AI-Based Myocardial Segmentation and Attenuation Mapping Improved Detection of Myocardial Ischemia and Infarction on Emergency CT Angiography
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. CT Examinations
2.3. Radiology Reports
2.4. Invasive Coronary Angiography
2.5. Myocardial Segmentation and Color Mapping
2.6. MRMC Study Design
2.7. Reading
2.8. Statistical Analysis
3. Results
3.1. Reports: Myocardial Infarction
3.2. Reading: Myocardial Infarction Detection
3.3. Sub-Classification of STEMI vs. NSTEMI and Vessel Territory
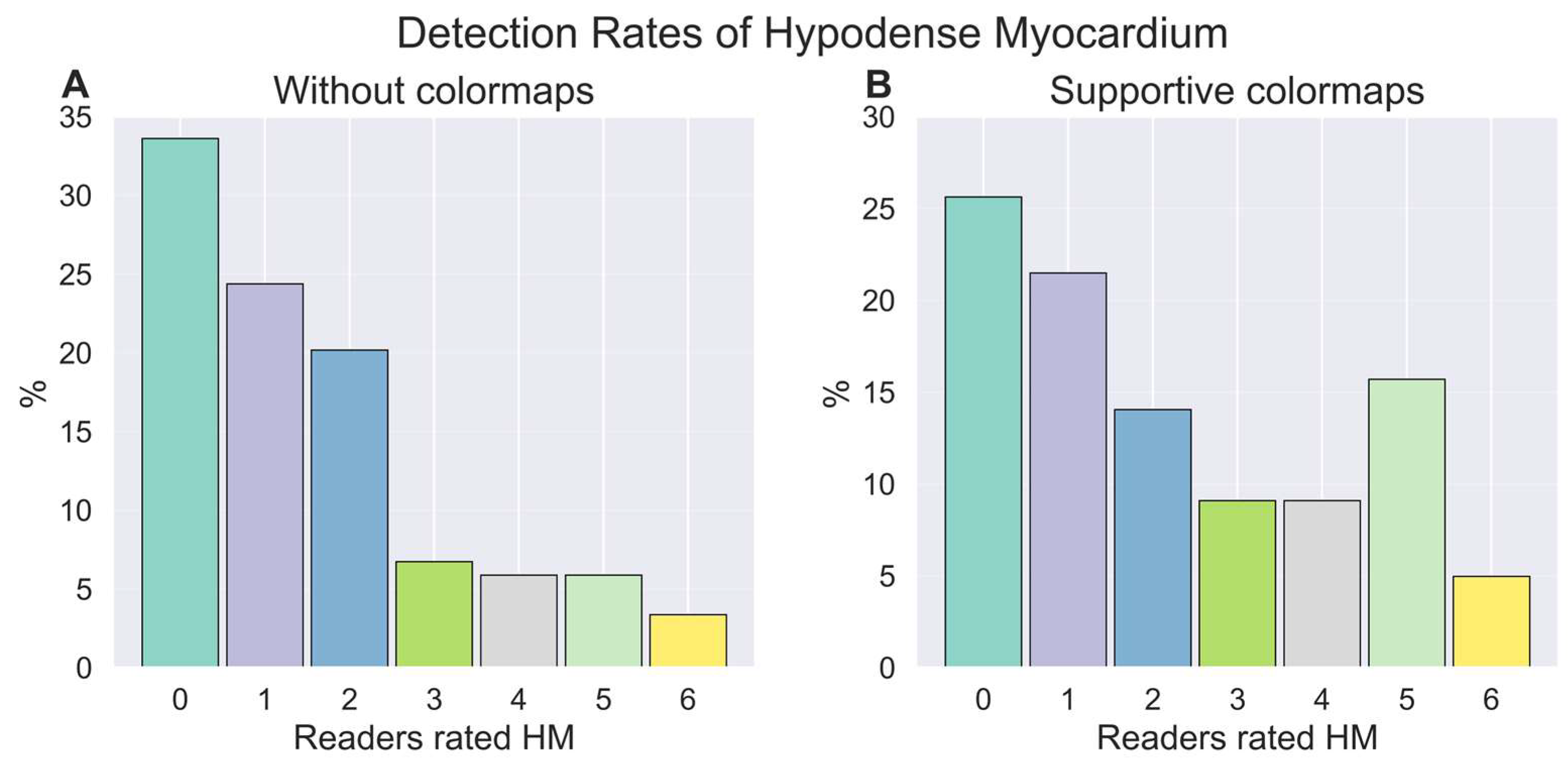
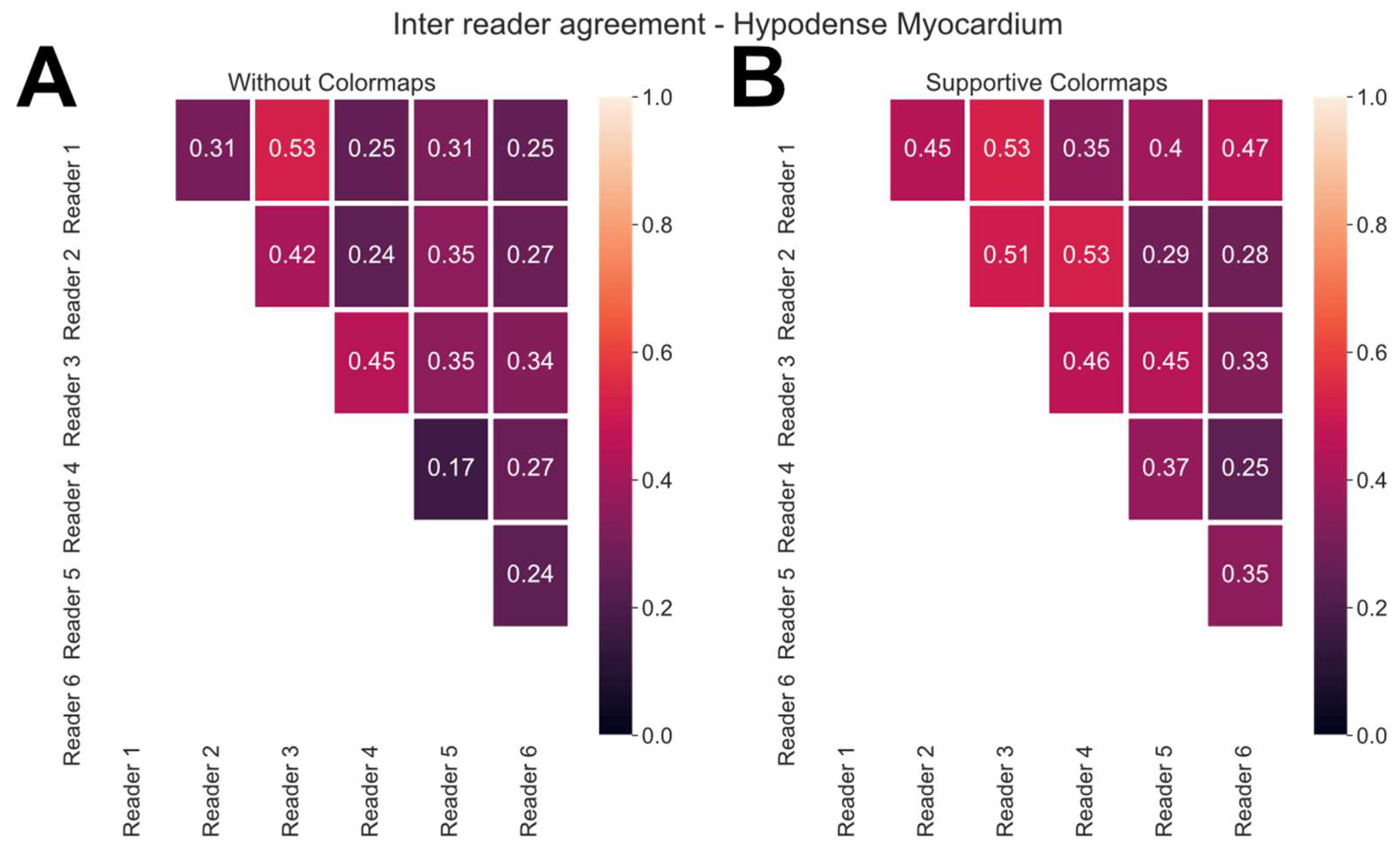
3.4. Inter-Reader Agreement
3.5. Time
4. Discussion
- First, in only a fraction of the peer-reviewed radiology reports written in clinical routine (7/119), the findings were clearly interpreted as a myocardial ischemia/infarction.
