Lab-on-a-Chip and Microfluidics Technologies for Nano Drug Delivery
Abstract
1. Introduction
1.1. Background on Nano Drug Delivery Systems (DDSs)
1.2. Emergence and Evolution of Lab-on-a-Chip (LoC) Technology
1.3. Scope and Organization of the Review
2. Fundamentals of Lab-on-a-Chip (LoC) and Microfluidic Technologies
2.1. Principles of LoC and Microfluidics
2.2. Materials and Fabrication Techniques
2.2.1. Materials
2.2.2. Fabrication Techniques
2.3. Fluid Handling and Flow Control Mechanisms
2.3.1. Pressure-Driven Flow (PDF)
2.3.2. Electroosmotic Flow (EOF)
2.3.3. Passive Flow Control Methods
2.3.4. Droplet Microfluidics
2.4. Integration of Detection and Sensing Modules
2.4.1. Optical Sensors
2.4.2. Electrochemical Sensors
2.4.3. Spectrometric-Based LoC Sensing Platforms
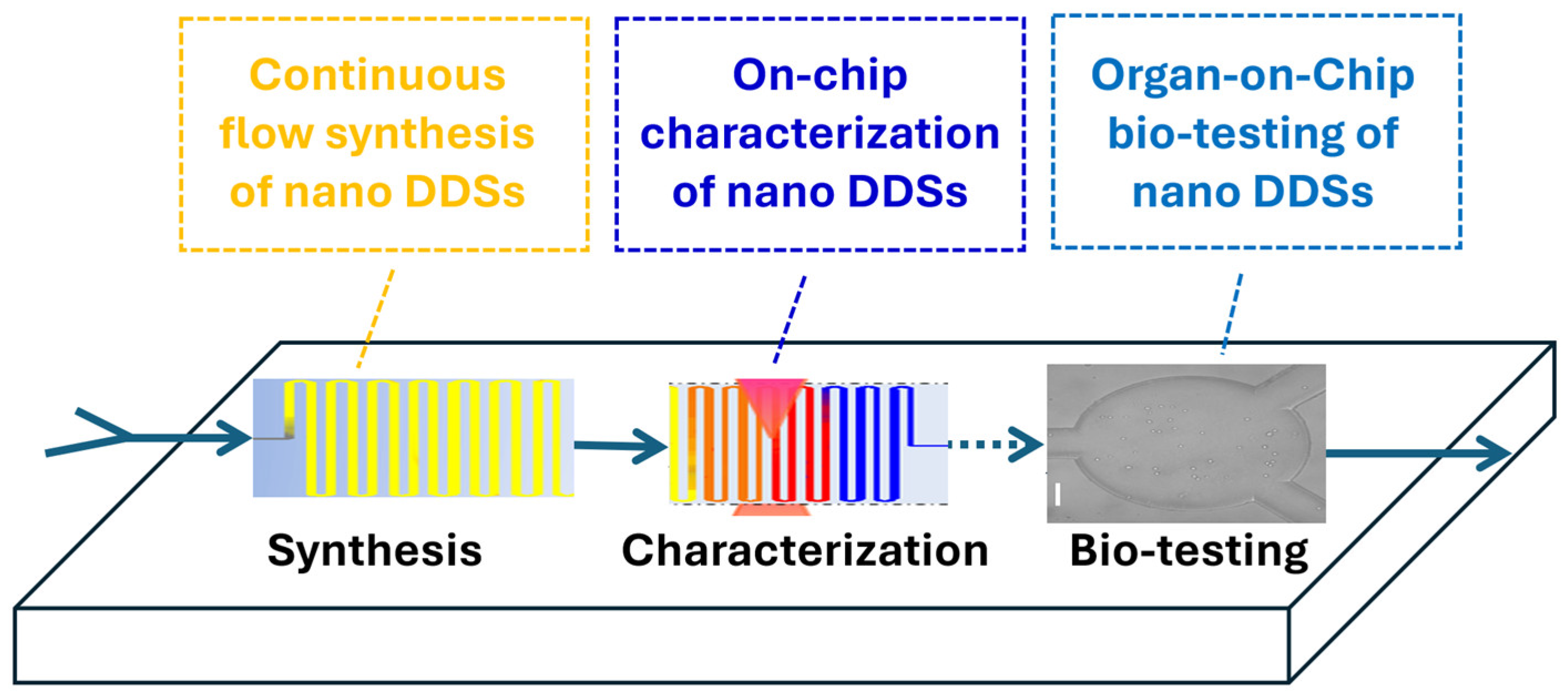
3. LoC-Based Synthesis and Characterization of Nanoparticle DDSs
3.1. Continuous Flow Synthesis of Nano DDSs (Liposomes, Polymeric NPs, Metallic NPs)
3.2. In Situ Characterization Tools (Spectroscopy, Imaging, Scattering)
3.3. Controlled Drug Loading On-Chip
3.4. Scalability of Nano DDSs Production
3.5. Clinically Relevant Nanomedicines Produced Using Microfluidics
4. Synergy Between LoC and Nano DDSs
4.1. On-Chip Screening and Optimization of Formulations
4.2. Real-Time Monitoring and Control of Pharmacokinetics and Drug Release
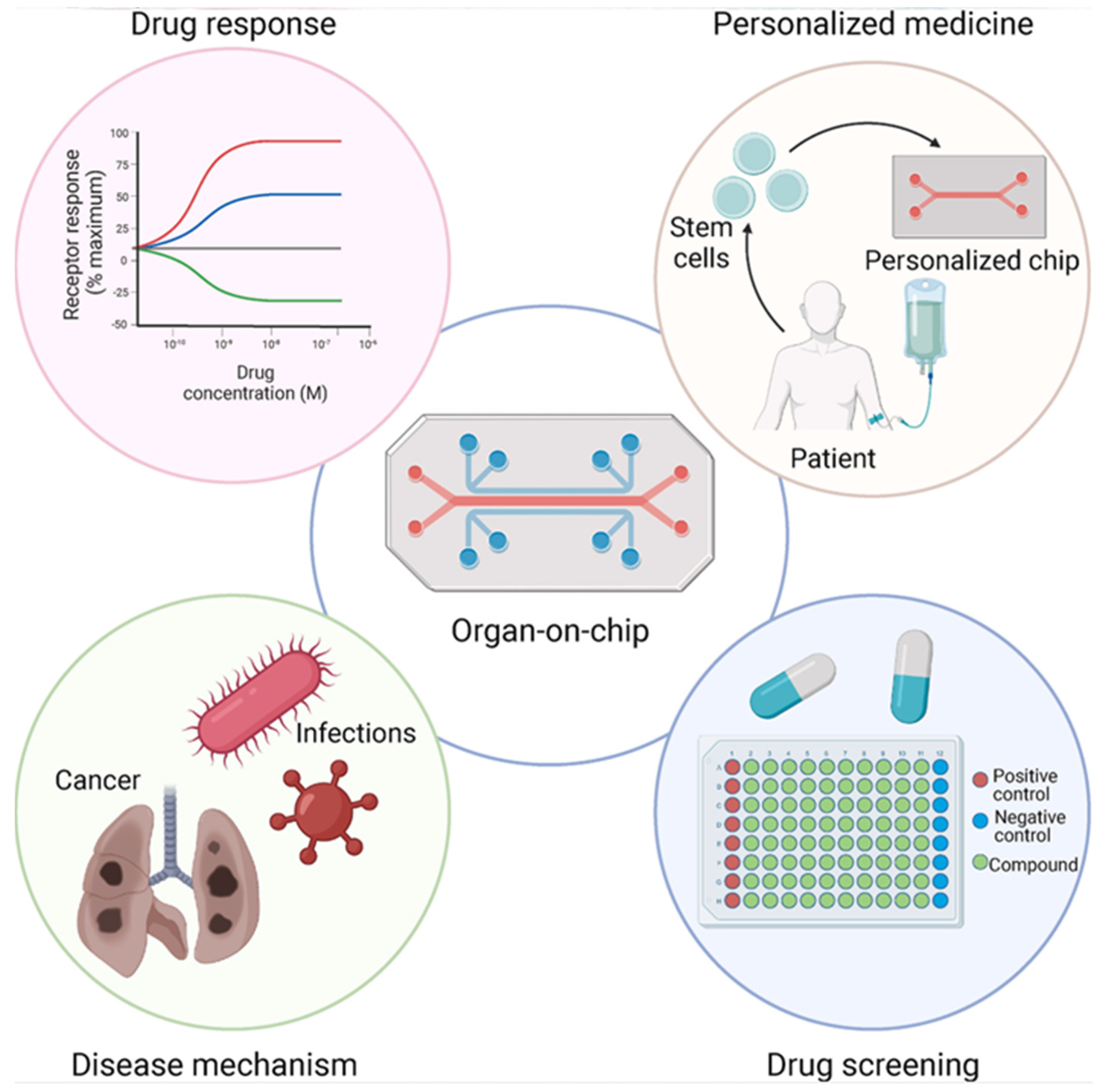
4.3. Organ-on-a-Chip (OoC) Models for Systemic Response to Nano DDSs
4.4. In Vitro and In Vivo Simulation On-Chip
5. Current Challenges and Limitations
5.1. Biocompatibility and Scaling Issues
5.2. Limited Physiological Relevance of Chip-Based Models
5.3. Standardization and Reproducibility
5.4. Data Integration and Analytical Complexity
5.5. Cost and Translation to Clinic
6. Emerging Trends and Future Perspectives
6.1. AI and Machine Learning in LoC–Nanomedicine Integration
6.2. Stimuli-Responsive Nanocarriers
6.3. 3D Printing and Hybrid Microfluidic Systems
6.4. Self-Powered and Portable LoC Systems
6.5. Translation to Clinical and Pharmaceutical Practice
6.6. Toward Fully Integrated Nano-Pharmaceutics Platforms
6.7. Regulatory Considerations for Microfluidic-Produced Nanomedicines
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wilczewska, A.Z.; Niemirowicz, K.; Markiewicz, K.H.; Car, H. Nanoparticles as drug delivery systems. Pharmacol. Rep. 2012, 64, 1020–1037. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S. Nano-based drug delivery system for therapeutics: A comprehensive review. Biomed. Phys. Eng. Express 2023, 9, 052002. [Google Scholar] [CrossRef] [PubMed]
- Sultana, A.; Zare, M.; Thomas, V.; Kumar, T.S.S.; Ramakrishna, S. Nano-based drug delivery systems: Conventional drug delivery routes, recent developments and future prospects. Med. Drug Discov. 2022, 15, 100134. [Google Scholar] [CrossRef]
- Tripathy, S.; Das, M.K. Dendrimers and their applications as novel drug delivery carriers. J. Appl. Pharm. Sci. 2013, 3, 142–149. [Google Scholar] [CrossRef]
- Ullah, M.; Shah, M.I.; Hasan, M.W.; Jamshed, M.; Mustafa, U.; Inam, M. Engineered metal nanoparticles for precision drug delivery: Pioneering the future of medicine: Mini review. J. Chin. Chem. Soc. 2024, 71, 1358–1367. [Google Scholar] [CrossRef]
- Yang, J.; Jia, C.; Yang, J. Designing Nanoparticle-based Drug Delivery Systems for Precision Medicine. Int. J. Med. Sci. 2021, 18, 2943–2949. [Google Scholar] [CrossRef]
- Mittal, M.; Juneja, S.; Pandey, N.; Mittal, R. Nanoparticle-Based Drug Delivery Systems: Current Advances and Future Directions. Curr. Drug Targets 2025, in press. [Google Scholar] [CrossRef]
- Geng, T.; Ding, L.; Liu, M.; Zou, X.; Gu, Z.; Lin, H.; Sun, L. Preservation of extracellular vesicles for drug delivery: A comparative evaluation of storage buffers. J. Drug Deliv. Sci. Technol. 2025, 107, 106850. [Google Scholar] [CrossRef]
- Hu, H.Y.; Xu, Z.M.; Liu, J.T.; Hu, H.D.; Yang, H.Y.; Ma, J.; Chen, J.Y.; Xu, H.W.; Cai, Z.H.; Wang, X.G.; et al. Light-Responsive Hydrogel with Nano-Microstructures for Bacteria Eradication and Tumor Therapy. Acs Appl. Nano Mater. 2025, 8, 6242–6254. [Google Scholar] [CrossRef]
- Sethuraman, V.; Janakiraman, K.; Krishnaswami, V.; Kandasamy, R. Recent Progress in Stimuli-Responsive Intelligent Nano Scale Drug Delivery Systems: A Special Focus Towards pH-Sensitive Systems. Curr. Drug Targets 2021, 22, 947–966. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.; Xu, Y.; Liu, M.M.; Wang, Y.M.; Wu, G.L. Nano-injectable pH/NIR-responsive hydrogel for chemo-photothermal synergistic drug delivery. J. Biomater. Appl. 2023, 38, 614–628. [Google Scholar] [CrossRef]
- Li, Z.Q.; Wang, X.; Wan, W.M.; Zhang, N.; Zhang, L.M.; Wang, X.Y.; Lin, K.; Yang, J.; Hao, J.; Tian, F. Rational design of pH-responsive nano-delivery system with improved biocompatibility and targeting ability from cellulose nanocrystals via surface polymerization for intracellular drug delivery. Int. J. Biol. Macromol. 2024, 281, 136435. [Google Scholar] [CrossRef] [PubMed]
- Farjadian, F.; Rezaeifard, S.; Naeimi, M.; Ghasemi, S.; Mohammadi-Samani, S.; Welland, M.E.; Tayebi, L. Temperature and pH-responsive nano-hydrogel drug delivery system based on lysine-modified poly (vinylcaprolactam). Int. J. Nanomed. 2019, 14, 6901–6915. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, X.; Hu, B.; Zou, B.; Xu, Y. Advancements in nanomedicine delivery systems: Unraveling immune regulation strategies for tumor immunotherapy. Nanomedicine 2024, 19, 1821–1840. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Suzuki, T.; Farsad, K. Nanoparticles as Multifunctional Drug-Delivery Systems: A Comprehensive Review and Integration into Interventional Oncology. J. Vasc. Interv. Radiol. 2025, 36, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Rahmati, M.; Mozafari, M. Nano-immunoengineering: Opportunities and challenges. Curr. Opin. Biomed. Eng. 2019, 10, 51–59. [Google Scholar] [CrossRef]
