Ultrasound Patches Toward Intelligent Theranostics: From Flexible Materials to Closed-Loop Biomedical Systems
Abstract
1. Introduction
2. Material Selection
3. Structure Design
3.1. Transducer Structural Innovation and Miniaturization
3.2. Array Configuration and Acoustic Field Control
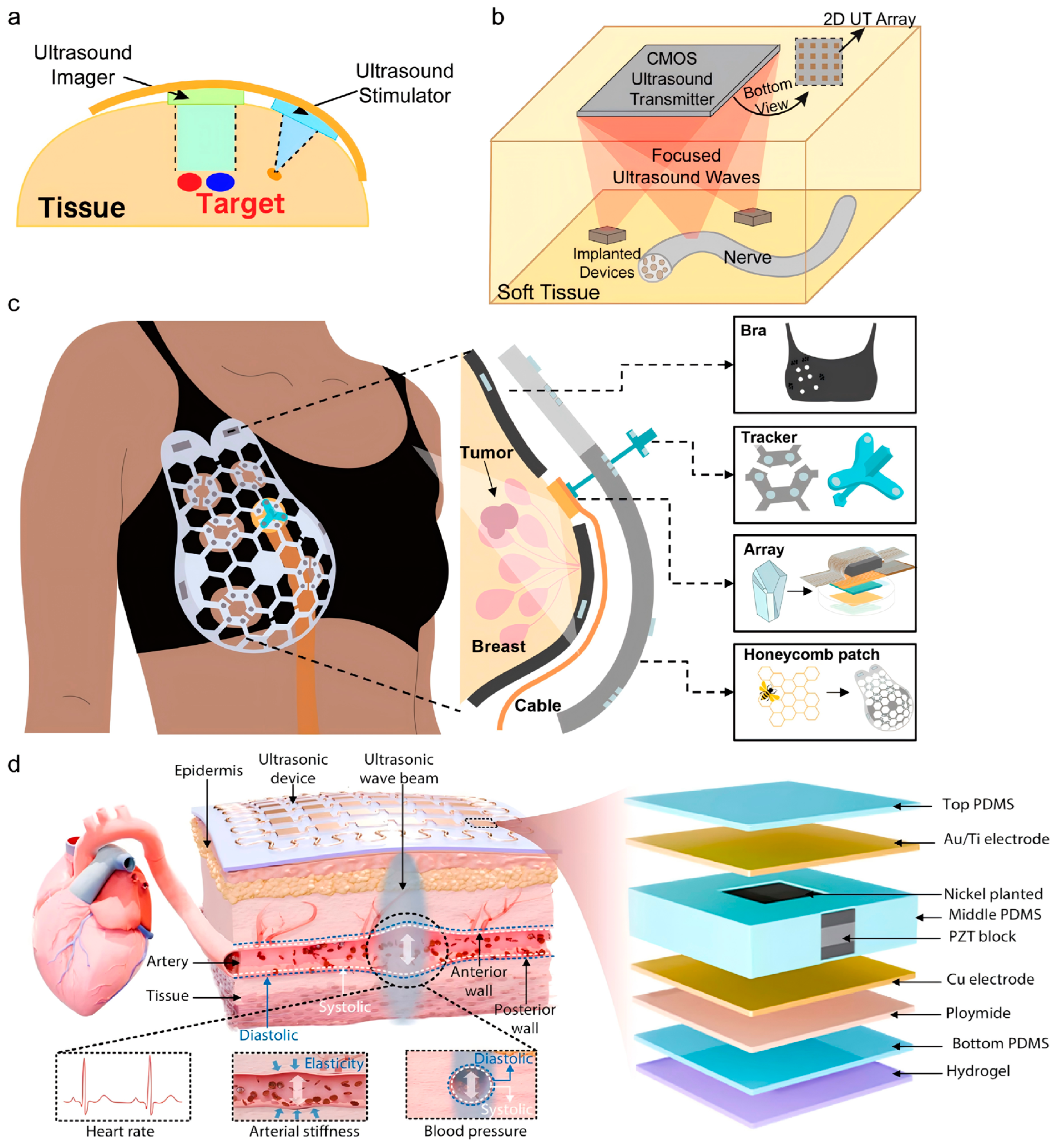
3.3. Lightweight Structures and Biointerfaces
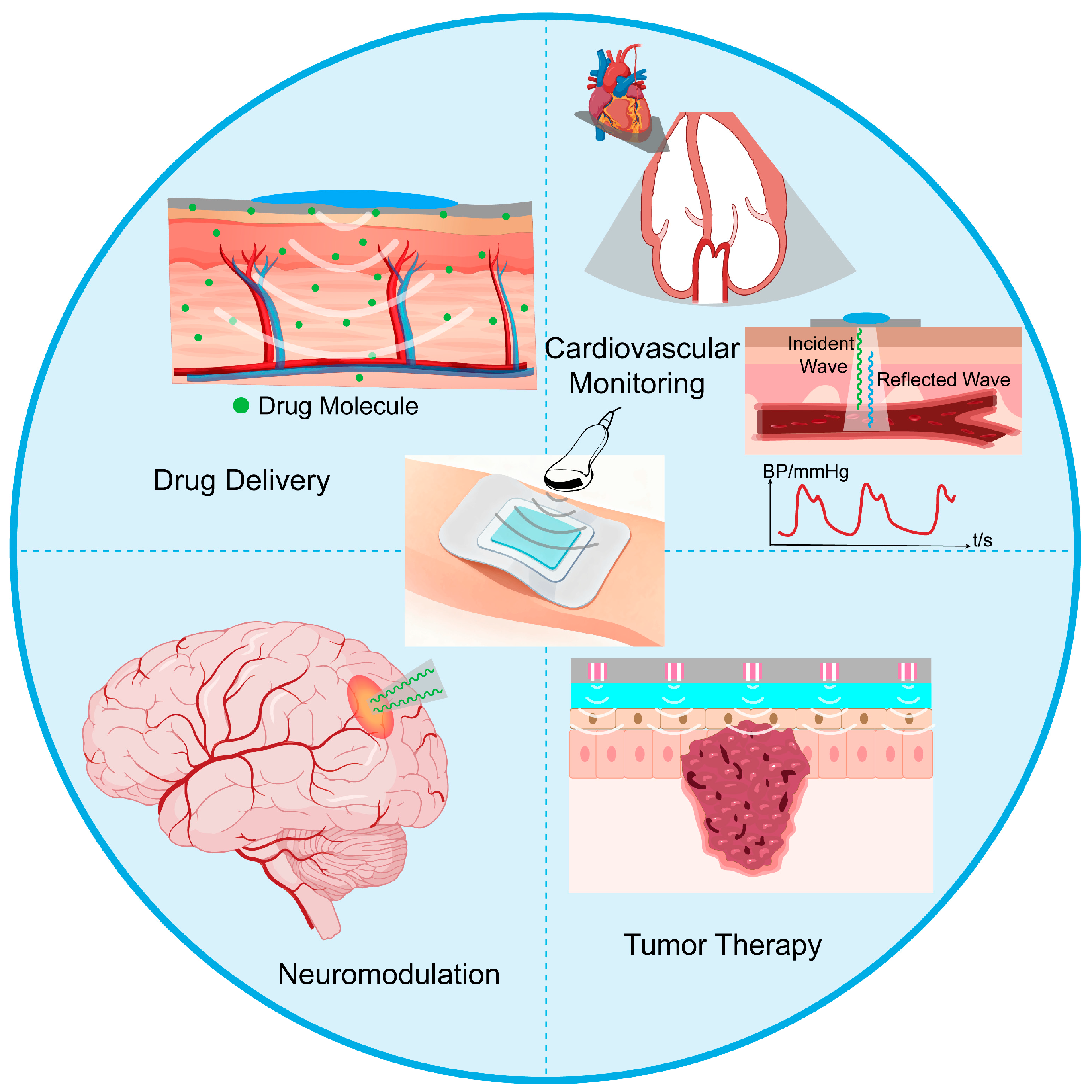
4. Application Scenarios
4.1. Disease Diagnosis and Imaging
4.2. Drug Delivery
4.3. Neuromodulation
4.4. Tumor Diagnosis and Therapy
4.5. Other Applications
5. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Aβ | β-Amyloid |
| AI | Artificial Intelligence |
| ASIC | Application-specific Integrated Circuit |
| BP | Blood Pressure |
| BTO | Barium Titanate |
| CHT | Chitosan |
| CMOS | Complementary Metal-oxide-semiconductor |
| CMUT | Capacitive Micromachined Ultrasonic Transducer |
| CNT | Carbon Nanotube |
| CWUS Patch | Conformal Wearable Ultrasound Patch |
| DS | Diclofenac Sodium |
| EPSC | Excitatory Postsynaptic Spontaneous Current |
| FAD | Familial Alzheimer’s Disease |
| FES | Functional Electrical Stimulation |
| FPCB | Flexible Printed Circuit Board |
| FUST | Flexible Ultrasound Transducer |
| GelMA | Gelatin Methacryloyl |
| HfO2 | Hafnium Oxide |
| HR | Heart Rate |
| KA | Kainic Acid |
| KNN | Potassium Sodium Niobate |
| K2P channels | two-pore domain potassium channels |
| LIPUS | Low-intensity Pulsed Ultrasound |
| LTCC | L-type Calcium Channel |
| MEMS | Microelectromechanical Systems |
| MG-SOG | MscL-G22S-mediated Sonogenetics |
| MOF | Metal–organic Framework |
| MTX | Methotrexate |
| NP | Nanoparticle |
| PCL | polycaprolactone |
| PDMA | Piezoelectric-driven Microneedle Array |
| PDMS | polydimethylsiloxane |
| PEG | Polyethyleneglycol |
| PI | Polyimide |
| PLGA | Poly(lactic-co-glycolic acid) |
| PMN | Pb(Mg1/3Nb2/3)O3 |
| PMUT | Piezoelectric Micromachined Ultrasonic Transducer |
| PT | PbTiO3 |
| PV | Parvalbumin |
| PVDF | Polyvinylidene Fluoride |
| PWI | Pulse Wave Imaging |
| PZT | Lead Zirconate Titanate |
| RA | Rheumatoid Arthritis |
| RCT | Randomized Controlled Trial |
| REP | Repaglinide |
| rGO | Reduced Graphene Oxide |
| ROS | Reactive Oxygen Species |
| SDT | Sonodynamic Therapy |
| SE | Status Epilepticus |
| SLN | Solid Lipid Nanoparticle |
| sn-CMUT | Silicon Nanopillar Capacitive Micromachined Ultrasonic Transducer |
| SST | Somatostatin |
| TDDS | Transdermal Delivery System |
| TPU | Thermoplastic Polyurethane |
| TUT | Transparent Ultrasonic Transducer |
| UsoP | Ultrasound Patch |
| VCSEL | Vertical-cavity Surface Emitting Laser |
| wf-UMP | Wearable Flexible Ultrasound Microneedle Patch |
| WUS | Wearable Ultrasound Sensor |
References
- Beard, J.R.; Officer, A.; de Carvalho, I.A.; Sadana, R.; Pot, A.M.; Michel, J.P.; Lloyd-Sherlock, P.; Epping-Jordan, J.E.; Peeters, G.; Mahanani, W.R.; et al. The World report on ageing and health: A policy framework for healthy ageing. Lancet 2016, 387, 2145–2154. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhang, J.; Xie, Y.; Gao, F.; Xu, S.; Wu, X.; Ye, Z. Wearable Health Devices in Health Care: Narrative Systematic Review. JMIR Mhealth Uhealth 2020, 8, e18907. [Google Scholar] [CrossRef] [PubMed]
- Mattison, G.; Canfell, O.; Forrester, D.; Dobbins, C.; Smith, D.; Töyräs, J.; Sullivan, C. The Influence of Wearables on Health Care Outcomes in Chronic Disease: Systematic Review. J. Med. Internet Res. 2022, 24, e36690. [Google Scholar] [CrossRef] [PubMed]
- Rabiee, N.; Rabiee, M. Wearable Aptasensors. Anal. Chem. 2024, 96, 19160–19182. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, J.; Chen, W.; Shang, D.; Zhang, Q.; Li, Y.; Zheng, H.; Gu, D.; Wu, D.; Ma, T. Skin-Conformable Flexible and Stretchable Ultrasound Transducer for Wearable Imaging. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2024, 71, 811–820. [Google Scholar] [CrossRef]
- Bashatah, A.; Mukherjee, B.; Rima, A.; Patwardhan, S.; Otto, P.; Sutherland, R.; King, E.L.; Lancaster, B.; Aher, A.; Gibson, G.; et al. Wearable Ultrasound System Using Low-Voltage Time Delay Spectrometry for Dynamic Tissue Imaging. IEEE Trans. Biomed. Eng. 2024, 71, 3232–3243. [Google Scholar] [CrossRef]
