A Review of Synthetic Bone Grafts in Lumbar Interbody Fusion
Abstract
1. Introduction
2. Methods
2.1. Review Design and Methodological Framework
2.2. Literature Search Strategy
2.3. Eligibility Criteria and Study Selection
2.4. Data Extraction and Synthesis Approach
2.5. Assessment of Reporting Quality
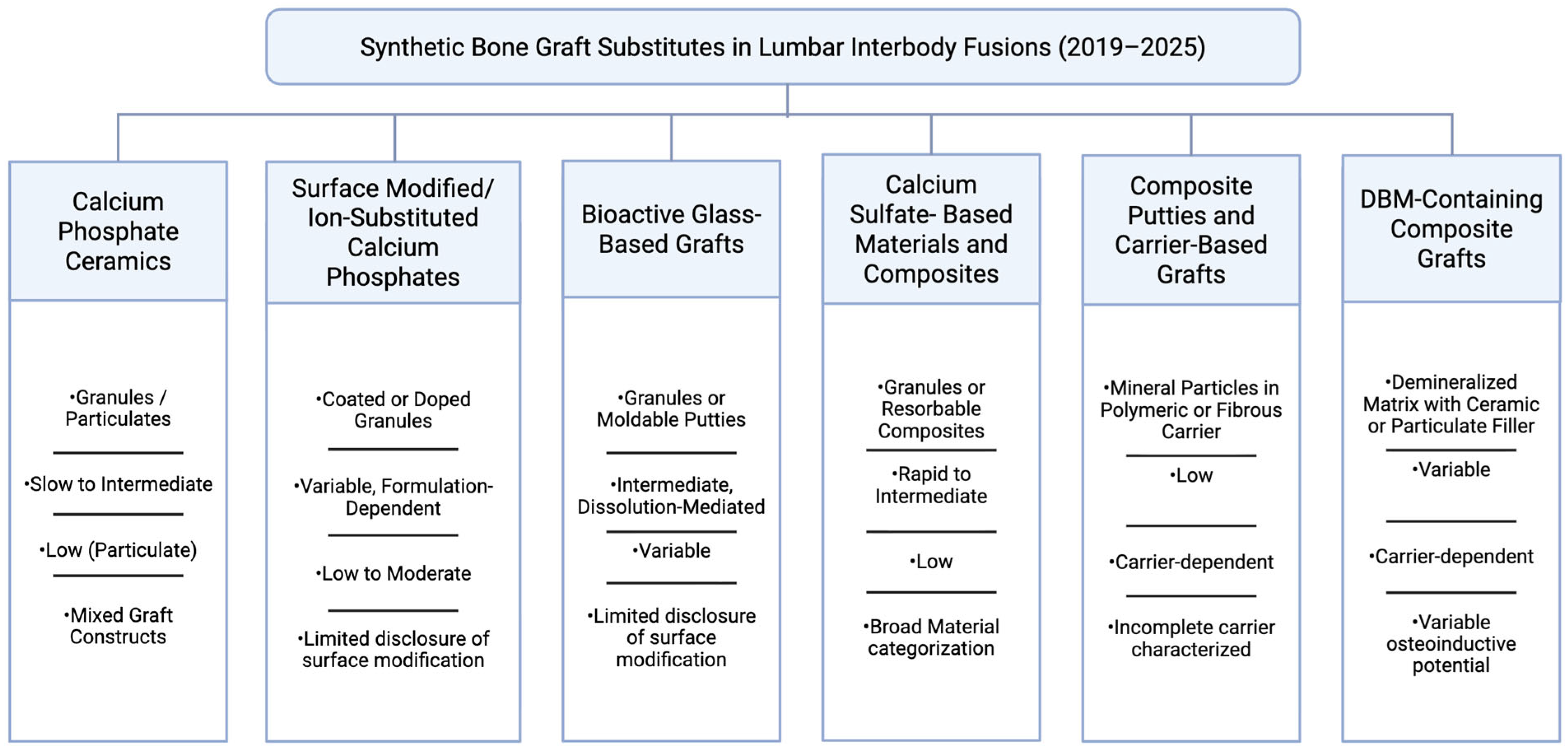
3. Material Classes of Synthetic Bone Graft Substitutes
3.1. Calcium Phosphate Ceramics
3.2. Surface-Modified and Ion-Substituted Calcium Phosphate Ceramics
3.3. Bioactive Glass-Based Graft Materials
3.4. Calcium Sulfate-Based Materials and Composites
3.5. Composite Putties, Carriers, and Cohesion-Focused Architectures
3.6. Demineralized Bone Matrix as a Frequent Composite Component
4. Early Mechanical Failure Modes at the Graft Site
4.1. Graft Displacement, Migration, and Washout
4.2. Loss of Graft–Endplate Contact and Void-Related Geometric Failure
4.3. Endplate Injury and Cage Subsidence as Coupled Mechanical Events
5. Synthesis of Recent Clinical and Translational Evidence
| Study | Fusion Approach | Graft Material Class | Adjuncts Used | Cage/Implant Context | Fusion Assessment | Follow-Up | Mechanical Events Reported |
|---|---|---|---|---|---|---|---|
| Biddau et al., 2024 [8] | ALIF | Mixed synthetic graft substitutes | ±Autograft/DBM | Interbody cages | CT | 12 months | Variably reported |
| Davis et al., 2025 [13] | TLIF | CaP composite putty | None (standalone) | Standalone interbody cage | CT | 12 months | Low subsidence incidence reported |
| Hashimoto et al., 2023 [26] | PLIF | Synthetic graft fragments | None reported | Interbody cage | CT/imaging | Acute | Migration reported |
| Kumagai et al., 2019 [38] | LLIF | Porous β-TCP | None reported | Interbody cage | CT | 12 months | Not reported |
| Nunley et al., 2024 [51] | Mixed interbody | Biphasic calcium phosphate | ±Autograft/BMP-2 | Interbody cages | CT + radiographs | 12 months | Not reported |
| Wakelin et al., 2025 [75] | Lumbar interbody | Biphasic CaP (submicron surface) | Local autograft | Interbody cages | CT | 12 months | Not reported |
| Westerlund & Borden, 2020 [77] | Lumbar interbody | Bioactive glass putty | ±Autograft | Interbody cages | Radiographs ± CT | 12 months | Not reported |
| Xie et al., 2023 [78] | Endoscopic LIF | Synthetic graft (NR) | None reported | Interbody cage | CT | Acute | Migration reported |
5.1. Definitions of Fusion and Outcome Reporting
5.2. Interpretation of Evidence by Material Class
5.3. Demineralized Bone Matrix in Composite Graft Strategies
5.4. Early Mechanical Events and Downstream Interpretation
5.5. Translational Relevance of Preclinical and Computational Studies
5.6. Summary of What Current Evidence Supports
6. Conclusions and Future Directions
Limitations
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| BMA | Bone marrow aspirtate |