- Chuang, S.T.; Conklin, B.; Stein, J.B.; Pan, G.; Lee, K.-B. Nanotechnology-enabled immunoengineering approaches to advance therapeutic applications. Nano Converg. 2022, 9, 19. [Google Scholar] [CrossRef]
- Khawar, M.B.; Afzal, A.; Abbasi, M.H.; Sheikh, N.; Sun, H. Nano-immunoengineering of CAR-T cell therapy against tumor microenvironment: The way forward in combating cancer. OpenNano 2023, 10, 100124. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.; Das, T.; Nandy, S.; Sahib, S.; Preetam, S.; Gopalakrishnan, A.V.; Dey, A. Ligand-based active targeting strategies for cancer theranostics. Naunyn-Schmiedebergs Arch. Pharmacol. 2023, 396, 3417–3441. [Google Scholar] [CrossRef]
- Picheth, G.F.; Ganzella, F.A.d.O.; Filizzola, J.O.; Canquerino, Y.K.; Cardoso, G.C.; Collini, M.B.; Colauto, L.B.; Figueroa-Magalhaes, M.C.; Cavalieri, E.A.; Klassen, G. Ligand-mediated nanomedicines against breast cancer: A review. Nanomedicine 2022, 17, 645–664. [Google Scholar] [CrossRef] [PubMed]
- Bajracharya, R.; Song, J.G.; Patil, B.R.; Lee, S.H.; Noh, H.-M.; Kim, D.-H.; Kim, G.-L.; Seo, S.-H.; Park, J.-W.; Jeong, S.H. Functional ligands for improving anticancer drug therapy: Current status and applications to drug delivery systems. Drug Deliv. 2022, 29, 1959–1970. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Li, W.; Li, Y.; Yin, S. Nanoparticle-based drug delivery systems targeting cancer cell surfaces. RSC Adv. 2023, 13, 21365–21382. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, K.; Tagami, T.; Ozeki, T. Targeted Drug Delivery for Cancer Therapy Using Functionalized Nanocarrier. In Nanomedicines for Effective Cancer Therapy; Springer: New York, NY, USA, 2024; pp. 35–49. [Google Scholar]
- Liu, L.C.; Chen, Y.H.; Lu, D.W. Overview of Recent Advances in Nano-Based Ocular Drug Delivery. Int. J. Mol. Sci. 2023, 24, 15352. [Google Scholar] [CrossRef]
- Li, S.; Chen, L.; Fu, Y. Nanotechnology-based ocular drug delivery systems: Recent advances and future prospects. J. Nanobiotechnol. 2023, 21, 232. [Google Scholar] [CrossRef] [PubMed]
- Khiev, D.; Mohamed, Z.A.; Vichare, R.; Paulson, R.; Bhatia, S.; Mohapatra, S.; Lobo, G.P.; Valapala, M.; Kerur, N.; Passaglia, C.L. Emerging nano-formulations and nanomedicines applications for ocular drug delivery. Nanomaterials 2021, 11, 173. [Google Scholar] [CrossRef]
- John, J. Advancements in nano-based drug delivery systems for therapeutics: A comprehensive review. RSC Pharm. 2025, 3, 43–59. [Google Scholar] [CrossRef]
- Islam, S.; Ahmed, M.M.S.; Islam, M.A.; Hossain, N.; Chowdhury, M.A. Advances in nanoparticles in targeted drug delivery—A review. Results Surf. Interf. 2025, 19, 100529. [Google Scholar] [CrossRef]
- Daw, R.; Finkelstein, J. Lab on a chip. Nature 2006, 442, 367. [Google Scholar] [CrossRef]
- Capretto, L.; Carugo, D.; Cheng, W.; Hill, M.; Zhang, X.L. Continuous-flow production of polymeric micelles in microreactors: Experimental and computational analysis. J. Colloid. Interface Sci. 2011, 357, 243–251. [Google Scholar] [CrossRef]
- Capretto, L.; Cheng, W.; Carugo, D.; Hill, M.; Zhang, X. Microfluidic reactors for controlled synthesis of polymeric micelles. J. Control. Release 2010, 148, e25–e26. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, P.D.I.; Haswell, S.J.; Pombo-Villar, E.; Warrington, B.H.; Watts, P.; Wong, S.Y.F.; Zhang, X. Micro reactors: Principles and applications in organic synthesis. Tetrahedron 2002, 58, 4735–4757. [Google Scholar] [CrossRef]
- Mathew, A.; Liu, D.; Yu, M.; Haasbroek, P.; Wang, X.; Guan, T.; Zaki, S.; Su, Q.; Li, J.; Jagannath, A. Lab-on-a-chip: From Prototyping to Scale-up Production. In Lab-on-a-Chip Devices for Advanced Biomedicines: Laboratory Scale Engineering to Clinical Ecosystem; Parihar, A., Mehta, P.P., Eds.; Detection Science Series; Royal Society of Chemistry: London, UK, 2024; Volume 25, pp. 505–559. [Google Scholar]
- McCreedy, T. Rapid prototyping of glass and PDMS microstructures for micro total analytical systems and micro chemical reactors by microfabrication in the general laboratory. Anal. Chim. Acta 2001, 427, 39–43. [Google Scholar] [CrossRef]
- Broadwell, I.; Fletcher, P.D.I.; Haswell, S.J.; McCreedy, T.; Zhang, X.L. Quantitative 3-dimensional profiling of channel networks within transparent ‘lab-on-a-chip’ microreactors using a digital imaging method. Lab Chip 2001, 1, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.L.; Haswell, S.J. Materials matter in microfluidic devices. MRS Bull. 2006, 31, 95–99. [Google Scholar] [CrossRef]
- Zhang, X.L.; Cooper, J.M.; Monaghan, P.B.; Haswell, S.J. Continuous flow separation of particles within an asymmetric microfluidic device. Lab Chip 2006, 6, 561–566. [Google Scholar] [CrossRef]
- Xia, Y.; Whitesides, G.M. Soft Lithography. Angew. Chem. Int. Ed. Engl. 1998, 37, 550–575. [Google Scholar] [CrossRef]
- Mitrogiannopoulou, A.-M.; Tselepi, V.; Ellinas, K. Polymeric and paper-based lab-on-a-chip devices in food safety: A review. Micromachines 2023, 14, 986. [Google Scholar] [CrossRef]