- Lin, M.; Zhang, Z.; Gao, X.; Bian, Y.; Wu, R.S.; Park, G.; Lou, Z.; Zhang, Z.; Xu, X.; Chen, X.; et al. A fully integrated wearable ultrasound system to monitor deep tissues in moving subjects. Nat. Biotechnol. 2024, 42, 448–457. [Google Scholar] [CrossRef]
- Bourbakis, N.; Tsakalakis, M. A 3-D Ultrasound Wearable Array Prognosis System with Advanced Imaging Capabilities. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2021, 68, 1062–1072. [Google Scholar] [CrossRef]
- Hu, Z.; Yang, Y.; Yang, L.; Gong, Y.; Chukwu, C.; Ye, D.; Yue, Y.; Yuan, J.; Kravitz, A.V.; Chen, H. Airy-beam holographic sonogenetics for advancing neuromodulation precision and flexibility. Proc. Natl. Acad. Sci. USA 2024, 121, e2402200121. [Google Scholar] [CrossRef]
- Kang, D.H.; Cho, S.; Kim, H.Y.; Shim, S.; Kim, D.H.; Jeong, B.; Lee, Y.S.; Park, E.A.; Lee, W.; Kim, H.; et al. Silicon nanocolumn-based disposable and flexible ultrasound patches. Nat. Commun. 2025, 16, 6609. [Google Scholar] [CrossRef]
- Barbarevech, K.; Schafer, M.E.; DiMaria-Ghalili, R.A.; Hyatt, J.; Lewin, P.A. Design of Point-of-Care Ultrasound Device to be Used in At-Home Setting-A Holistic Approach. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2024, 71, 821–830. [Google Scholar] [CrossRef]
- Cai, P.Y.; Logvinenko, T.; Omar, B.; Oottamasathien, S.; Kurtz, M.; Estrada, C.; Bauer, S.; Nelson, C.P. Real-Time Bladder Volume Monitoring for Pediatric Patients Using a Commercially Available Wearable Ultrasound Device. Neurourol. Urodyn. 2025, 44, 1188–1192. [Google Scholar] [CrossRef]
- Kenny, J.S. Functional Hemodynamic Monitoring with a Wireless Ultrasound Patch. J. Cardiothorac. Vasc. Anesth. 2021, 35, 1509–1515. [Google Scholar] [CrossRef] [PubMed]
- Kenny, J.S.; Munding, C.E.; Eibl, J.K.; Eibl, A.M.; Long, B.F.; Boyes, A.; Yin, J.; Verrecchia, P.; Parrotta, M.; Gatzke, R.; et al. A novel, hands-free ultrasound patch for continuous monitoring of quantitative Doppler in the carotid artery. Sci. Rep. 2021, 11, 7780. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Qu, M.; Liu, W.; Wang, Y.; Zhang, G.; Zheng, Y.; Xie, J. Continuous Monitoring of Blood Pressure by Measuring Local Pulse Wave Velocity Using Wearable Micromachined Ultrasonic Probes. IEEE Trans. Biomed. Eng. 2025, 72, 1615–1624. [Google Scholar] [CrossRef]
- Zou, F.; Luo, Y.; Zhuang, W.; Xu, T. A Fully Integrated Conformal Wearable Ultrasound Patch for Continuous Sonodynamic Therapy. Adv. Mater. 2024, 36, e2409528. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Anguluan, E.; Kum, J.; Sanchez-Casanova, J.; Park, T.Y.; Kim, J.G.; Kim, H. Wearable Transcranial Ultrasound System for Remote Stimulation of Freely Moving Animal. IEEE Trans. Biomed. Eng. 2021, 68, 2195–2202. [Google Scholar] [CrossRef]
- Zhong, C.; Guo, N.; Hu, C.; Ni, R.; Zhang, X.; Meng, Z.; Liu, T.; Ding, S.; Ding, W.; Zhao, Y.; et al. Efficacy of Wearable low-intensity pulsed Ultrasound treatment in the Movement disorder in Parkinson’s disease (the SWUMP trial): Protocol for a single-site, double-blind, randomized controlled trial. Trials 2024, 25, 275. [Google Scholar] [CrossRef]
- Zhou, H.; Niu, L.; Xia, X.; Lin, Z.; Liu, X.; Su, M.; Guo, R.; Meng, L.; Zheng, H. Wearable Ultrasound Improves Motor Function in an MPTP Mouse Model of Parkinson’s Disease. IEEE Trans. Biomed. Eng. 2019, 66, 3006–3013. [Google Scholar] [CrossRef]
- Kim, J.H.; Yoon, H.; Viswanath, S.; Dagdeviren, C. Conformable Piezoelectric Devices and Systems for Advanced Wearable and Implantable Biomedical Applications. Annu. Rev. Biomed. Eng. 2025, 27, 255–282. [Google Scholar] [CrossRef]
- Chen, B.; Feng, Z.; Yao, F.-Z.; Zhang, M.-H.; Wang, K.; Wei, Y.; Gong, W.; Rödel, J. Flexible piezoelectrics: Integration of sensing, actuating and energy harvesting. npj Flex. Electron. 2025, 9, 58. [Google Scholar] [CrossRef]
- Wang, C.; Chen, X.; Wang, L.; Makihata, M.; Liu, H.C.; Zhou, T.; Zhao, X. Bioadhesive ultrasound for long-term continuous imaging of diverse organs. Science 2022, 377, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, J.; Zhong, C.; Zhang, Y.; Qiu, Y.; Qin, L. Flexible Electronics: Advancements and Applications of Flexible Piezoelectric Composites in Modern Sensing Technologies. Micromachines 2024, 15, 982. [Google Scholar] [CrossRef]