| β-TCP | Beta-tricalcium phosphate |
| CT | Computed tomography |
| DBM | Demineralized bone matrix |
| LLIF | Lateral lumbar interbody fusion |
| OLIF | Oblique lumbar interbody fusion |
| PLIF | Posterior lumbar interbody fusion |
| TLIF | Transforaminal lumbar interbody fusion |
References
- Aguirre, A.O.; Soliman, M.A.R.; Kuo, C.C.; Kassay, A.; Parmar, G.; Kruk, M.D.; Quiceno, E.; Khan, A.; Lim, J.; Hess, R.M.; et al. Defining cage subsidence in anterior, oblique, and lateral lumbar spine fusion approaches: A systematic review of the literature. Neurosurg. Rev. 2024, 47, 332. [Google Scholar] [CrossRef]
- Ambrosio, L.; Schol, J.; Tamagawa, S.; Muthu, S.; Sakai, D.; Papalia, R.; Vadalà, G.; Denaro, V. Efficacy and Safety of Osteobiologics for Lumbar Spinal Fusion. J. Bone Jt. Surg. Am. 2025, 107, 2110–2121. [Google Scholar] [CrossRef]
- Amorim-Barbosa, T.; Pereira, C.; Catelas, D.; Rodrigues, C.; Costa, P.; Rodrigues-Pinto, R.; Neves, P. Risk factors for cage subsidence and clinical outcomes after transforaminal and posterior lumbar interbody fusion. Eur. J. Orthop. Surg. Traumatol. 2022, 32, 1291–1299. [Google Scholar] [CrossRef] [PubMed]
- Antonacci, C.L.; Davey, A.P.; Kia, C.; Zhou, H. Use of ceramic synthetic allografts in spine surgery: A narrative review with early basic science and clinic data of novel nanosynthetic bone graft. J. Spine Surg. 2024, 10, 715–723. [Google Scholar] [CrossRef]
- Antoniac, I.; Manescu (Paltanea), V.; Paltanea, G.; Antoniac, A.; Fosca, M.; Laptoiu, D.; Rau, J.V. Advancements in biomaterials and bioactive solutions for lumbar spine fusion cages: Current trends and future perspectives. Bioact. Mater. 2025, 53, 656–703. [Google Scholar] [CrossRef]
- Barone, A.W.; Andreana, S.; Dziak, R. Current Use of Calcium Sulfate Bone Grafts. Med. Res. Arch. 2020, 8. [Google Scholar] [CrossRef]
- Bi, F.; Chen, W.; Yu, J. Risk Factors for Cage Subsidence After Transforaminal Lumbar Interbody Fusion: A Systematic Review and Meta-Analysis. Clin Spine Surg. 2025. [Google Scholar] [CrossRef]
- Biddau, D.T.; Wang, Z.A.; Faulks, C.R.; Mobbs, R.J.; Malham, G.M. Bone graft substitutes used in anterior lumbar interbody fusion: A contemporary systematic review of fusion rates and complications. J. Spine Surg. 2024, 10, 548–561. [Google Scholar] [CrossRef]
- Chen, K.J.; Huang, Y.C.; Lin, H.H.; Chou, P.H.; Wang, S.T.; Wang, C.Y.; Chang, M.C.; Yao, Y.C. The Impact of Cage and End plate-Related Factors on Cage Subsidence in Oblique Lateral Interbody Fusion. World Neurosurg. 2023, 173, e629–e638. [Google Scholar] [CrossRef]
- Chen, R.; Xu, Z.; Chen, Q.; Li, H. Insufficient endplate-bone graft contact is a risk factor for high-grade cage subsidence occurring after lateral lumbar interbody fusion supplemented with lateral plate: An analysis of 121 cases. J. Clin. Neurosci. 2024, 129, 110818. [Google Scholar] [CrossRef]