- Alexandre-Franco, M.F.; Kouider, R.; Kassir Al-Karany, R.; Cuerda-Correa, E.M.; Al-Kassir, A. Recent advances in polymer science and fabrication processes for enhanced microfluidic applications: An overview. Micromachines 2024, 15, 1137. [Google Scholar] [CrossRef]
- Cristaldi, D.A.; Yanar, F.; Mosayyebi, A.; García-Manrique, P.; Stulz, E.; Carugo, D.; Zhang, X. Easy-to-perform and cost-effective fabrication of continuous-flow reactors and their application for nanomaterials synthesis. New Biotechnol. 2018, 47, 1–7. [Google Scholar] [CrossRef]
- Cristaldi, D.A.; Labanca, A.; Pottinger, T.D.; Owen, J.; Stulz, E.; Zhang, X.L.; Carugo, D. 3D printed reactor-in-a-centrifuge (RIAC): Making flow-synthesis of nanoparticles pump-free and cost-effective. Chem. Eng. J. 2021, 425, 130656. [Google Scholar] [CrossRef]
- Tice, J.D.; Song, H.; Lyon, A.D.; Ismagilov, R.F. Formation of Droplets and Mixing in Multiphase Microfluidics at Low Values of the Reynolds and the Capillary Numbers. Langmuir 2003, 19, 9127–9133. [Google Scholar] [CrossRef]
- Teh, S.Y.; Lin, R.; Hung, L.H.; Lee, A.P. Droplet microfluidics. Lab Chip 2008, 8, 198–220. [Google Scholar] [CrossRef] [PubMed]
- Utada, A.S.; Lorenceau, E.; Link, D.R.; Kaplan, P.D.; Stone, H.A.; Weitz, D. Monodisperse double emulsions generated from a microcapillary device. Science 2005, 308, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Kaspar, O.; Koyuncu, A.H.; Pittermannova, A.; Ulbrich, P.; Tokarova, V. Governing factors for preparation of silver nanoparticles using droplet-based microfluidic device. Biomed. Microdevices 2019, 21, 88. [Google Scholar] [CrossRef]
- Zou, L.; Huang, B.B.; Zheng, X.Y.; Pan, H.L.; Zhang, Q.; Xie, W.H.; Zhao, Z.J.; Li, X. Microfluidic synthesis of magnetic nanoparticles in droplet-based microreactors. Mater. Chem. Phys. 2022, 276, 125384. [Google Scholar] [CrossRef]
- Erdem, E.Y. Droplet-based Microfluidic Device for the Synthesis of Silica Nanoparticles. Celal Bayar Univ. J. Sci. 2020, 16, 245–249. [Google Scholar] [CrossRef]
- Deng, H.; Nguyen, L.; Shenoda, A.; Banerjee, P.; Mehlawat, N.; Cao, C.; Sharma, K.; Sharma, M.; Brenker, J.; Martino, M.M.; et al. Tunable passive chaotic mixing in droplet microfluidics for controlled nanomaterial synthesis. Phys. Fluids 2025, 37. [Google Scholar] [CrossRef]
- Wikswo, J.P.; Block, F.E., 3rd; Cliffel, D.E.; Goodwin, C.R.; Marasco, C.C.; Markov, D.A.; McLean, D.L.; McLean, J.A.; McKenzie, J.R.; Reiserer, R.S.; et al. Engineering challenges for instrumenting and controlling integrated organ-on-chip systems. IEEE Trans. Biomed. Eng. 2013, 60, 682–690. [Google Scholar] [CrossRef]
- Stavrou, M.; Phung, N.; Grimm, J.; Andreou, C. Organ-on-chip systems as a model for nanomedicine. Nanoscale 2023, 15, 9927–9940. [Google Scholar] [CrossRef]
- Piergiovanni, M.; Leite, S.B.; Corvi, R.; Whelan, M. Standardisation needs for organ on chip devices. Lab Chip 2021, 21, 2857–2868. [Google Scholar] [CrossRef] [PubMed]
- Caballero, D.; Kaushik, S.; Correlo, V.M.; Oliveira, J.M.; Reis, R.L.; Kundu, S.C. Organ-on-chip models of cancer metastasis for future personalized medicine: From chip to the patient. Biomaterials 2017, 149, 98–115. [Google Scholar] [CrossRef] [PubMed]
- Kieninger, J.; Weltin, A.; Flamm, H.; Urban, G.A. Microsensor systems for cell metabolism—From 2D culture to organ-on-chip. Lab Chip 2018, 18, 1274–1291. [Google Scholar] [CrossRef] [PubMed]
- Dornhof, J.; Kieninger, J.; Rupitsch, S.J.; Weltin, A. Microsensor systems for cell metabolism—From 2D culture to organ-on-chip (2019-2024). Lab. Chip 2025, 25, 1149–1168. [Google Scholar] [CrossRef]
- Weidema, J.; de Vries, M.; Mummery, C.; de Graeff, N. The ethical aspects of human organ-on-chip models: A mapping review. Stem Cell Rep. 2025, 20, 102686. [Google Scholar] [CrossRef]
- Gangwal, A.; Lavecchia, A. Artificial intelligence in preclinical research: Enhancing digital twins and organ-on-chip to reduce animal testing. Drug Discov. Today 2025, 30, 104360. [Google Scholar] [CrossRef]
- Zhou, J.; Dong, J.; Hou, H.; Huang, L.; Li, J. High-throughput microfluidic systems accelerated by artificial intelligence for biomedical applications. Lab Chip 2024, 24, 1307–1326. [Google Scholar] [CrossRef]
- Asadian, E.; Bahramian, F.; Siavashy, S.; Movahedi, S.; Keçili, R.; Hussain, C.M.; Ghorbani-Bidkorpeh, F. A review on recent advances of AI-integrated microfluidics for analytical and bioanalytical applications. TrAC Trends Anal. Chem. 2024, 181, 118004. [Google Scholar] [CrossRef]
- Dong, H.; Lin, J.; Tao, Y.; Jia, Y.; Sun, L.; Li, W.J.; Sun, H. AI-enhanced biomedical micro/nanorobots in microfluidics. Lab Chip 2024, 24, 1419–1440. [Google Scholar] [CrossRef]
- Rui, Z.; Mehdizadeh, S.; Abasi, M.; Pilehvar, Y. Advances in nanorobotics for gastrointestinal surgery: A new frontier in precision medicine and minimally invasive therapeutics. J. Robot. Surg. 2025, 19, 390. [Google Scholar] [CrossRef]
- Zhang, D.; Lin, L.; Deng, C.; Osman, M.S.; Rodriguez, P.E.S.; Han, F.; Li, M.; Wang, L. Advanced Imaging Strategies Based on Intelligent Micro/Nanomotors. Cyborg Bionic Syst. 2025, 6, 0384. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.M.; Carthy, E.; Dunne, N.; Kinahan, D. Advances in nanoparticle synthesis assisted by microfluidics. Lab Chip 2025, 25, 3060–3093. [Google Scholar] [CrossRef] [PubMed]
- Guevara, M.L.; Persano, F.; Persano, S. Advances in Lipid Nanoparticles for mRNA-Based Cancer Immunotherapy. Front. Chem. 2020, 8, 589959. [Google Scholar] [CrossRef] [PubMed]
