, M.; Jayne, D.R.W.; Lightstone, L.; Rovin, B.H.; Seshan, S.V.; Vaglio, A.; Parodis, I.; et al. Biomarkers of Lupus Nephritis Histopathology: Where Do We Stand? Arthritis Rheumatol. 2026, 78, 548–557. [Google Scholar] [CrossRef]
- Dubois, E.L. Prednisone and prednisolone in the treatment of systemic lupus erythematosus. JAMA 1956, 161, 427. [Google Scholar] [CrossRef]
- Kasturi, S.; Sammaritano, L.R. Corticosteroids in lupus. Rheum. Dis. Clin. N. Am. 2016, 42, 47–62. [Google Scholar] [CrossRef] [PubMed]
- Arévalo, A.M.T.; Quaye, A.; Rojas-Rodríguez, L.C.; Poole, B.D.; Baracaldo-Santamaría, D.; Tellez Freitas, C.M. Synthetic pharmacotherapy for systemic lupus erythematosus: Potential mechanisms of action, efficacy, and safety. Medicina 2022, 59, 56. [Google Scholar] [CrossRef] [PubMed]
- Donadio, J.V.; Holley, K.E.; Ferguson, R.H.; Ilstrup, D.M. Treatment of diffuse proliferative lupus nephritis with prednisone and combined prednisone and cyclophosphamide. N. Engl. J. Med. 1978, 299, 1151–1155. [Google Scholar] [CrossRef] [PubMed]
- Austin, H.A.; Klippel, J.H.; Balow, J.E.; Le Riche, N.G.; Steinberg, A.D.; Plotz, P.H.; Decker, J.L. Therapy of lupus nephritis. N. Engl. J. Med. 1986, 314, 614–619. [Google Scholar] [CrossRef]
- Mok, C.C. Con: Cyclophosphamide for the treatment of lupus nephritis. Nephrol. Dial. Transplant. 2016, 31, 1053–1057. [Google Scholar] [CrossRef]
- Houssiau, F.A.; Vasconcelos, C.; D’Cruz, D.; Sebastiani, G.D.; Garrido, E.d.R.; Danieli, M.G.; Abramovicz, D.; Blockmans, D.; Mathieu, A.; Direskeneli, H.; et al. Immunosuppressive therapy in lupus nephritis: The Euro-Lupus Nephritis Trial, a randomized trial of low-dose versus high-dose intravenous cyclophosphamide. Arthritis Rheum. 2002, 46, 2121–2131. [Google Scholar] [CrossRef]
- Abu-Shakra, M.; Shoenfeld, Y. Azathioprine therapy for patients with systemic lupus erythematosus. Lupus 2001, 10, 152–153. [Google Scholar] [CrossRef]
- Nossent, H.C.; Koldingsnes, W. Long-term efficacy of azathioprine treatment for proliferative lupus nephritis. Rheumatology 2000, 39, 969–974. [Google Scholar] [CrossRef][Green Version]
- Houssiau, F.A. Management of lupus nephritis: An update. J. Am. Soc. Nephrol. 2004, 15, 2694–2704. [Google Scholar] [CrossRef]
- Martin-Suarez, I.; D’Cruz, D.; Mansoor, M.; Fernandes, A.P.; Khamashta, M.A.; Hughes, G.R.V. Immunosuppressive treatment in severe connective tissue diseases: Effects of low dose intravenous cyclophosphamide. Ann. Rheum. Dis. 1997, 56, 481–487. [Google Scholar] [CrossRef]
- Moore, R.A.; Derry, S. Systematic review and meta-analysis of randomised trials and cohort studies of mycophenolate mofetil in lupus nephritis. Arthritis Res. Ther. 2006, 8, R182. [Google Scholar] [CrossRef]
- Touma, Z.; Gladman, D.D.; Urowitz, M.B.; Beyene, J.; Uleryk, E.M.; Shah, P.S. Mycophenolate mofetil for induction treatment of lupus nephritis: A systematic review and metaanalysis. J. Rheumatol. 2010, 38, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.M.; Li, F.K.; Tang, C.S.; Wong, R.W.; Fang, G.X.; Ji, Y.L.; Lau, C.S.; Wong, A.K.; Tong, M.K.; Chan, K.W.; et al. Efficacy of mycophenolate mofetil in patients with diffuse proliferative lupus nephritis. N. Engl. J. Med. 2000, 343, 1156–1162. [Google Scholar] [CrossRef] [PubMed]
