A PHMB-Functionalized Fully Absorbable Synthetic Matrix as a Novel Alternative to Biologics: Balancing Antibacterial Efficacy, Tissue Repair, and Safety
Highlights
- •
- A fully absorbable PHMB-functionalized synthetic matrix mimicking extracellular matrix (ECM) architecture enables synchronized biphasic release and degradation, ensuring sustained antibacterial efficacy throughout wound healing while facilitating tissue repair.
- •
- AATCC 100 assessments confirmed that the matrix maintains robust broad-spectrum bactericidal potency (log10 reduction values (LRV) > 4.0; >99.99% reduction) against six clinically relevant pathogens after 15 months of real-time aging, demonstrating superior stability for clinical translation.
- •
- The PHMB Matrix demonstrates significantly accelerated healing compared to Xenograft AM in porcine full-thickness wound models by Day 22, with improved wound surface quality and histologically confirmed tissue remodeling and biocompatibility.
- •
- The PHMB Matrix demonstrates sustained wound-site protection via a localized “enrichment effect” while maintaining a robust systemic safety profile through rapid, biphasic elimination.
Abstract
1. Introduction
2. Experimental Section
2.1. Preparation, Characterization, and In Vitro Performance of the PHMB Matrix
2.1.1. Preparation and Characterization of the PHMB Matrix
2.1.2. In Vitro Release Kinetics of PHMB
2.1.3. Quantitative Antibacterial Efficacy and Stability
2.1.4. In Vitro Biodegradation and pH Monitoring
2.2. Safety and Effectiveness Evaluation of PHMB Matrix in Porcine Full-Thickness Defects
2.3. Pharmacokinetic (PK) Study in SD Rats
2.4. Statistical Analysis
2.5. Quality Assurance
3. Results
3.1. Characterization of the PHMB Matrix
3.1.1. Structural, Morphological and Mechanical Characterization
3.1.2. In Vitro Drug Release Kinetics
3.1.3. Broad-Spectrum Antibacterial Efficacy and Stability
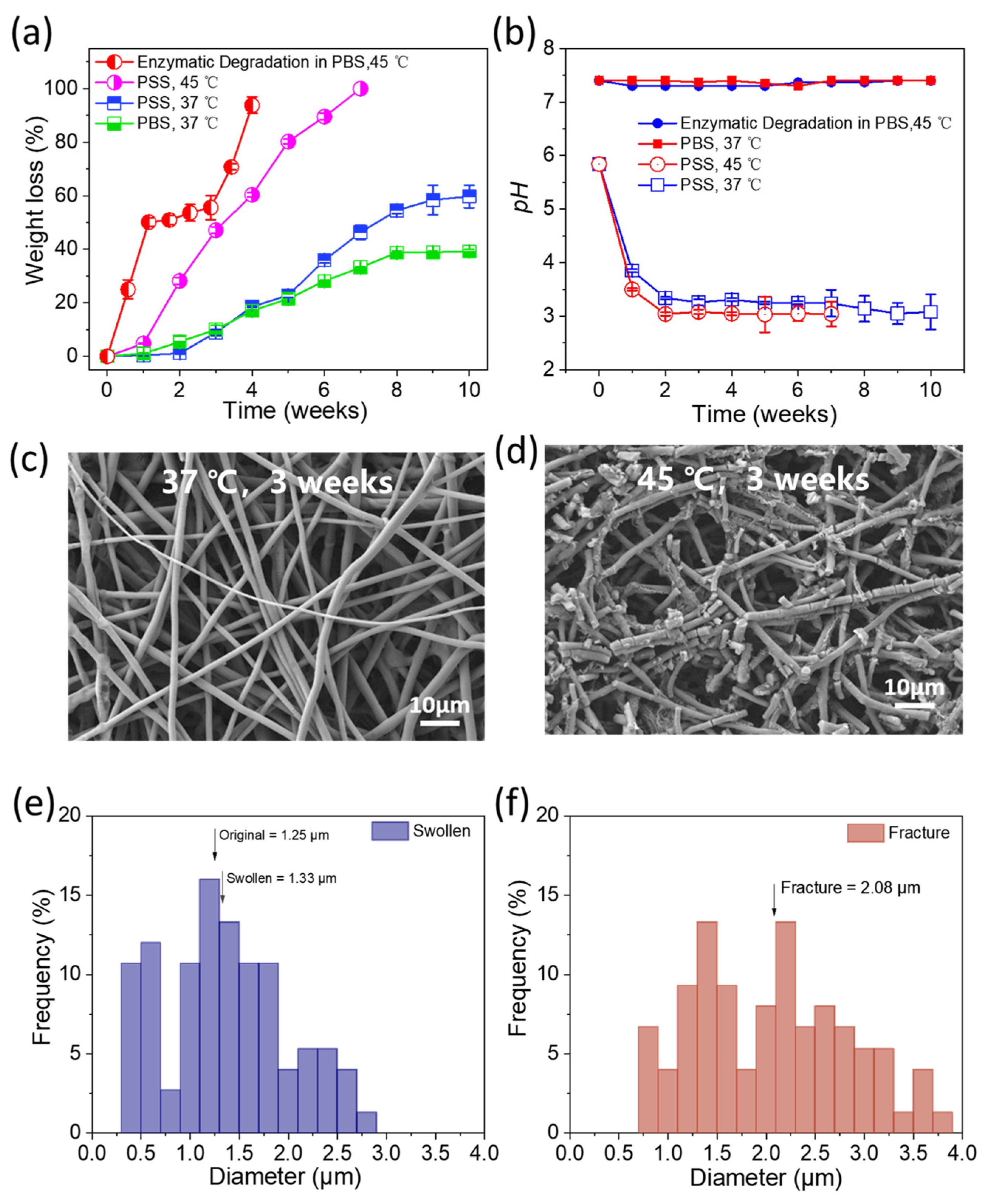
3.1.4. In Vitro Degradation and Microenvironmental Stability
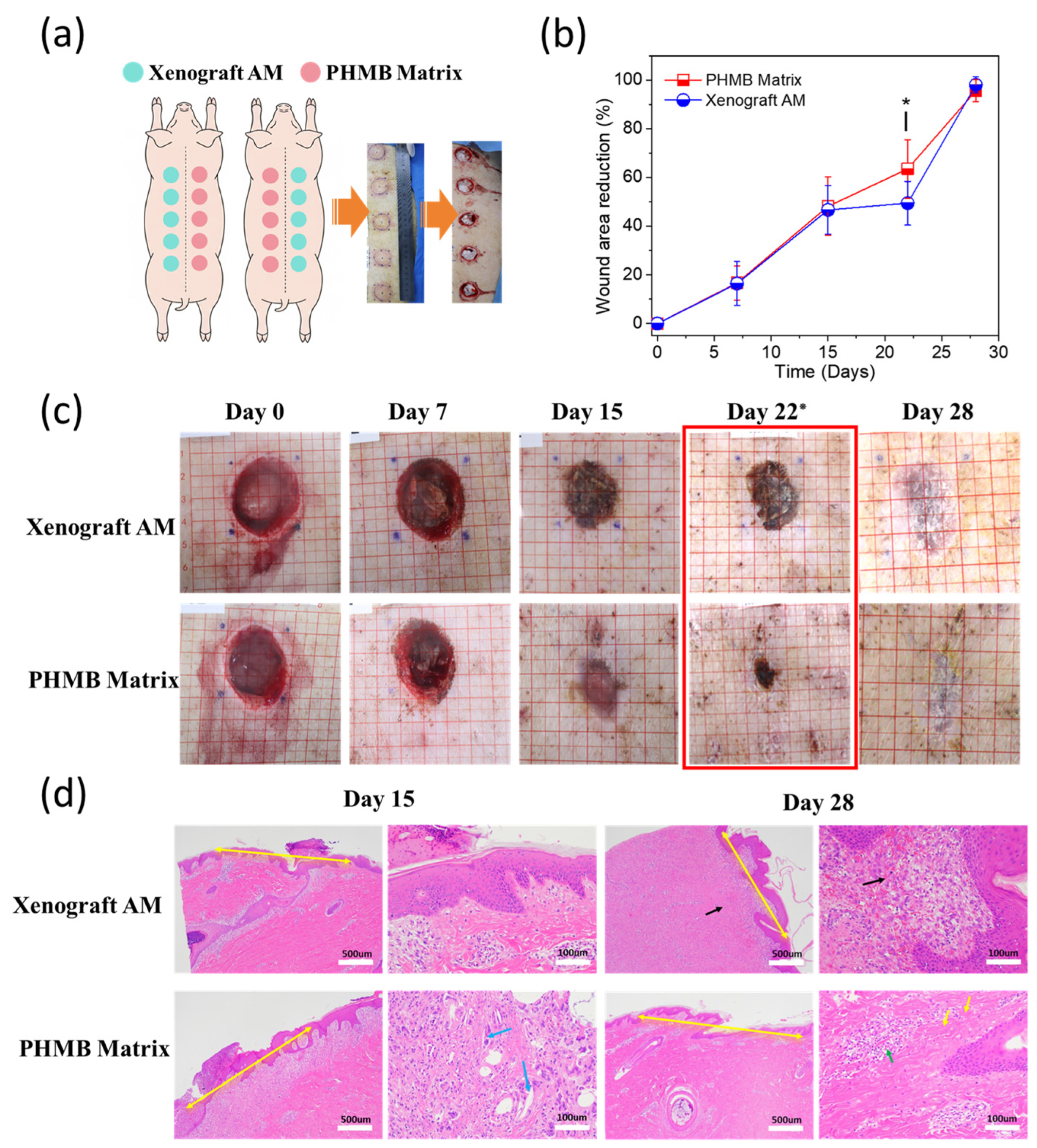
3.2. Efficacy and Biocompatibility in Porcine Wound Models