- Ball, R.L.; Hajj, K.A.; Vizelman, J.; Bajaj, P.; Whitehead, K.A. Lipid Nanoparticle Formulations for Enhanced Co-delivery of siRNA and mRNA. Nano Lett. 2018, 18, 3814–3822. [Google Scholar] [CrossRef]
- Lopes, C.; Cristóvão, J.; Silvério, V.; Lino, P.R.; Fonte, P. Microfluidic production of mRNA-loaded lipid nanoparticles for vaccine applications. Expert. Opin. Drug Deliv. 2022, 19, 1381–1395. [Google Scholar] [CrossRef]
- Zhao, S.; Huang, P.H.; Zhang, H.; Rich, J.; Bachman, H.; Ye, J.; Zhang, W.; Chen, C.; Xie, Z.; Tian, Z.; et al. Fabrication of tunable, high-molecular-weight polymeric nanoparticles via ultrafast acoustofluidic micromixing. Lab Chip 2021, 21, 2453–2463. [Google Scholar] [CrossRef]
- Maged, A.; Abdelbaset, R.; Mahmoud, A.A.; Elkasabgy, N.A. Merits and advances of microfluidics in the pharmaceutical field: Design technologies and future prospects. Drug Deliv. 2022, 29, 1549–1570. [Google Scholar] [CrossRef]
- Yu, Z.; Chen, Y.; Li, J.; Chen, C.; Lu, H.; Chen, S.; Zhang, T.; Guo, T.; Zhu, Y.; Jin, J.; et al. A tempo-spatial controllable microfluidic shear-stress generator for in-vitro mimicking of the thrombus. J. Nanobiotechnol. 2024, 22, 187. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, J.; Sun, R.; Han, S.; Yang, Z.; Teng, L. Microfluidics for nano-drug delivery systems: From fundamentals to industrialization. Acta Pharm. Sin. B 2023, 13, 3277–3299. [Google Scholar] [CrossRef]
- Ottino, J.M.; Wiggins, S.R.; Ottino, J.M.; Wiggins, S. Introduction: Mixing in microfluidics. Philos. Trans. R. Soc. London. Ser. A Math. Phys. Eng. Sci. 2004, 362, 923–935. [Google Scholar] [CrossRef]
- Capretto, L.; Cheng, W.; Hill, M.; Zhang, X.L. Micromixing within microfluidic devices. Top. Curr. Chem. 2011, 304, 27–68. [Google Scholar] [CrossRef]
- Zhang, X.; Haswell, S.J. Micro-Fluidic and Lab-on-a-Chip Technology. In New Avenues to Efficient Chemical Synthesis; Springer: Berlin/Heidelberg, Germany, 2007; pp. 21–37. [Google Scholar]
- Francesko, A.; Cardoso, V.F.; Lanceros-Méndez, S. Chapter 1—Lab-on-a-chip technology and microfluidics. In Microfluidics for Pharmaceutical Applications; Santos, H.A., Liu, D., Zhang, H., Eds.; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 3–36. [Google Scholar]
- Mathur, A.; Roy, S. 10—Microfluidics and lab-on-a-chip. In Electrochemical Sensors; Maruccio, G., Narang, J., Eds.; Woodhead Publishing: Cambridge, UK, 2022; pp. 261–287. [Google Scholar]
- Fletcher, P.D.I.; Haswell, S.J.; Zhang, X.L. Electrical currents and liquid flow rates in micro-reactors. Lab Chip 2001, 1, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, P.D.I.; Haswell, S.J.; Zhang, X.L. Electrokinetic control of a chemical reaction in a lab-on-a-chip micro-reactor: Measurement and quantitative modelling. Lab Chip 2002, 2, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Spanner, D. Electroosmotic flow. In Transport in Plants I: Phloem Transport; Springer: Berlin/Heidelberg, Germany, 1975; pp. 301–327. [Google Scholar]
- Alizadeh, A.; Hsu, W.L.; Wang, M.; Daiguji, H. Electroosmotic flow: From microfluidics to nanofluidics. Electrophoresis 2021, 42, 834–868. [Google Scholar] [CrossRef] [PubMed]
- Gurkan, U.A.; Wood, D.K.; Carranza, D.; Herbertson, L.H.; Diamond, S.L.; Du, E.; Guha, S.; Di Paola, J.; Hines, P.C.; Papautsky, I.; et al. Next generation microfluidics: Fulfilling the promise of lab-on-a-chip technologies. Lab Chip 2024, 24, 1867–1874. [Google Scholar] [CrossRef] [PubMed]
- Apoorva, S.; Nguyen, N.T.; Sreejith, K.R. Recent developments and future perspectives of microfluidics and smart technologies in wearable devices. Lab Chip 2024, 24, 1833–1866. [Google Scholar] [CrossRef]
- Akther, F.; Yakob, S.B.; Nguyen, N.-T.; Ta, H.T. Surface Modification Techniques for Endothelial Cell Seeding in PDMS Microfluidic Devices. Biosensors 2020, 10, 182. [Google Scholar] [CrossRef]
- Gökaltun, A.; Kang, Y.B.; Yarmush, M.L.; Usta, O.B.; Asatekin, A. Simple Surface Modification of Poly(dimethylsiloxane) via Surface Segregating Smart Polymers for Biomicrofluidics. Sci. Rep. 2019, 9, 7377. [Google Scholar] [CrossRef]
- Lin, L.; Chung, C.-K. PDMS Microfabrication and Design for Microfluidics and Sustainable Energy Application: Review. Micromachines 2021, 12, 1350. [Google Scholar] [CrossRef]
- Wang, Z.; Shah, A.; Lee, H.; Lee, C.H. Microfluidic technologies for wearable and implantable biomedical devices. Lab Chip 2025, 25, 4542–4576. [Google Scholar] [CrossRef]
- Su, R.; Wang, F.; McAlpine, M.C. 3D printed microfluidics: Advances in strategies, integration, and applications. Lab Chip 2023, 23, 1279–1299. [Google Scholar] [CrossRef]
- Ahmadianyazdi, A.; Miller, I.J.; Folch, A. Tunable resins with PDMS-like elastic modulus for stereolithographic 3D-printing of multimaterial microfluidic actuators. Lab Chip 2023, 23, 4019–4032. [Google Scholar] [CrossRef] [PubMed]
- Venzac, B. Light-based 3D printing and post-treatments of moulds for PDMS soft lithography. Lab Chip 2025, 25, 2129–2147. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Fu, H. Precise measurement and control of the pressure-driven flows for microfluidic systems. Electrophoresis 2020, 41, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Gao, Y.; Nguyen, N.-T.; Wong, T.N.; Yang, C.; Ooi, K.T. Interface control of pressure-driven two-fluid flow in microchannels using electroosmosis. J. Micromech. Microeng. 2005, 15, 2289. [Google Scholar] [CrossRef]
- Lake, J.R.; Heyde, K.C.; Ruder, W.C. Low-cost feedback-controlled syringe pressure pumps for microfluidics applications. PLoS ONE 2017, 12, e0175089. [Google Scholar] [CrossRef]