- Appel, G.B.; Contreras, G.; Dooley, M.A.; Ginzler, E.M.; Isenberg, D.; Jayne, D.; Li, L.-S.; Mysler, E.; Solomons, N.; Wofsy, D.; et al. Mycophenolate mofetil versus cyclophosphamide for induction treatment of lupus nephritis. J. Am. Soc. Nephrol. 2009, 20, 1103–1112. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Chen, N.; Lin, Y.; Ren, H.; Zhang, W.; Wang, W.; Pan, X.; Yu, H. Mycophenolate mofetil in induction and maintenance therapy of severe lupus nephritis: A meta-analysis of randomized controlled trials. Nephrol. Dial. Transplant. 2007, 22, 1933–1942. [Google Scholar] [CrossRef]
- Ma, Y.; Li, X.; Yu, X.; Lan, P.; Liang, Y.; Lu, W.; Sun, J. Efficacy and safety of belimumab in refractory and newly diagnosed active lupus nephritis patients: A real-world observational study. Clin. Kidney J. 2025, 18, sfaf103. [Google Scholar] [CrossRef]
- Furie, R.; Rovin, B.H.; Houssiau, F.; Malvar, A.; Teng, Y.O.; Contreras, G.; Amoura, Z.; Yu, X.; Mok, C.-C.; Santiago, M.B.; et al. Two-year, randomized, controlled trial of belimumab in lupus nephritis. N. Engl. J. Med. 2020, 383, 1117–1128. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, H.; Liu, Z.; Xing, C.; Fu, P.; Ni, Z.; Chen, J.; Lin, H.; Liu, F.; He, Y.; et al. Multitarget therapy for induction treatment of lupus nephritis: A randomized trial. Ann. Intern. Med. 2015, 162, 18–26. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Z.; Zhou, M.; Liu, Z.; Chen, J.; Xing, C.; Lin, H.; Ni, Z.; Fu, P.; Liu, F.; et al. Multitarget therapy for maintenance treatment of lupus nephritis. J. Am. Soc. Nephrol. 2017, 28, 3671–3678. [Google Scholar] [CrossRef]
- Tian, G.-Q.; Li, Z.-Q. Efficacy and safety of biologics, multitarget therapy, and standard therapy for lupus nephritis: A systematic review and network meta-analysis. Ren. Fail. 2024, 46, 2395451. [Google Scholar] [CrossRef]
- Kale, A.; Shelke, V.; Lei, Y.; Gaikwad, A.B.; Anders, H.-J. Voclosporin: Unique chemistry, pharmacology and toxicity profile, and possible options for implementation into the management of lupus nephritis. Cells 2023, 12, 2440. [Google Scholar] [CrossRef]
- Rovin, B.H.; Teng, Y.K.O.; Ginzler, E.M.; Arriens, C.; Caster, D.J.; Romero-Diaz, J.; Gibson, K.; Kaplan, J.; Lisk, L.; Navarra, S.; et al. Efficacy and safety of voclosporin versus placebo for lupus nephritis (AURORA 1): A double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 2021, 397, 2070–2080. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.; Ginzler, E.M.; Gibson, K.; Satirapoj, B.; Santillán, A.E.Z.; Levchenko, O.; Navarra, S.; Atsumi, T.; Yasuda, S.; Chavez-Perez, N.N.; et al. Safety and efficacy of long-term voclosporin treatment for lupus nephritis in the phase 3 AURORA 2 clinical trial. Arthritis Rheumatol. 2023, 76, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Menn-Josephy, H.; Hodge, L.S.; Birardi, V.; Leher, H. Efficacy of voclosporin in proliferative lupus nephritis with high levels of proteinuria. Clin. J. Am. Soc. Nephrol. 2023, 19, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.C.W.; Cambridge, G. B-cell targeting in rheumatoid arthritis and other autoimmune diseases. Nat. Rev. Immunol. 2006, 6, 394–403. [Google Scholar] [CrossRef]
- Rovin, B.H.; Furie, R.; Latinis, K.; Looney, R.J.; Fervenza, F.C.; Sanchez-Guerrero, J.; Maciuca, R.; Zhang, D.; Garg, J.P.; Brunetta, P.; et al. Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: The lupus nephritis assessment with rituximab study. Arthritis Rheum. 2012, 64, 1215–1226. [Google Scholar] [CrossRef]