3.3. Systemic Pharmacokinetics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hubner, N.O.; Kramer, A. Review on the efficacy, safety and clinical applications of polihexanide, a modern wound antiseptic. Skin Pharmacol. Physiol. 2010, 23, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Bjarnsholt, T.; Kirketerp-Moller, K.; Jensen, P.O.; Madsen, K.G.; Phipps, R.; Krogfelt, K.; Hoiby, N.; Givskov, M. Why chronic wounds will not heal: A novel hypothesis. Wound Repair Regen. 2008, 16, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Gordillo, G.M.; Roy, S.; Kirsner, R.; Lambert, L.; Hunt, T.K.; Gottrup, F.; Gurtner, G.C.; Longaker, M.T. Human skin wounds: A major and snowballing threat to public health and the economy. Wound Repair Regen. 2009, 17, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V.; Isseroff, R.R.; Soulika, A.M.; Romanelli, M.; Margolis, D.; Kapp, S.; Granick, M.; Harding, K. Chronic wounds. Nat. Rev. Dis. Primers 2022, 8, 50. [Google Scholar] [CrossRef]
- Brett, D. A Review of Collagen and Collagen-based Wound Dressings. Wounds 2008, 20, 347–356. [Google Scholar]
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials 2010, 3, 1863–1887. [Google Scholar] [CrossRef]
- Zilberman, M.; Elsner, J.J. Antibiotic-eluting medical devices for various applications. J. Control Release 2008, 130, 202–215. [Google Scholar] [CrossRef]
- Gou, Y.; Hu, L.; Liao, X.; He, J.; Liu, F. Advances of antimicrobial dressings loaded with antimicrobial agents in infected wounds. Front. Bioeng. Biotechnol. 2024, 12, 1431949. [Google Scholar] [CrossRef]
- Gul, A.; Gallus, I.; Tegginamath, A.; Maryska, J.; Yalcinkaya, F. Electrospun Antibacterial Nanomaterials for Wound Dressings Applications. Membranes 2021, 11, 908. [Google Scholar] [CrossRef]
- Katti, D.S.; Robinson, K.W.; Ko, F.K.; Laurencin, C.T. Bioresorbable nanofiber-based systems for wound healing and drug delivery: Optimization of fabrication parameters. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 70, 286–296. [Google Scholar] [CrossRef]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.; Eccleston, G.M. Wound healing dressings and drug delivery systems: A review. J. Pharm. Sci. 2008, 97, 2892–2923. [Google Scholar] [CrossRef]
- Zeng, J.; Xu, X.; Chen, X.; Liang, Q.; Bian, X.; Yang, L.; Jing, X. Biodegradable electrospun fibers for drug delivery. J. Control Release 2003, 92, 227–231. [Google Scholar] [CrossRef]