- Cottrill, E.; Pennington, Z.; Lankipalle, N.; Ehresman, J.; Valencia, C.; Schilling, A.; Feghali, J.; Perdomo-Pantoja, A.; Theodore, N.; Sciubba, D.M.; et al. The effect of bioactive glasses on spinal fusion: A cross-disciplinary systematic review and meta-analysis of the preclinical and clinical data. J. Clin. Neurosci. 2020, 78, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Courvoisier, A.; Maximin, M.C.; Baroncini, A. Safety and Efficacy of Stand-Alone Bioactive Glass Injectable Putty or Granules in Posterior Vertebral Fusion for Adolescent Idiopathic and Non-Idiopathic Scoliosis. Children 2023, 10, 398. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.; Everist, B.; Hatfield, C.; Sage, K. A Retrospective Review of MagnetOs Easypack PuttyTM Bone Graft Used Standalone in Transforaminal Lumbar Interbody Fusion|Published in Orthopedic Reviews. Available online: https://orthopedicreviews.openmedicalpublishing.org/article/133986-a-retrospective-review-of-magnetos-easypack-putty-tm-bone-graft-used-standalone-in-transforaminal-lumbar-interbody-fusion?utm_source=chatgpt.com (accessed on 31 December 2025).
- Dinh, H.T.P.; Ushirozako, H.; Hasegawa, T.; Ebata, S.; Ohba, T.; Oba, H.; Mukaiyama, K.; Yamato, Y.; Yoshida, G.; Banno, T.; et al. Risk Factors and Consequences of Cage Subsidence after Single-level Posterior or Transforaminal Lumbar Interbody Fusion: A Retrospective Multicenter Study. Spine Surg. Relat. Res. 2024, 9, 339–349. [Google Scholar] [CrossRef]
- Dong, R.; Wu, S.; Zhong, D.; Zhao, G.; Liu, Y.; Wang, Y. Unilateral biportal endoscopic lumbar interbody fusion vs. posterior lumbar interbody fusion for the treatment of bilateral lumbar spinal stenosis. Front. Surg. 2025, 12, 1533458. [Google Scholar] [CrossRef]
- Duits, A.A.A.; van Urk, P.R.; Lehr, A.M.; Nutzinger, D.; Reijnders, M.R.L.; Weinans, H.; Foppen, W.; Oner, F.C.; van Gaalen, S.M.; Kruyt, M.C. Radiologic Assessment of Interbody Fusion: A Systematic Review on the Use, Reliability, and Accuracy of Current Fusion Criteria. JBJS Rev. 2024, 12, e23.00065. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.J.R.; Gee, A.; Kanawati, A.J.; Siddiqi, F.; Rasoulinejad, P.; Zdero, R.; Bailey, C.S. Evaluation of the contact surface between vertebral endplate and 3D printed patient-specific cage vs commercial cage. Sci. Rep. 2022, 12, 12505. [Google Scholar] [CrossRef]
- Ferrari, R. Writing narrative style literature reviews. Med. Writ. 2015, 24, 230–235. [Google Scholar] [CrossRef]
- Fiani, B.; Jarrah, R.; Shields, J.; Sekhon, M. Enhanced biomaterials: Systematic review of alternatives to supplement spine fusion including silicon nitride, bioactive glass, amino peptide bone graft, and tantalum. Neurosurg. Focus 2021, 50, E10. [Google Scholar] [CrossRef]