- AlMohimeed, I.; Ono, Y. Ultrasound Measurement of Skeletal Muscle Contractile Parameters Using Flexible and Wearable Single-Element Ultrasonic Sensor. Sensors 2020, 20, 3616. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wu, D. Low Temperature Adhesive Bonding-Based Fabrication of an Air-Borne Flexible Piezoelectric Micromachined Ultrasonic Transducer. Sensors 2020, 20, 3333. [Google Scholar] [CrossRef]
- Joshi, S.V.; Sadeghpour, S.; Kraft, M. Flexible PZT-Based Row-Column Addressed 2-D PMUT Array. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2024, 71, 1616–1626. [Google Scholar] [CrossRef]
- Song, Z.; Wang, B.; Zhang, Z.; Yu, Y.; Lin, D. A Highly Flexible Piezoelectric Ultrasonic Sensor for Wearable Bone Density Testing. Micromachines 2023, 14, 1798. [Google Scholar] [CrossRef]
- Wang, W.; Li, K.; Ma, W.; Li, Y.; Liu, F.; Kong, Y.; Wang, L.; Yi, F.; Sang, Y.; Li, G.; et al. Ultrasound-activated piezoelectric nanostickers for neural stem cell therapy of traumatic brain injury. Nat. Mater. 2025, 24, 1137–1150. [Google Scholar] [CrossRef]
- Sun, Y.; Quan, Y.; Xing, J.; Tan, Z.; Sun, X.; Lou, L.; Fei, C.; Zhu, J.; Yang, Y. Lead-Free Potassium Sodium Niobate-Based Wearable Ultrasonic Patches for Blood Pressure Detection. Micromachines 2025, 16, 392. [Google Scholar] [CrossRef]
- Chakraborty, S.; Debnath, S.; Mahipal Malappuram, K.; Parasuram, S.; Chang, H.T.; Chatterjee, K.; Nain, A. Flexible and Robust Piezoelectric Chitosan Films with Enhanced Bioactivity. Biomacromolecules 2025, 26, 1128–1140. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Z.; Zhao, Z.; Xu, Y.; Duan, X.; Zhao, Y.; Ma, W.; Yang, Y.; Yang, Y.; Liu, Z. An Easy Nanopatch Promotes Peripheral Nerve Repair through Wireless Ultrasound-Electrical Stimulation in a Band-Aid-Like Way. Adv. Funct. Mater. 2024, 34, 2407411. [Google Scholar] [CrossRef]
- Feng, Y.; Zhang, Q.; Li, H.; Qi, Q.; Tong, Z.; Rong, D.; Zhou, Z. Design and characteristic analysis of flexible CNT film patch for potential application in ultrasonic therapy. Nanotechnology 2023, 34, 195502. [Google Scholar] [CrossRef] [PubMed]
- Gerardo, C.D.; Cretu, E.; Rohling, R. Fabrication and testing of polymer-based capacitive micromachined ultrasound transducers for medical imaging. Microsyst. Nanoeng. 2018, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, B.; Wang, S.; Li, B.; Chen, Y.; Ye, W.; Luan, S.; Wang, L.; Shi, H. Tenon-and-Mortise Structure-Inspired MOF/PVDF Composites with Enhanced Piezocatalytic Performance via Dipole-Engineering Strategy. Small 2025, 21, e2409314. [Google Scholar] [CrossRef]
- Zhang, Y.; Jin, T.; Deng, Y.; Zhao, Z.; Wang, R.; He, Q.; Luo, J.; Li, J.; Du, K.; Wu, T.; et al. A low-voltage-driven MEMS ultrasonic phased-array transducer for fast 3D volumetric imaging. Microsyst. Nanoeng. 2024, 10, 128. [Google Scholar] [CrossRef]
- Gami, P.; Roy, T.; Liang, P.; Kemper, P.; Travagliati, M.; Baldasarre, L.; Bart, S.; Konofagou, E.E. In Vivo Characterization of Central Arterial Properties Using a Miniaturized pMUT Array Compared to a Clinical Transducer: A Feasibility Study Towards Wearable Pulse Wave Imaging. IEEE Trans. Biomed. Eng. 2025, 72, 2769–2780. [Google Scholar] [CrossRef]
- Pashaei, V.; Dehghanzadeh, P.; Enwia, G.; Bayat, M.; Majerus, S.J.A.; Mandal, S. Flexible Body-Conformal Ultrasound Patches for Image-Guided Neuromodulation. IEEE Trans. Biomed. Circuits Syst. 2020, 14, 305–318. [Google Scholar] [CrossRef]
- Huan, J.; Pashaei, V.; Majerus, S.J.A.; Bhunia, S.; Mandal, S. A Wearable Dual-Mode Probe for Image-Guided Closed-Loop Ultrasound Neuromodulation. IEEE Trans. Biomed. Circuits Syst. 2025, 19, 357–373. [Google Scholar] [CrossRef]
- Costa, T.; Shi, C.; Tien, K.; Elloian, J.; Cardoso, F.A.; Shepard, K.L. An Integrated 2D Ultrasound Phased Array Transmitter in CMOS with Pixel Pitch-Matched Beamforming. IEEE Trans. Biomed. Circuits Syst. 2021, 15, 731–742. [Google Scholar] [CrossRef]
- Chen, H.; Agrawal, S.; Osman, M.; Minotto, J.; Mirg, S.; Liu, J.; Dangi, A.; Tran, Q.; Jackson, T.; Kothapalli, S.R. A Transparent Ultrasound Array for Real-Time Optical, Ultrasound, and Photoacoustic Imaging. BME Front. 2022, 2022, 9871098. [Google Scholar] [CrossRef]