- Makadia, H.K.; Siegel, S.J. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.S.; Catanzano, O. Advanced Therapeutic Dressings for Effective Wound Healing—A Review. J. Pharm. Sci. 2015, 104, 3653–3680. [Google Scholar] [CrossRef] [PubMed]
- Kenry; Lim, C.T. Nanofiber technology: Current status and emerging developments. Prog. Polym. Sci. 2017, 70, 1–17. [Google Scholar] [CrossRef]
- Kou, J.; Li, Y.; Zhou, C.; Wang, X.; Ni, J.; Lin, Y.; Ge, H.; Zheng, D.; Chen, G.; Sun, X.; et al. Electrospinning in promoting chronic wound healing: Materials, process, and applications. Front. Bioeng. Biotechnol. 2025, 13, 1550553. [Google Scholar] [CrossRef]
- Broxton, P.; Woodcock, P.M.; Heatley, F.; Gilbert, P. Interaction of some polyhexamethylene biguanides and membrane phospholipids in Escherichia coli. J. Appl. Bacteriol. 1984, 57, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Messick, C.R.; Pendland, S.L.; Moshirfar, M.; Fiscella, R.G.; Losnedahl, K.J.; Schriever, C.A.; Schreckenberger, P.C. In-vitro activity of polyhexamethylene biguanide (PHMB) against fungal isolates associated with infective keratitis. J. Antimicrob. Chemother. 1999, 44, 297–298. [Google Scholar] [CrossRef]
- Allen, M.J.; Morby, A.P.; White, G.F. Cooperativity in the binding of the cationic biocide polyhexamethylene biguanide to nucleic acids. Biochem. Biophys. Res. Commun. 2004, 318, 397–404. [Google Scholar] [CrossRef]
- Chindera, K.; Mahato, M.; Sharma, A.K.; Horsley, H.; Kloc-Muniak, K.; Kamaruzzaman, N.F.; Kumar, S.; McFarlane, A.; Stach, J.; Bentin, T.; et al. The antimicrobial polymer PHMB enters cells and selectively condenses bacterial chromosomes. Sci. Rep. 2016, 6, 23121. [Google Scholar] [CrossRef]
- Kaehn, K. Polihexanide: A safe and highly effective biocide. Skin. Pharmacol. Physiol. 2010, 23, 7–16. [Google Scholar] [CrossRef]
- Fjeld, H.; Lingaas, E. Polyhexanide—Safety and efficacy as an antiseptic. Tidsskr. Nor. Legeforen. 2016, 136, 707–711. [Google Scholar] [CrossRef]
- Kamaruzzaman, N.F.; Chong, S.Q.Y.; Edmondson-Brown, K.M.; Ntow-Boahene, W.; Bardiau, M.; Good, L. Bactericidal and Anti-biofilm Effects of Polyhexamethylene Biguanide in Models of Intracellular and Biofilm of Staphylococcus aureus Isolated from Bovine Mastitis. Front. Microbiol. 2017, 8, 1518. [Google Scholar] [CrossRef]