- Wang, J.; Cui, X.; Wang, W.; Wang, J.; Zhang, Q.; Guo, X.; Liang, Y.; Lin, S.; Chu, B.; Cui, D. Microfluidic-based electrically driven particle manipulation techniques for biomedical applications. RSC Adv. 2025, 15, 167–198. [Google Scholar] [CrossRef]
- Movahed, S.; Kamali, R.; Eghtesad, M.; Khosravifard, A. Analytical study of mixed electroosmotic-pressure-driven flow in rectangular micro-channels. Theor. Comput. Fluid. Dyn. 2013, 27, 599–616. [Google Scholar] [CrossRef]
- Yuan, S.; Zhou, M.; Liu, X.; Jiang, B. Effect of pressure-driven flow on electroosmotic flow and electrokinetic mass transport in microchannels. Int. J. Heat. Mass. Transf. 2023, 206, 123925. [Google Scholar] [CrossRef]
- Hassan, S.U.; Tariq, A.; Noreen, Z.; Donia, A.; Zaidi, S.Z.J.; Bokhari, H.; Zhang, X. Capillary-Driven Flow Microfluidics Combined with Smartphone Detection: An Emerging Tool for Point-of-Care Diagnostics. Diagnostics 2020, 10, 509. [Google Scholar] [CrossRef]
- Hassan, S.U.; Zhang, X. Design and Fabrication of Capillary-Driven Flow Device for Point-Of-Care Diagnostics. Biosensors 2020, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Songok, J.; Toivakka, M. Enhancing Capillary-Driven Flow for Paper-Based Microfluidic Channels. ACS Appl. Mater. Interfaces 2016, 8, 30523–30530. [Google Scholar] [CrossRef] [PubMed]
- Songok, J.; Toivakka, M. Modelling of capillary-driven flow for closed paper-based microfluidic channels. J. Micromech. Microeng. 2017, 27, 065001. [Google Scholar] [CrossRef]
- Filatov, N.A.; Evstrapov, A.A.; Bukatin, A.S. Negative Pressure Provides Simple and Stable Droplet Generation in a Flow-Focusing Microfluidic Device. Micromachines 2021, 12, 662. [Google Scholar] [CrossRef]
- Fergola, A.; Ballesio, A.; Frascella, F.; Napione, L.; Cocuzza, M.; Marasso, S.L. Droplet Generation and Manipulation in Microfluidics: A Comprehensive Overview of Passive and Active Strategies. Biosensors 2025, 15, 345. [Google Scholar] [CrossRef]
- Ali, M.; Kim, W.; Khan, M.S.; Sahin, M.A.; Destgeer, G.; Park, J. Droplet acoustofluidics: Recent progress and challenges. Biomicrofluidics 2025, 19, 031502. [Google Scholar] [CrossRef]
- Yuan, T. Microfluidic Fiber and Its Sensing Laboratory. In How to Build a Lab-on/in-Fiber; Yuan, L., Ed.; Springer Nature: Singapore, 2025; pp. 227–281. [Google Scholar]
- Zmijan, R.; Carboni, M.; Capretto, L.; Stulz, E.; Zhang, X. In situ microspectroscopic monitoring within a microfluidic reactor. RSC Adv. 2014, 4, 14569–14572. [Google Scholar] [CrossRef]
- Wang, W.; Xia, L.; Xiao, X.; Li, G. Recent Progress on Microfluidics Integrated with Fiber-Optic Sensors for On-Site Detection. Sensors 2024, 24, 2067. [Google Scholar] [CrossRef]
- Torres-Cubillo, A.; Teuber, A.; Halir, R.; Mizaikoff, B. Optimized waveguides for mid-infrared lab-on-chip systems: A rigorous design approach. Sens. Actuat A-Phys. 2024, 378, 115797. [Google Scholar] [CrossRef]
- Glatz, R.T.; Ates, H.C.; Mohsenin, H.; Weber, W.; Dincer, C. Designing electrochemical microfluidic multiplexed biosensors for on-site applications. Anal. Bioanal. Chem. 2022, 414, 6531–6540. [Google Scholar] [CrossRef]
- Abouhagger, A.; Celiesiute-Germaniene, R.; Bakute, N.; Stirke, A.; Melo, W. Electrochemical biosensors on microfluidic chips as promising tools to study microbial biofilms: A review. Front. Cell Infect. Microbiol. 2024, 14, 1419570. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, M.B.; Ayachit, N.H.; Aminabhavi, T.M. Recent Advances in Microfluidics-Based Electrochemical Sensors for Foodborne Pathogen Detection. Biosensors 2023, 13, 246. [Google Scholar] [CrossRef] [PubMed]
- Ouedraogo, L.J.G.; Kling, M.; Hashemi, N.N. Graphene microelectrodes for real-time impedance spectroscopy of neural cells in organ-on-a-chip. APL Mater. 2025, 13, 021113. [Google Scholar] [CrossRef]
- Obeid, P.J.; Sari-Chmayssem, N.; Yammine, P.; Homsi, D.; El-Nakat, H.; Matar, Z.; Hamieh, S.; Koumeir, D.; Chmayssem, A. Designs and Materials of Electrodes for Electrochemical Sensors. Chemelectrochem 2025, 12, e202500230. [Google Scholar] [CrossRef]
- Wang, C.; Weng, G.; Li, J.; Zhu, J.; Zhao, J. A review of SERS coupled microfluidic platforms: From configurations to applications. Anal. Chim. Acta 2024, 1296, 342291. [Google Scholar] [CrossRef]
- Yu, X.; Park, S.; Lee, S.; Joo, S.-W.; Choo, J. Microfluidics for disease diagnostics based on surface-enhanced raman scattering detection. Nano Converg. 2024, 11, 17. [Google Scholar] [CrossRef]
- Li, D.-L.; Huang, W.-S.; Wu, Y.H.; Jen, C.-P. Microspectrometer-Enabled Real-Time Concentration Monitoring in the Microfluidic Protein Enrichment Chip. Biosensors 2025, 15, 1. [Google Scholar] [CrossRef]
- Li, H.; Ma, Y.; Fu, R.; Peng, J.; Zhai, Y.; Li, J.; Xu, W.; Hu, S.; Ma, H.; Wheeler, A.R.; et al. Droplet-Based Microfluidics with Mass Spectrometry for Microproteomics. Engineering 2024, 43, 37–53. [Google Scholar] [CrossRef]
- Bogdanowicz, R.; Jönsson-Niedziółka, M.; Vereshchagina, E.; Dettlaff, A.; Boonkaew, S.; Pierpaoli, M.; Wittendorp, P.; Jain, S.; Tyholdt, F.; Thomas, J.; et al. Microfluidic devices for photo-and spectroelectrochemical applications. Curr. Opin. Electrochem. 2022, 36, 101138. [Google Scholar] [CrossRef]
- Mehraji, S.; DeVoe, D.L. Microfluidic synthesis of lipid-based nanoparticles for drug delivery: Recent advances and opportunities. Lab Chip 2024, 24, 1154–1174. [Google Scholar] [CrossRef]
- Capretto, L.; Cheng, W.; Carugo, D.; Katsamenis, O.L.; Hill, M.; Zhang, X.L. Mechanism of co-nanoprecipitation of organic actives and block copolymers in a microfluidic environment. Nanotechnology 2012, 23, 375602. [Google Scholar] [CrossRef] [PubMed]