- Du, W.; Zhang, L.; Suh, E.; Lin, D.; Marcus, C.; Ozkan, L.; Ahuja, A.; Fernandez, S.; Shuvo, I.I.; Sadat, D.; et al. Conformable ultrasound breast patch for deep tissue scanning and imaging. Sci. Adv. 2023, 9, eadh5325. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Li, Z.; Zhao, Y.; Luo, R.; Qin, S.; Li, J.; Li, M.; Han, G.; Li, Z.; Zhao, Z.; et al. Skin-adaptive focused flexible micromachined ultrasound transducers for wearable cardiovascular health monitoring. Sci. Adv. 2025, 11, eadw7632. [Google Scholar] [CrossRef] [PubMed]
- Ngo, O.; Niemann, E.; Gunasekaran, V.; Sankar, P.; Putterman, M.; Lafontant, A.; Nadkarni, S.; DiMaria-Ghalili, R.A.; Neidrauer, M.; Zubkov, L.; et al. Development of Low Frequency (20-100 kHz) Clinically Viable Ultrasound Applicator for Chronic Wound Treatment. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2019, 66, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Lyu, W.; Ma, Y.; Chen, S.; Li, H.; Wang, P.; Chen, Y.; Feng, X. Flexible Ultrasonic Patch for Accelerating Chronic Wound Healing. Adv. Healthc. Mater. 2021, 10, e2100785. [Google Scholar] [CrossRef]
- Huang, D.; Sun, M.; Bu, Y.; Luo, F.; Lin, C.; Lin, Z.; Weng, Z.; Yang, F.; Wu, D. Microcapsule-embedded hydrogel patches for ultrasound responsive and enhanced transdermal delivery of diclofenac sodium. J. Mater. Chem. B 2019, 7, 2330–2337. [Google Scholar] [CrossRef]
- Ma, H.; Liu, Z.; Lu, X.; Zhang, S.; Tang, C.; Cheng, Y.; Zhang, H.; Liu, G.; Sui, C.; Ding, C.; et al. 3D printed multi-coupled bioinspired skin-electronic interfaces with enhanced adhesion for monitoring and treatment. Acta Biomater. 2024, 187, 183–198. [Google Scholar] [CrossRef]
- Xue, H.; Jin, J.; Huang, X.; Tan, Z.; Zeng, Y.; Lu, G.; Hu, X.; Chen, K.; Su, Y.; Hu, X.; et al. Wearable flexible ultrasound microneedle patch for cancer immunotherapy. Nat. Commun. 2025, 16, 2650. [Google Scholar] [CrossRef]
- Sempionatto, J.R.; Lin, M.; Yin, L.; De la Paz, E.; Pei, K.; Sonsa-Ard, T.; de Loyola Silva, A.N.; Khorshed, A.A.; Zhang, F.; Tostado, N.; et al. An epidermal patch for the simultaneous monitoring of haemodynamic and metabolic biomarkers. Nat. Biomed. Eng. 2021, 5, 737–748. [Google Scholar] [CrossRef]
- Amado-Rey, A.B.; Goncalves Seabra, A.C.; Stieglitz, T. Towards Ultrasound Wearable Technology for Cardiovascular Monitoring: From Device Development to Clinical Validation. IEEE Rev. Biomed. Eng. 2025, 18, 93–112. [Google Scholar] [CrossRef]
- Gao, X.; Chen, X.; Hu, H.; Wang, X.; Yue, W.; Mu, J.; Lou, Z.; Zhang, R.; Shi, K.; Chen, X.; et al. A photoacoustic patch for three-dimensional imaging of hemoglobin and core temperature. Nat. Commun. 2022, 13, 7757. [Google Scholar] [CrossRef]
- Ali, H.S.M.; Namazi, N.; Elbadawy, H.M.; El-Sayed, A.A.A.; Ahmed, S.A.; Bafail, R.; Almikhlafi, M.A.; Alahmadi, Y.M. Repaglinide-Solid Lipid Nanoparticles in Chitosan Patches for Transdermal Application: Box-Behnken Design, Characterization, and In Vivo Evaluation. Int. J. Nanomed. 2024, 19, 209–230. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Huang, G.; Ma, Z.; Qin, S. Ultrasound-assisted transdermal delivery of alendronate for the treatment of osteoporosis. Acta Biochim. Pol. 2020, 67, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Bok, M.; Zhao, Z.J.; Hwang, S.H.; Jeong, Y.; Ko, J.; Ahn, J.; Lee, J.H.; Jeon, S.; Jeong, J.H. Biocompatible All-in-One Adhesive Needle-Free Cup Patch for Enhancing Transdermal Drug Delivery. ACS Appl. Mater. Interfaces 2021, 13, 58220–58228. [Google Scholar] [CrossRef]
- Bok, M.; Zhao, Z.J.; Jeon, S.; Jeong, J.H.; Lim, E. Ultrasonically and Iontophoretically Enhanced Drug-Delivery System Based on Dissolving Microneedle Patches. Sci. Rep. 2020, 10, 2027. [Google Scholar] [CrossRef]
- Li, L.; Kai, Y.; Wang, Y.; Chen, Z. Piezoelectric-Driven Microneedle Array Delivery of Methotrexate for Enhanced Psoriasis Treatment. ACS Biomater. Sci. Eng. 2025, 11, 4515–4522. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Y.; Chen, Y.; Cheng, J.; Zhang, J.; Shang, J.; Chen, Y.; Liu, Q.; An, Q.; Feng, Z. Microbubble-Enhanced Transdermal Drug Delivery Sonoelectric Patch. ACS Appl. Mater. Interfaces 2024, 16, 49069–49082. [Google Scholar] [CrossRef]