- Zhong, Z.; Li, H.; Zhong, H.; Zhou, T. Clinical efficacy and safety of rituximab in lupus nephritis. Drug Des. Devel. Ther. 2019, 13, 845–856. [Google Scholar] [CrossRef]
- Fanouriakis, A.; Kostopoulou, M.; Andersen, J.; Aringer, M.; Arnaud, L.; Bae, S.-C.; Boletis, J.; Bruce, I.N.; Cervera, R.; Doria, A.; et al. EULAR recommendations for the management of systemic lupus erythematosus: 2023 update. Ann. Rheum. Dis. 2024, 83, 15–29. [Google Scholar] [CrossRef]
- Furie, R.A.; Rovin, B.H.; Garg, J.P.; Santiago, M.B.; Aroca-Martínez, G.; Zuta Santillán, A.E.; Alvarez, D.; Navarro Sandoval, C.; Lila, A.M.; Tumlin, J.A.; et al. Efficacy and safety of obinutuzumab in active lupus nephritis. N. Engl. J. Med. 2025, 392, 1471–1483. [Google Scholar] [CrossRef]
- Liu, X.; Chen, X.; Yang, C.; Li, R.; Chen, X.; Li, Q. Biologicals for the treatment of lupus nephritis: A Bayesian network meta-regression analysis. Front. Immunol. 2024, 15, 1445814. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, S.-Q.; Li, M.; Wang, Y.-G. Effectiveness and safety of B cell-targeting biologics in the treatment of lupus nephritis: A systematic review and network metaanalysis. Ren. Fail. 2024, 46, 2416605. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Song, G.G. Comparative efficacy and safety of biological agents in the treatment of lupus nephritis: A network meta-analysis. Pharmacology 2022, 108, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Lai, B.; Luo, S.-F.; Lai, J.-H. Therapeutically targeting proinflammatory type I interferons in systemic lupus erythematosus: Efficacy and insufficiency with a specific focus on lupus nephritis. Front. Immunol. 2024, 15, 1489205. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Tummala, R. Anifrolumab, a monoclonal antibody to the type I interferon receptor subunit 1, for the treatment of systemic lupus erythematosus: An overview from clinical trials. Mod. Rheumatol. 2020, 31, 1–12. [Google Scholar] [CrossRef]
- Jayne, D.; Rovin, B.; Mysler, E.F.; Furie, R.A.; Houssiau, F.A.; Trasieva, T.; Knagenhjelm, J.; Schwetje, E.; Chia, Y.L.; Tummala, R.; et al. Phase II randomised trial of type I interferon inhibitor anifrolumab in patients with active lupus nephritis. Ann. Rheum. Dis. 2022, 81, 496–506. [Google Scholar] [CrossRef]
- Askanase, A.D.; Dall’Era, M.; Almaani, S. Insights into future management of lupus nephritis. Front. Lupus 2024, 2, 1334932. [Google Scholar] [CrossRef]
- Jayne, D.; Rovin, B.; Mysler, E.; Furie, R.; Houssiau, F.; Trasieva, T.; Knagenhjelm, J.; Schwetje, E.; Tang, W.; Tummala, R.; et al. Anifrolumab in lupus nephritis: Results from second-year extension of a randomised phase II trial. Lupus Sci. Med. 2023, 10, e000910. [Google Scholar] [CrossRef]
- Fan, Y.; Gao, D.; Zhang, Z. Telitacicept, a novel humanized, recombinant TACI-Fc fusion protein, for the treatment of systemic lupus erythematosus. Drugs Today 2022, 58, 23–32. [Google Scholar] [CrossRef]
- Wang, L.; Li, J.; Xu, D.; Fang, J.; Van Vollenhoven, R.; Zhang, F. Efficacy and safety of telitacicept, a novel BLyS/APRIL dual inhibitor, in patients with systemic lupus erythematosus: A phase 3, randomized, placebo-controlled 52-week study. Ann. Rheum. Dis. 2023, 82, 90–91. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, G.; Li, X.; Xie, C. Efficacy of telitacicept in the treatment of lupus nephritis: A 52-week, multicenter observational study. Ann. Rheum. Dis. 2024, 83, 994. [Google Scholar] [CrossRef]