- Second, the use of supportive colormaps led to an improved detection of myocardial hypodensities (11% of the study population were correctly reclassified) compared to a scenario without colormaps, with a significant increase in sensitivity for three readers.
- Third, the use of additional colormaps allowed the readers to increase their sensitivity in cases with STEMI by 15% (49% vs. 34%) and in cases with NSTEMIs by 11% (44% vs. 33%) compared to a reading scenario without colormaps.
- Fourth, particularly senior residents, who are more frequently exposed to the emergency setting, benefited most from the colormaps, with significant increases in sensitivity for both readers.
- Fifth, the use of colormaps allowed less experienced readers to achieve a higher agreement with expert readers concerning the detection of a hypodense myocardium.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACS | acute coronary syndrome |
| AAS | acute aortic syndrome |
| ED | emergency department |
| PE | pulmonary embolism |
| TRO | triple-rule out |
| ICA | invasive coronary angiography |
| STEMI | ST-elevation myocardial infarction |
| NSTEMI | non-ST-elevation myocardial infarction |
References
- Bhuiya, F.A.; Pitts, S.R.; McCaig, L.F. Emergency Department Visits for Chest Pain and Abdominal Pain: United States, 1999–2008; US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics: Hyattsville, MD, USA, 2010; pp. 1–8.
- Chang, A.M.; Fischman, D.L.; Hollander, J.E. Evaluation of Chest Pain and Acute Coronary Syndromes. Cardiol. Clin. 2018, 36, 1–12. [Google Scholar] [CrossRef]
- Gulati, M.; Levy, P.D.; Mukherjee, D.; Amsterdam, E.; Bhatt, D.L.; Birtcher, K.K.; Blankstein, R.; Boyd, J.; Bullock-Palmer, R.P.; Conejo, T.; et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation and Diagnosis of Chest Pain: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2021, 144, e368–e454. [Google Scholar] [CrossRef]
- Halpern, E.J. Triple-Rule-Out CT Angiography for Evaluation of Acute Chest Pain and Possible Acute Coronary Syndrome. Radiology 2009, 252, 332–345. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; de Ferranti, S.; Després, J.-P.; Fullerton, H.J.; Howard, V.J.; et al. Heart Disease and Stroke Statistics—2015 Update. Circulation 2015, 131, e29–e322. [Google Scholar] [CrossRef]
- Pope, J.H.; Selker, H.P. Acute Coronary Syndromes in the Emergency Department: Diagnostic Characteristics, Tests, and Challenges. Cardiol. Clin. 2005, 23, 423–451. [Google Scholar] [CrossRef]
- Rusnak, R.A.; Stair, T.O.; Hansen, K.; Fastow, J.S. Litigation against the emergency physician: Common features in cases of missed myocardial infarction. Ann. Emerg. Med. 1989, 18, 1029–1034. [Google Scholar] [CrossRef]
- Canto, J.G.; Rogers, W.J.; Goldberg, R.J.; Peterson, E.D.; Wenger, N.K.; Vaccarino, V.; Kiefe, C.I.; Frederick, P.D.; Sopko, G.; Zheng, Z.-J.; et al. Association of Age and Sex With Myocardial Infarction Symptom Presentation and In-Hospital Mortality. JAMA 2012, 307, 813–822. [Google Scholar] [CrossRef]