- Ganguly, S.; Wulff, D.; Phan, C.M.; Jones, L.W.; Tang, X.S. Injectable and 3D Extrusion Printable Hydrophilic Silicone-Based Hydrogels for Controlled Ocular Delivery of Ophthalmic Drugs. ACS Appl. Bio Mater. 2024, 7, 6286–6296. [Google Scholar] [CrossRef]
- Vaidya, J.; Shende, P. Potential of Sonophoresis as a Skin Penetration Technique in the Treatment of Rheumatoid Arthritis with Transdermal Patch. AAPS PharmSciTech 2020, 21, 180. [Google Scholar] [CrossRef]
- Cui, K.; Zhang, S.; Sun, J.; Zhang, X.; Ding, C.; Xu, G. Inhibitory effect of ultrasonic stimulation on the voltage-dependent potassium currents in rat hippocampal CA1 neurons. BMC Neurosci. 2019, 20, 3. [Google Scholar] [CrossRef]
- Prieto, M.L.; Firouzi, K.; Khuri-Yakub, B.T.; Madison, D.V.; Maduke, M. Spike frequency-dependent inhibition and excitation of neural activity by high-frequency ultrasound. J. Gen. Physiol. 2020, 152, e202012672. [Google Scholar] [CrossRef]
- Sorum, B.; Docter, T.; Panico, V.; Rietmeijer, R.A.; Brohawn, S.G. Pressure and ultrasound activate mechanosensitive TRAAK K+ channels through increased membrane tension. bioRxiv 2023. [Google Scholar] [CrossRef]
- Sorum, B.; Docter, T.; Panico, V.; Rietmeijer, R.A.; Brohawn, S.G. Tension activation of mechanosensitive two-pore domain K+ channels TRAAK, TREK-1, and TREK-2. Nat. Commun. 2024, 15, 3142. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.Y.; Chen, Y.M.; Wang, Y.F.; Wang, Y.Q.; Hu, J.Q.; Tang, W.X.; Feng, Y.; Cheng, Q.; Xue, L. L-Type Calcium Channel Modulates Low-Intensity Pulsed Ultrasound-Induced Excitation in Cultured Hippocampal Neurons. Neurosci. Bull. 2024, 40, 921–936. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Jiang, H.; Lin, J.; Qiao, C.; Augustine, G.J. Low Intensity Pulsed Ultrasound Activates Excitatory Synaptic Networks in Cultured Hippocampal Neurons. Ultrasound Med. Biol. 2025, 51, 1250–1260. [Google Scholar] [CrossRef]
- Lin, Z.; Meng, L.; Zou, J.; Zhou, W.; Huang, X.; Xue, S.; Bian, T.; Yuan, T.; Niu, L.; Guo, Y.; et al. Non-invasive ultrasonic neuromodulation of neuronal excitability for treatment of epilepsy. Theranostics 2020, 10, 5514–5526. [Google Scholar] [CrossRef]
- Xu, T.; Tan, D.; Wang, Y.; Gong, C.; Yuan, J.; Yang, X.; Wen, Y.; Ban, Y.; Liang, M.; Hu, Y.; et al. Targeted sonogenetic modulation of GABAergic interneurons in the hippocampal CA1 region in status epilepticus. Theranostics 2024, 14, 6373–6391. [Google Scholar] [CrossRef]
- Zou, F.; Liu, Y.; Luo, Y.; Xu, T. A wearable spatiotemporal controllable ultrasonic device with amyloid-β disaggregation for continuous Alzheimer’s disease therapy. Sci. Adv. 2025, 11, eadw1732. [Google Scholar] [CrossRef]
- Siboro, P.Y.; Sharma, A.K.; Lai, P.J.; Jayakumar, J.; Mi, F.L.; Chen, H.L.; Chang, Y.; Sung, H.W. Harnessing HfO2 Nanoparticles for Wearable Tumor Monitoring and Sonodynamic Therapy in Advancing Cancer Care. ACS Nano 2024, 18, 2485–2499. [Google Scholar] [CrossRef]
- Bai, X.; Kang, J.; Wei, S.; Wang, Y.; Liu, Y.; Yuan, B.; Lu, Q.; Li, H.; Yan, J.; Yang, X.; et al. A pH responsive nanocomposite for combination sonodynamic-immunotherapy with ferroptosis and calcium ion overload via SLC7A11/ACSL4/LPCAT3 pathway. Exploration 2025, 5, 20240002. [Google Scholar] [CrossRef]
- Wang, F.; Fan, Y.; Liu, Y.; Lou, X.; Sutrisno, L.; Peng, S.; Li, J. Oxygen-carrying semiconducting polymer nanoprodrugs induce sono-pyroptosis for deep-tissue tumor treatment. Exploration 2024, 4, 20230100. [Google Scholar] [CrossRef]
- Zhang, H.; Pan, X.; Wu, Q.; Guo, J.; Wang, C.; Liu, H. Manganese carbonate nanoparticles-mediated mitochondrial dysfunction for enhanced sonodynamic therapy. Exploration 2021, 1, 20210010. [Google Scholar] [CrossRef]
- Cao, L.L.; Wang, F.W.; Xue, J.W.; Liu, F.L.; Jin, M.L. Integrated Ultrasound Device for Precision Bladder Volume Monitoring via Acoustic Focusing and Machine Learning. Adv. Sci. 2026, e20926, early view. [Google Scholar] [CrossRef] [PubMed]
- Pu, C.; Fu, B.; Guo, L.H.; Xu, H.X.; Peng, C. A Stretchable and Wearable Ultrasonic Transducer Array for Bladder Volume Monitoring Application. IEEE Sens. J. 2024, 24, 15875–15883. [Google Scholar] [CrossRef]