- Fitzgerald, A.; McCool, R.; Carr, E.; Miller, P.; Reddish, K.; Lohr, C.C.; Annoni, E.; Lawrence, B. A systematic review of bone graft products used in lumbar interbody fusion procedures for degenerative disc disease. N. Am. Spine Soc. J. 2025, 21, 100579. [Google Scholar] [CrossRef]
- Foong, B.C.M.; Wong, J.Y.H.; Betzler, B.; Oh, J.Y.L. Cage Obliquity in Oblique Lumbar Interbody Fusion—How Common Is It and What Are the Effects on Fusion Rates, Subsidence, and Sagittal Alignment? A Computed Tomography-Based Analysis. Int. J. Spine Surg. 2024, 18, 595–602. [Google Scholar] [CrossRef]
- Ge, T.; Xu, Z.; Wu, J.; Sun, Y. Pear-Shaped Disk as a Risk Factor for Intraoperative End Plate Injury in Oblique Lumbar Interbody Fusion. World Neurosurg. 2022, 165, e43–e50. [Google Scholar] [CrossRef] [PubMed]
- González-Gallardo, E.; Ekert, J.O.; Iyer, H.G.; Navarro-García de Llano, J.P.; Sánchez-Garavito, J.E.; Loizos, M.; Ríos-Zermeño, J.; Buchanan, I.A.; Abode-Iyamah, K.O.; Nottmeier, E.W.; et al. Radiological Assessment of Lumbar Fusion Status: Which Imaging Modality is Best Assessing Non-union in Lumbar Spine Pseudarthrosis? Glob. Spine J. 2025, 15, 3465–3478. [Google Scholar] [CrossRef] [PubMed]
- Green, B.N.; Johnson, C.D.; Adams, A. Writing narrative literature reviews for peer-reviewed journals: Secrets of the trade. J. Chiropr. Med. 2006, 5, 101–117. [Google Scholar] [CrossRef]
- Ham, D.W.; Park, S.M.; Kim, Y.B.; Chang, D.G.; Yang, J.J.; Kwon, B.T.; Song, K.S. Feasibility of the Non-Window-Type 3D-Printed Porous Titanium Cage in Posterior Lumbar Interbody Fusion: A Randomized Controlled Multicenter Trial. J. Bone Jt. Surg. Am. 2024, 106, 2102–2110. [Google Scholar] [CrossRef]
- Hashimoto, M.; Hirata, H.; Tsukamoto, M.; Tomohito, Y.; Mawatari, M.; Morimoto, T. Anterior graft migration in posterior lumbar interbody fusion: A case report and literature review. Clin. Case Rep. 2023, 11, e7847. [Google Scholar] [CrossRef]
- Heard, J.C.; Lee, Y.; Lee, Y.; Lambrechts, M.J.; Berthiaume, E.; D’Antonio, N.D.; Bodnar, J.; Paulik, J.; Mangan, J.J.; Canseco, J.A.; et al. The impact of demineralized bone matrix characteristics on pseudarthrosis and surgical outcomes after posterolateral lumbar decompression and fusion. J. Craniovertebral Junction Spine 2023, 14, 194. [Google Scholar] [CrossRef]
- Hefny, S.; El Gayar, A.; Ghobashy, M.K.; Youssef, M.E.; AbdelFatah, M.A.; Elshazly, K. Is the size of the transforaminal lumbar cage a risk factor for cage subsidence? A retrospective cohort study. Neurosurg. Rev. 2025, 48, 427. [Google Scholar] [CrossRef]
- Higgins, J.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.; Welch, V. Cochrane Handbook for Systematic Reviews of Interventions (Current Version)|Cochrane. Available online: https://www.cochrane.org/authors/handbooks-and-manuals/handbook/current (accessed on 31 December 2025).