- Podoll, A.; Furie, R.; Kim, F.; Chou, J.; Sengupta, R.; Bayer, R.; Gutman, J. P95 first two US patients with lupus nephritis (LN) treated with anti-CD19 chimeric antigen receptor (CAR) t-cell therapy: Preliminary results from the KYSA-1 phase 1, multicenter study of KYV-101. Lupus 2024, 33, A109–A110. [Google Scholar] [CrossRef]
- Mackensen, A.; Müller, F.; Mougiakakos, D.; Böltz, S.; Wilhelm, A.; Aigner, M.; Völkl, S.; Simon, D.; Kleyer, A.; Munoz, L.; et al. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nat. Med. 2022, 28, 2124–2132. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Hu, Y.; Chang, A.H.; Huang, H. CD19/BCMA CAR-T cell therapy for refractory systemic lupus erythematosus-safety and preliminary efficacy data from a phase I clinical study. Blood 2023, 142, 4835. [Google Scholar] [CrossRef]
- Figueroa-Parra, G.; Bautista-Vargas, M.; Navarro-Mendoza, E.; Duarte-García, A. Optimal glucocorticoid therapy in lupus nephritis. Nephrol. Dial. Transplant. 2024, 40, 1284–1293. [Google Scholar] [CrossRef]
- Isenberg, D.; Appel, G.B.; Contreras, G.; Dooley, M.A.; Ginzler, E.M.; Jayne, D.; Sánchez-Guerrero, J.; Wofsy, D.; Yu, X.; Solomons, N. Influence of race/ethnicity on response to lupus nephritis treatment: The ALMS study. Rheumatology 2009, 49, 128–140. [Google Scholar] [CrossRef]
- Sammaritano, L.R.; Askanase, A.; Bermas, B.L.; Dall’Era, M.; Duarte-García, A.; Hiraki, L.T.; Rovin, B.H.; Son, M.B.F.; Alvarado, A.; Aranow, C.; et al. 2024 American College of Rheumatology (ACR) guideline for the screening, treatment, and management of lupus nephritis. Arthritis Rheumatol. 2025, 77, 1115. [Google Scholar] [CrossRef]
- Ginzler, E.M.; Dooley, M.A.; Aranow, C.; Kim, M.Y.; Buyon, J.; Merrill, J.T.; Petri, M.; Gilkeson, G.S.; Wallace, D.J.; Weisman, M.H.; et al. Mycophenolate mofetil or intravenous cyclophosphamide for lupus nephritis. N. Engl. J. Med. 2005, 353, 2219–2228. [Google Scholar] [CrossRef]
- Chen, W.; Liu, Q.; Chen, W.; Tang, X.; Fu, P.; Liu, F.; Liao, Y.; Yang, Z.; Zhang, J.; Chen, J.; et al. Outcomes of maintenance therapy with tacrolimus versus azathioprine for active lupus nephritis: A multicenter randomized clinical trial. Lupus 2012, 21, 944–952. [Google Scholar] [CrossRef]
- Gunnarsson, I.; Sundelin, B.; Jónsdóttir, T.; Jacobson, S.H.; Henriksson, E.W.; van Vollenhoven, R.F. Histopathologic and clinical outcome of rituximab treatment in patients with cyclophosphamide-resistant proliferative lupus nephritis. Arthritis Rheum. 2007, 56, 1263–1272. [Google Scholar] [CrossRef]
- Yap, D.Y.; Tang, C.; Ma, M.K.; Mok, M.M.; Chan, G.C.; Kwan, L.P.; Chan, T.M. Longterm data on disease flares in patients with proliferative lupus nephritis in recent years. J. Rheumatol. 2017, 44, 1375–1383. [Google Scholar] [CrossRef]
- Joseph, V.M.K.; Reddy, V.; Fisher, C.; Pepper, R.; Ehrenstein, M.; Ciurtin, C.; Leandro, M. Evaluating outcomes for patients with systemic lupus erythematosus treated with obinutuzumab, a humanised anti-CD20 monoclonal antibody, following secondary non-depletion of B cells and non-response with rituximab. Rheumatology 2025, 64, keaf142.189. [Google Scholar] [CrossRef]
- Sinha, R.; Raut, S. Pediatric lupus nephritis: Management update. World J. Nephrol. 2014, 3, 16–23. [Google Scholar] [CrossRef]
- Trindade, V.C.; Carneiro-Sampaio, M.; Bonfa, E.; Silva, C.A. An update on the management of childhood-onset systemic lupus erythematosus. Pediatr. Drugs 2021, 23, 331–347. [Google Scholar] [CrossRef]