- Zhang, L.; Marcus, C.; Lin, D.B.; Mejorado, D.; Schoen, S.J., Jr.; Pierce, T.T.; Kumar, V.; Fernandez, S.V.; Hunt, D.; Li, Q.; et al. A conformable phased-array ultrasound patch for bladder volume monitoring. Nat. Electron. 2024, 7, 77–90. [Google Scholar] [CrossRef]
- Tang, X.Y.; Liu, P.L.; Liu, X.; Qiu, L. Dynamic Evaluation of Skeletal Muscle Voluntary Contraction Function Using Pulsed Wave Doppler Imaging: An Exploratory Study Based on Wearable Ultrasound. Ultrason. Imaging 2026, 48, 67–73. [Google Scholar] [CrossRef]
- King, E.L.; Patwardhan, S.; Bashatah, A.; Magee, M.; Jones, M.T.; Wei, Q.; Sikdar, S.; Chitnis, P.V. Distributed Wearable Ultrasound Sensors Predict Isometric Ground Reaction Force. Sensors 2024, 24, 5023. [Google Scholar] [CrossRef]
- Cao, Y.D.; Lu, Y.; Wang, W.P.; Xu, P.; Yang, X.L.; Zhang, S.W.; Wu, M.; Gong, X.L.; Sun, S.S. Development of a wearable ultrasound-FES integrated rehabilitation and motor-functional reconstruction system for post-stroke patients. Biomed. Signal Process. And. Control 2025, 100, 106846. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, G.J.; Song, J.Q.; Song, H.B.; Chu, Q.J. Innovative wearable technology for continuous echocardiographic monitoring: First-in-neonate case report. Front. Pediatr. 2025, 13, 1567386. [Google Scholar] [CrossRef]
- Currens, J.B.; Moon, R.E.; Makowski, M.S.; Natoli, M.J.; Leypoldt, J.; Woolard, J.; Brown, D.; Azarang, A.; Brown, R.; Schinazi, E.; et al. Ultrasound detection of lymphatic bubbles in a porcine dive model. J. Appl. Physiol. 2025, 139, 365–375. [Google Scholar] [CrossRef]
- Yang, Y.; Jin, Z.; Zhou, H.; Li, Y.; Zhang, W.; Xiang, Y.; Wu, D.; Li, C.; Li, F. Wearable ultrasound regulation of blood glucose levels in type 2 diabetic db/db mice. Ultrasonics 2025, 155, 107739. [Google Scholar] [CrossRef]
- Luo, X.; Xie, J.; Yang, L.; Cui, Y. An intelligent wearable artificial pancreas patch based on a microtube glucose sensor and an ultrasonic insulin pump. Talanta 2024, 273, 125879. [Google Scholar] [CrossRef]
- Oh, C.; Kim, Y.M.; Lee, T.; Lee, S.M.; Jung, J.; Bae, H.M.; Kim, C.; Lee, H.J. Patch-type capacitive micromachined ultrasonic transducer for ultrasonic power and data transfer. Microsyst. Nanoeng. 2025, 11, 124. [Google Scholar] [CrossRef]
- Aafizaa, K.; Uma Haimavathi, K.; Saravanan, S. Recent Innovations in Microstrip Patch Antennas: Biomedical Uses and Wireless Integration. Biomed. Mater. Devices 2025, 4, 326–341. [Google Scholar] [CrossRef]


| Material Type | Examples | Advantages | Disadvantages |
|---|---|---|---|
| Piezoelectric Polymers | Polyvinylidene fluoride (PVDF) [24,25] | High flexibility and conformability, lightweight, good biocompatibility, simple and low-cost fabrication process. | Lower piezoelectric output compared to ceramics, which may limit sensitivity for some deep-tissue applications. |
| Lead-Containing Piezoelectric Ceramics | Lead zirconate titanate (PZT) [26] PMN-PZT [27] | Exceptionally high piezoelectric coefficients and electromechanical coupling, enabling high-sensitivity imaging and actuation. | Inherent rigidity requires composite design for wearability; contains toxic lead, raising biosafety and environmental concerns. |
| Lead-Free Piezoelectric Ceramics | Barium titanate (BTO) [28] Potassium sodium niobate (KNN) [29] | Excellent biosafety and environmental friendliness due to lead-free composition, with moderate to good piezoelectric performance. | Piezoelectric properties generally inferior to PZT; fabrication can be more challenging to achieve comparable performance. |
| Natural Biomaterials | Chitosan (CHT) [30] | Inherent biocompatibility, biodegradability, and bioactivity (e.g., antibacterial, anti-inflammatory), suitable for bioactive interfaces. | Mechanically weak and hydrolytically unstable unless chemically crosslinked; piezoelectric response is typically weak. |
| Piezoelectric Composites | BTO@PCL/GO@GelMA [31] | Design versatility to tailor flexibility, piezoelectricity, and additional functionalities (e.g., conductivity, bioadhesion) in a single system. | Complex fabrication; long-term reliability of the material interface under dynamic conditions needs further validation. |