- Gdowski, A.; Johnson, K.; Shah, S.; Gryczynski, I.; Vishwanatha, J.; Ranjan, A. Optimization and scale up of microfluidic nanolipomer production method for preclinical and potential clinical trials. J. Nanobiotechnol. 2018, 16, 12. [Google Scholar] [CrossRef] [PubMed]
- Gharatape, A.; Niasari-Naslaji, Z.; Leblond Chain, J.; Tabatabaei, N.; Faridi-Majidi, R. Microfluidic-based nanocarriers for overcoming biological barriers in therapeutic delivery systems. Nanoscale 2025, 17, 23822–23853. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Zhang, L.; Liu, C.; Li, X.; Hu, G.; Sun, J.; Jiang, X. Microfluidic based high throughput synthesis of lipid-polymer hybrid nanoparticles with tunable diameters. Biomicrofluidics 2015, 9, 052604. [Google Scholar] [CrossRef]
- Chen, K.; Yang, H.; Cai, R. Microfluidics for Nanomedicine Delivery. ACS Biomater. Sci. Eng. 2025, 11, 774–783. [Google Scholar] [CrossRef]
- Pal, S.; Gkogkos, G.; Piovesan, J.; Whiteley, Z.; Besenhard, M.O.; Storozhuk, L.; Lees, M.R.; Thanh, N.T.K.; Craig, D.Q.M.; MacRobert, A.J.; et al. Multistep non-fouling continuous flow synthesis and PEG-functionalisation of biocompatible iron oxide nanoparticles for magnetic hyperthermia, photothermal heating and antifungal activity. J. Flow Chem. 2025, 15, 185–196. [Google Scholar] [CrossRef]
- Gimondi, S.; Ferreira, H.; Reis, R.L.; Neves, N.M. Microfluidic devices: A tool for nanoparticle synthesis and performance evaluation. Acs Nano 2023, 17, 14205–14228. [Google Scholar] [CrossRef]
- Martín-Asensio, A.; Pardo, I.; Mesa, R.; Pardo, D.; Fernández-Blázquez, J.P.; Martínez-Guil, J.C.; Castellanos, M.; Hernández, J.J.; Somoza, Á.; Rodríguez, I. Investigating non fluorescence nanoparticle transport in Matrigel-filled microfluidic devices using synchrotron X-ray scattering. Micro Nano Syst. Lett. 2024, 12, 22. [Google Scholar] [CrossRef]
- Šimić, M.; Auer, D.; Neuper, C.; Šimić, N.; Prossliner, G.; Prassl, R.; Hill, C.; Hohenester, U. Real-Time Nanoparticle Characterization Through Optofluidic Force Induction. Phys. Rev. Appl. 2022, 18, 024056. [Google Scholar] [CrossRef]
- Chakra, A.; Singh, N.; Vladisavljevic, G.T.; Nadal, F.; Cottin-Bizonne, C.; Pirat, C.; Bolognesi, G. Continuous Manipulation and Characterization of Colloidal Beads and Liposomes via Diffusiophoresis in Single- and Double-Junction Microchannels. Acs Nano 2023, 17, 14644–14657. [Google Scholar] [CrossRef]
- Fabozzi, A.; Della Sala, F.; di Gennaro, M.; Barretta, M.; Longobardo, G.; Solimando, N.; Pagliuca, M.; Borzacchiello, A. Design of functional nanoparticles by microfluidic platforms as advanced drug delivery systems for cancer therapy. Lab Chip 2023, 23, 1389–1409. [Google Scholar] [CrossRef] [PubMed]
- Koswig, T.; Geitner, R.; Anufriev, I.; Nischang, I.; Zechel, S.; Schubert, U.S.; Junkers, T. 3D-printed microfluidic dialysis apparatus for efficient polymer purification. Polym. Chem. 2025, 16, 4171–4179. [Google Scholar] [CrossRef]
- Carvell, T.; Burgoyne, P.; Fraser, A.R.; Bridle, H. Microfluidic technologies: Buffer exchange in bioprocessing, a mini review. Microfluid. Nanofluid 2024, 28, 79. [Google Scholar] [CrossRef]
- Seder, I.; Zheng, T.; Zhang, J.; Rojas, C.C.; Helalat, S.H.; Tellez, R.C.; Sun, Y. A Scalable Microfluidic Platform for Nanoparticle Formulation: For Exploratory- and Industrial-Level Scales. Nano Lett. 2024, 24, 5132–5138. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.; Jang, S.; Park, D.; Bae, N.H.; Han, C.S.; Ryu, S.; Lim, E.-K.; Lee, K.G. Automated Microfluidic Systems Facilitating the Scalable and Reliable Production of Lipid Nanoparticles for Gene Delivery. BioChip J. 2025, 19, 79–90. [Google Scholar] [CrossRef]
- Barenholz, Y. Doxil®--the first FDA-approved nano-drug: Lessons learned. J. Control. Release Off. J. Control. Release Soc. 2012, 160, 117–134. [Google Scholar] [CrossRef]
- Lee, C.-Y.; Tsai, T.; Peng, P.-C.; Chen, C.-T. Fabrication of Doxorubicin-Loaded Lipid-Based Nanocarriers by Microfluidic Rapid Mixing. Biomedicines 2022, 10, 1259. [Google Scholar] [CrossRef]
- Cheung, C.C.L.; Ma, G.; Ruiz, A.; Al-Jamal, W.T. Microfluidic Production of Lysolipid-Containing Temperature-Sensitive Liposomes. J. Vis. Exp. 2020, 157, e60907. [Google Scholar] [CrossRef]
- Buttitta, G.; Bonacorsi, S.; Barbarito, C.; Moliterno, M.; Pompei, S.; Saito, G.; Oddone, I.; Verdone, G.; Secci, D.; Raimondi, S. Scalable microfluidic method for tunable liposomal production by a design of experiment approach. Int. J. Pharm. 2024, 662, 124460. [Google Scholar] [CrossRef]
- Sun, Y.; Jin, F.; Gao, D. Microfluidic chip-based nano-carrier-based drug delivery systems and their efficacy assessment using tumor organ chips. BME Horiz. 2024, 2, 136. [Google Scholar] [CrossRef]
- Kang, S.; Park, S.E.; Huh, D.D. Organ-on-a-chip technology for nanoparticle research. Nano Converg. 2021, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Chai, D.; Wang, Q.; Wang, X.; Yong, Q.; Cheng, Z.; Zhang, C.; Zhang, D.; Liu, B.; Liu, R.; et al. Sensor-combined organ-on-a-chip for pharmaceutical and medical sciences: From design and materials to typical biomedical applications. Mater. Horiz. 2025, 12, 2161–2178. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Ai, J.; Ye, Z.; Chen, K.; Lin, J.; Zhang, Z.; Luo, M.; Zhou, B.; Xiang, S.; Zhou, J.; et al. Fully Automated Deep Learning Enabled Miniature Mass Spectrometry System for Psychoactive Therapeutic Drug Monitoring. Adv. Sci. 2025, 12, e02721. [Google Scholar] [CrossRef] [PubMed]