- Guiomar, A.J.; Urbano, A.M. Polyhexanide-Releasing Membranes for Antimicrobial Wound Dressings: A Critical Review. Membranes 2022, 12, 1281. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Dissemond, J.; Kim, S.; Willy, C.; Mayer, D.; Papke, R.; Tuchmann, F.; Assadian, O. Consensus on Wound Antisepsis: Update 2018. Skin Pharmacol. Physiol. 2018, 31, 28–58. [Google Scholar] [CrossRef]
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A Cellular Perspective. Physiol. Rev. 2019, 99, 665–706. [Google Scholar] [CrossRef]
- Wallace, H.A.; Basehore, B.M.; Zito, P.M. Wound Healing Phases; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Wilkinson, H.N.; Hardman, M.J. Wound healing: Cellular mechanisms and pathological outcomes. Open Biol. 2020, 10, 200223. [Google Scholar] [CrossRef]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Church, D.; Elsayed, S.; Reid, O.; Winston, B.; Lindsay, R. Burn wound infections. Clin. Microbiol. Rev. 2006, 19, 403–434. [Google Scholar] [CrossRef] [PubMed]
- Llorens, E.; Calderon, S.; del Valle, L.J.; Puiggali, J. Polybiguanide (PHMB) loaded in PLA scaffolds displaying high hydrophobic, biocompatibility and antibacterial properties. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 50, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lin, T.; Gao, Y.; Xu, Z.; Huang, C.; Yao, G.; Jiang, L.; Tang, Y.; Wang, X. Antimicrobial electrospun nanofibers of cellulose acetate and polyester urethane composite for wound dressing. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 1556–1565. [Google Scholar] [CrossRef] [PubMed]
- Fahimirad, S.; Satei, P.; Latifi, A.; Changizi-Ashtiyani, S.; Bahrami, M.; Abtahi, H. Electrospun PCL/PVA/PHMB nanofibers incorporating Ziziphus jujuba fruit extract as promising wound dressings with potent antibacterial and antidiabetic properties. J. Biomater. Sci. Polym. Ed. 2024, 35, 2484–2505. [Google Scholar] [CrossRef]
- Li, J.; Ding, Q.; Zha, Y.; Xie, J.; Li, F.; Li, R.; Ao, N. The silk fibroin nanofibrous membrane loaded with polyhexamethyl biguanide for promoting infected wound healing. Eur. Polym. J. 2024, 202, 112666. [Google Scholar] [CrossRef]