- Himawan, F.; Widhiyanto, L. Investigating the effect of combination between demineralized bone matrix and hydroxyapatite in spinal fusion: A systematic review and meta analysis. J. Orthop. Rep. 2025, 100787. [Google Scholar] [CrossRef]
- Hou, Y.; Shi, H.; Shi, H.; Zhao, T.; Shi, J.; Shi, G. A meta-analysis of risk factors for cage migration after lumbar fusion surgery. World Neurosurg. X 2023, 18, 100152. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A. How to Make More Published Research True. PLoS Med. 2014, 11, e1001747. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Park, M.K.; Park, Y.J.; Cho, D.C.; San Ko, Y. Reduction-First Technique of Unilateral Biportal Endoscopy Lumbar Interbody Fusion for Spondylolisthesis. World Neurosurg. 2025, 198, 124005. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Ha, K.Y.; Kim, K.T.; Chang, D.G.; Park, H.Y.; Yoon, E.J.; Kim, S.I. Risk factors for intraoperative endplate injury during minimally-invasive lateral lumbar interbody fusion. Sci. Rep. 2021, 11, 20149. [Google Scholar] [CrossRef]
- Koo, B.; Park, J.; Hong, J.Y. Predictors of Cage Subsidence After Oblique Lumbar Interbody Fusion. J. Clin. Med. 2025, 14, 8956. [Google Scholar] [CrossRef] [PubMed]
- Kotheeranurak, V.; Jitpakdee, K.; Lin, G.X.; Mahatthanatrakul, A.; Singhatanadgige, W.; Limthongkul, W.; Yingsakmongkol, W.; Kim, J.S. Subsidence of Interbody Cage Following Oblique Lateral Interbody Fusion: An Analysis and Potential Risk Factors. Glob. Spine J. 2023, 13, 1981–1991. [Google Scholar] [CrossRef]
- Kucko, N.W.; Barrère-de Groot, F.; Wills, D.; Wang, T.; Pelletier, M.; de Bruijn, J.D.; Walsh, W.R. Preclinical Evaluation of Synthetic Bone Graft Materials in an Instrumented Sheep Posterolateral Lumbar Spinal Fusion Model. Clin. Spine Surg. 2025. [Google Scholar] [CrossRef]
- Kumagai, H.; Abe, T.; Koda, M.; Nagashima, K.; Miura, K.; Mataki, K.; Fujii, K.; Noguchi, H.; Funayama, T.; Yamazaki, M. Unidirectional porous β-tricalcium phosphate induces bony fusion in lateral lumbar interbody fusion. J. Clin. Neurosci. 2019, 59, 232–235. [Google Scholar] [CrossRef]
- Lee, B.J.; Seok, M.C.; Koo, H.W.; Jeong, J.H.; Ko, M.J. Bone Substitute Options for Spine Fusion in Patients with Spine Trauma-Part I: Fusion Biology, Autografts, Allografts, Demineralized Bone Matrix, and Ceramics. Korean J. Neurotrauma 2023, 19, 446. [Google Scholar] [CrossRef]
- Lehr, A.M.; Duits, A.A.A.; Reijnders, M.R.L.; Nutzinger, D.; Castelein, R.M.; Oner, F.C.; Kruyt, M.C. Assessment of Posterolateral Lumbar Fusion: A Systematic Review of Imaging-Based Fusion Criteria. JBJS Rev. 2022, 10, e22.00129. [Google Scholar] [CrossRef]
- Li, Q.; Long, X.; Shi, L.; Wang, Y.; Guan, T.; Lv, J.; Cai, L. Prevalence and risk factors for cage subsidence after lumbar interbody fusion. Medicine 2021, 100, e28085. [Google Scholar] [CrossRef]
- Lin, G.X.; He, L.R.; Nan, J.N.; Xu, W.B. Comparing Outcomes of Banana-Shaped and Straight Cages in Transforaminal Lumbar Interbody Fusion for Lumbar Degenerative Diseases: A Systematic Review and Meta-Analysis. Neurospine. 2024, 21, pp. 261–272. [CrossRef]
- Lun, D.; Li, S.; Li, N.; Mou, L.M.; Li, H.Q.; Zhu, W.P.; Li, H.F.; Hu, Y.C. Limitations and modifications in the clinical application of calcium sulfate. Front Surg. 2024, 11, 1278421. [Google Scholar] [CrossRef] [PubMed]
- Maeno, T.; Okuda, S.; Haku, T.; Yamashita, T.; Matsumoto, T.; Sugiura, T.; Oda, T.; Iwasaki, M. Anterior migration of an interbody graft in posterior lumbar interbody fusion: Report of three cases without removal of the migrated graft. J. Orthop. Sci. 2019, 24, 742–745. [Google Scholar] [CrossRef]