- Gholizadeh Ghozloujeh, Z.; Singh, T.; Jhaveri, K.D.; Shah, S.; Lerma, E.; Abdipour, A.; Norouzi, S. Lupus nephritis: Management challenges during pregnancy. Front. Nephrol. 2024, 4, 1390783. [Google Scholar] [CrossRef]
- Juliao, P.; Wurst, K.; Pimenta, J.M.; Gemzoe, K.; Landy, H.; Moody, M.A.; Tilson, H.; Covington, D.; Moore, T.; Marino, R.; et al. Belimumab use during pregnancy: Interim results of the belimumab pregnancy registry. Birth Defects Res. 2022, 115, 188–204. [Google Scholar] [CrossRef]
- Korbet, S.M.; Schwartz, M.M.; Evans, J.; Lewis, E.J. Severe lupus nephritis: Racial differences in presentation and outcome. J. Am. Soc. Nephrol. 2007, 18, 244–254. [Google Scholar] [CrossRef]
- Hanly, J.G.; O’Keeffe, A.G.; Su, L.; Urowitz, M.B.; Romero-Diaz, J.; Gordon, C.; Bae, S.-C.; Bernatsky, S.; Clarke, A.E.; Wallace, D.J.; et al. The frequency and outcome of lupus nephritis: Results from an international inception cohort study. Rheumatology 2015, 55, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Burger, C.; Benichou, N.; Narjoz, C.; Costedoat-Chalumeau, N.; Guern, V.L.; Hummel, A.; Jourde-Chiche, N.; Chezel, J.; Thervet, E.; Pallet, N.; et al. APOL1 genotype is a major determinant of lupus nephritis severity in patients of African ancestry. Nephrol. Dial. Transplant. 2023, 38, gfad063a_3155. [Google Scholar] [CrossRef]
- Lanata, C.M.; Nititham, J.; Taylor, K.E.; Chung, S.A.; Torgerson, D.G.; Seldin, M.F.; Pons-Estel, B.A.; Tusié-Luna, T.; Tsao, B.P.; Morand, E.F.; et al. Genetic contributions to lupus nephritis in a multi-ethnic cohort of systemic lupus erythematous patients. PLoS ONE 2018, 13, e0199003. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Jawad, A.S. The effect of ethnicity and genetic ancestry on the epidemiology, clinical features and outcome of systemic lupus erythematosus. Rheumatology 2017, 56, i67–i77. [Google Scholar] [CrossRef]
- Hahn, B.H.; McMahon, M.A.; Wilkinson, A.; Wallace, W.D.; Daikh, D.I.; FitzGerald, J.D.; Karpouzas, G.A.; Merrill, J.T.; Wallace, D.J.; Yazdany, J.; et al. American College of Rheumatology guidelines for screening, treatment, and management of lupus nephritis. Arthritis Care Res. 2012, 64, 797–808. [Google Scholar] [CrossRef]
- Mejía-Vilet, J.M.; Córdova-Sánchez, B.M.; Uribe-Uribe, N.O.; Correa-Rotter, R. Immunosuppressive treatment for pure membranous lupus nephropathy in a Hispanic population. Clin. Rheumatol. 2016, 35, 2219–2227. [Google Scholar] [CrossRef]
- Palmer, S.C.; Tunnicliffe, D.J.; Singh-Grewal, D.; Correa-Rotter, R. Induction and maintenance immunosuppression treatment of proliferative lupus nephritis: A network meta-analysis of randomized trials. Am. J. Kidney Dis. 2017, 70, 324–336. [Google Scholar] [CrossRef]
- Müller, F.; Boeltz, S.; Knitza, J.; Aigner, M.; Völkl, S.; Kharboutli, S.; Reimann, H.; Taubmann, J.; Kretschmann, S.; Rösler, W.; et al. CD19-targeted CAR T cells in refractory antisynthetase syndrome. Lancet 2023, 401, 815–818. [Google Scholar] [CrossRef]

| Drug | Mechanism of Action | Key Trial/Efficacy | Adverse Effects | Recommendation |
|---|---|---|---|---|
| Glucocorticoids | Broad anti-inflammatory and immunosuppressive [30]. | Foundational therapy; decades of clinical use. | Hyperglycemia, osteoporosis, infections, CVD [31]. | Taper to ≤5 mg/d; aim for withdrawal [20]. |