- Chereddy, K.K.; Payen, V.L.; Preat, V. PLGA: From a classic drug carrier to a novel therapeutic activity contributor. J. Control Release 2018, 289, 10–13. [Google Scholar] [CrossRef]
- Percival, S.L.; McCarty, S.; Hunt, J.A.; Woods, E.J. The effects of pH on wound healing, biofilms, and antimicrobial efficacy. Wound Repair Regen. 2014, 22, 174–186. [Google Scholar] [CrossRef]
- Abdullahi, A.; Amini-Nik, S.; Jeschke, M.G. Animal models in burn research. Cell. Mol. Life Sci. 2014, 71, 3241–3255. [Google Scholar] [CrossRef]
- TSullivan, P.; Eaglstein, W.H.; Davis, S.C.; Mertz, P. The pig as a model for human wound healing. Wound Repair Regen. 2001, 9, 66–76. [Google Scholar] [CrossRef]
- Seaton, M.; Hocking, A.; Gibran, N.S. Porcine models of cutaneous wound healing. ILAR J. 2015, 56, 127–138. [Google Scholar] [CrossRef]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications. Chem. Rev. 2019, 119, 5298–5415. [Google Scholar] [CrossRef]
- YY/T 0471.1-2004; Test Methods for Primary Wound Dressings—Part 1: Aspects of Absorbency. National Medical Products Administration: Beijing, China, 2004.
- AATCC Test Method 100-2019; Assessment of Antibacterial Finishes on Textile Materials. AATCC: Research Triangle Park, NC, USA, 2019.
- Svensby, A.U.; Nygren, E.; Gefen, A.; Cullen, B.; Ronkvist, A.M.; Gergely, A.; Craig, M.D. The importance of the simulated wound fluid composition and properties in the determination of the fluid handling performance of wound dressings. Int. Wound J. 2024, 21, e14861. [Google Scholar] [CrossRef] [PubMed]
- ISO 10993-13:2010; Biological Evaluation of Medical Devices—Part 13: Identification and Quantification of Degradation Products from Polymeric Medical Devices. ISO: Geneva, Switzerland, 2010.
- ASTM F1635-16; Standard Test Method for in vitro Degradation Testing of Hydrolytically Degradable Polymer Resins and Fabricated Forms for Surgical Implants. ASTM International: West Conshohocken, PA, USA, 2016.
- Cai, Q.; Shi, G.; Bei, J.; Wang, S. Enzymatic degradation behavior and mechanism of poly(lactide-co-glycolide) foams by trypsin. Biomaterials 2003, 24, 629–638. [Google Scholar] [CrossRef]
- ISO 10993-6:2016; Biological Evaluation of Medical Devices—Part 6: Tests for Local Effects After Implantation. International Organization for Standardization (ISO): Geneva, Switzerland, 2016.