- Ayaram, D.; Bellolio, M.F.; Murad, M.H.; Laack, T.A.; Sadosty, A.T.; Erwin, P.J.; Hollander, J.E.; Montori, V.M.; Stiell, I.G.; Hess, E.P. Triple Rule-out Computed Tomographic Angiography for Chest Pain: A Diagnostic Systematic Review and Meta-Analysis. Acad. Emerg. Med. 2013, 20, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Chae, M.K.; Kim, E.K.; Jung, K.-Y.; Shin, T.G.; Sim, M.S.; Jo, I.-J.; Song, K.J.; Chang, S.-A.; Bin Song, Y.; Hahn, J.-Y.; et al. Triple rule-out computed tomography for risk stratification of patients with acute chest pain. J. Cardiovasc. Comput. Tomogr. 2016, 10, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.E.; Wann, S. Evaluation of Acute Chest Pain in the Emergency Department: “Triple Rule-Out” Computed Tomography Angiography. Cardiol. Rev. 2011, 19, 115–121. [Google Scholar] [CrossRef]
- Higashigaito, K.; Hinzpeter, R.; Baumueller, S.; Benz, D.; Manka, R.; Keller, D.I.; Alkadhi, H.; Morsbach, F. Chest pain CT in the emergency department: Watch out for the myocardium. Eur. J. Radiol. Open 2018, 5, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Blankstein, R.; Rogers, I.S.; Cury, R.C. Practical tips and tricks in cardiovascular computed tomography: Diagnosis of myocardial infarction. J. Cardiovasc. Comput. Tomogr. 2009, 3, 104–111. [Google Scholar] [CrossRef]
- Varga-Szemes, A.; Meinel, F.G.; De Cecco, C.N.; Fuller, S.R.; Bayer, R.R.; Schoepf, U.J. CT Myocardial Perfusion Imaging. Am. J. Roentgenol. 2015, 204, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Austen, W.; Edwards, J.; Frye, R.; Gensini, G.; Gott, V.; Griffith, L.; McGoon, D.; Murphy, M.; Roe, B. A reporting system on patients evaluated for coronary artery disease. Report of the Ad Hoc Committee for Grading of Coronary Artery Disease, Council on Cardiovascular Surgery, American Heart Association. Circulation 1975, 51, 5–40. [Google Scholar] [CrossRef] [PubMed]
- MM-WHS: Multi-Modality Whole Heart Segmentation. Available online: https://zmiclab.github.io/zxh/0/mmwhs/ (accessed on 1 January 2021).
- Obuchowski, N.A.; Bullen, J. Multireader Diagnostic Accuracy Imaging Studies: Fundamentals of Design and Analysis. Radiology 2022, 303, 26–34. [Google Scholar] [CrossRef]
- Anastasopoulos, C.; Reisert, M.; Kellner, E. “Nora Imaging”: A Web-Based Platform for Medical Imaging. Neuropediatrics 2017, 48, S1–S45. [Google Scholar] [CrossRef]
- Jewell, E.S.; Maile, M.D.; Engoren, M.; Elliott, M. Net Reclassification Improvement. Anesth. Analg. 2016, 122, 818–824. [Google Scholar] [CrossRef]
- Leening, M.J.G.; Vedder, M.M.; Witteman, J.C.M.; Pencina, M.J.; Steyerberg, E.W. Net Reclassification Improvement: Computation, Interpretation, and Controversies. Ann. Intern. Med. 2014, 160, 122–131. [Google Scholar] [CrossRef]
- Adams, D.F.; Hessel, S.J.; Judy, P.F.; Stein, J.A.; Abrams, H.L. Computed tomography of the normal and infarcted myocardium. Am. J. Roentgenol. 1976, 126, 786–791. [Google Scholar] [CrossRef]
- Bischoff, B.; Deseive, S.; Rampp, M.; Todica, A.; Wermke, M.; Martinoff, S.; Massberg, S.; Reiser, M.F.; Becker, H.-C.; Hausleiter, J. Myocardial ischemia detection with single-phase CT perfusion in symptomatic patients using high-pitch helical image acquisition technique. Int. J. Cardiovasc. Imaging 2017, 33, 569–576. [Google Scholar] [CrossRef]
- Nieman, K.; Balla, S. Dynamic CT myocardial perfusion imaging. J. Cardiovasc. Comput. Tomogr. 2020, 14, 303–306. [Google Scholar] [CrossRef]
- Kim, K.W.; Lee, J.M.; Klotz, E.; Park, H.S.; Lee, D.H.; Kim, J.Y.; Kim, S.J.; Kim, S.H.; Lee, J.Y.; Han, J.K.; et al. Quantitative CT Color Mapping of the Arterial Enhancement Fraction of the Liver to Detect Hepatocellular Carcinoma. Radiology 2009, 250, 425–434. [Google Scholar] [CrossRef]