- Michael Cheers, G.; Philipp Weimer, L.; Neuerburg, C.; Arnholdt, J.; Gilbert, F.; Thorwächter, C.; Holzapfel, B.M.; Mayer-Wagner, S.; Laubach, M. Advances in implants and bone graft types for lumbar spinal fusion surgery. Biomater. Sci. 2024, 12, 4875–4902. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Munn, Z.; Peters, M.D.J.; Stern, C.; Tufanaru, C.; McArthur, A.; Aromataris, E. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med. Res. Methodol. 2018, 18, 143. [Google Scholar] [CrossRef]
- Murad, M.H.; Asi, N.; Alsawas, M.; Alahdab, F. New evidence pyramid. Evid. Based Med. 2016, 21, 125–127. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, H.; Ito, S.; Segi, N.; Ouchida, J.; Yamauchi, I.; Morita, Y.; Kanemura, T.; Imagama, S. Current status of indirect decompression with lateral lumbar interbody fusion. Nagoya J. Med. Sci. 2025, 87, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Nery Navarro, J.; Colaço Aguiar, N.R.; Roxo Fernandes, A.; Santos Baptista, V.; Bertolini, M.G.V.B.M.; Rubens Dias Pereira Filho, A. Posterior cage migration after transforaminal lumbar interbody fusion: Risk factors and treatment. Surg. Neurol. Int. 2025, 16, 266. [Google Scholar] [CrossRef]
- Nunley, P.; Sanda, M.; Googe, H.; Cavanaugh, D.; Sage, K.; Ryaby, J.; Stone, M.B. Biphasic Calcium Phosphate Bone Graft with a Unique Surface Topography: A Single-Center Ambispective Study for Degenerative Disease of the Lumbar Spine. Cureus 2024, 16, e58218. [Google Scholar] [CrossRef]
- Ohnmeiss, D.D.; Stastny, D.C.; Buser, Z.; Ferrara, L.A. Section on innovative spine research and novel technologies: Fusion cage design, materials, and coatings: Science versus hype. N. Am. Spine Soc. J. 2025, 24, 100814. [Google Scholar] [CrossRef]
- Olivos-Meza, A.; Mata-Miranda, M.M.; Robles-Rodríguez, M.; Vázquez-Zapién, G.J.; Guerrero-Ruiz, M.; Landa-Solís, C. Osteogenic Potential and Bone Matrix Maturity: Comparison of Demineralized Bone Matrix and P15 Polypeptide iFactor® in an In Vitro Study. Medicina 2025, 61, 914. [Google Scholar] [CrossRef]
- Pace, R.D.; Molinari, S.; Mazzoni, E.; Perale, G. Bone Regeneration: A Review of Current Treatment Strategies. J. Clin. Med. 2025, 14, 1838. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Parisien, A.; Wai, E.K.; ElSayed, M.S.A.; Frei, H. Subsidence of Spinal Fusion Cages: A Systematic Review. Int. J. Spine Surg. 2022, 16, 1103–1118. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Shin, J.I.; You, K.H.; Yang, J.I.; Kim, N.; Kim, Y.H.; Kang, M.S.; Park, S.M. Biportal Endoscopic Transforaminal Lumbar Interbody Fusion: How to Improve Fusion Rate? Int. J. Spine Surg. 2024, 18, 582–588. [Google Scholar] [CrossRef]
- Park, S.M.; Park, H.J.; You, K.H.; Kim, H.J.; Yeom, J.S. Biportal endoscopic lumbar interbody fusion using a large polyetheretherketone cage: Preliminary results. Asian Spine J. 2025, 19, 252–258. [Google Scholar] [CrossRef]
- Pholprajug, P.; Kotheeranurak, V.; Liu, Y.; Kim, J.S. The Endoscopic Lumbar Interbody Fusion: A Narrative Review, and Future Perspective. Neurospine 2023, 20, 1224–1245. [Google Scholar] [CrossRef] [PubMed]
- Plantz, M.A.; Gerlach, E.B.; Hsu, W.K. Synthetic bone graft materials in spine fusion: Current evidence and future trends. Int. J. Spine Surg. 2021, 15, 104. [Google Scholar] [CrossRef]
- Raabe, C.; Cafferata, E.A.; Zhou, W.; Müller, K.M.; Lingwal, N.; Ramanauskaite, A.; Schwarz, F.; Couso-Queiruga, E. The Effect of Defect Morphology and Membrane Fixation on 3D Graft Material Displacement During Primary Wound Closure in Horizontal Bone Augmentation-An Ex Vivo Study. Clin. Oral Implant. Res. 2025. [Google Scholar] [CrossRef]