- Turner, P.V.; Brabb, T.; Pekow, C.; Vasbinder, M.A. Administration of Substances to Laboratory Animals: Routes of Administration and Factors to Consider. J. Am. Assoc. Lab. Anim. Sci. 2011, 50, 600–613. [Google Scholar]
- Johnson, W., Jr.; Boyer, I.; Zhu, J.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G., Jr.; Shank, R.C.; et al. Safety Assessment of Polyaminopropyl Biguanide (Polyhexamethylene Biguanide Hydrochloride) as Used in Cosmetics. Int. J. Toxicol. 2020, 39, 26S–73S. [Google Scholar] [CrossRef]
- International Council for Harmonisation (ICH). M10: Bioanalytical Method Validation and Study Sample Analysis. ICH Harmonised Guideline, Step 4 Version. 2022. Available online: https://database.ich.org/sites/default/files/M10_Guideline_Step4_2022_0524.pdf (accessed on 25 February 2026).
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef]
- Sell, S.A.; Wolfe, P.S.; Garg, K.; McCool, J.M.; Rodriguez, I.A.; Bowlin, G.L. The Use of Natural Polymers in Tissue Engineering: A Focus on Electrospun Extracellular Matrix Analogues. Polymers 2010, 2, 522–553. [Google Scholar] [CrossRef]
- Norouzi, M.; Boroujeni, S.M.; Omidvarkordshouli, N.; Soleimani, M. Advances in skin regeneration: Application of electrospun scaffolds. Adv. Healthc. Mater. 2015, 4, 1114–1133. [Google Scholar] [CrossRef]
- Luraghi, A.; Peri, F.; Moroni, L. Electrospinning for drug delivery applications: A review. J. Control Release 2021, 334, 463–484. [Google Scholar] [CrossRef] [PubMed]
- Minsart, M.; Van Vlierberghe, S.; Dubruel, P.; Mignon, A. Commercial wound dressings for the treatment of exuding wounds: An in-depth physico-chemical comparative study. Burns Trauma 2022, 10, tkac024. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, C.; Abel, M.; Ruth, P.; Elsner, P.; Hipler, U.C. pH influence on antibacterial efficacy of common antiseptic substances. Skin Pharmacol. Physiol. 2015, 28, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Worsley, A.; Vassileva, K.; Tsui, J.; Song, W.; Good, L. Polyhexamethylene Biguanide:Polyurethane Blend Nanofibrous Membranes for Wound Infection Control. Polymers 2019, 11, 915. [Google Scholar] [CrossRef]
- Serra, R.; Grande, R.; Butrico, L.; Rossi, A.; Settimio, U.F.; Caroleo, B.; Amato, B.; Gallelli, L.; de Franciscis, S. Chronic wound infections: The role of Pseudomonas aeruginosa and Staphylococcus aureus. Expert. Rev. Anti-Infect. Ther. 2015, 13, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Iizaka, S.; Sanada, H.; Nakagami, G.; Koyanagi, H.; Konya, C.; Sugama, J. Quantitative estimation of exudate volume for full-thickness pressure ulcers: The ESTimation method. J. Wound Care 2011, 20, 453–454, 458–463. [Google Scholar] [CrossRef]
- Knipper, J.A.; Willenborg, S.; Brinckmann, J.; Bloch, W.; Maass, T.; Wagener, R.; Krieg, T.; Sutherland, T.; Munitz, A.; Rothenberg, M.E.; et al. Interleukin-4 Receptor alpha Signaling in Myeloid Cells Controls Collagen Fibril Assembly in Skin Repair. Immunity 2015, 43, 803–816. [Google Scholar] [CrossRef] [PubMed]
- Vaz, L.M.; Branco, R.; Morais, P.V.; Guiomar, A.J. Sterilized Polyhexanide-Releasing Chitosan Membranes with Potential for Use in Antimicrobial Wound Dressings. Membranes 2023, 13, 877. [Google Scholar] [CrossRef]
- Ulery, B.D.; Kan, H.M.; Williams, B.A.; Narasimhan, B.; Lo, K.W.; Nair, L.S.; Laurencin, C.T. Facile fabrication of polyanhydride/anesthetic nanoparticles with tunable release kinetics. Adv. Healthc. Mater. 2014, 3, 843–847. [Google Scholar] [CrossRef]