- Cognetti, J.S.; Miller, B.L. Real-Time, Continuous Monitoring of Tissue Chips as an Emerging Opportunity for Biosensing. Sensors 2025, 25, 5153. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Paul, S.; Biswas, S.; Jana, B.; Singh, N.D.P. Green-light responsive fluorescein-based photoremovable protecting group: Nanoparticle formulation for controlled release of bioactive molecules with real-time-monitoring ability. J. Mater. Chem. B 2025, 13, 7172–7180. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, R.; Zhu, Y.; de Barros, N.R. Microfluidics and Organ-on-a-Chip for Disease Modeling and Drug Screening. Biosensors 2024, 14, 86. [Google Scholar] [CrossRef]
- Khurram, M.M.; Cinel, G.; Yesil Celiktas, O.; Bedir, E. Organ-on-a-chip platforms for drug development, cellular toxicity assessment, and disease modeling. Turk. J. Biol. 2024, 48, 348–363. [Google Scholar] [CrossRef]
- Zhou, Y.; Jeong, M.; Zhang, M.; Duan, X.; Qiu, T. A Miniaturized Device for Ultrafast On-Demand Drug Release Based on a Gigahertz Ultrasonic Resonator. Adv. Eng. Mater. 2024, 26, 2302253. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Foo, G.W.; Aggarwal, N.; Chang, M.W. Organ-on-chip technology: Opportunities and challenges. Biotechnol. Notes 2024, 5, 8–12. [Google Scholar] [CrossRef]
- Li, Z.; Hui, J.; Yang, P.; Mao, H. Microfluidic Organ-on-a-Chip System for Disease Modeling and Drug Development. Biosensors 2022, 12, 370. [Google Scholar] [CrossRef]
- Jang, K.-J.; Otieno, M.A.; Ronxhi, J.; Lim, H.-K.; Ewart, L.; Kodella, K.R.; Petropolis, D.B.; Kulkarni, G.; Rubins, J.E.; Conegliano, D.; et al. Reproducing human and cross-species drug toxicities using a Liver-Chip. Sci. Transl. Med. 2019, 11, eaax5516. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, A.R.; Maia, R.F.; Ciardulli, M.C.; Santos, H.A.; Sarmento, B. Organ-on-chip platforms for nanoparticle toxicity and efficacy assessment: Advancing beyond traditional in vitro and in vivo models. Mater. Today Bio 2025, 33, 102053. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Xu, N.; Zhang, W.; Wang, P. Integrated microfluidic three-organ chip for real-time toxicity analysis of fluorotelomer alcohols in the gut-vascular-nerve axis. Lab Chip 2025, 25, 6170–6176. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Gwak, H.; Han, B. Advanced manufacturing of nanoparticle formulations of drugs and biologics using microfluidics. Analyst 2024, 149, 614–637. [Google Scholar] [CrossRef]
- Shaw, J.R.; Vaidya, R.; Xu, F.; Dharmaraj, S.; Pearson, R.M. Microfluidics-generated PLA nanoparticles: Impact of purification method on macrophage interactions, anti-inflammatory effects, biodistribution, and protein corona formation. Rsc Pharm. 2025, 2, 135–146. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, C.; Feng, Q.; Sun, J. Microfluidic synthesis of nanomaterials for biomedical applications. Nanoscale Horiz. 2023, 8, 1610–1627. [Google Scholar] [CrossRef]
- Leung, C.M.; de Haan, P.; Ronaldson-Bouchard, K.; Kim, G.-A.; Ko, J.; Rho, H.S.; Chen, Z.; Habibovic, P.; Jeon, N.L.; Takayama, S.; et al. A guide to the organ-on-a-chip. Nat. Rev. Methods Primers 2022, 2, 33. [Google Scholar] [CrossRef]
- Saliba, J.; Daou, A.; Damiati, S.; Saliba, J.; El-Sabban, M.; Mhanna, R. Development of Microplatforms to Mimic the In Vivo Architecture of CNS and PNS Physiology and Their Diseases. Genes 2018, 9, 285. [Google Scholar] [CrossRef]
- Mofazzal Jahromi, M.A.; Abdoli, A.; Rahmanian, M.; Bardania, H.; Bayandori, M.; Moosavi Basri, S.M.; Kalbasi, A.; Aref, A.R.; Karimi, M.; Hamblin, M.R. Microfluidic Brain-on-a-Chip: Perspectives for Mimicking Neural System Disorders. Mol. Neurobiol. 2019, 56, 8489–8512. [Google Scholar] [CrossRef]
- Gupte, B.; Jadhav, U.; Gosavi, S.; Jagtap, S. Lab on chip for medical and clinical applications. Sens. Diagn. 2025, 4, 939–965. [Google Scholar] [CrossRef]
- Venugopal Menon, N.; Lee, J.; Tang, T.; Lim, C.T. Microfluidics for morpholomics and spatial omics applications. Lab Chip 2025, 25, 752–763. [Google Scholar] [CrossRef] [PubMed]
- Ducree, J. Efficient Development of Integrated Lab-On-A-Chip Systems Featuring Operational Robustness and Manufacturability. Micromachines 2019, 10, 886. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-F.; Podder, S.; Chen, P.-Y. Microsystem Advances through Integration with Artificial Intelligence. Micromachines 2023, 14, 826. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.; Pandya, A. Chapter Twelve—Challenges and opportunities in micro/nanofluidic and lab-on-a-chip. In Progress in Molecular Biology and Translational Science; Pandya, A., Singh, V., Eds.; Academic Press: Cambridge, MA, USA, 2022; Volume 186, pp. 289–302. [Google Scholar]
- Park, J.; Kim, Y.W.; Jeon, H.-J. Machine Learning-Driven Innovations in Microfluidics. Biosensors 2024, 14, 613. [Google Scholar] [CrossRef] [PubMed]
- Bellotti, M.; Chiesa, E.; Conti, B.; Genta, I.; Conti, M.; Auricchio, F.; Caimi, A. Computational-Aided Approach for the Optimization of Microfluidic-Based Nanoparticles Manufacturing Process. Ann. Biomed. Eng. 2024, 52, 3240–3252. [Google Scholar] [CrossRef]
- FDA. Considerations for the Use of Artificial Intelligence to Support Regulatory Decision-Making for Drug and Biological Products; FDA: Silver Spring, MD, USA, 2025.
- EMA. Reflection Paper on the Use of Artificial Intelligence (AI) in the Medicinal Product Lifecycle; EMA: Amsterdam, The Netherlands, 2024.