| Application Scenarios | Study | Material/Structure | Dimensions/Thickness | Target/Function | Power/Voltage | Biocompatibility/Wearability Features | Working Principle | Intended Application |
|---|---|---|---|---|---|---|---|---|
| Disease Diagnosis and Imaging | USoP (Lin et al., 2024) [7] | Flexible US transducer array + integrated control circuit | Not specified | Deep tissue (up to 164 mm) physiological signals | a power consumption of ~614 mW | Fully integrated, wearable, continuous 12 h operation, motion tracking | Ultrasound imaging + machine learning | Continuous deep-tissue monitoring in moving subjects |
| snCMUT (Kang et al., 2025) [10] | Silicon nanopillar CMUT array, flexible packaging | Overall thickness < 1 mm | Carotid artery imaging, BP waveform | 8.9 Vpp (operating voltage) | Flexible, stretchable (except FPCB), disposable, no ASIC needed | Capacitive micromachined ultrasound transduction | Wearable cardiovascular monitoring patch | |
| BP Monitoring Patch (Sun et al., 2025) [29] | Lead-free piezoelect. (KNN-Cr), silicone rubber packaging | 2.8 mm × 2.8 mm, center freq. 5 MHz | Radial artery, blood pressure detection | Not specified | Flexible, wearable, biocompatible, environmentally friendly | US measurement of vessel diameter change | Non-invasive continuous BP monitoring | |
| Multimodal Patch (Sempionatto et al., 2021) [48] | Integrated US transducer & electrochemical sensors | Not specified | BP, HR, and multiple biomarkers (glucose, lactate, etc.) | Not specified | Flexible epidermal patch, anti-crosstalk design, iontophoresis sampling | US monitoring + electrochemical sensing | Multimodal physiological & biochemical monitoring | |
| Photoacoustic Patch (Gao et al., 2022) [50] | VCSEL diode array + piezoelectric transducer | 2.0 cm × 1.6 cm overall footprint, thickness 1.2 mm | Deep tissue hemoglobin imaging & core temperature | Not specified | Wearable, first continuous deep-tissue biomolecule monitoring | Photoacoustic imaging & thermometry | 3D tissue imaging & core temperature measurement | |
| Drug Delivery | Ultrasound-Responsive TDDS (Huang et al., 2019) [45] | PEG-PLGA microcapsules embedded in 4-arm-PEG hydrogel | Microcapsule diam. ~3.5 μm; patch diameter 10 mm; height 2 mm (for in vitro test) | Skin, transdermal drug delivery | 2 W/cm2 (ultrasound intensity) | Good biocompatibility, excellent skin adhesion | Ultrasound-triggered drug release & permeation enhancement | Controlled transdermal delivery (e.g., arthritis) |
| Transdermal REP-SLN (Ali et al., 2024) [51] | Chitosan-based transdermal system (Solid Lipid Nanoparticles) | Nanoparticle size ~249 nm | Skin, repaglinide delivery | Not specified | Transdermal patch, enhanced bioavailability | SLN prepared by ultrasound melt-emulsification | Transdermal delivery of antidiabetic drug | |
| PDMA (Li et al., 2025) [55] | PDMA (piezoelectric ceramic PZT8 + 3D-printed hollow microneedle array) | Device: φ44 mm × 26 mm; Needle: inner diameter 120 μm, outer diameter 180 μm, 1 mm height (3 × 3 array) | MTX, psoriasis treatment | 200 Vpp, ~120 kHz, 0.56 W/cm2 | Minimally invasive, no skin irritation, safe temp rise (5 °C) | Ultrasound cavitation-enhanced penetration (sonophoresis + microneedles) | Psoriasis therapy (9× penetration, 50% oral dose with better efficacy) | |
| Energy-Converting TDDS (Zhang et al., 2024) [56] | Microbubble & piezoelectric soft structure composite patch | PVDF film thickness 200 μm; patch size 2 × 2 cm | Skin, transdermal drug delivery | Not specified | Thermochromic indicator, safe operating temperature | US energy to electricity conversion, multi-field synergy for permeation | Enhanced & controlled transdermal delivery (e.g., pain) | |
| Neuromodulation | Neuro-Modulation Patch (Pashaei et al., 2020) [37] | 64-element imaging array + 8-element modulation array | Imaging: 0.4 mm thick; Modulation: 1.5 mm thick | Nerve localization & modulation (e.g., vagus) | Sensitivity: ~80 kPa/V (modulation array) | Body-conformal, integrated strain sensor for closed-loop feedback | Image-guided focused ultrasound neuromodulation | Neuromodulation therapy |