- Grayson, A.C.; Voskerician, G.; Lynn, A.; Anderson, J.M.; Cima, M.J.; Langer, R. Differential degradation rates in vivo and in vitro of biocompatible poly(lactic acid) and poly(glycolic acid) homo- and co-polymers for a polymeric drug-delivery microchip. J. Biomater. Sci. Polym. Ed. 2004, 15, 1281–1304. [Google Scholar] [CrossRef]
- Su, J.; Yu, W.; Guo, X.; Wang, C.; Wang, Q.; Chen, B.; Hu, Y.; Dai, H. Development and Evaluation of a Novel Antibacterial Wound Dressing: A Powder Preparation Based on Cross-Linked Pullulan with Polyhexamethylene Biguanide for Hydrogel-Transition in Advanced Wound Management and Infection Control. Polymers 2024, 16, 1352. [Google Scholar] [CrossRef] [PubMed]
- Göpferich, A. Mechanisms of polymer degradation and erosion. Biomaterials 1996, 17, 103–114. [Google Scholar] [CrossRef]
- Schneider, L.A.; Korber, A.; Grabbe, S.; Dissemond, J. Influence of pH on wound-healing: A new perspective for wound-therapy? Arch. Dermatol. Res. 2007, 298, 413–420. [Google Scholar] [CrossRef]
- Jones, E.M.; Cochrane, C.A.; Percival, S.L. The Effect of pH on the Extracellular Matrix and Biofilms. Adv. Wound Care 2015, 4, 431–439. [Google Scholar] [CrossRef]
- Memic, A.; Abudula, T.; Mohammed, H.S.; Navare, K.J.; Colombani, T.; Bencherif, S.A. Latest Progress in Electrospun Nanofibers for Wound Healing Applications. ACS Appl. Bio Mater. 2019, 2, 952–969. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). 510(k) Premarket Notification: FortaDerm Antimicrobial Wound Dressing (K051647). Organogenesis, Inc. 2005. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K051647 (accessed on 25 February 2026).
- Bain, M.A.; Koullias, G.J.; Morse, K.; Wendling, S.; Sabolinski, M.L. Type I collagen matrix plus polyhexamethylene biguanide antimicrobial for the treatment of cutaneous wounds. J. Comp. Eff. Res. 2020, 9, 691–703. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). 510(k) Premarket Notification: PolyMem Silver Wound Dressing (K031307). Ferris Mfg. Corp. 2003. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K031307 (accessed on 25 February 2026).
- U.S. Food and Drug Administration (FDA). 510(k) Premarket Notification: PolyMem Hydrophilic Wound Dressing (K932913). Ferris Mfg. Corp. 1993. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K932913 (accessed on 25 February 2026).
- U.S. Food and Drug Administration (FDA). 510(k) Premarket Notification: PolyMem Cavity Wound Filler (K990906). Ferris Mfg. Corp. 1999. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K990906 (accessed on 25 February 2026).
- Moloughney, J.G.; Weisleder, N. Poloxamer 188 (p188) as a membrane resealing reagent in biomedical applications. Recent. Pat. Biotechnol. 2012, 6, 200–211. [Google Scholar] [CrossRef]
- Khandelwal, P.; Das, A.; Sen, C.K.; Srinivas, S.P.; Roy, S.; Khanna, S. A surfactant polymer wound dressing protects human keratinocytes from inducible necroptosis. Sci. Rep. 2021, 11, 4357. [Google Scholar] [CrossRef] [PubMed]
- Robson, M.C.; Hill, D.P.; Woodske, M.E.; Steed, D.L. Wound healing trajectories as predictors of effectiveness of therapeutic agents. Arch. Surg. 2000, 135, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Cardinal, M.; Eisenbud, D.E.; Phillips, T.; Harding, K. Early healing rates and wound area measurements are reliable predictors of later complete wound closure. Wound Repair Regen. 2008, 16, 19–22. [Google Scholar] [CrossRef] [PubMed]
- ISO 10993-2:2022; Biological Evaluation of Medical Devices—Part 2: Animal Welfare Requirements. International Organization for Standardization (ISO): Geneva, Switzerland, 2022.
- ISO 10993-16:2017; Biological Evaluation of Medical Devices—Part 16: Toxicokinetic Study Design for Degradation Products and Leachables. International Organization for Standardization (ISO): Geneva, Switzerland, 2017.