- Rethlefsen, M.L.; Kirtley, S.; Waffenschmidt, S.; Ayala, A.P.; Moher, D.; Page, M.J.; Koffel, J.B.; PRISMA-S Group. PRISMA-S: An extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Syst. Rev. 2021, 10, 39. [Google Scholar] [CrossRef]
- Sato, K.; Funayama, T.; Noguchi, H.; Asada, T.; Kono, M.; Eto, F.; Shibao, Y.; Miura, K.; Kikuchi, N.; Yoshioka, T.; et al. Efficacy of platelet-rich plasma impregnation for unidirectional porous β-tricalcium phosphate in lateral lumbar interbody fusion: Study protocol for a prospective controlled trial. Trials 2022, 23, 908. [Google Scholar] [CrossRef]
- Shabani, S.; Yoshida, B.; Ton, A.; Karakash, W.; Avetisian, H.; Rusu, D.; Lindgren, A.; Mills, E.; Wang, J.C.; Hah, R.J.; et al. The Effect of Positional Graft Placement on Pseudarthrosis Rates in Anterior Cervical Discectomy and Fusion. Spine 2025. [Google Scholar] [CrossRef]
- Sanghvi, P.A.; Wiener, J.M.; Meade, S.M.; Boden, L.M.; Shost, M.D.; Steinmetz, M.P. Development of a unified and comprehensive definition of successful spinal fusion: A systematic review. J. Neurosurg. Spine 2025, 42, 403–412. [Google Scholar] [CrossRef]
- Shen, S.; You, X.; Ren, Y.; Ye, S. Risk Factors of Cage Subsidence Following Oblique Lumbar Interbody Fusion: A Meta-analysis and Systematic Review. World Neurosurg. 2024, 183, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Shepard, N.A.; Rush, A.J.; Scarborough, N.L.; Carter, A.J.; Phillips, F.M. Demineralized Bone Matrix in Spine Surgery: A Review of Current Applications and Future Trends. Int. J. Spine Surg. 2021, 15, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Wang, X.H.; Zhu, L.; Chen, L.; Jiang, Z.L.; Wu, X.T. Intraoperative Endplate Injury Following Transforaminal Lumbar Interbody Fusion. World Neurosurg. 2022, 168, e110–e118. [Google Scholar] [CrossRef] [PubMed]
- Stempels, H.W.; Lehr, A.M.; Delawi, D.; Hoebink, E.A.; Wiljouw, I.A.A.A.; Kempen, D.H.R.; van Susante, J.L.C.; Kruyt, M.C.; Dutch Clinical Spine Research Group. Efficacy of Biphasic Calcium Phosphate Ceramic with a Needle-Shaped Surface Topography Versus Autograft in Instrumented Posterolateral Spinal Fusion. Spine 2024, 49, 1323. [Google Scholar] [CrossRef]
- Sukhera, J. Narrative Reviews in Medical Education: Key Steps for Researchers. J. Grad. Med. Educ. 2022, 14, 418–419. [Google Scholar] [CrossRef]
- Tian, X.; Vater, C.; Raina, D.B.; Findeisen, L.; Matuszewski, L.M.; Tägil, M.; Lidgren, L.; Winkler, A.; Gottwald, R.; Modler, N.; et al. Co-delivery of rhBMP-2 and zoledronic acid using calcium sulfate/hydroxyapatite carrier as a bioactive bone substitute to enhance and accelerate spinal fusion. Bioact. Mater. 2024, 36, 256–271. [Google Scholar] [CrossRef]
- Trygonis, N.; Daskalakis, I.I.; Tsagkaris, C.; Trygonis, N.; Daskalakis, I.I.; Tsagkaris, C. The Application of Biologic and Synthetic Bone Grafts in Scoliosis Surgery: A Scoping Review of Emerging Technologies. Healthcare 2025, 13, 2359. [Google Scholar] [CrossRef]
- Ushirozako, H.; Hasegawa, T.; Ebata, S.; Ohba, T.; Oba, H.; Mukaiyama, K.; Shimizu, S.; Yamato, Y.; Ide, K.; Shibata, Y.; et al. Impact of sufficient contact between the autograft and endplate soon after surgery to prevent nonunion at 12 months following posterior lumbar interbody fusion. J. Neurosurg. Spine 2020, 33, 796–805. [Google Scholar] [CrossRef]
- Velluto, C.; Mundis, G.; Scaramuzzo, L.; Perna, A.; Capece, G.; Cruciani, A.; Inverso, M.; Borruto, M.I.; Proietti, L. Radiological evaluation of fusion patterns after Lateral Lumbar Interbody fusion with 3D-printed porous titanium cages vs. conventional titanium cages. Front. Surg. 2024, 11, 1446792. [Google Scholar] [CrossRef]