- Bedekar, D.; Jensen, T.; Schmainda, K.M. Standardization of relative cerebral blood volume (rCBV) image maps for ease of both inter- and intrapatient comparisons. Magn. Reson. Med. 2010, 64, 907–913. [Google Scholar] [CrossRef]
- Takakuwa, K.M.; Halpern, E.J. Evaluation of a “Triple Rule-Out” Coronary CT Angiography Protocol: Use of 64-Section CT in Low-to-Moderate Risk Emergency Department Patients Suspected of Having Acute Coronary Syndrome. Radiology 2008, 248, 438–446. [Google Scholar] [CrossRef]
- Budoff, M.J.; Dowe, D.; Jollis, J.G.; Gitter, M.; Sutherland, J.; Halamert, E.; Scherer, M.; Bellinger, R.; Martin, A.; Benton, R.; et al. Diagnostic Performance of 64-Multidetector Row Coronary Computed Tomographic Angiography for Evaluation of Coronary Artery Stenosis in Individuals Without Known Coronary Artery Disease: Results From the Prospective Multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of Individuals Undergoing Invasive Coronary Angiography) Trial. J. Am. Coll. Cardiol. 2008, 52, 1724–1732. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.; Rideau, A.M.; Paulson, E.K.; Bisset, G.S. Emergency Department Imaging: Current Practice. J. Am. Coll. Radiol. 2008, 5, 811–816.e2. [Google Scholar] [CrossRef]
- Russo, V.; Sportoletti, C.; Scalas, G.; Attinà, D.; Buia, F.; Niro, F.; Modolon, C.; De Luca, C.; Monteduro, F.; Lovato, L. The triple rule out CT in acute chest pain: A challenge for emergency radiologists? Emerg. Radiol. 2021, 28, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Jung, J.I.; Choi, Y.-S.; Ann, S.H.; Youn, H.-J.; Jeon, G.N.; Choi, H.C. Coronary CT angiography in patients with high calcium score: Evaluation of plaque characteristics and diagnostic accuracy. Int. J. Cardiovasc. Imaging 2011, 27, 43–51. [Google Scholar] [CrossRef]
- Mahnken, A.H.; Lautenschläger, S.; Fritz, D.; Koos, R.; Scheuering, M. Perfusion weighted color maps for enhanced visualization of myocardial infarction by MSCT: Preliminary experience. Int. J. Cardiovasc. Imaging 2008, 24, 883–890. [Google Scholar] [CrossRef] [PubMed]



| Experience Level | Reader | Sensitivity | Specificity | Accuracy | Positive Predictive Value | Negative Predictive Value | McNemar-Test | Net Reclassification Index |
|---|---|---|---|---|---|---|---|---|
| Junior resident | Reader 1 wc | 46.3% | 92.3% | 66.4% | 88.6% | 57.1% | Sensitivity: p = 1 Specificity: p = 0.22 | −6% |
| Reader 1 sc | 46.4% | 84.6% | 62.8% | 80.0% | 54.3% | |||
| Junior resident | Reader 2 wc | 55.2% | 55.8% | 55.5% | 61.7% | 49.2% | Sensitivity: p = 0.30 Specificity: p = 1 | 8% |
| Reader 2 sc | 62.7% | 55.8% | 59.7% | 64.6% | 53.7% | |||
| Experienced resident | Reader 3 wc | 28.4% | 94.2% | 57.1% | 86.4% | 50.5% | Sensitivity: p = 0.006 Specificity: p = 0.016 | 7% |
| Reader 3 sc | 43.3% | 80.8% | 59.7% | 74.4% | 52.5% | |||
| Experienced resident | Reader 4 wc | 29.9% | 76.9% | 50.4% | 62.5% | 46.0% | Sensitivity: p = 0.008 Specificity: p = 0.12 | 21% |
| Reader 4 sc | 52.2% | 63.5% | 57.1% | 64.8% | 50.8% | |||
| Expert | Reader 5 wc | 13.4% | 92.3% | 47.9% | 69.2% | 45.3% | Sensitivity: p < 0.0001 Specificity: p = 0.022 | 35% |
| Reader 5 sc | 44.9% | 75.0% | 57.8% | 70.5% | 50.6% | |||
| Expert | Reader 6 wc | 28.4% | 86.5% | 53.8% | 73.1% | 48.4% | Sensitivity: p = 0.80 Specificity: p = 0.69 | 1% |