- Wang, N.; Wei, Y.; Hu, Y.; Sun, X.; Wang, X. Microfluidic Preparation of pH-Responsive Microsphere Fibers and Their Controlled Drug Release Properties. Molecules 2024, 29, 193. [Google Scholar] [CrossRef]
- Morozov, V.N.; Groves, S.; Turell, M.J.; Bailey, C. Three minutes-long electrophoretically assisted zeptomolar microfluidic immunoassay with magnetic-beads detection. J. Am. Chem. Soc. 2007, 129, 12628–12629. [Google Scholar] [CrossRef]
- Kolahi Azar, H.; Sabouri, L.; Beheshtizadeh, N.; Maleki, R. pH-stimuli-responsive doxorubicin release and stability in chitosan-Eudragit nanocarriers. Sci. Rep. 2025, 15, 45428. [Google Scholar] [CrossRef]
- Raza, A.; Hayat, U.; Rasheed, T.; Bilal, M.; Iqbal, H.M.N. Redox-responsive nano-carriers as tumor-targeted drug delivery systems. Eur. J. Med. Chem. 2018, 157, 705–715. [Google Scholar] [CrossRef]
- Carugo, D.; Capretto, L.; Roy, B.; Carboni, M.; Caine, M.; Lewis, A.L.; Hill, M.; Chakraborty, S.; Zhang, X. Spatiotemporal dynamics of doxorubicin elution from embolic beads within a microfluidic network. J. Control. Release 2015, 214, 62–75. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, L.; Bi, M.; Wang, Y.; Liu, J.; Jiang, X.; Xu, Z.; Zhang, X. Artificial intelligence-powered microfluidics for nanomedicine and materials synthesis. Nanoscale 2021, 13, 19352–19366. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Qiu, Y.; Chen, C.Y.; Carton, M.; Campbell, P.M.R.; Chowdhury, A.M.; Bandyopadhyay, B.C.; Bentley, W.E.; Smith, B.R.; Sochol, R.D. 3D nanoprinting of PDMS microvessels with tailored tortuosity and microporosity via direct laser writing. Lab Chip 2025, 25, 1947–1958. [Google Scholar] [CrossRef] [PubMed]
- Young, O.M.; Xu, X.; Sarker, S.; Sochol, R.D. Direct laser writing-enabled 3D printing strategies for microfluidic applications. Lab Chip 2024, 24, 2371–2396. [Google Scholar] [CrossRef] [PubMed]
- Oldach, B.; Fortmann, R.; Pleie, T.; Timm, P.; Kockmann, N. Design and Rapid Prototyping of 3D-Printed Microfluidic Systems for Multiphase Flow. Chemistry 2024, 6, 1458–1476. [Google Scholar] [CrossRef]
- Li, X.; Wan, S.; Pronay, T.S.; Yang, X.; Gao, B.; Lim, C.T. Toward next-generation smart medical care wearables: Where microfluidics meet microneedles. Nanoscale Horiz. 2025, 10, 1815–1837. [Google Scholar] [CrossRef]
- Shepherd, S.J.; Issadore, D.; Mitchell, M.J. Microfluidic formulation of nanoparticles for biomedical applications. Biomaterials 2021, 274, 120826. [Google Scholar] [CrossRef]
- Agrahari, V.; Agrahari, V. Facilitating the translation of nanomedicines to a clinical product: Challenges and opportunities. Drug Discov. Today 2018, 23, 974–991. [Google Scholar] [CrossRef]
- Mangla, B.; Kumar, P.; Javed, S.; Pathan, T.; Ahsan, W.; Aggarwal, G. Regulating nanomedicines: Challenges, opportunities, and the path forward. Nanomedicine 2025, 20, 1911–1927. [Google Scholar] [CrossRef]
- Foulkes, R.; Man, E.; Thind, J.; Yeung, S.; Joy, A.; Hoskins, C. The regulation of nanomaterials and nanomedicines for clinical application: Current and future perspectives. Biomater. Sci. 2020, 8, 4653–4664. [Google Scholar] [CrossRef]



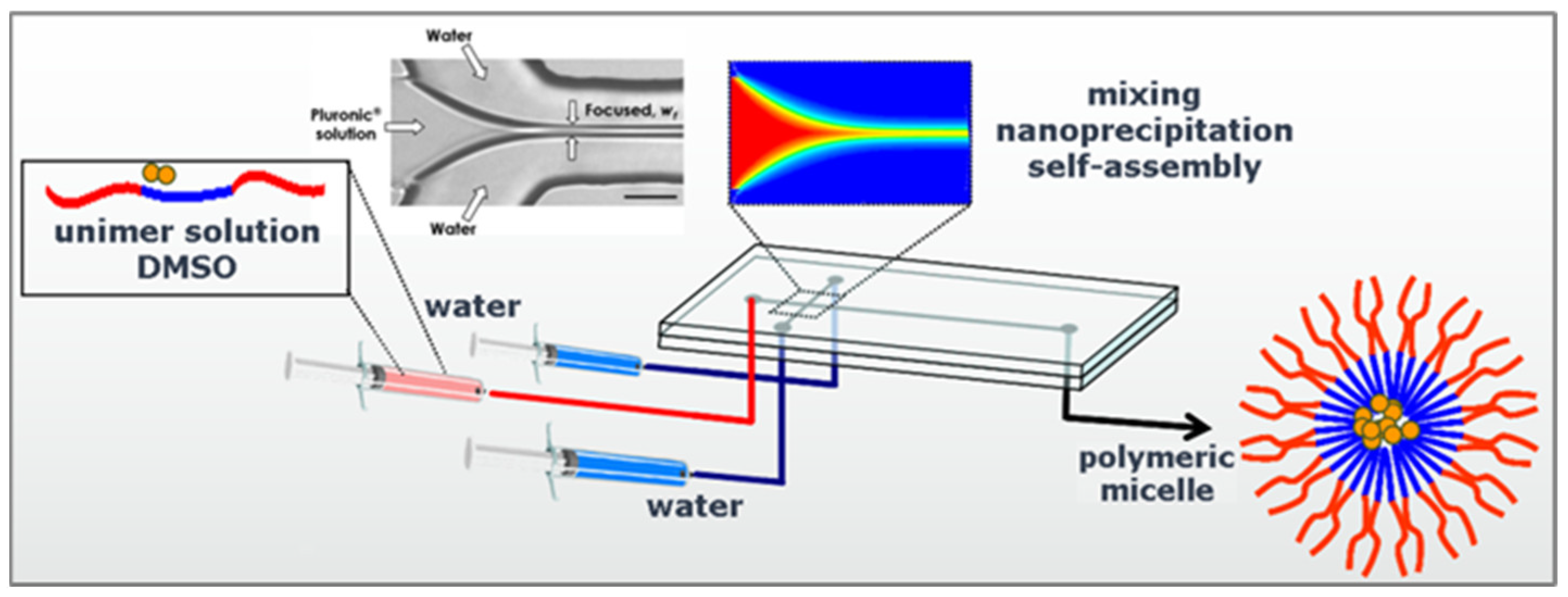
| Performance Metric | Conventional Batch (e.g., Stirred Tank) | LoC/Microfluidic Systems | Significance in DDS Application |
|---|---|---|---|
| Particle Size (nm) | 10–300 nm (high variance) | 20–120 nm (highly tunable) | Smaller and more uniform nanoparticles enhance cellular uptake. [64,65] |
| Polydispersity Index (PDI) | 0.2–0.5 (broad distribution) | <0.1–0.2 (monodisperse) | Predictable PK/PD profiles. [66,67] |
| Mixing Time (τmix) | Seconds–minutes | Milliseconds | Rapid mixing prevents uncontrolled particle growth. [68] |
| Flow Rate (mL/min) | N/A (static/bulk) | 0.01–10 mL/min | High precision over residence time. [69] |
| Shear Stress | Non-uniform | Controlled/predictable | Regulates impacts of shear stress on biologics. [70] |
| Encapsulation Efficiency (%) | 40–70% | 80–95% | Improved drug loading and reduced waste of expensive APIs. [71] |
| Batch-to-Batch Variability | High (operator-dependent) | Minimal (digital/automated) | Essential for clinical/regulatory approval. [64] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Guo, B.; Zhao, Y.; Zhang, X. Lab-on-a-Chip and Microfluidics Technologies for Nano Drug Delivery. Bioengineering 2026, 13, 363. https://doi.org/10.3390/bioengineering13030363
Guo B, Zhao Y, Zhang X. Lab-on-a-Chip and Microfluidics Technologies for Nano Drug Delivery. Bioengineering. 2026; 13(3):363. https://doi.org/10.3390/bioengineering13030363
Chicago/Turabian StyleGuo, Bochun, Yuchao Zhao, and Xunli Zhang. 2026. "Lab-on-a-Chip and Microfluidics Technologies for Nano Drug Delivery" Bioengineering 13, no. 3: 363. https://doi.org/10.3390/bioengineering13030363
APA StyleGuo, B., Zhao, Y., & Zhang, X. (2026). Lab-on-a-Chip and Microfluidics Technologies for Nano Drug Delivery. Bioengineering, 13(3), 363. https://doi.org/10.3390/bioengineering13030363