- Wakelin, S.H.; Cobourn, K.D.; Stirrat, T.; Sayah, A.; Sage, K.; Ryaby, J.; Sandhu, F.A. Fusion Rate of Biphasic Calcium Phosphate Bone Graft with Needle-Shaped Submicron Surface Topography in Interbody Lumbar Fusion for Degenerative Disc Disease: A Single-Center Retrospective Review. World Neurosurg. 2025, 196, 123759. [Google Scholar] [CrossRef]
- Wang, J.L.; Eaton, R.G.; Harrigan, M.E.; Munjal, V.; Sette, K.N.; Wilson, S.B.; Grossbach, A.J. Demineralized Bone Matrix and Fibers in Spinal Fusion. Int. J. Spine Surg. 2023, 17, S28. [Google Scholar] [CrossRef] [PubMed]
- Westerlund, L.E.; Borden, M. Clinical experience with the use of a spherical bioactive glass putty for cervical and lumbar interbody fusion. J. Spine Surg. 2020, 6, 49–61. [Google Scholar] [CrossRef]
- Xie, Y.; Zhou, Q.; Wang, Y.; Feng, C.; Fan, X.; Yu, Y. Postoperative bone graft migration into the thecal sac and shifting down to the lower level after an endoscopic lumbar interbody fusion: A case report. BMC Musculoskelet. Disord. 2023, 24, 143. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Li, S.; Wang, W.; Li, J.; Zhang, W. Demineralized bone matrix combined with concentrated growth factors promotes intervertebral fusion in a novel rat extreme lateral interbody fusion model. J. Orthop. Surg. Res. 2025, 20, 529. [Google Scholar] [CrossRef]
- Yao, Y.C.; Chou, P.H.; Lin, H.H.; Wang, S.T.; Liu, C.L.; Chang, M.C. Risk Factors of Cage Subsidence in Patients Received Minimally Invasive Transforaminal Lumbar Interbody Fusion. Spine 2020, 45, E1279–E1285. [Google Scholar] [CrossRef]
- You, K.H.; Cho, S.K.; Hwang, J.Y.; Cha, S.H.; Kang, M.S.; Park, S.M.; Park, H.J. Effect of Cage Material and Size on Fusion Rate and Subsidence Following Biportal Endoscopic Transforaminal Lumbar Interbody Fusion. Neurospine 2024, 21, 973–983. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.; Tiao, J.; Cai, C.W.; Huang, J.J.; Mohamed, K.; Hoang, R.; Hong, J.; Berman, D.; Lee, J.; Ambrosio, L.; et al. Radiographic Assessment of Successful Lumbar Spinal Fusion: A Systematic Review of Fusion Criteria in Randomized Trials. Glob. Spine J. 2025, 21925682251384662. [Google Scholar] [CrossRef]
- Zhang, Y.; Shu, T.; Wang, S.; Liu, Z.; Cheng, Y.; Li, A.; Pei, D. The Osteoinductivity of Calcium Phosphate-Based Biomaterials: A Tight Interaction with Bone Healing. Front. Bioeng. Biotechnol. 2022, 10, 911180. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wise, J.; Merem, I.; Wrubluski, D.; Zhang, X.; Weston, R.; Shi, M.; Lin, M.; Vrionis, F.D. A Review of Synthetic Bone Grafts in Lumbar Interbody Fusion. Bioengineering 2026, 13, 262. https://doi.org/10.3390/bioengineering13030262
Wise J, Merem I, Wrubluski D, Zhang X, Weston R, Shi M, Lin M, Vrionis FD. A Review of Synthetic Bone Grafts in Lumbar Interbody Fusion. Bioengineering. 2026; 13(3):262. https://doi.org/10.3390/bioengineering13030262
Chicago/Turabian StyleWise, Jaden, Isabella Merem, Dahlia Wrubluski, Xuanzong Zhang, Ridge Weston, Min Shi, Maohua Lin, and Frank D. Vrionis. 2026. "A Review of Synthetic Bone Grafts in Lumbar Interbody Fusion" Bioengineering 13, no. 3: 262. https://doi.org/10.3390/bioengineering13030262
APA StyleWise, J., Merem, I., Wrubluski, D., Zhang, X., Weston, R., Shi, M., Lin, M., & Vrionis, F. D. (2026). A Review of Synthetic Bone Grafts in Lumbar Interbody Fusion. Bioengineering, 13(3), 262. https://doi.org/10.3390/bioengineering13030262