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biol. 2020, 18, e3000410. [Google Scholar]
- van Ravenzwaay, B.; Leibold, E. A comparison between in vitro rat and human and in vivo rat skin absorption studies. Hum. Exp. Toxicol. 2004, 23, 421–430. [Google Scholar] [CrossRef]
- Gizaw, M.; Thompson, J.; Faglie, A.; Lee, S.Y.; Neuenschwander, P.; Chou, S.F. Electrospun Fibers as a Dressing Material for Drug and Biological Agent Delivery in Wound Healing Applications. Bioengineering 2018, 5, 9. [Google Scholar] [CrossRef]
- Chen, Z.; Zhou, D.; Fu, Q.; Wang, X. Enhanced wound healing and antimicrobial efficacy of PHMB-loaded injectable chitosan/quaternary chitosan/NaHCO3/β-glycerophosphate hydrogels for seawater-infected wounds. Int. J. Biol. Macromol. 2025, 310, 143418. [Google Scholar] [CrossRef]
- Farahani, M.; Shafiee, A. Wound Healing: From Passive to Smart Dressings. Adv. Healthc. Mater. 2021, 10, e2100477. [Google Scholar] [CrossRef]
- Müller, G.; Kramer, A. Biocompatibility index of antiseptic agents by parallel assessment of antimicrobial activity and cellular cytotoxicity. J. Antimicrob. Chemother. 2008, 61, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, T.; Jacobsen, F.; Rittig, A.; Goertz, O.; Niederbichler, A.; Steinau, H.U.; Seipp, H.M.; Steinstraesser, L. A comparative in vitro study of cell toxicity of clinically used antiseptics. Hautarzt 2009, 60, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.C.; Maibach, H.I. Animal models for percutaneous absorption. J. Appl. Toxicol. 2015, 35, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Powers, J.G.; Higham, C.; Broussard, K.; Phillips, T.J. Wound healing and treating wounds: Chronic wound care and management. J. Am. Acad. Dermatol. 2016, 74, 607–625. [Google Scholar] [CrossRef]


| Property | PHMB Matrix | Xenograft AM |
|---|---|---|
| Structure | Synthetic Composite Fiber Matrix | Cross-linked Collagen Sheet |
| Composition | PLGA, PDO, Poloxamer 188, PHMB | Type I Collagen, PHMB |
| Antibacterial Agent | PHMB | PHMB |
| Concentration | 2.5% (w/w) | 0.1% (w/w) |
| Integration Method | Embedded in Fiber | Interfacial Coating |
| Release Behavior | Biphasic controlled, sustained | Monophasic, rapid depletion |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chen, S.; Bibbo, C.; Starinski, J.; Xu, X.; Wang, C. A PHMB-Functionalized Fully Absorbable Synthetic Matrix as a Novel Alternative to Biologics: Balancing Antibacterial Efficacy, Tissue Repair, and Safety. Bioengineering 2026, 13, 353. https://doi.org/10.3390/bioengineering13030353
Chen S, Bibbo C, Starinski J, Xu X, Wang C. A PHMB-Functionalized Fully Absorbable Synthetic Matrix as a Novel Alternative to Biologics: Balancing Antibacterial Efficacy, Tissue Repair, and Safety. Bioengineering. 2026; 13(3):353. https://doi.org/10.3390/bioengineering13030353
Chicago/Turabian StyleChen, Sean, Christopher Bibbo, John Starinski, Xianghua Xu, and Chenhong Wang. 2026. "A PHMB-Functionalized Fully Absorbable Synthetic Matrix as a Novel Alternative to Biologics: Balancing Antibacterial Efficacy, Tissue Repair, and Safety" Bioengineering 13, no. 3: 353. https://doi.org/10.3390/bioengineering13030353
APA StyleChen, S., Bibbo, C., Starinski, J., Xu, X., & Wang, C. (2026). A PHMB-Functionalized Fully Absorbable Synthetic Matrix as a Novel Alternative to Biologics: Balancing Antibacterial Efficacy, Tissue Repair, and Safety. Bioengineering, 13(3), 353. https://doi.org/10.3390/bioengineering13030353