| Reader 6 sc | 26.1% | 90.4% | 53.7% | 78.3% | 48.0% | |||
| Average | Average wc | 33.6% | 83.0% | 55.1% | 73.5% | 49.4% | 11% | |
| Average sc | 45.9% | 75.0% | 58.4% | 72.1% | 51.65% |
| Case | CT | Total, Split | Total | ||
|---|---|---|---|---|---|
| Non Case | Abnormal | Normal | |||
| CT Colormaps | Abnormal | 13 | 22 | 35 | 71 |
| 25 | 11 | 36 | |||
| Normal | 7 | 8 | 15 | 48 | |
| 4 | 29 | 33 | |||
| Total, split | 20 | 30 | |||
| 29 | 40 | ||||
| Total | 49 | 70 | |||
| NRI | 0.21 | ||||
| Sensitivity | Positive Predictive Value | ||
|---|---|---|---|
| STEMI | Reader 1 | 63.6% (50.0%) | 35.0% (31.4%) |
| Reader 2 | 63.6% (54.5%) | 21.5% (20.0%) | |
| Reader 3 | 50% (31.8%) | 28.2% (31.8%) | |
| Reader 4 | 59.1% (31.8%) | 24.1% (21.9%) | |
| Reader 5 | 36.4% (13.6%) | 18.2% (18.2%) | |
| Reader 6 | 22.7% (22.7%) | 21.7% (19.2%) | |
| Average | 49.2% (34.1%) | 24.8% (23.8%) | |
| NSTEMI | Reader 1 | 38.3% (44.4%) | 45.0% (57.1%) |
| Reader 2 | 62.2% (55.6%) | 43.1% (41.7%) | |
| Reader 3 | 40.0% (26.7%) | 46.2% (54.5%) | |
| Reader 4 | 48.9% (28.9%) | 40.7% (40.6%) | |
| Reader 5 | 48.9% (13.3%) | 52.3% (46.2%) | |
| Reader 6 | 27.7% (31.1%) | 56.5% (53.8%) | |
| Average | 44.3% (33.3%) | 47.3% (49%) | |
| Sensitivity | Specificity | Accuracy | Positive Predictive Value | Negative Predictive Value | ||
|---|---|---|---|---|---|---|
| LAD | Average | 35.0% (34.2%) | 73.9% (77.5%) | 54.3% (56.0%) | 63.2% (62.4%) | 52.8% (54.7%) |
| CX | Average | 32.5% (22.6%) | 77.2% (90.2%) | 62.4% (68.1%) | 49.6% (61.7%) | 69.7% (70.6%) |
| RCA | Average | 11.1% (9.1%) | 95.4% (94.4%) | 66.1% (64.3%) | 63.7% (38.8%) | 66.9% (65.6%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Segeroth, M.; Vosshenrich, J.; Breit, H.-C.; Krebs-Fleischmann, H.W.A.; Abel, L.; Obmann, M.; Yang, S.; Cyriac, J.; Wasserthal, J.; Indrakanti, A.K.; et al. AI-Based Myocardial Segmentation and Attenuation Mapping Improved Detection of Myocardial Ischemia and Infarction on Emergency CT Angiography. Bioengineering 2026, 13, 355. https://doi.org/10.3390/bioengineering13030355
Segeroth M, Vosshenrich J, Breit H-C, Krebs-Fleischmann HWA, Abel L, Obmann M, Yang S, Cyriac J, Wasserthal J, Indrakanti AK, et al. AI-Based Myocardial Segmentation and Attenuation Mapping Improved Detection of Myocardial Ischemia and Infarction on Emergency CT Angiography. Bioengineering. 2026; 13(3):355. https://doi.org/10.3390/bioengineering13030355
Chicago/Turabian StyleSegeroth, Martin, Jan Vosshenrich, Hanns-Christian Breit, Helge Walter Anand Krebs-Fleischmann, Lorraine Abel, Markus Obmann, Shan Yang, Joshy Cyriac, Jakob Wasserthal, Ashraya Kumar Indrakanti, and et al. 2026. "AI-Based Myocardial Segmentation and Attenuation Mapping Improved Detection of Myocardial Ischemia and Infarction on Emergency CT Angiography" Bioengineering 13, no. 3: 355. https://doi.org/10.3390/bioengineering13030355
APA StyleSegeroth, M., Vosshenrich, J., Breit, H.-C., Krebs-Fleischmann, H. W. A., Abel, L., Obmann, M., Yang, S., Cyriac, J., Wasserthal, J., Indrakanti, A. K., Bach, M., Zellweger, M. J., Sauter, A., Bremerich, J., Haaf, P., & Winkel, D. J. (2026). AI-Based Myocardial Segmentation and Attenuation Mapping Improved Detection of Myocardial Ischemia and Infarction on Emergency CT Angiography. Bioengineering, 13(3), 355. https://doi.org/10.3390/bioengineering13030355