| Epilepsy Therapy (LIPUS) (Lin et al., 2020) [65] | Conventional US transducer (non-patch form) | Not specified | Brain, neural excitability modulation | Low-intensity pulsed US | Non-patch form, investigational application | Low-intensity US neuromodulation | Epilepsy treatment | |
| Sonogenetics (MG-SOG) (Xu et al., 2024) [66] | Sonogenetics technique (non-device description) | Not specified | Hippocampal CA1 PV interneurons | 650 kHz, 0.38 MPa | Research technique, non-wearable device | Sonogenetic control of specific neurons | Research on status epilepticus treatment | |
| AD Therapy Patch (Zou et al., 2025) [67] | Flexible honeycomb US array patch + flexible circuit | 2 mm thick | Brain, amyloid-β plaque disaggregation | 1 MHz, 1.7 W/cm2 | Wearable, spatiotemporally controllable, non-invasive | US-induced protein disaggregation & immune regulation | Alzheimer’s disease therapy | |
| Tumor Diagnosis and Therapy | CWUS Patch (Zou et al., 2024) [16] | Fully integrated conformal wearable US patch system | 2 mm thick | Tumor site, sonodynamic therapy | 2.0 W/cm2 (intensity) | Good mechanical conformability, biocompatibility, portable, non-invasive | Focused US activation of sonosensitizer for ROS generation | Continuous SDT for deep-seated tumors |
| wf-UMP (Xue et al., 2025) [47] | Lead-free US array + bioadhesive hydrogel + dissolvable microneedles | Microneedle height 600 μm, hydrogel thickness ~1 mm | Tumor, drug delivery & immunotherapy | 10–120 Vpp driving voltage, 1.2 MHz center frequency | Flexible, wearable, bioadhesive, stable on dynamic tissue | US-enhanced drug delivery & immunomodulation | Cancer immunotherapy | |
| Tumor Monitoring & SDT (Siboro et al., 2024) [68] | Flexible TPU/HfO2 NPs sensor platform | Not specified | Tumor volume monitoring & sonodynamic therapy | 1.0 W/cm2, 3 MHz | Flexible wearable, wireless data transmission | US volume monitoring + SDT (HfO2 as sonosensitizer) | Cancer theranostics & telemedicine | |
| Other Applications | cUSB-Patch (Zhang et al., 2024) [74] | Sm/La-doped PMN-PT ceramic phased array | Overall thickness < 4.5 mm, single array 20.0 mm × 20.0 mm × 4.0 mm | Bladder volume monitoring | 50 V, 3.5 MHz | Conformable, no manual operation/coupling gel needed (validated), wide field-of-view | Phased-array US volumetric imaging | Bladder volume monitoring (e.g., urinary retention) |
| FES-Rehab System (Cao et al. 2025) [77] | Wearable musculoskeletal US + Functional Electrical Stimulation (FES) | Not specified | Muscle intent recognition, motor function assistance | US: 60 V driving voltage; FES: 10–21 mA, 30–40 Hz | Wearable integrated system, improved robustness & SNR | US-based intent recognition + synchronized FES | Post-stroke rehabilitation & motor function restoration | |
| Glucose Regulation (Yang et al., 2025) [80] | 1–3 composite, Cu/PI electrodes, Ecoflex substrate | Not specified | Hepatic-pancreatic area, blood glucose regulation | 986 kHz, 86.81 mW/cm2 (ISATA), 30% duty cycle, pulse repetition frequency of 2 kHz | Wearable patch + driver, biosafety validated in vivo | Low-intensity pulsed ultrasound therapy | Type 2 diabetes management |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhao, J.; Huang, Y.; Zhang, Y.; Xie, Y.; Guo, W.; Li, Y.; Wang, S. Ultrasound Patches Toward Intelligent Theranostics: From Flexible Materials to Closed-Loop Biomedical Systems. Bioengineering 2026, 13, 345. https://doi.org/10.3390/bioengineering13030345
Zhao J, Huang Y, Zhang Y, Xie Y, Guo W, Li Y, Wang S. Ultrasound Patches Toward Intelligent Theranostics: From Flexible Materials to Closed-Loop Biomedical Systems. Bioengineering. 2026; 13(3):345. https://doi.org/10.3390/bioengineering13030345
Chicago/Turabian StyleZhao, Jinpeng, Yi Huang, Yuan Zhang, Yuhang Xie, Wei Guo, Yang Li, and Shidong Wang. 2026. "Ultrasound Patches Toward Intelligent Theranostics: From Flexible Materials to Closed-Loop Biomedical Systems" Bioengineering 13, no. 3: 345. https://doi.org/10.3390/bioengineering13030345
APA StyleZhao, J., Huang, Y., Zhang, Y., Xie, Y., Guo, W., Li, Y., & Wang, S. (2026). Ultrasound Patches Toward Intelligent Theranostics: From Flexible Materials to Closed-Loop Biomedical Systems. Bioengineering, 13(3), 345. https://doi.org/10.3390/bioengineering13030345