| Cyclophosphamide (CYC) | Alkylating agent; broad lymphocyte and neutrophil suppression [32]. | NIH trials [33,34,35]; Euro-Lupus: comparable efficacy with lower toxicity [36]. | Gonadal toxicity, infections, myelosuppression. * | Low-dose IV CYC for induction (KDIGO 2024) [20]. |
| Azathioprine (AZA) | Purine analog; inhibits B- and T-cell proliferation [32]. | Similar efficacy to CYC for maintenance [36,39,40]; comparable to MMF [45]. | Myelosuppression, hepatotoxicity. * | Maintenance if MMF unavailable/intolerable [20]. |
| Mycophenolate Mofetil (MMF) | Selective inhibitor of de novo purine synthesis [32]. | ALMS: non-inferior to IV CYC for induction [44,45]. | GI symptoms, leukopenia, serious infections. [41] | First-line induction and maintenance (KDIGO 2024) [20]. |
| Belimumab | Anti-BLyS monoclonal antibody; reduces autoreactive B-cells [46]. | BLISS-LN: CRR 30% vs. 20% at 2 yr; PERR 43% vs. 32% [47]. | Infections, infusion reactions. * | First-line add-on (KDIGO 2024) [20]. |
| Tacrolimus | CNI; suppresses T-cell activation, and acts as a podocyte stabilizer [19]. | Multi-target trial: CRR 45.9% vs. 25.6% at 24 wk [48,49]. | Nephrotoxicity, glucose intolerance, tremor. * | Multi-target induction; mainly Asia evidence [50]. |
| Voclosporin | Next-generation CNI; no drug monitoring required [51]. | AURORA 1: CRR 41% vs. 23% at 52 wk [52]; AURORA 2: sustained at 3 yr [53]. | Hypertension, GFR decline, diarrhea. * | First-line triple therapy (KDIGO 2024) [20,54]. |
| Rituximab | Chimeric anti-CD20 antibody; depletes circulating B-cells [55]. | LUNAR: ORR 56.9% vs. 45.8% (p = 0.18, not sig.) [56]. | Infusion reactions, infections, PML (rare). * | Off-label for refractory LN [58]. |
| Obinutuzumab | Humanized anti-CD20; enhanced B-cell depletion vs. rituximab. | REGENCY: CRR 46.4% vs. 33.1% at 76 wk (p = 0.02) [59]. | Infections, infusion reactions [59]. | FDA-approved for LN [59]. |
| Anifrolumab | Anti-IFNAR1; blocks type I IFN signaling [63,64]. | TULIP-LN (Phase II): primary endpoint not met [65]. | Herpes zoster, URI. * | Approved for non-renal SLE; Phase III IRIS ongoing [67]. |
| Telitacicept | TACI-Fc fusion protein; dual BLyS/APRIL blockade [68]. | Phase III SLE: SRI-4 82.6% [69]; LN real-world study: PERR 73.3%, CRR 68.4% [70]. | Infections, injection site reactions. * | NMPA-approved for SLE; Phase 2 RCT in LN ongoing (NCT05680480). |
| CAR-T-Cell therapy | Anti-CD19 CAR-T; deep B-cell depletion, including tissue niches. | Phase 1: clinical improvement in Class IV LN [71]; drug-free remission [72,73]. | CRS, neurotoxicity, infections. * | Experimental; refractory SLE/LN. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhu, W.; He, H.; Huang, X.; Zhang, L.; Pai, P. The Evolution of Lupus Nephritis Therapy from the 1960s to the Present. Bioengineering 2026, 13, 428. https://doi.org/10.3390/bioengineering13040428
Zhu W, He H, Huang X, Zhang L, Pai P. The Evolution of Lupus Nephritis Therapy from the 1960s to the Present. Bioengineering. 2026; 13(4):428. https://doi.org/10.3390/bioengineering13040428
Chicago/Turabian StyleZhu, Wenjuan, Haiyan He, Xiaoyan Huang, Lijun Zhang, and Pearl Pai. 2026. "The Evolution of Lupus Nephritis Therapy from the 1960s to the Present" Bioengineering 13, no. 4: 428. https://doi.org/10.3390/bioengineering13040428
APA StyleZhu, W., He, H., Huang, X., Zhang, L., & Pai, P. (2026). The Evolution of Lupus Nephritis Therapy from the 1960s to the Present. Bioengineering, 13(4), 428. https://doi.org/10.3390/bioengineering13040428





